Diffusive Motion of Linear Microgel Assemblies in Solution
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Microgel Preparation
2.3. Substrate Preparation
2.4. Fluorescence-Labelling of Microgel Assemblies
2.5. Formation of Microgel Assemblies and Microscopy Sample Preparation
2.6. Confocal Microscopy
2.7. Widefield Fluorescence Microscopy
2.8. Analysis of Microgel Assembly Conformation
2.9. Dynamic Light Scattering
2.10. Atomic Force Microscopy (AFM)
2.11. Transmission Electron Microscopy (TEM)
3. Results and Discussion
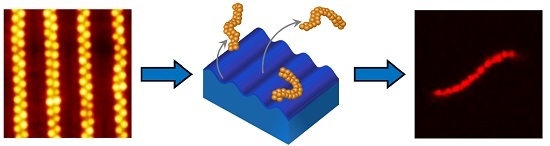
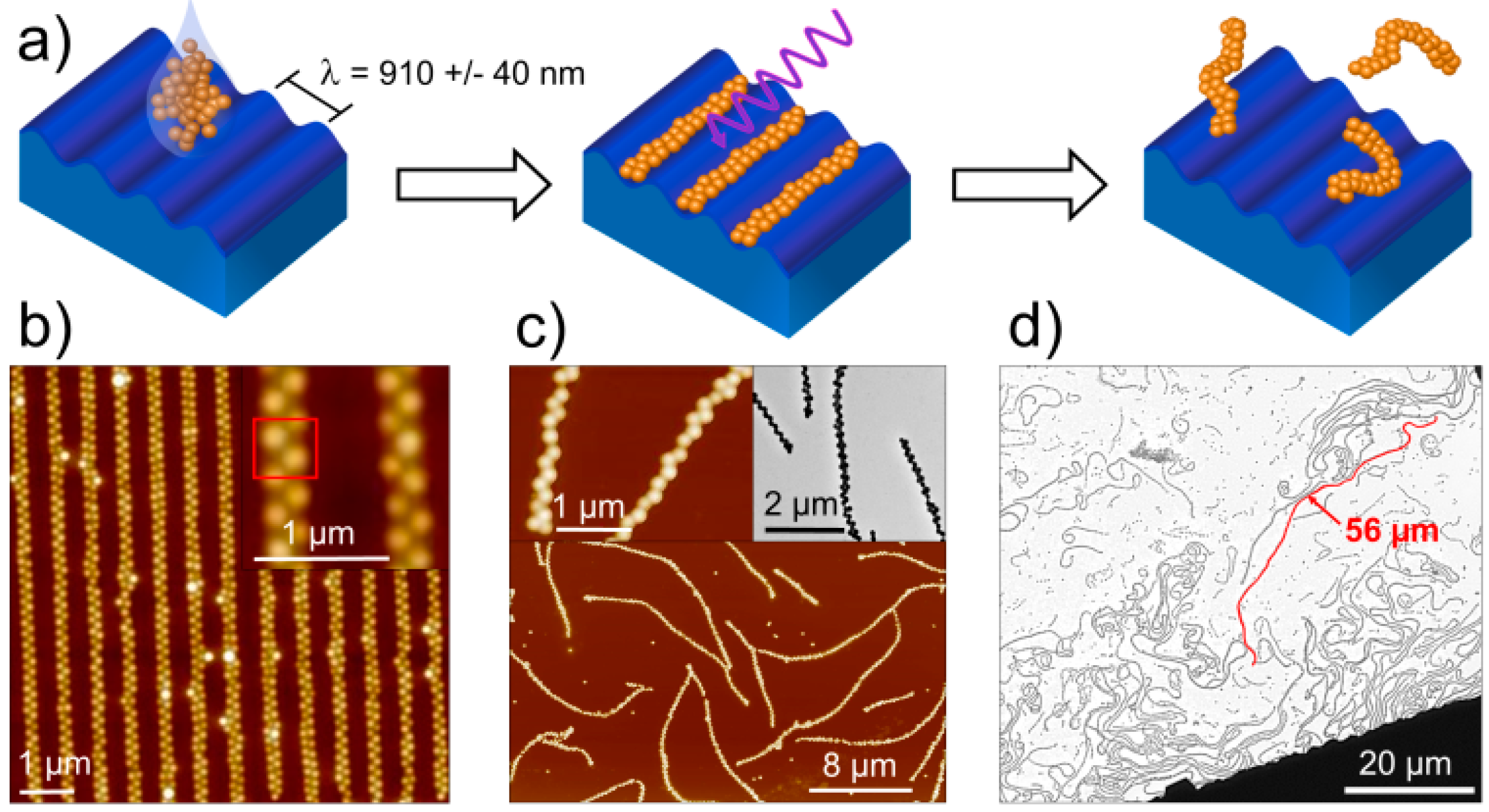
3.1. Synthesis of Microgel Assemblies
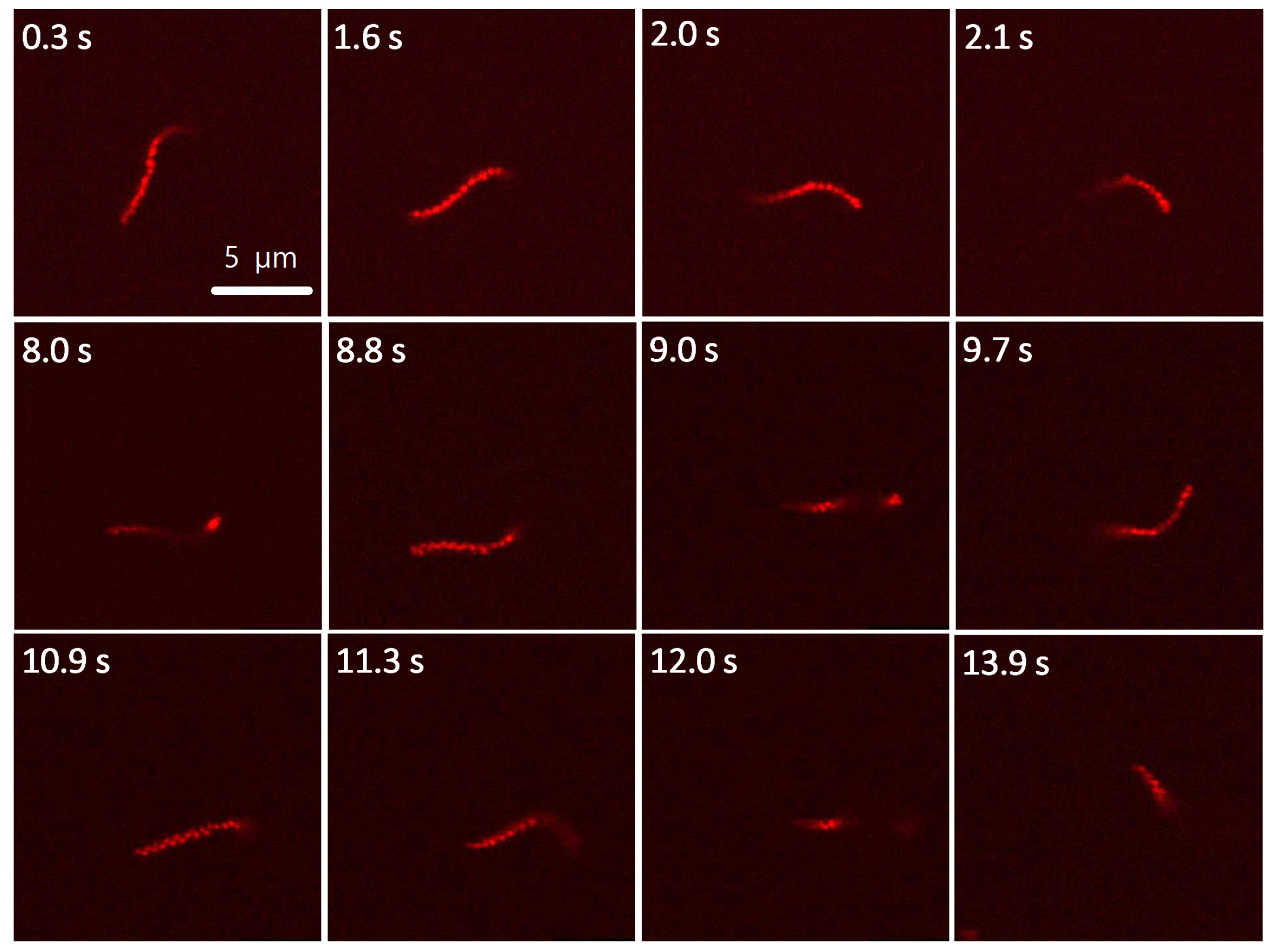
3.2. Visualization of Microgel Assemblies with Fluorescence Microscopy
3.3. Analysis of the 2D Projections of Microgel Chains
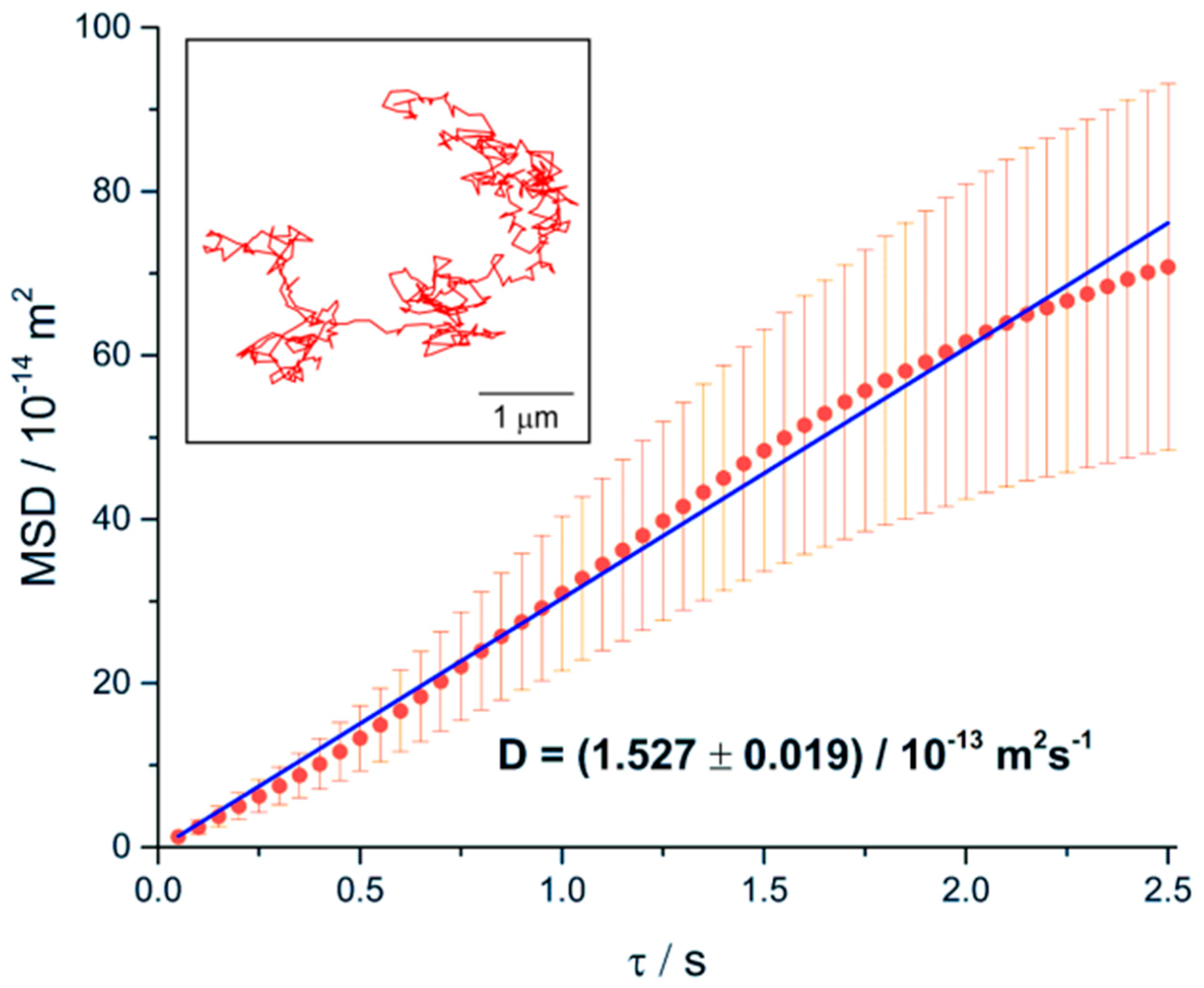
3.4. Translational Diffusion of Microgel Chains
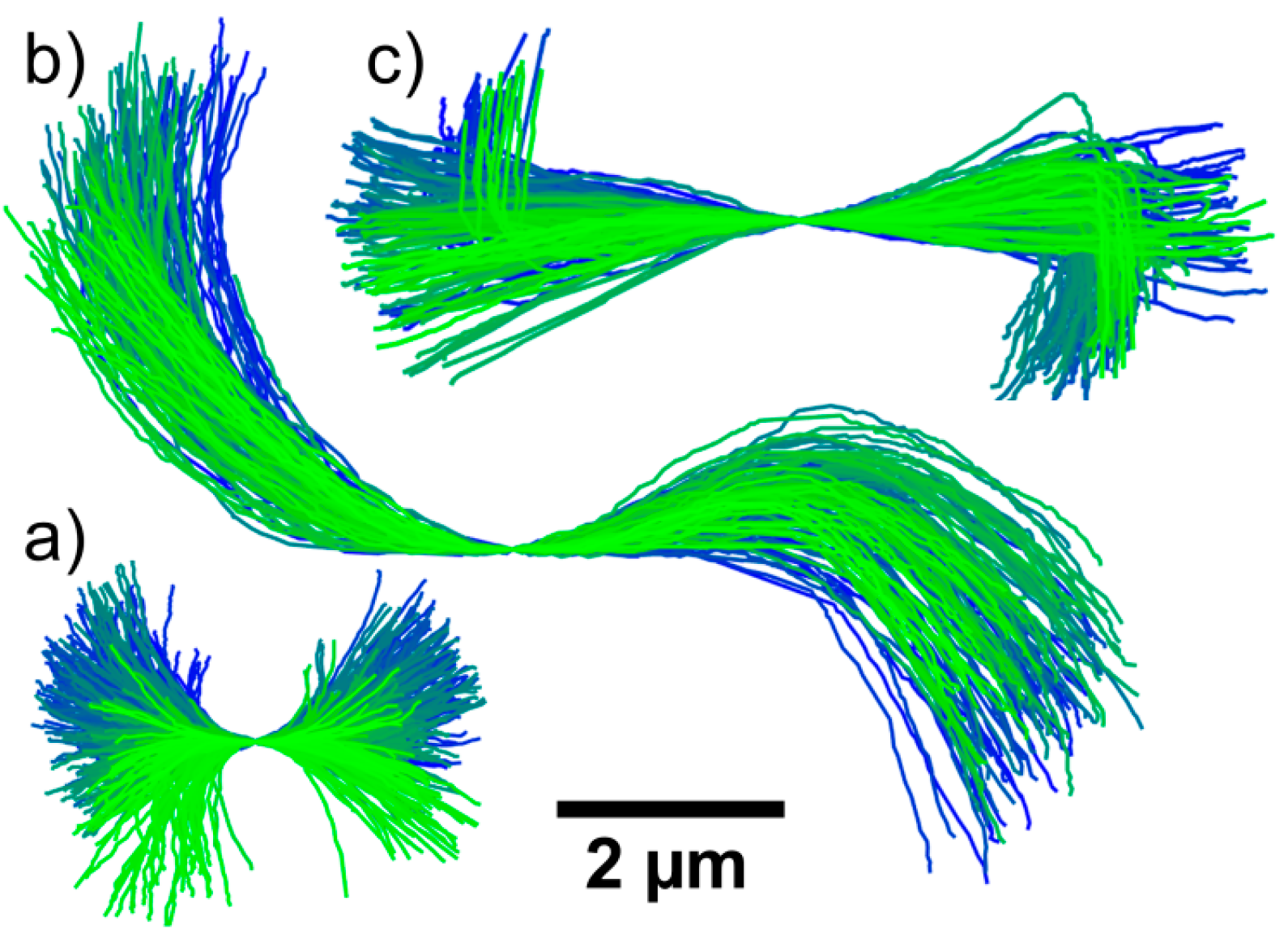
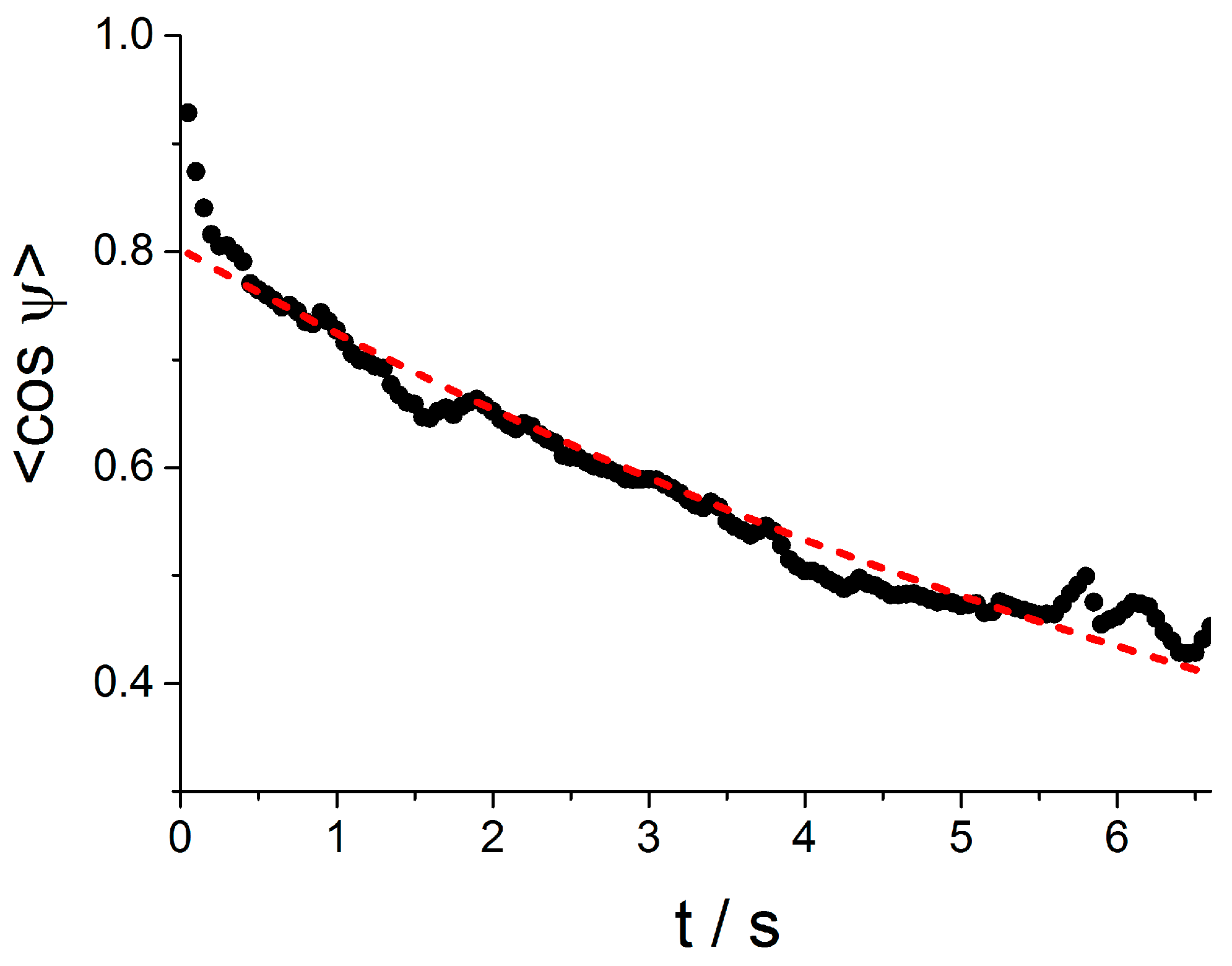
3.5. Rotational Diffusion of Microgel Chains
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cohen Stuart, M.A.; Huck, W.T.; Genzer, J.; Müller, M.; Ober, C.; Stamm, M.; Sukhorukov, G.B.; Szleifer, I.; Tsukruk, V.V.; Urban, M.; et al. Emerging applications of stimuli-responsive polymer materials. Nat. Mater. 2010, 9, 101–113. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-B.; Choi, J.-K.; Yu, J.-A.; Hong, J.-W. Synthesis and characterization of a non-aqueous conductive microgel coated with poly(aniline)—DBSA in a colloidal dispersion. Synth. Met. 2002, 131, 79–85. [Google Scholar] [CrossRef]
- Kim, Y.-B.; Park, C.-H.; Hong, J.-W. Electrically Conductive Microgel and Method for Preparing the Same. U.S. Patent 6,399,675 B1, 4 June 2002. [Google Scholar]
- Clara-Rahola, J.; Contreras-Caceres, R.; Sierra-Martin, B.; Maldonado-Valdivia, A.; Hund, M.; Fery, A.; Hellweg, T.; Fernandez-Barbero, A. Structure and plasmon coupling of gold-poly(N-isopropylacrylamide) core–shell microgel arrays with thermally controlled interparticle gap. Colloids Surf. A 2014, 463, 18–27. [Google Scholar] [CrossRef]
- Müller, M.; Karg, M.; Fortini, A.; Hellweg, T.; Fery, A. Wrinkle-assisted linear assembly of hard-core/soft-shell particles: Impact of the soft shell on the local structure. Nanoscale 2012, 4, 2491–2499. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Zhang, J.; Paquet, C.; Lin, Y.; Kumacheva, E. From hybrid microgels to photonic crystals. Adv. Funct. Mater. 2003, 13, 468–472. [Google Scholar] [CrossRef]
- Napolitano, A.P.; Chai, P.; Dean, D.M.; Morgan, J.R. Dynamics of the self-assembly of complex cellular aggregates on micromolded nonadhesive hydrogels. Tissue Eng. 2007, 13, 2087–2094. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; He, X.; Cao, M.; Wang, X.; Sun, Y.; He, H.; Xu, H.; Lu, J.R. Self-assembled two-dimensional thermoresponsive microgel arrays for cell growth/detachment control. Biomacromolecules 2014, 15, 4021–4031. [Google Scholar] [CrossRef] [PubMed]
- Harmon, M.E.; Tang, M.; Frank, C.W. A microfluidic actuator based on thermoresponsive hydrogels. Polymer 2003, 44, 4547–4556. [Google Scholar] [CrossRef]
- Liu, R.H.; Yu, Q.; Beebe, D.J. Fabrication and characterization of hydrogel-based microvalves. J. Microelectromech. Syst. 2002, 11, 45–53. [Google Scholar] [CrossRef]
- Liu, Z.; Calvert, P. Multilayer hydrogels as muscle-like actuators. Adv. Mater. 2000, 12, 288–291. [Google Scholar] [CrossRef]
- Das, M.; Mardyani, S.; Chan, W.C.; Kumacheva, E. Biofunctionalized pH-responsive microgels for cancer cell targeting: Rational design. Adv. Mater. 2006, 18, 80–83. [Google Scholar] [CrossRef]
- Tanaka, T.; Fillmore, D.J. Kinetics of swelling of gels. J. Chem. Phys. 1979, 70, 1214–1218. [Google Scholar] [CrossRef]
- Gestos, A.; Whitten, P.G.; Wallace, G.G.; Spinks, G.M. Actuating individual electrospun hydrogel nanofibres. Soft Matter 2012, 8, 8082–8087. [Google Scholar] [CrossRef]
- Hiltl, S.; Schürings, M.-P.; Balaceanu, A.; Mayorga, V.; Liedel, C.; Pich, A.; Böker, A. Guided self-assembly of microgels: From particle arrays to anisotropic nanostructures. Soft Matter 2011, 7, 8231–8238. [Google Scholar] [CrossRef]
- Biswal, S.L.; Gast, A.P. Mechanics of semiflexible chains formed by poly(ethylene glycol)-linked paramagnetic particles. Phys. Rev. E 2003, 68, 021402. [Google Scholar] [CrossRef] [PubMed]
- Goubault, C.; Jop, P.; Fermigier, M.; Baudry, J.; Bertrand, E.; Bibette, J. Flexible magnetic filaments as micromechanical sensors. Phys. Rev. Lett. 2003, 91, 260802. [Google Scholar] [CrossRef] [PubMed]
- Dreyfus, R.; Baudry, J.; Roper, M.L.; Fermigier, M.; Stone, H.A.; Bibette, J. Microscopic artificial swimmers. Nature 2005, 437, 862–865. [Google Scholar] [CrossRef] [PubMed]
- Reineck, P.; Lee, G.P.; Brick, D.; Karg, M.; Mulvaney, P.; Bach, U. A solid-state plasmonic solar cell via metal nanoparticle self-assembly. Adv. Mater. 2012, 24, 4750–4755. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.Y.; Rettner, C.T.; Sanders, D.P.; Kim, H.C.; Hinsberg, W.D. Dense self-assembly on sparse chemical patterns: Rectifying and multiplying lithographic patterns using block copolymers. Adv. Mater. 2008, 20, 3155–3158. [Google Scholar] [CrossRef]
- Srinivasan, S.; Praveen, V.K.; Philip, R.; Ajayaghosh, A. Bioinspired superhydrophobic coatings of carbon nanotubes and linear π systems based on the “bottom-up” self-assembly approach. Angew. Chem. Int. Ed. 2008, 47, 5750–5754. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, R.; Hataguchi, Y.; Kimura, R.; Tsuchiya, K.; Mori, Y. Stripe and network formation of particle arrays fabricated by convective self-assembly. Chem. Lett. 2012, 41, 1207–1209. [Google Scholar] [CrossRef]
- Han, W.; Byun, M.; Lin, Z. Assembling and positioning latex nanoparticles via controlled evaporative self-assembly. J. Mater. Chem. 2011, 21, 16968–16972. [Google Scholar] [CrossRef]
- Xia, Y.; Yin, Y.; Lu, Y.; McLellan, J. Template-assisted self-assembly of spherical colloids into complex and controllable structures. Adv. Funct. Mater. 2003, 13, 907–918. [Google Scholar] [CrossRef]
- Yin, Y.; Lu, Y.; Gates, B.; Xia, Y. Template-assisted self-assembly: A practical route to complex aggregates of monodispersed colloids with well-defined sizes, shapes, and structures. J. Am. Chem. Soc. 2001, 123, 8718–8729. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Lu, Y.; Xia, Y. Assembly of monodispersed spherical colloids into one-dimensional aggregates characterized by well-controlled structures and lengths. J. Mater. Chem. 2001, 11, 987–989. [Google Scholar] [CrossRef]
- Yin, Y.; Xia, Y. Self-assembly of spherical colloids into helical chains with well-controlled handedness. J. Am. Chem. Soc. 2003, 125, 2048–2049. [Google Scholar] [CrossRef] [PubMed]
- Efimenko, K.; Rackaitis, M.; Manias, E.; Vaziri, A.; Mahadevan, L.; Genzer, J. Nested self-similar wrinkling patterns in skins. Nat. Mater. 2005, 4, 293–297. [Google Scholar] [CrossRef] [PubMed]
- Chiche, A.; Stafford, C.M.; Cabral, J.T. Complex micropatterning of periodic structures on elastomeric surfaces. Soft Matter 2008, 4, 2360–2364. [Google Scholar] [CrossRef]
- Gittes, F.; Mickey, B.; Nettleton, J.; Howard, J. Flexural rigidity of microtubules and actin-filaments measured from thermal fluctuations in shape. J. Cell Biol. 1993, 120, 923–934. [Google Scholar] [CrossRef] [PubMed]
- Fakhri, N.; Tsyboulski, D.A.; Cognet, L.; Weisman, R.B.; Pasquali, M. Diameter-dependent bending dynamics of single-walled carbon nanotubes in liquids. Proc. Natl. Acad. Sci. USA 2009, 106, 14219–14223. [Google Scholar] [CrossRef] [PubMed]
- Tsyboulski, D.A.; Bachilo, S.M.; Kolomeisky, A.B.; Weisman, R.B. Translational and rotational dynamics of individual single-walled carbon nanotubes in aqueous suspension. ACS Nano 2008, 2, 1770–1776. [Google Scholar] [CrossRef] [PubMed]
- Keaveny, E.E.; Shelley, M.J. Hydrodynamic mobility of chiral colloidal aggregates. Phys. Rev. E 2009, 79, 051405. [Google Scholar] [CrossRef] [PubMed]
- Boyko, V.; Pich, A.; Lu, Y.; Richter, S.; Arndt, K.-F.; Adler, H.-J.P. Thermo-sensitive poly(N-vinylcaprolactam-co-acetoacetoxyethyl methacrylate) microgels: 1—Synthesis and characterization. Polymer 2003, 44, 7821–7827. [Google Scholar] [CrossRef]
- Genzer, J.; Groenewold, J. Soft matter with hard skin: From skin wrinkles to templating and material characterization. Soft Matter 2006, 2, 310–323. [Google Scholar] [CrossRef]
- Lamour, G.; Kirkegaard, J.B.; Li, H.; Knowles, T.P.; Gsponer, J. Easyworm: An open-source software tool to determine the mechanical properties of worm-like chains. Source Code Biol. Med. 2014, 9, 16. [Google Scholar] [CrossRef] [PubMed]
- Knowles, T.P.; Fitzpatrick, A.W.; Meehan, S.; Mott, H.R.; Vendruscolo, M.; Dobson, C.M.; Welland, M.E. Role of intermolecular forces in defining material properties of protein nanofibrils. Science 2007, 318, 1900–1903. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Akiyama, M.; Totani, K.; Kuebler, S.M.; Stellacci, F.; Wenseleers, W.; Braun, K.; Marder, S.R.; Perry, J.W. Photoresponsive hydrogel microstructure fabricated by two-photon initiated polymerization. Adv. Funct. Mater. 2002, 12, 611–614. [Google Scholar] [CrossRef]
- Hollmann, F.; Arends, I.W.C.E. Enzyme initiated radical polymerizations. Polymers 2012, 4, 759–793. [Google Scholar] [CrossRef]
- Han, Y.; Alsayed, A.M.; Nobili, M.; Zhang, J.; Lubensky, T.C.; Yodh, A.G. Brownian motion of an ellipsoid. Science 2006, 314, 626–630. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Bates, M.; Zhuang, X.W. Super-resolution fluorescence microscopy. Annu. Rev. Biochem. 2009, 78, 993–1016. [Google Scholar] [CrossRef] [PubMed]
- Saxton, M.J. Single-particle tracking: The distribution of diffusion coefficients. Biophys. J. 1997, 72, 1744–1753. [Google Scholar] [CrossRef]
- Broersma, S. Viscous force constant for a closed cylinder. J. Chem. Phys. 1960, 32, 1632–1635. [Google Scholar] [CrossRef]
- Broersma, S. Viscous force and torque constants for a cylinder. J. Chem. Phys. 1981, 74, 6989–6990. [Google Scholar] [CrossRef]
- Duggal, R.; Pasquali, M. Dynamics of individual single-walled carbon nanotubes in water by real-time visualization. Phys. Rev. Lett. 2006, 96, 246104. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Ortiz, C.; Xu, Y.; Willenbring, J.; Jerolmack, D. In situ liquid cell observations of asbestos fiber diffusion in water. Environ. Sci. Technol. 2015, 49, 13340–13349. [Google Scholar] [CrossRef] [PubMed]
- De La Torre, J.G.; Martinez, M.C.L.; Tirado, M.M. Dimensions of short, rodlike macromolecules from translational and rotational diffusion coefficients. Study of the gramicidin dimer. Biopolymers 1984, 23, 611–615. [Google Scholar] [CrossRef]
- Saragosti, J.; Silberzan, P.; Buguin, A. Modeling E. coli tumbles by rotational diffusion. Implications for chemotaxis. PLoS ONE 2012, 7, e35412. [Google Scholar] [CrossRef] [PubMed]

© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schürings, M.-P.; Nevskyi, O.; Eliasch, K.; Michel, A.-K.; Liu, B.; Pich, A.; Böker, A.; Von Plessen, G.; Wöll, D. Diffusive Motion of Linear Microgel Assemblies in Solution. Polymers 2016, 8, 413. https://doi.org/10.3390/polym8120413
Schürings M-P, Nevskyi O, Eliasch K, Michel A-K, Liu B, Pich A, Böker A, Von Plessen G, Wöll D. Diffusive Motion of Linear Microgel Assemblies in Solution. Polymers. 2016; 8(12):413. https://doi.org/10.3390/polym8120413
Chicago/Turabian StyleSchürings, Marco-Philipp, Oleksii Nevskyi, Kamill Eliasch, Ann-Katrin Michel, Bing Liu, Andrij Pich, Alexander Böker, Gero Von Plessen, and Dominik Wöll. 2016. "Diffusive Motion of Linear Microgel Assemblies in Solution" Polymers 8, no. 12: 413. https://doi.org/10.3390/polym8120413