Stabilization of Enzymes by Multipoint Covalent Attachment on Aldehyde-Supports: 2-Picoline Borane as an Alternative Reducing Agent
Abstract
:1. Introduction
- The poor solubility of 2-PB in aqueous solutions.
- The necessity for obtaining an inert support after the immobilization process, reducing the remaining aldehydes to hydroxyls, or blocking them with other substances.
- The ability of the 2-PB to reduce all the Schiff bases that are formed between the enzyme and the support. 2-PB molecules, which are much larger molecules than the hydrides, have to go through the region that lies between the enzyme and the support to reduce all the Schiff base (the width of this region is approximately 2 nm) [25].
2. Results
2.1. Immobilization of PGA, DAAO, ADH2, GlyDH, and Xys1Δ on Glyoxyl-Activated Agarose
2.2. Reduction of the Immobilized Enzymes with 2-Picoline Borane
2.3. Comparison of Immobilized Enzymes Reduced with Borohydride and 2-Picoline Borane: Expressed Activity and Thermal Stability
2.4. Evaluation of the Correlation between Thermal Stability and Number of Enzyme–Support
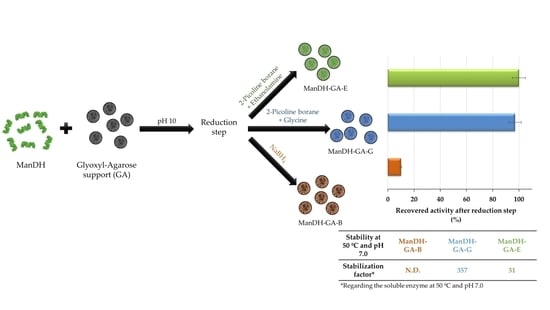
2.5. Immobilization–Stabilization of a Bacterial Mandelate Dehydrogenase: Optimization of the Eduction Step
3. Materials and Methods
3.1. Materials
3.2. Methods
3.2.1. Protein Production
3.2.2. Enzymatic Assays
3.2.3. Support Immobilization Preparation
3.2.4. Protein Immobilization
Reduction of Schiff Base with Sodium Borohydride
Reduction of Schiff Bases with the 2-Picoline Borane Complex
3.2.5. Thermal Inactivation Assays
3.2.6. Estimation of the Lysine Residues Involved in the Immobilization of the Enzyme on the GA Support
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Paradisi, F. Biocatalysis takes to flow. Chim. Oggi/Chem. Today 2017, 35, 38–40. [Google Scholar]
- Choi, J.M.; Han, S.S.; Kim, H.S. Industrial applications of enzyme biocatalysis: Current status and future aspects. Biotechnol. Adv. 2015, 33, 1443–1454. [Google Scholar] [CrossRef] [PubMed]
- DiCosimo, R.; McAuliffe, J.; Poulose, A.J.; Bohlmann, G. Industrial use of immobilized enzymes. Chem. Soc. Rev. 2013, 42, 6437–6474. [Google Scholar] [PubMed]
- Wells, A.; Meyer, H.P. Biocatalysis as a strategic green technology for the chemical industry. ChemCatChem 2014, 6, 918–920. [Google Scholar] [CrossRef]
- Valavanidis, A. Biocatalysis and directed evolution in the pharmaceutical industry. New developments for ‘green’ synthetic methods of high value pharmaceuticals. Pharmakeftiki 2016, 28, 117–130. [Google Scholar]
- Homaei, A.A.; Sariri, R.; Vianello, F.; Stevanato, R. Enzyme immobilization: An update. J. Chem. Biol. 2013, 6, 185–205. [Google Scholar] [CrossRef] [PubMed]
- Brady, D.; Jordaan, J. Advances in enzyme immobilisation. Biotechnol. Lett 2009, 31, 1639–1650. [Google Scholar] [CrossRef] [PubMed]
- Mateo, C.; Palomo, J.M.; Fernandez-Lorente, G.; Guisan, J.M.; Fernandez-Lafuente, R. Improvement of enzyme activity, stability and selectivity via immobilization techniques. Enzym. Microb. Technol. 2007, 40, 1451–1463. [Google Scholar] [CrossRef]
- Fernández-Lorente, G.; Lopez-Gallego, F.; Bolivar, J.M.; Rocha-Martin, J.; Moreno-Perez, S.; Guisán, J.M. Immobilization of proteins on highly activated glyoxyl supports: Dramatic increase of the enzyme stability via multipoint immobilization on pre-existing carriers. Curr. Org. Chem. 2015, 19, 1–13. [Google Scholar] [CrossRef]
- Fernández-Lorente, G.; Betancor, L.; Carrascosa, A.V.; Palomo, J.M.; Guisan, J.M. Modulation of the selectivity of immobilized lipases by chemical and physical modifications: Release of omega-3 fatty acids from fish oil. J. Am. Oil Chem. Soc. 2012, 89, 97–102. [Google Scholar] [CrossRef]
- Rocha-Martin, J.; Acosta, A.; Berenguer, J.; Guisan, J.M.; Lopez-Gallego, F. Selective oxidation of glycerol to 1,3-dihydroxyacetone by covalently immobilized glycerol dehydrogenases with higher stability and lower product inhibition. Bioresour. Technol. 2014, 170, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Liese, A.; Hilterhaus, L. Evaluation of immobilized enzymes for industrial applications. Chem. Soc. Rev. 2013, 42, 6236–6249. [Google Scholar] [CrossRef] [PubMed]
- Eş, I.; Vieira, J.D.G.; Amaral, A.C. Principles, techniques, and applications of biocatalyst immobilization for industrial application. Appl. Microbiol. Biotechnol. 2015, 99, 2065–2082. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brena, B.; González-Pombo, P.; Batista-Viera, F. Immobilization of Enzymes: A Literature Survey. In Immobilization of Enzymes and Cells: Third Edition; Guisan, J.M., Ed.; Humana Press: Totowa, NJ, USA; New York, NY, USA, 2013; pp. 15–31. [Google Scholar]
- Datta, S.; Christena, L.R.; Rajaram, Y.R.S. Enzyme immobilization: An overview on techniques and support materials. 3 Biotech. 2013, 3, 1–9. [Google Scholar] [PubMed]
- Garcia-Galan, C.; Berenguer-Murcia, Á.; Fernandez-Lafuente, R.; Rodrigues, R.C. Potential of different enzyme immobilization strategies to improve enzyme performance. Adv. Synth. Catal. 2011, 353, 2885–2904. [Google Scholar]
- Bernal, C.; Guzman, F.; Illanes, A.; Wilson, L. Selective and eco-friendly synthesis of lipoaminoacid-based surfactants for food, using immobilized lipase and protease biocatalysts. Food Chem. 2018, 239, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Guisán, J.M. Aldehyde-agarose gels as activated supports for immobilization-stabilization of enzymes. Enzym. Microb. Technol. 1988, 10, 375–382. [Google Scholar] [CrossRef]
- Benítez-Mateos, A.I.; Sebastian, E.S.; Ríos-Lombardía, N.; Morís, F.; González-Sabín, J.; López-Gallego, F. asymmetric reduction of prochiral ketones by using self-sufficient heterogeneous biocatalysts based on NADPH-dependent ketoreductases. Chem. Eur. J. 2017, 23, 16843–16852. [Google Scholar] [CrossRef] [PubMed]
- Romero-Fernández, M.; Moreno-Perez, S.; Orrego, A.H.; Martins de Oliveira, S.; Santamaría, R.I.; Díaz, M.; Guisan, J.M.; Rocha-Martin, J. Designing continuous flow reaction of xylan hydrolysis for xylooligosaccharides production in packed-bed reactors using xylanase immobilized on methacrylic polymer-based supports. Bioresour. Technol. 2018, 266, 249–258. [Google Scholar] [CrossRef] [PubMed]
- Marques Netto, C.G.C.; da Silva, D.G.; Toma, S.H.; Andrade, L.H.; Nakamura, M.; Araki, K.; Toma, H.E. Bovine glutamate dehydrogenase immobilization on magnetic nanoparticles: Conformational changes and catalysis. RSC Adv. 2016, 6, 12977–12992. [Google Scholar] [CrossRef]
- Bassan, J.; de Souza Bezerra, T.; Peixoto, G.; da Cruz, C.; Galán, J.; Vaz, A.; Garrido, S.; Filice, M.; Monti, R. Immobilization of trypsin in lignocellulosic waste material to produce peptides with bioactive potential from whey protein. Materials 2016, 9, 357. [Google Scholar] [CrossRef] [PubMed]
- Bezerra, T.M.d.S.; Bassan, J.C.; Santos, V.T.d.O.; Ferraz, A.; Monti, R. Covalent immobilization of laccase in green coconut fiber and use in clarification of apple juice. Process. Biochem. 2015, 50, 417–423. [Google Scholar] [CrossRef]
- Mateo, C.; Bolivar, J.M.; Godoy, C.A.; Rocha-Martin, J.; Pessela, B.C.; Curiel, J.A.; Muñoz, R.; Guisan, J.M.; Fernández-Lorente, G. Improvement of enzyme properties with a two-step immobilizaton process on novel heterofunctional supports. Biomacromolecules 2010, 11, 3112–3117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Orrego, A.H.; Garcia, C.; Mancheno, J.M.; Guisan, J.M.; Lillo, M.P.; Lopez-Gallego, F. Two-Photon Fluorescence Anisotropy Imaging to Elucidate the Dynamics and the Stability of Immobilized Proteins. J. Phys. Chem. B 2016, 120, 485–491. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ottone, C.; Bernal, C.; Serna, N.; Illanes, A.; Wilson, L. Enhanced long-chain fatty alcohol oxidation by immobilization of alcohol dehydrogenase from S. cerevisiae. Appl. Microbiol. Biotechnol. 2018, 102, 237–247. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, S.M.; Moreno-Perez, S.; Terrasan, C.R.F.; Romero-Fernández, M.; Vieira, M.F.; Guisan, J.M.; Rocha-Martin, J. Covalent immobilization-stabilization of β-1,4-endoxylanases from Trichoderma reesei: Production of xylooligosaccharides. Process. Biochem. 2018, 64, 170–176. [Google Scholar] [CrossRef]
- Trobo-Maseda, L.; Orrego, A.H.; Moreno-Pérez, S.; Fernández-Lorente, G.; Guisan, J.M.; Rocha-Martin, J. Stabilization of multimeric sucrose synthase from Acidithiobacillus caldus via immobilization and post-immobilization techniques for synthesis of UDP-glucose. Appl. Microbiol. Biotechnol. 2018, 102, 773–787. [Google Scholar] [CrossRef] [PubMed]
- Rocha-Martín, J.; Vega, D.; Bolivar, J.M.; Hidalgo, A.; Berenguer, J.; Guisán, J.M.; López-Gallego, F. Characterization and further stabilization of a new anti-prelog specific alcohol dehydrogenase from Thermus thermophilus HB27 for asymmetric reduction of carbonyl compounds. Bioresour. Technol. 2012, 103, 343–350. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Orrego, A.H.; Trobo-Maseda, L.; Rocha-Martin, J.; Guisan, J.M. Immobilization-stabilization of a complex multimeric sucrose synthase from Nitrosomonas europaea. Synthesis of UDP-glucose. Enzym. Microb. Technol. 2017, 105, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Jackson, E.; López-Gallego, F.; Guisan, J.M.; Betancor, L. Enhanced stability of L-lactate dehydrogenase through immobilization engineering. Process. Biochem. 2016, 51, 1248–1255. [Google Scholar] [CrossRef]
- López-Gallego, F.; Fernandez-Lorente, G.; Rocha-Martin, J.; Bolivar, J.M.; Mateo, C.; Guisan, J.M. Stabilization of enzymes by multipoint covalent immobilization on supports activated with glyoxyl groups. Methods Mol. Biol. 2013, 1051, 59–71. [Google Scholar] [PubMed]
- Sato, S.; Sakamoto, T.; Miyazawa, E.; Kikugawa, Y. One-pot reductive amination of aldehydes and ketones with α-picoline-borane in methanol, in water, and in neat conditions. Tetrahedron 2004, 60, 7899–7906. [Google Scholar] [CrossRef]
- Ruhaak, L.R.; Steenvoorden, E.; Koeleman, C.A.M.; Deelder, A.M.; Wuhrer, M. 2-Picoline-borane: A non-toxic reducing agent for oligosaccharide labeling by reductive amination. Proteomics 2010, 10, 2330–2336. [Google Scholar] [CrossRef] [PubMed]
- Cosenza, V.A.; Navarro, D.A.; Stortz, C.A. Usage of α-picoline borane for the reductive amination of carbohydrates. ARKIVOC 2011, 2011, 182–194. [Google Scholar]
- Ambrogelly, A.; Cutler, C.; Paporello, B. Screening of reducing agents for the pegylation of recombinant human IL-10. Protein J. 2013, 32, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Aragon, C.C.; Mateo, C.; Ruiz-Matute, A.I.; Corzo, N.; Fernandez-Lorente, G.; Sevillano, L.; Díaz, M.; Monti, R.; Santamaría, R.I.; Guisan, J.M. Production of xylo-oligosaccharides by immobilized-stabilized derivatives of endo-xylanase from Streptomyces halstedii. Process. Biochem. 2013, 48, 478–483. [Google Scholar] [CrossRef] [Green Version]
- Betancor, L.; Hidalgo, A.; Fernández-Lorente, G.; Mateo, C.; Rodríguez, V.; Fuentes, M.; López-Gallego, F.; Fernández-Lafuente, R.; Guisan, J.M. Use of physicochemical tools to determine the choice of optimal enzyme: Stabilization of D-amino acid oxidase. Biotechnol. Prog. 2003, 19, 784–788. [Google Scholar] [CrossRef] [PubMed]
- Alvaro, G.; Fernandez-Lafuente, R.; Blanco, R.M.; Guisán, J.M. Immobilization-stabilization of Penicillin G acylase from Escherichia coli. Appl. Biochem. Biotechnol. 1990, 26, 181–195. [Google Scholar] [CrossRef] [PubMed]
- Romero-Fernández, M.; Moreno-Perez, S.; Martins de Oliveira, S.; Santamaría, R.I.; Guisan, J.M.; Rocha-Martin, J. Preparation of a robust immobilized biocatalyst of β-1,4-endoxylanase by surface coating with polymers for production of xylooligosaccharides from different xylan sources. New Biotechnol. 2018, 44, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Kunduru, K.R.; Katzhendler, J.; Domb, A.J. Poly(α-hydroxy acid)s and poly(α-hydroxy acid-co-α-amino acid)s derived from amino acid. Adv. Drug Del. Rev. 2016, 107, 82–96. [Google Scholar] [CrossRef] [PubMed]
- Kneedler, J.A.; Sky, S.S.; Sexton, L.R. Understanding alpha-hydroxy acids. Dermatol. Nurs./Dermatol. Nurses’ Assoc. 1998, 10, 247–254, 259–262, quiz 265–266. [Google Scholar]
- Howard, P.C.; Sams Ii, R.L.; Dennis, D.A.; Wamer, W.G. Alpha-hydroxy acids: Consideration of the biological effects and possible role in photocarcinogenesis. J. Food Drug Anal. 2002, 10, 258–261. [Google Scholar]
- Kawase, Y.; Yamagishi, T.; Kutsuma, T.; Ueda, K.; Iwakuma, T.; Nakata, T.; Yokomatsu, T. One-pot synthesis of alkoxyamine derivatives by reductive alkoxyamination with a 2-picoline-borane complex. Heterocycles 2009, 78, 463–470. [Google Scholar] [CrossRef]
- Tamashima, E.; Hayama, T.; Yoshida, H.; Imakyure, O.; Yamaguchi, M.; Nohta, H. Direct tandem mass spectrometric analysis of amino acids in plasma using fluorous derivatization and monolithic solid-phase purification. J. Pharm. Biomed. Anal. 2015, 115, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Duff, R.J.; Smith, E.; Li, W.; Fodor, S. Evidence for an imidazoline by-product from glycans using tandem mass spectrometry. J. Chromatogr. A 2017, 1501, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Pedroche, J.; del Mar Yust, M.; Mateo, C.; Fernández-Lafuente, R.; Girón-Calle, J.; Alaiz, M.; Vioque, J.; Guisán, J.M.; Millán, F. Effect of the support and experimental conditions in the intensity of the multipoint covalent attachment of proteins on glyoxyl-agarose supports: Correlation between enzyme–support linkages and thermal stability. Enzym. Microb. Technol. 2007, 40, 1160–1166. [Google Scholar] [CrossRef]
- Grazú, V.; López-Gallego, F.; Montes, T.; Abian, O.; González, R.; Hermoso, J.A.; García, J.L.; Mateo, C.; Guisán, J.M. Promotion of multipoint covalent immobilization through different regions of genetically modified penicillin G acylase from E. coli. Process. Biochem. 2010, 45, 390–398. [Google Scholar] [CrossRef]
- López-Gallego, F.; Betancor, L.; Hidalgo, A.; Alonso, N.; Fernandez-Lorente, G.; Guisan, J.M.; Fernandez-Lafuente, R. Preparation of a robust biocatalyst of d-amino acid oxidase on sepabeads supports using the glutaraldehyde crosslinking method. Enzym. Microb. Technol. 2005, 37, 750–756. [Google Scholar] [CrossRef]
- Miller, G.L. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Romero, O.; Guisán, J.M.; Illanes, A.; Wilson, L. Reactivation of penicillin acylase biocatalysts: Effect of the intensity of enzyme–support attachment and enzyme load. J. Mol. Catal. B: Enzym. 2012, 74, 224–229. [Google Scholar] [CrossRef]





| Enzyme | Conjugate 1 | Immobilization Yield (%) 5 | Expressed Activity before Reduction Step (%) 6 | Expressed Activity after Reduction Step (%) 7 | Half-Life (h) 8 | Stability Factor 9 |
|---|---|---|---|---|---|---|
| PGA (dimer) | GA-B 2 | 97 | 43 | 43 | 70 | 1 |
| GA-G 3 | 48.6 | 6 | 0.09 | |||
| GA-E 4 | 48.6 | 1 | 0.01 | |||
| DAAO (dimer) | GA-B | 99 | 25 | 20.7 | 15 | 1 |
| GA-G | 25 | 3 | 0.2 | |||
| GA-E | 27.5 | 1.5 | 0.1 | |||
| GlyDH (octamer) | GA-B | 100 | 35 | 35 | 10 | 1 |
| GA-G | 25.5 | 11 | 1.1 | |||
| GA-E | 53.5 | 1 | 0.1 | |||
| ADH2 (dimer) | GA-B | 98 | 65 | 69.5 | 4.5 | 1 |
| GA-G | 87.7 | 20 | 4.4 | |||
| GA-E | 107 | 18 | 4 | |||
| Xys1Δ (monomer) | GA-B | 75 | 60 | 60 | 3.5 | 1 |
| GA-G | 53 | 5 | 1.4 | |||
| GA-E | 66 | 2.5 | 0.7 |
| Enzyme | GA-B 1 | GA-G 2 |
|---|---|---|
| PGA | 7 | 9 |
| DAAO | 4 | 5 |
| Xys1Δ | 5 | 4 |
| Conjugate | ManDH-GA-B 1 | ManDH-GA-E 2 | ManDH-GA-G1 3 | ManDH-GA-G2 4 | ManDH-GA-G3 5 |
|---|---|---|---|---|---|
| Immobilization yield (%) 6 | 99 | 99 | 99 | 99 | 99 |
| Expressed activity before reduction step (%) 7 | 15 | 14 | 17 | 50 | 39 |
| Recovered activity after reduction step (%) 8 | 1.5 | 14 | 16.5 | 16 | 19.5 |
| Half-life (h) 9 | N.D. | 19 | 221 | 43 | 82 |
| Stabilization factor 10 | N.D. | 31 | 357 | 69 | 132 |
| Enzyme | Buffer | Substrate 1 | Temperature (°C) 2 | Wavelength (nm) |
|---|---|---|---|---|
| PGA | 50 mM sodium phosphate pH 7 | 0.15 mM NIPAB | 25 | 405 |
| DAAO 3 | 50 mM sodium phosphate pH 7.5 | 10 mM D-phenylalanine | 25 | 450 |
| ADH2 | 50 mM sodium phosphate pH 7 | 10 mM 2,2,2-trifluoroacetophenone and 0.25 mM NADH | 65 | 340 |
| GlyDH | 50 mM sodium phosphate pH 7 | 100 mM glycerol and 2.5 mM NAD+ | 25 | 340 |
| ManDH | 0.1 M potassium phosphate pH 7 | 1.5 mM phenylglyoxylate and 0.25 mM NADH | 30 | 340 |
| Enzyme | Immobilization Time (h) | Immobilization Buffer | Offered Protein per g of Support (mg) | Temperature (°C) |
|---|---|---|---|---|
| PGA | 1 | 0.1 M sodium bicarbonate pH 10, 0.1 M phenylacetic and 25% glycerol | 5 | 25 |
| DAAO | 1 | 0.1 M sodium bicarbonate buffer pH 10 | 2.7 | 25 |
| ADH2 | 6 | 0.1 M sodium bicarbonate buffer pH 10 | 2 | 25 |
| GlyDH | 3 | 0.1 M sodium bicarbonate buffer pH 10 | 2.5 | 25 |
| Xys1Δ | 24 | 0.1 M sodium bicarbonate buffer pH 10 | 10 | 4 |
| ManDH | 1–3 | 0.1 M potassium bicarbonate buffer pH 10 and 40% trehalose | 0.5 | 4–25 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
H. Orrego, A.; Romero-Fernández, M.; Millán-Linares, M.D.C.; Yust, M.D.M.; Guisán, J.M.; Rocha-Martin, J. Stabilization of Enzymes by Multipoint Covalent Attachment on Aldehyde-Supports: 2-Picoline Borane as an Alternative Reducing Agent. Catalysts 2018, 8, 333. https://doi.org/10.3390/catal8080333
H. Orrego A, Romero-Fernández M, Millán-Linares MDC, Yust MDM, Guisán JM, Rocha-Martin J. Stabilization of Enzymes by Multipoint Covalent Attachment on Aldehyde-Supports: 2-Picoline Borane as an Alternative Reducing Agent. Catalysts. 2018; 8(8):333. https://doi.org/10.3390/catal8080333
Chicago/Turabian StyleH. Orrego, Alejandro, Maria Romero-Fernández, María Del Carmen Millán-Linares, María Del Mar Yust, José M. Guisán, and Javier Rocha-Martin. 2018. "Stabilization of Enzymes by Multipoint Covalent Attachment on Aldehyde-Supports: 2-Picoline Borane as an Alternative Reducing Agent" Catalysts 8, no. 8: 333. https://doi.org/10.3390/catal8080333