Impact of Endometallofullerene on P84 Copolyimide Transport and Thermomechanical Properties
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Membrane Preparation
2.3. Sorption Test
2.4. Pervaporation Test
2.5. Membrane Characterization
3. Results
3.1. Mechanical and Thermal Properties
3.2. Transport Properties
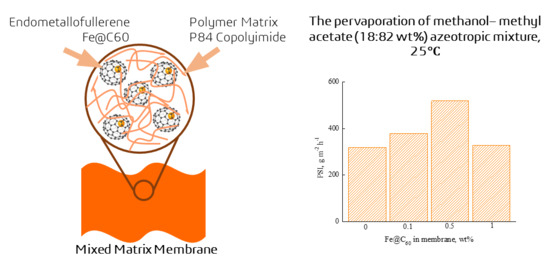
3.3. Pervaporation of Methanol—Methyl Acetate Mixture
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Baker, R.W. Research needs in the membrane separation industry: Looking back, looking forward. J. Membr. Sci. 2010, 362, 134–136. [Google Scholar] [CrossRef]
- Koros, W.J.; Zhang, C. Materials for next-generation molecularly selective synthetic membranes. Nat. Mater. 2017, 16, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; He, G.; Wang, S.; Yu, S.; Pan, F.; Wu, H.; Jiang, Z. Recent advances in the fabrication of advanced composite membranes. J. Mater. Chem. A 2013, 1, 10058–10077. [Google Scholar] [CrossRef]
- Meguid, S.A. Advances in Nanocomposites: Modeling, Characterization and Applications; Springer: Berlin, Germany, 2016; ISBN 9783319316604. [Google Scholar]
- Popov, A.A.; Yang, S.; Dunsch, L. Endohedral Fullerenes. Chem. Rev. 2013, 113, 5989–6113. [Google Scholar] [CrossRef] [PubMed]
- Weiske, T.; Böhme, D.K.; Hrušák, J.; Krätschmer, W.; Schwarz, H. Endohedral Cluster Compounds: Inclusion of Helium within C60•⊕ and C70•⊕ through Collision Experiments. Angew. Chem. Int. Ed. Engl. 1991, 30, 884–886. [Google Scholar] [CrossRef]
- Krätschmer, W.; Lamb, L.D.; Fostiropoulos, K.; Huffman, D.R. Solid C60: A new form of carbon. Nature 1990, 347, 354–358. [Google Scholar] [CrossRef]
- Shinohara, H. Endohedral metallofullerenes. Rep. Prog. Phys. 2000, 63, 843–892. [Google Scholar] [CrossRef]
- Eletskii, A.V. Endohedral structures. Phys.-Uspekhi 2000, 43, 111–137. [Google Scholar] [CrossRef]
- Rudolf, M.; Wolfrum, S.; Guldi, D.M.; Feng, L.; Tsuchiya, T.; Akasaka, T.; Echegoyen, L. Endohedral Metallofullerenes-Filled Fullerene Derivatives towards Multifunctional Reaction Center Mimics. Chem. A Eur. J. 2012, 18, 5136–5148. [Google Scholar] [CrossRef] [PubMed]
- Dorn, H.C.; Fatouros, P.P. Endohedral Metallofullerenes: Applications of a New Class of Carbonaceous Nanomaterials. Nanosci. Nanotechnol. Lett. 2010, 2, 65–72. [Google Scholar] [CrossRef]
- Ananta, J.S.; Wilson, L.J. Gadonanostructures as Magnetic Resonance Imaging Contrast Agents. In Chemistry of Nanocarbons; John Wiley & Sons, Ltd.: Chichester, UK, 2010; pp. 287–300. [Google Scholar]
- Bolskar, R.D. Gadolinium Endohedral Metallofullerene-Based MRI Contrast Agents; Springer: Dordrecht, The Netherlands, 2008; pp. 157–180. [Google Scholar]
- Lebedev, V.T.; Szhogina, A.A.; Yu Bairamukov, V. Small angle neutron and X-ray studies of carbon structures with metal atoms. J. Phys. Conf. Ser. 2017, 848, 012005. [Google Scholar] [CrossRef] [Green Version]
- Polotskaya, G.A.; Lebedev, V.T.; Gofman, I.V.; Vinogradova, L.V. Composite films based on polyphenylene oxide modified with endofullerenes C60 with encapsulated iron atoms. Russ. J. Appl. Chem. 2017, 90, 1549–1557. [Google Scholar] [CrossRef]
- Beerlage, M.A.M.; Heijnen, M.L.; Mulder, M.H.V.; Smolders, C.A.; Strathmann, H.; Smolders, C.A.; Strathmann, H. Non-aqueous retention measurements: Ultrafiltration behaviour of polystyrene solutions and colloidal silver particles. J. Membr. Sci. 1996, 113, 259–273. [Google Scholar] [CrossRef]
- White, L.S. Transport properties of a polyimide solvent resistant nanofiltration membrane. J. Membr. Sci. 2002, 205, 191–202. [Google Scholar] [CrossRef]
- Barsema, J.N.; Kapantaidakis, G.C.; van der Vegt, N.F.A.; Koops, G.H.; Wessling, M. Preparation and characterization of highly selective dense and hollow fiber asymmetric membranes based on BTDA-TDI/MDI co-polyimide. J. Membr. Sci. 2003, 216, 195–205. [Google Scholar] [CrossRef]
- Qiao, X.; Chung, T.-S.; Pramoda, K.P. Fabrication and characterization of BTDA-TDI/MDI (P84) co-polyimide membranes for the pervaporation dehydration of isopropanol. J. Membr. Sci. 2005, 264, 176–189. [Google Scholar] [CrossRef]
- Liu, R.; Qiao, X.; Chung, T.-S. The development of high performance P84 co-polyimide hollow fibers for pervaporation dehydration of isopropanol. Chem. Eng. Sci. 2005, 60, 6674–6686. [Google Scholar] [CrossRef]
- Qiao, X.; Chung, T.S. Fundamental characteristics of sorption, swelling, and permeation of P84 Co-polyimide membranes for pervaporation dehydration of alcohols. Ind. Eng. Chem. Res. 2005, 44, 8938–8943. [Google Scholar] [CrossRef]
- Qiao, X.; Chung, T.-S.; Rajagopalan, R. Zeolite filled P84 co-polyimide membranes for dehydration of isopropanol through pervaporation process. Chem. Eng. Sci. 2006, 61, 6816–6825. [Google Scholar] [CrossRef]
- Hua, D.; Ong, Y.K.; Wang, Y.; Yang, T.; Chung, T.-S. ZIF-90/P84 mixed matrix membranes for pervaporation dehydration of isopropanol. J. Membr. Sci. 2014, 453, 155–167. [Google Scholar] [CrossRef]
- Torabi, B.; Ameri, E. Methyl acetate production by coupled esterification-reaction process using synthesized cross-linked PVA/silica nanocomposite membranes. Chem. Eng. J. 2016, 288, 461–472. [Google Scholar] [CrossRef]
- Assabumrungrat, S.; Phongpatthanapanich, J.; Praserthdam, P.; Tagawa, T.; Goto, S. Theoretical study on the synthesis of methyl acetate from methanol and acetic acid in pervaporation membrane reactors: Effect of continuous-flow modes. Chem. Eng. J. 2003, 95, 57–65. [Google Scholar] [CrossRef]
- Abdallah, H.; El-Gendi, A.; El-Zanati, E.; Matsuura, T. Pervaporation of methanol from methylacetate mixture using polyamide-6 membrane. Desalin. Water Treat. 2013, 51, 7807–7814. [Google Scholar] [CrossRef] [Green Version]
- Penkova, A.V.; Polotskaya, G.A.; Toikka, A.M. Separation of acetic acid-methanol-methyl acetate-water reactive mixture. Chem. Eng. Sci. 2013, 101, 586–592. [Google Scholar] [CrossRef]
- Genduso, G.; Farrokhzad, H.; Latré, Y.; Darvishmanesh, S.; Luis, P.; Van der Bruggen, B. Polyvinylidene fluoride dense membrane for the pervaporation of methyl acetate–methanol mixtures. J. Membr. Sci. 2015, 482, 128–136. [Google Scholar] [CrossRef]
- Steinigeweg, S.; Gmehling, J. Transesterification processes by combination of reactive distillation and pervaporation. Chem. Eng. Process. Process Intensif. 2004, 43, 447–456. [Google Scholar] [CrossRef]
- Ohe, S. Vapor-Liquid Equilibrium Data, 2nd ed.; Kodansha: New York, NY, USA, 1989; ISBN 0444988769. [Google Scholar]
- Pulyalina, A.; Polotskaya, G.; Goikhman, M.; Podeshvo, I.; Gulii, N.; Shugurov, S.; Tataurov, M.; Toikka, A. Preparation and characterization of methanol selective membranes based on polyheteroarylene−Cu(I) complexes for purification of methyl tertiary butyl ether. Polym. Int. 2017. [Google Scholar] [CrossRef]
- Pulyalina, A.Y.; Toikka, A.M.; Polotskaya, G.A. Investigation of pervaporation membranes based on polycarbamide: Effect of residual solvent. Pet. Chem. 2014, 54, 573–579. [Google Scholar] [CrossRef]
- Pulyalina, A.Y.; Polotskaya, G.A.; Kalyuzhnaya, L.M.; Saprykina, N.N.; Sushchenko, I.G.; Meleshko, T.K.; Toikka, A.M. The study of sorption and transport properties of membranes containing polyaniline. Polym. Sci. Ser. A 2010, 52, 856–863. [Google Scholar] [CrossRef]
- Pulyalina, A.Y.; Polotskaya, G.A.; Toikka, A.M. Membrane materials based on polyheteroarylenes and their application for pervaporation. Russ. Chem. Rev. 2016, 85. [Google Scholar] [CrossRef]
- Baker, R.W.; Wijmans, J.G.; Huang, Y. Permeability, permeance and selectivity: A preferred way of reporting pervaporation performance data. J. Membr. Sci. 2010, 348, 346–352. [Google Scholar] [CrossRef]
- Polotskaya, G.; Pulyalinа, A.; Goikhman, M.; Podeshvo, I.; Valieva, I.; Toikka, A.; Polotskaya, G.A.; Pulyalinа, A.Y.; Goikhman, M.Y.; Podeshvo, I.V.; et al. Aromatic Copolyamides with Anthrazoline Units in the Backbone: Synthesis, Characterization, Pervaporation Application. Polymers (Basel) 2016, 8, 362. [Google Scholar] [CrossRef]
- Polotskaya, G.A.; Avagimova, N.V.; Toikka, A.M.; Tsvetkov, N.V.; Lezov, A.A.; Strelina, I.A.; Gofman, I.V.; Pientka, Z. Optical, mechanical, and transport studies of nanodiamonds/poly(phenylene oxide) composites. Polym. Compos. 2017. [Google Scholar] [CrossRef]
- Ong, Y.K.; Shi, G.M.; Le, N.L.; Tang, Y.P.; Zuo, J.; Nunes, S.P.; Chung, T.S. Recent membrane development for pervaporation processes. Prog. Polym. Sci. 2016. [Google Scholar] [CrossRef]
- Dong, G.; Li, H.; Chen, V. Challenges and opportunities for mixed-matrix membranes for gas separation. J. Mater. Chem. A 2013, 1, 4610. [Google Scholar] [CrossRef]
- Yang, H.-C.; Hou, J.; Chen, V.; Xu, Z.-K. Surface and interface engineering for organic–inorganic composite membranes. J. Mater. Chem. A 2016, 4, 9716–9729. [Google Scholar] [CrossRef]
- Barton, A.F.M. CRC Handbook of Solubility Parameters and Other Cohesion Parameters; CRC Press: Boca Raton, FL, USA, 1991; ISBN 9780849301766. [Google Scholar]
- Park, C.H.; Tocci, E.; Fontananova, E.; Bahattab, M.A.; Aljlil, S.A.; Drioli, E. Mixed matrix membranes containing functionalized multiwalled carbon nanotubes: Mesoscale simulation and experimental approach for optimizing dispersion. J. Membr. Sci. 2016, 514, 195–209. [Google Scholar] [CrossRef]
- Brandrup, J.; Immergut, E.H.; Grulke, E.A. Polymer Handbook; Wiley-Interscience: Hoboken, NJ, USA, 1999; ISBN 9780471479369. [Google Scholar]
- Wijmans, J.G. Process performance = membrane properties + operating conditions. J. Membr. Sci. 2003, 220, 1–3. [Google Scholar] [CrossRef]
- Guo, W.F.; Chung, T.-S.; Matsuura, T. Pervaporation study on the dehydration of aqueous butanol solutions: A comparison of flux vs. permeance, separation factor vs. selectivity. J. Membr. Sci. 2004, 245, 199–210. [Google Scholar] [CrossRef]
- Gorri, D.; Ibáñez, R.; Ortiz, I. Comparative study of the separation of methanol–methyl acetate mixtures by pervaporation and vapor permeation using a commercial membrane. J. Membr. Sci. 2006, 280, 582–593. [Google Scholar] [CrossRef]
- Avagimova, N.; Polotskaya, G.; Toikka, A.; Pulyalina, A.; Morávková, Z.; Trchová, M.; Pientka, Z. Effect of nanodiamonds additives on structure and gas transport properties of poly(phenylene-iso-phtalamide) matrix. J. Appl. Polym. Sci. 2018. [Google Scholar] [CrossRef]
- Sain, S.; Dinçer, S.; Savaşçyı, Ö. Pervaporation of methanol–methyl acetate binary mixtures. Chem. Eng. Process. Process Intensif. 1998, 37, 203–206. [Google Scholar] [CrossRef]







| Membrane | E, GPa | σy, MPa | σb, MPa | εb, % | Tg, °C |
|---|---|---|---|---|---|
| P84 | 2.41 ± 0.09 | 113 ± 2 | 113 ± 2 | 7.4 ± 0.4 | 344 |
| P84/Fe@C60(0.1%) | 2.40 ± 0.07 | 112 ± 4 | 109 ± 4 | 7.8 ± 0.5 | 345 |
| P84/Fe@C60(0.5%) | 2.39 ± 0.13 | 113 ± 7 | 115 ± 4 | 8.9 ± 0.6 | 346 |
| P84 P84/Fe@C60(1%) | 2.39 ± 0.11 | 112 ± 4 | 97 ± 2 | 10 ± 1 | 346 |
| Membrane | τ1, °C | τ5, °C | τ10, °C |
|---|---|---|---|
| P84 | 400 | 466 | 507 |
| P84/Fe@C60(0.1%) | 406 | 472 | 508 |
| P84/Fe@C60(0.5%) | 410 | 476 | 509 |
| P84/Fe@C60(1%) | 400 | 463 | 503 |
| Liquid | MW | Density, g/cm3 | Molar Volume, m3/mol | Boiling Point, °C | Viscosity, mPa∙s | Solubility Parameter, (J/cm3)1/2 |
|---|---|---|---|---|---|---|
| Methanol | 32.0 | 0.792 | 40.4 | 64.7 | 0.54 | 29.7 |
| Methyl acetate | 74.1 | 0.933 | 79.4 | 57.1 | 0.36 | 19.6 |
| Membrane | Sorption Degree, % | |
|---|---|---|
| Methanol | Methyl Acetate | |
| P84 | 7.8 | 17.0 |
| P84/Fe@C60(0.1%) | 11.6 | 16.8 |
| P84/Fe@C60(0.5%) | 11.8 | 16.7 |
| P84/Fe@C60(1%) | 12.8 | 16.0 |
| Membrane | Methanol in Feed, wt % | T, °C | Flux, kg/(m2 h) | Methanol in Permeate, wt % | Separation Factor | Reference |
|---|---|---|---|---|---|---|
| Cuprophane | 19.9 | 45 | 0.453 | 66.3 | 7.9 | [48] |
| Cuprophane | 20.8 | 30 | 0.222 | 68.0 | 8.1 | [48] |
| Pervap 2255_40 | 21 | 45 | 4.1 | 45.5 | 3.1 | [29] |
| Pervap 2255_50 | 16 | 45 | 1.1 | 55.0 | 6.4 | [29] |
| Pervap 2255_30 | 20 | 40 | 2.44 | 54.4 | 4.8 | [46] |
| PA/ND(3%) | 18 | 25 | 0.22 | 74.0 | 13.0 | [47] |
| P84/Fe@C60(0.5%) | 18 | 25 | 0.072 | 64.3 | 8.2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Polotskaya, G.; Putintseva, M.; Pulyalina, A.; Gofman, I.; Toikka, A. Impact of Endometallofullerene on P84 Copolyimide Transport and Thermomechanical Properties. Polymers 2018, 10, 1108. https://doi.org/10.3390/polym10101108
Polotskaya G, Putintseva M, Pulyalina A, Gofman I, Toikka A. Impact of Endometallofullerene on P84 Copolyimide Transport and Thermomechanical Properties. Polymers. 2018; 10(10):1108. https://doi.org/10.3390/polym10101108
Chicago/Turabian StylePolotskaya, Galina, Maia Putintseva, Alexandra Pulyalina, Iosif Gofman, and Alexander Toikka. 2018. "Impact of Endometallofullerene on P84 Copolyimide Transport and Thermomechanical Properties" Polymers 10, no. 10: 1108. https://doi.org/10.3390/polym10101108