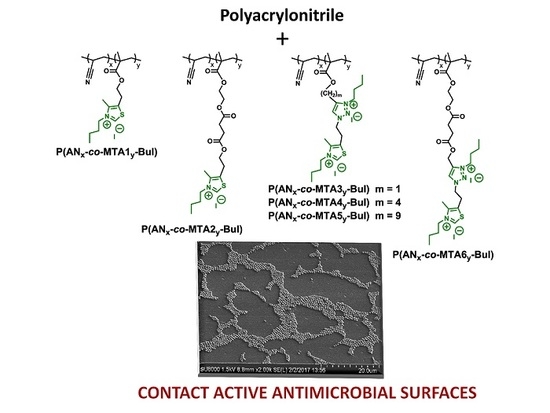
Tailoring Macromolecular Structure of Cationic Polymers towards Efficient Contact Active Antimicrobial Surfaces
Abstract
:1. Introduction
2. Experimental Part
2.1. Materials
2.2. Antimicrobial Films Preparation and Characterization
2.3. Evaluation of Antimicrobial Activity of the Films
2.4. Characterization of Microorganisms after Exposure to the Antimicrobial Surface by SEM
3. Results and Discussion
3.1. Antimicrobial Copolymer Design and Film Preparation
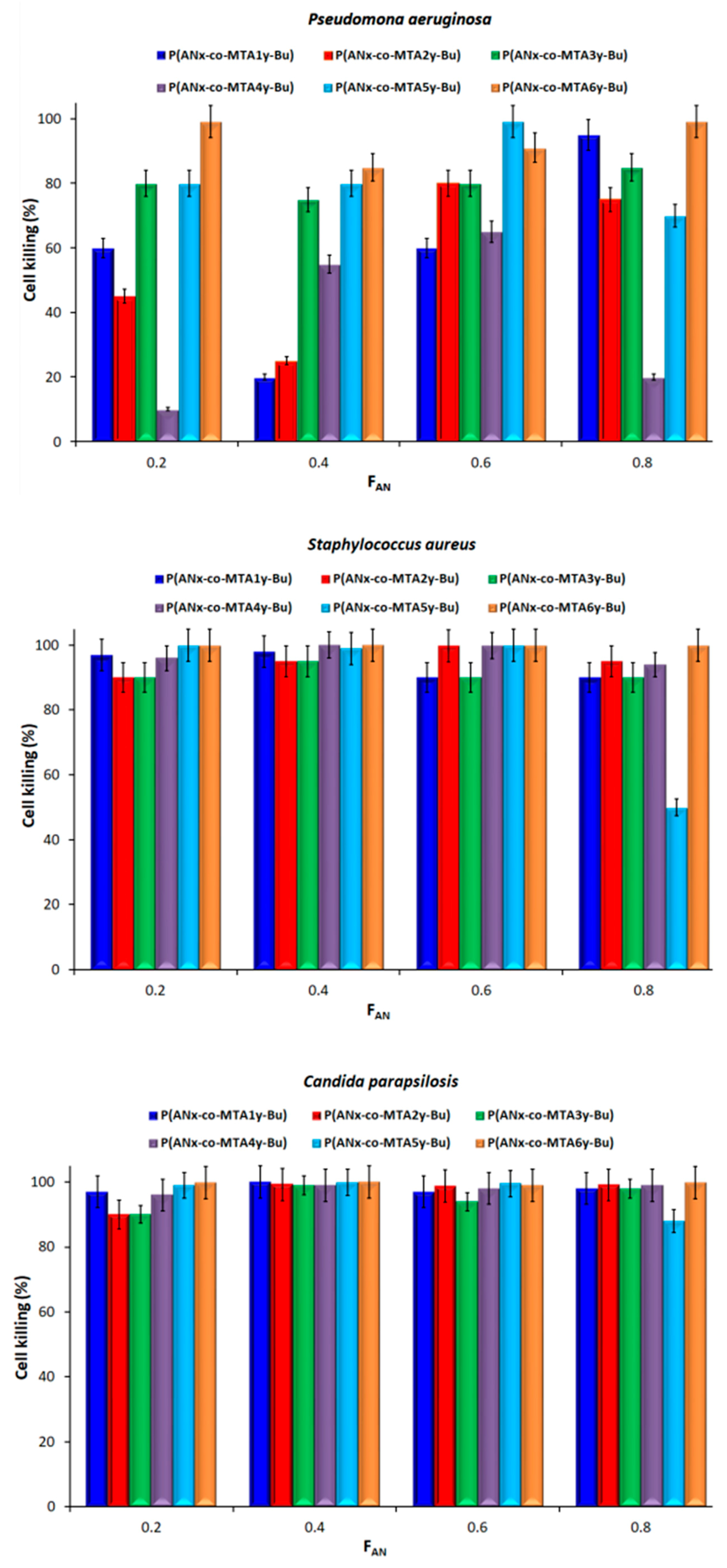
3.2. Evaluation of the Antimicrobial Activity of the Blend Films
3.3. Evaluation of the Morphological Changes of Microbes
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Magill, S.S.; Edwards, J.R.; Bamberg, W.; Beldavs, Z.G.; Dumyati, G.; Kainer, M.A.; Lynfield, R.; Maloney, M.; McAllister-Hollod, L.; Nadle, J.; et al. Multistate point-prevalence survey of health care–associated infections. N. Engl. J. Med. 2014, 370, 1198–1208. [Google Scholar] [CrossRef] [PubMed]
- ECDC. 2013. Available online: https://ecdc.europa.eu/en/healthcare-associated-infections-acute-care-hospitals (accessed on 4 July 2013).
- Page, K.; Wilson, M.; Parkin, I.P. Antimicrobial surfaces and their potential in reducing the role of the inanimate environment in the incidence of hospital-acquired infections. J. Mater. Chem. 2009, 19, 3819. [Google Scholar] [CrossRef]
- Hickok, N.J.; Shapiro, I.M. Immobilized antibiotics to prevent orthopaedic implant infections. Adv. Drug Del. Rev. 2012, 64, 1165–1176. [Google Scholar] [CrossRef] [PubMed]
- Milovic, N.M.; Wang, J.; Lewis, K.; Klibanov, A.M. Immobilized N-alkylated polyethylenimine avidly kills bacteria by rupturing cell membranes with no resistance developed. Biotechnol. Bioeng. 2005, 90, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Siedenbiedel, F.; Tiller, J.C. Antimicrobial polymers in solution and on surfaces: Overview and functional principles. Polymers 2012, 4, 46–71. [Google Scholar] [CrossRef]
- Álvarez-Paino, M.; Muñoz-Bonilla, A.; López-Fabal, F.; Gómez-Garcés, J.L.; Heuts, J.P.A.; Fernández-García, M. Functional surfaces obtained from emulsion polymerization using antimicrobial glycosylated block copolymers as surfactants. Polym. Chem. 2015, 6, 6171–6181. [Google Scholar] [CrossRef]
- Alvarez-Paino, M.; Bonilla, P.; Cuervo-Rodríguez, R.; López-Fabal, F.; Gómez-Garcés, J.L.; Muñoz-Bonilla, A.; Fernández-García, M. Antimicrobial surfaces obtained from blends of block copolymers synthesized by simultaneous atrp and click chemistry reactions. Eur. Polym. J. 2017, 93, 53–62. [Google Scholar] [CrossRef]
- Gao, J.; Huddleston, N.E.; White, E.M.; Pant, J.; Handa, H.; Locklin, J. Surface grafted antimicrobial polymer networks with high abrasion resistance. ACS Biomater. Sci. Eng. 2016, 2, 1169–1179. [Google Scholar] [CrossRef]
- Correia, V.G.; Ferraria, A.M.; Pinho, M.G.; Aguiar-Ricardo, A. Antimicrobial contact-active oligo(2-oxazoline)s-grafted surfaces for fast water disinfection at the point-of-use. Biomacromolecules 2015, 16, 3904–3915. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Bonilla, A.; Fernández-García, M. Polymeric materials with antimicrobial activity. Prog. Polym. Sci. 2012, 37, 281–339. [Google Scholar] [CrossRef]
- Kenawy, E.-R.; Worley, S.D.; Broughtonand, R. The chemistry and applications of antimicrobial polymers: A state-of-the-art review. Biomacromolecules 2007, 8, 1359–1384. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Xiao, H.; Zhang, Y. Antimicrobial polymeric materials with quaternary ammonium and phosphonium salts. Int J. Mol. Sci. 2015, 16, 3626–3655. [Google Scholar] [CrossRef] [PubMed]
- Jiao, Y.; Niu, L.-N.; Ma, S.; Li, J.; Tay, F.R.; Chen, J.-H. Quaternary ammonium-based biomedical materials: State-of-the-art, toxicological aspects and antimicrobial resistance. Prog. Polym. Sci. 2017, 71, 53–90. [Google Scholar] [CrossRef]
- Ganewatta, M.S.; Tang, C. Controlling macromolecular structures towards effective antimicrobial polymers. Polymer 2015, 63, A1–A29. [Google Scholar] [CrossRef]
- Palermo, E.F.; Sovadinova, I.; Kuroda, K. Structural determinants of antimicrobial activity and biocompatibility in membrane-disrupting methacrylamide random copolymers. Biomacromolecules 2009, 10, 3098–3107. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.-G.; Wan, L.-S.; Xu, Z.-K. Surface engineerings of polyacrylonitrile-based asymmetric membranes towards biomedical applications: An overview. J. Membr. Sci. 2007, 304, 8–23. [Google Scholar] [CrossRef]
- Tejero, R.; Gutierrez, B.; Lopez, D.; Lopez-Fabal, F.; Gomez-Garces, J.L.; Fernandez-Garcia, M. Copolymers of acrylonitrile with quaternizable thiazole and triazole side-chain methacrylates as potent antimicrobial and hemocompatible systems. Acta Biomater. 2015, 25, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Tejero, R.; López, D.; López-Fabal, F.; Gómez-Garcés, J.L.; Fernández-García, M. Antimicrobial polymethacrylates based on quaternized 1,3-thiazole and 1,2,3-triazole side-chain groups. Polym. Chem. 2015, 6, 3449–3459. [Google Scholar] [CrossRef]
- ASTM International. ASTM E2149-01, Standard Test Method for Determining the Antimicrobial Activity of Immobilized Antimicrobial Agents under Dynamic Contact Conditions (Withdrawn 2010); ASTM International: West Conshohocken, PA, USA, 2001. [Google Scholar]
- Tejero, R.; López, D.; Fernández-García, M. Influence of spacer group on the structure and thermal properties of copolymers based on acrylonitrile and methacrylic 1,3-thiazole and 1,2,3-triazole derivatives. Eur. Polym. J. 2015, 71, 401–411. [Google Scholar] [CrossRef]
- Bayramoğlu, G.; Metin, A.Ü.; Arıca, M.Y. Surface modification of polyacrylonitrile film by anchoring conductive polyaniline and determination of uricase adsorption capacity and activity. Appl. Surf. Sci. 2010, 256, 6710–6716. [Google Scholar] [CrossRef]
- Zou, P.; Laird, D.; Riga, E.K.; Deng, Z.; Dorner, F.; Perez-Hernandez, H.-R.; Guevara-Solarte, D.L.; Steinberg, T.; Al-Ahmad, A.; Lienkamp, K. Antimicrobial and cell-compatible surface-attached polymer networks—How the correlation of chemical structure to physical and biological data leads to a modified mechanism of action. J. Mater. Chem. B 2015, 3, 6224–6238. [Google Scholar] [CrossRef]
- Engler, A.C.; Tan, J.P.; Ong, Z.Y.; Coady, D.J.; Ng, V.W.; Yang, Y.Y.; Hedrick, J.L. Antimicrobial polycarbonates: Investigating the impact of balancing charge and hydrophobicity using a same-centered polymer approach. Biomacromolecules 2013, 14, 4331–4339. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Algburi, A.; Wang, N.; Kholodovych, V.; Oh, D.O.; Chikindas, M.; Uhrich, K.E. Self-assembled cationic amphiphiles as antimicrobial peptides mimics: Role of hydrophobicity, linkage type, and assembly state. Nanomedicine 2017, 13, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Bieser, A.M.; Tiller, J.C. Mechanistic considerations on contact-active antimicrobial surfaces with controlled functional group densities. Macromol. Biosci. 2011, 11, 526–534. [Google Scholar] [CrossRef] [PubMed]
- Hoque, J.; Akkapeddi, P.; Yadav, V.; Manjunath, G.B.; Uppu, D.S.; Konai, M.M.; Yarlagadda, V.; Sanyal, K.; Haldar, J. Broad spectrum antibacterial and antifungal polymeric paint materials: Synthesis, structure-activity relationship, and membrane-active mode of action. ACS Appl. Mater. Interfaces 2015, 7, 1804–1815. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Zhang, Y.; Li, J.; Gao, Y.; Luo, F.; Tan, H.; Wang, K.; Fu, Q. A novel surface structure consisting of contact-active antibacterial upper-layer and antifouling sub-layer derived from gemini quaternary ammonium salt polyurethanes. Sci. Rep. 2016, 6, 32140. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Paino, M.; Juan-Rodriguez, R.; Cuervo-Rodriguez, R.; Tejero, R.; Lopez, D.; Lopez-Fabal, F.; Gomez-Garces, J.L.; Munoz-Bonilla, A.; Fernandez-Garcia, M. Antimicrobial films obtained from latex particles functionalized with quaternized block copolymers. Colloids Surf. B Biointerfaces 2016, 140, 94–103. [Google Scholar] [CrossRef] [PubMed]




| Copolymer in the Blend | fAN | Tg (°C) a | θ (°) |
|---|---|---|---|
| P(ANx-co-MTA1y-Bu) | 0.2 | 48 | 69 ± 3 |
| 0.4 | 52 | 67 ± 2 | |
| 0.6 | 60 | 67 ± 2 | |
| 0.8 | 69 | 65 ± 1 | |
| P(ANx-co-MTA2y-Bu) | 0.2 | 10 | 66 ± 3 |
| 0.4 | 12 | 64 ± 3 | |
| 0.6 | 22 | 65 ± 2 | |
| 0.8 | 32 | 61 ± 2 | |
| P(ANx-co-MTA3y-Bu) | 0.2 | 61 | 64 ± 2 |
| 0.4 | 63 | 63 ± 2 | |
| 0.6 | 71 | 70 ± 4 | |
| 0.8 | 74 | 62 ± 3 | |
| P(ANx-co-MTA4y-Bu) | 0.2 | 25 | 79 ± 3 |
| 0.4 | 29 | 75 ± 3 | |
| 0.6 | 44 | 72 ± 3 | |
| 0.8 | 55 | 70 ± 3 | |
| P(ANx-co-MTA5y-Bu) | 0.2 | -7 | 73 ± 2 |
| 0.4 | 0 | 70 ± 3 | |
| 0.6 | 8 | 78 ± 3 | |
| 0.8 | 25 | 75 ± 3 | |
| P(ANx-co-MTA6y-Bu) | 0.2 | 20 | 60 ± 2 |
| 0.4 | 23 | 69 ± 3 | |
| 0.6 | 32 | 65 ± 2 | |
| 0.8 | 45 | 65 ± 2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tejero, R.; Gutiérrez, B.; López, D.; López-Fabal, F.; Gómez-Garcés, J.L.; Muñoz-Bonilla, A.; Fernández-García, M. Tailoring Macromolecular Structure of Cationic Polymers towards Efficient Contact Active Antimicrobial Surfaces. Polymers 2018, 10, 241. https://doi.org/10.3390/polym10030241
Tejero R, Gutiérrez B, López D, López-Fabal F, Gómez-Garcés JL, Muñoz-Bonilla A, Fernández-García M. Tailoring Macromolecular Structure of Cationic Polymers towards Efficient Contact Active Antimicrobial Surfaces. Polymers. 2018; 10(3):241. https://doi.org/10.3390/polym10030241
Chicago/Turabian StyleTejero, Rubén, Beatriz Gutiérrez, Daniel López, Fátima López-Fabal, José L. Gómez-Garcés, Alexandra Muñoz-Bonilla, and Marta Fernández-García. 2018. "Tailoring Macromolecular Structure of Cationic Polymers towards Efficient Contact Active Antimicrobial Surfaces" Polymers 10, no. 3: 241. https://doi.org/10.3390/polym10030241
APA StyleTejero, R., Gutiérrez, B., López, D., López-Fabal, F., Gómez-Garcés, J. L., Muñoz-Bonilla, A., & Fernández-García, M. (2018). Tailoring Macromolecular Structure of Cationic Polymers towards Efficient Contact Active Antimicrobial Surfaces. Polymers, 10(3), 241. https://doi.org/10.3390/polym10030241