Potential of Temperate, Tropical, and Sub-Tropical Exotic Maize Germplasm for Increased Gains in Yield Performance in Sub-Tropical Breeding Programs
Abstract
:1. Introduction
2. Methodology
2.1. Germplasm and Methodology
2.2. Trial Establishment, Experimental Design, and Site Management
2.3. Data Collection
2.4. Data Analysis
3. Results
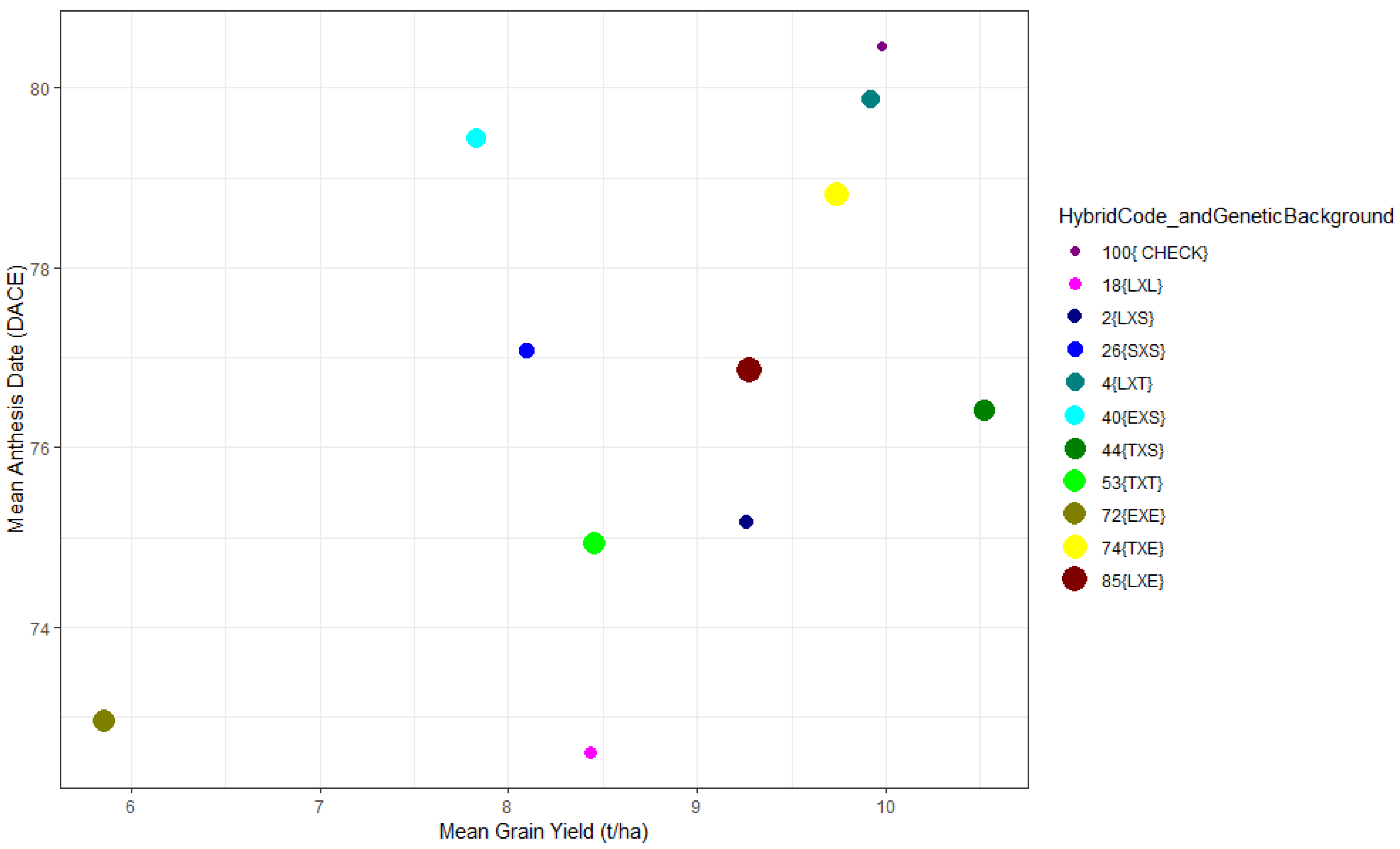
3.1. Agronomic Performance of the F1s within and across Locations
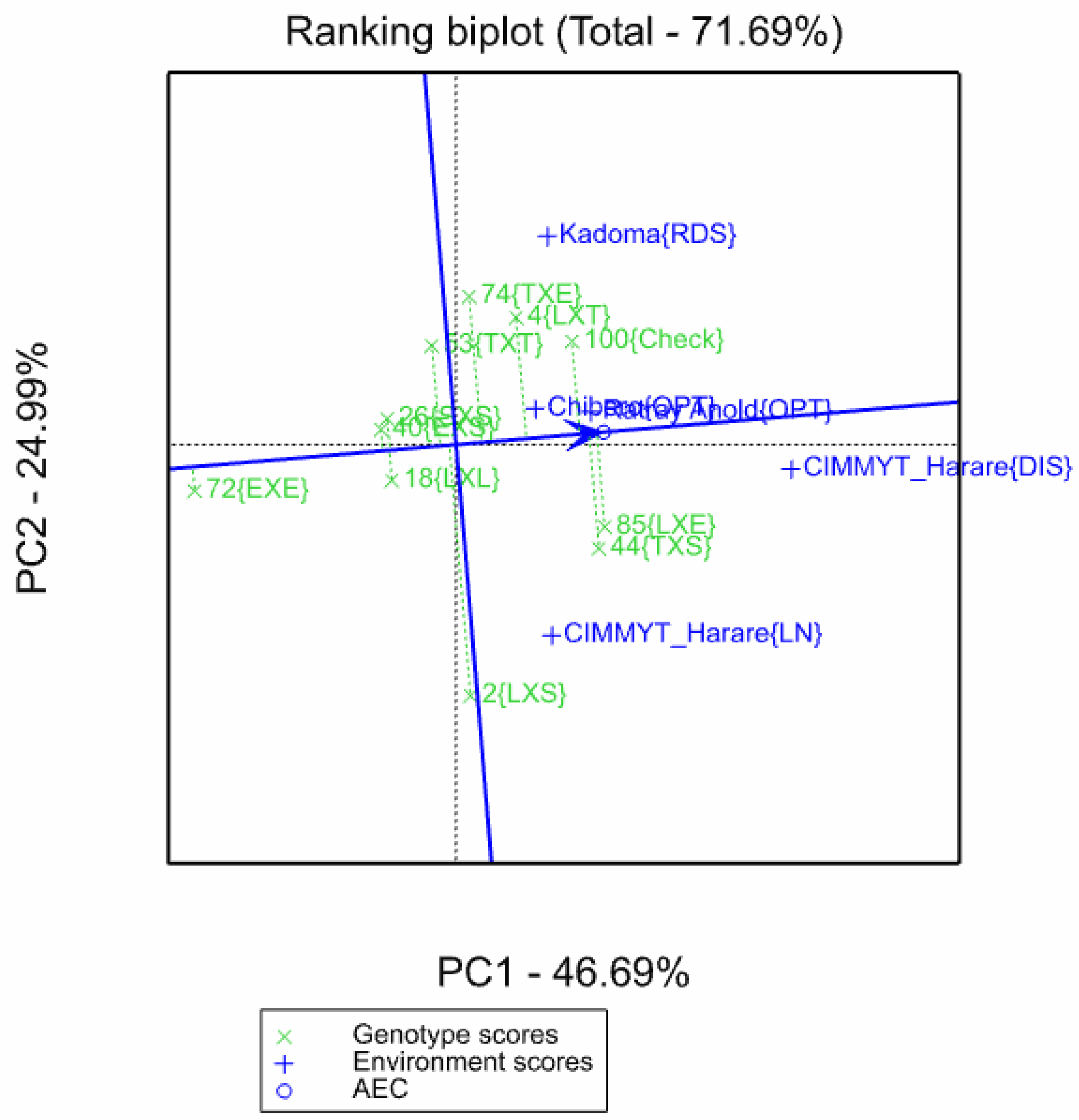
3.2. Identification of Ideal Exotic Germplasm for Use in Sub-Tropical Breeding Programs
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Aslam, M.; Cengiz, R. Effects of Drought on Maize. In Drought Stress in Maize (Zea mays L.); Springer International Publishing: Cham, Switzerland, 2015; pp. 5–17. [Google Scholar]
- Doswell, C. Maize in the Third World; CRC Press: Boca Raton, FL, USA, 2019. [Google Scholar]
- Ekpa, O.; Palacios-Rojas, N. Sub-Saharan African maize-based foods: Technological perspectives to increase the food and nutrition security impacts of maize breeding programmes. Glob. Food Secur. 2018, 17, 48–56. [Google Scholar] [CrossRef]
- Lydonn, Sub-Saharan-Africa-Falling-Short-in-Grain-Production. Available online: https://www.world-grain.com/articles/10887-sub-saharan-africa-falling-short-in-grain-production (accessed on 28 August 2018).
- Nelimor, C.; Badu-Apraku, B. Phenotypic Characterization of Maize Landraces from Sahel and Coastal West Africa Reveals Marked Diversity and Potential for Genetic Improvement. J. Crop Improv. 2019, 34, 122–138. [Google Scholar] [CrossRef]
- Bradshaw, C.D.; Pope, E.; Kay, G.; Davie, J.C.; Cottrell, A.; Bacon, J.; Cosse, A.; Dunstone, N.; Jennings, S.; Challinor, A.; et al. Unprecedented climate extremes in South Africa and implications for maize production. Environ. Res. Lett. 2022, 17, 084028. [Google Scholar] [CrossRef]
- Ortiz-Bobea, A.; Ault, T.R.; Carrillo, C.M.; Chambers, R.G.; Lobell, D.B. Anthropogenic climate change has slowed global agricultural productivity growth. Nat. Clim. Chang. 2021, 11, 306–312. [Google Scholar] [CrossRef]
- Muitire, C.; Kamutando, C.; Moyo, M. Building Stress Resilience of Cereals under Future Climatic Scenarios: ‘The Case of Maize, Wheat, Rice and Sorghum’. In Cereal Grains-Volume 1; IntechOpen: London, UK, 2021. [Google Scholar]
- Shiferaw, B.; Prasanna, B.M.; Hellin, J.; Bänziger, M. Crops that feed the world 6. Past successes and future challenges to the role played by maize in global food security. Food Secur. 2011, 3, 307. [Google Scholar] [CrossRef]
- Grafton, R.; Williams, Q.J.; Jiang, Q. Food and Water Gaps to 2050: Preliminary Results from the Global Food and Water System (GFWS) Platform. Food Secur. 2015, 7, 209–210. [Google Scholar] [CrossRef]
- Prasanna, B.M. Developing and Deploying Abiotic Stress-Tolerant Maize Varieties in the Tropics: Challenges and Opportunities. In Molecular Breeding for Sustainable Crop Improvement. Sustainable Development and Biodiversity; Rajpal, V., Rao, S., Raina, S., Eds.; Springer: Cham, Switzerland, 2016; Volume 11. [Google Scholar] [CrossRef]
- Campos, H.; Cooper, M.; Habben, J.E.; Edmeades, G.O.; Schussler, J.R. Improving drought tolerance in maize: A view from industry. Field Crops Res. 2004, 90, 19–34. [Google Scholar] [CrossRef]
- Cairns, J.E.; Hellin, J.; Sonder, K.; Araus, J.L.; Macrobert, J.F.; Thierfelder, C.; Prasanna, B.M. Adapting maize production to climate change in sub-Saharan Africa. Food Secur. 2013, 5, 345–360. [Google Scholar] [CrossRef]
- Prasanna, B.M. Diversity in global maize germplasm: Characterization and utilization. J. Biosci. 2012, 37, 843–855. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, F.C.; Caton, H.G.; Hallauer, A.R.; Vasal, S.; Cordova, H. Incorporation of elite subtropical and tropical maize germplasm into elite temperate germplasm. Maydica 2006, 51, 43–56. [Google Scholar]
- Lanes, E.C.M.; Viana, J.M.S.; Paes, G.P.; Paula, M.F.B.; Maia, C. Population structure and genetic diversity of maize inbreds derived from tropical hybrids. Genet. Mol. Res. 2014, 13, 7365–7376. [Google Scholar] [CrossRef]
- Adebayo, A.M.; Menkir, A.; Blay, E.; Gracen, V.; Danquah, E. Combining ability and heterosis of elite drought-tolerant maize inbred lines evaluated in diverse environments of lowland tropics. Euphytica 2017, 213, 43. [Google Scholar] [CrossRef]
- Meseka, S.; Williams, W.P.; Warburton, M.L.; Brown, R.L.; Augusto, J.; Ortega-Beltran, A.; Bandyopadhyay, R.; Menki, A. Heterotic affinity and combining ability of exotic maize inbred lines for resistance to aflatoxin accumulation. Euphytica 2018, 214, 184. [Google Scholar] [CrossRef]
- Wang, C.; Gardner, C.; Lubberstedt, T. Emerging Avenues for Utilization of Exotic Germplasm Emerging Avenues for Utilization of Exotic Germplasm. Trends Plant Sci. 2017, 22, 624–637. [Google Scholar] [CrossRef]
- Griffing, B. Concept of general and specific combining ability in relation to diallel crossing systems. Aust. J. Biol. Sci. 1956, 9, 463–493. [Google Scholar] [CrossRef]
- Bajet, N.B.; Renfro, B.L. Inoculation Methods for Maize Diseases; CIMMYT Maize Program: Harare, Zimbabwe, 2002. [Google Scholar]
- Vivek, B.; Kasango, J.; Chisoro, S.; Magorokosho, C. Fieldbook: Software for Managing a Maize Breeding Program: A Hookbook for Handling Field Experiments, Data Stocks and Pedigree Information; CIMMYT-Zimbabwe: Harare, Zimbabwe, 2007. [Google Scholar]
- Payne, R.; Murray, D.; Harding, S.; Baird, D.; Soutar, D. GenStat for Windows Introduction; VSN International: Hemel Hempstead, UK, 2009; p. 204. [Google Scholar]
- Barreto, H.; Edmeades, G.O.; Chapman, S.C.; Crossa, J. The alpha lattice design in plant breeding and agronomy: Generation and analysis; Drought-and Low-N tolerant maize. Agronomy 1996, 54. [Google Scholar]
- Wickham, H. GGplot2: Elegant Graphics for Data Analysis. Use R; Springer: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Lemon, J. Plotrix: A package in the red light district of R. R-News 2006, 6, 8–12. [Google Scholar]
- Rodríguez, F.; Alvarado, G.; Pacheco, Á.; Crossa, J.; Burgueño, J. AGD-R (Analysis of Genetic Designs with R for Windows) Version 5.0.; CIMMYT: Texcoco, México, 2018. [Google Scholar]
- Yan, W.; Kang, M.S. GGE Biplot Analysis: A Graphical Tool for Breeders, Geneticists, and Agronomists; CRC Press: Boca Raton, FL, USA, 2003. [Google Scholar]
- Holland, J.B. Breeding: Incorporation of exotic germplasm. In Encyclopedia of Plant and Crop Science; Marcel Dekker, Inc.: New York, NY, USA, 2004; pp. 222–224. [Google Scholar]
- Tahir, M.; Tanveer, A.; Ali, A.; Abbas, M.; Wasaya, A. Comparative Yield Performance of Different Maize (Zea mays L.) hybrids under Local Conditions of Faisalabad-Pakistan. Pak. J. Life Soc. Sci. 2008, 6, 118–120. [Google Scholar]
- Xavier, M.; Pollak, L.M.; Antônio, C.; Pacheco, P. Incorporating different proportions of exotic maize germplasm into two adapted populations. Genet. Mol. Biol. 2000, 451, 445–451. [Google Scholar]
- Nastasić, A.; Ivanović, M.; Stojaković, M.; Stanisavljević, D.; Treskić, S.; Mitrović, B.; Dražić, S. Effect of different proportions of exotic germplasm on grain yield and grain moisture in maize. Genetika 2011, 43, 67–73. [Google Scholar] [CrossRef]
- Abadassi, J. Maize agronomic traits needed in tropical zone. Int. J. Sci. Environ. Technol. 2015, 4, 371–392. [Google Scholar]
- Michelini, L.A.; Hallauer, A.R. Evaluation of exotic and adapted maize (Zea mays L.) germplasm crosses. Maydica 1993, 38, 275–282. [Google Scholar]
- Lana, M.A.; Eulenstein, F.; Schlindwein, S.L.; Graef, F.; Sieber, S.; von Hertwig Bittencourt, H. Yield stability and lower susceptibility to abiotic stresses of improved open-pollinated and hybrid maize cultivars. Agron. Sustain. Dev. 2017, 37, 30. [Google Scholar] [CrossRef]
- MacRoberts, J.; Setimela, P.S. Maize Hybrid Seed Production Manual; CIMMYT: Texcoco, México, 2014; pp. 27–31. [Google Scholar]
- Wen, W.; Guo, T.; Tovar, V.; Li, H. The strategy and potential utilization of temperate germplasm for tropical germplasm improvement: A case study of maize (Zea mays L.). Mol. Breed. 2012, 29, 951–962. [Google Scholar] [CrossRef]

| Parental Number | Name | Genetic Background | Genetic Background [Coded] |
|---|---|---|---|
| P1 | CL115324 | Pedigree start = STAL × Temperate | Temperate (T) |
| P2 | CL1210884 | Pedigree start = STAL × Temperate | Temperate (T) |
| P3 | DJ-154 | Pedigree start = STAL × Temperate | Temperate (T) |
| P4 | CL1210969 | Pedigree start = STAL × Temperate | Temperate (T) |
| P5 | C1008-1 | Pedigree start = STAL × Tropical | Tropical (E) |
| P6 | CL1211291 | Pedigree start = STAL × Sub-tropical | Sub-tropical (S) |
| P7 | CL1211293 | Pedigree start = STAL × Sub-tropical | Sub-tropical (S) |
| P8 | CL1212428 | Pedigree start = STAL × Tropical | Sub-tropical (S) |
| P9 | CL1214868 | Pedigree start = STAL × STAL | Local (L) |
| P10 | CL1210571 | Pedigree start = STAL × STAL | Local (L) |
| P11 | CL1310262 | Pedigree start = STAL × STAL | Local (L) |
| P12 | DJ9-5 | Pedigree start = STAL × STAL | Local (L) |
| P13 | DJ9-1 | Pedigree start = STAL × STAL | Local (L) |
| P14 | CML444 | Pedigree start = STAL × STAL | Local (L) |
| Site | Chiredzi | CIMMYT-Harare | Chibhero | Kadoma | Ratray Anold |
|---|---|---|---|---|---|
| Latitude | 21°1′10′′ S | 17°49′ S | 17°26′ S | 18°20′ S | 15°49′ S |
| Longitude | 31°34′23′′ E | 31°01′ E | 31°05′ E | 30°97′ E | 20°01′ E |
| Soil type | Paragneiss clay | Red clay soil | Sandy loam soil | Red Clay | Red Clay |
| Type of Irrigation | Overhead sprinkler | Overhead sprinkler | Overheard sprinkler | Overheard sprinkler | Overheard sprinkler |
| Rainfall received | <450 mm | 750–1000 mm | 750–1000 mm | 650–800 mm | 550–800 mm |
| Altitude (mas) | 445 | 1480 | 1480 | 1149 | |
| Mega Environment | E | A | C | C | B |
| Natural region | IV | IIa | IIa | III | IIb |
| GY (Tha−1) | AD (Days) | ASI (Days) | EPO (cm) | HC (%) | ER (%) | ET (Score 1–5) | GLS (Score 1–5) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Source of Variation | Df | Mean Sq | Df | Mean Sq | Df | Mean Sq | Df | Mean Sq | DF | Mean Sq | Df | Mean Sq | DF | Mean Sq | Df | Mean Sq |
| Location | 4 | 802.9 *** | 4 | 14,242.7 *** | 3 | 214.968 *** | 6 | 5,252,421 *** | 4 | 5156.8 *** | 3 | 0 | 3 | 0 | 3 | 28.4028 *** |
| Genotype | 99 | 16.76 *** | 99 | 42.8 *** | 99 | 28.003 *** | 99 | 519,550 *** | 99 | 729.9 *** | 99 | 227.501 *** | 99 | 0.69171 *** | 99 | 1.8292 *** |
| Replication (Location) | 5 | 9.65 * | 5 | 26.5 *** | 4 | 87.008 *** | 7 | 585,832 *** | 5 | 458.3 *** | 4 | 26.091 | 4 | 0.06564 | 4 | 0.5879 *** |
| Genotype × Location | 396 | 8.16 *** | 391 | 10.2 *** | 297 | 17.975 *** | 594 | 135,493 *** | 396 | 175.9 *** | 297 | 0.061 | 297 | 0.00063 | 292 | 0.2338 *** |
| Block (Replication × Location) | 190 | 4.48 | 190 | 5.9 | 152 | 11.243 | 266 | 101,578 * | 190 | 100.2 | 152 | 65.085 | 152 | 0.13882 | 152 | 0.0962 |
| Residuals | 305 | 3.93 | 283 | 5.9 | 244 | 10.636 | 427 | 79891 | 305 | 100.5 | 244 | 63.541 | 240 | 0.13078 | 221 | 0.1178 |
| Grand mean | 7.4821 | 75.6232 | 1.6028 | 0.5339 | 7.0942 | 7.1903 | 2.2072 | 2.0842 | ||||||||
| Broad sense heritability | 0.5133 | 0.7699 | 0.2823 | 0.744 | 0.7594 | 0 | 0.1559 | 0.8774 | ||||||||
| LSD (5 %) | 3.9872 | 4.5967 | 3.4582 | 0.0822 | 19.5947 | 15.7009 | 0.524 | 0.6507 | ||||||||
| CV | 27.1885 | 3.1013 | 110.0849 | 7.8539 | 140.9215 | 111.4093 | 12.1133 | 15.929 | ||||||||
| Genotype | Cross | HybridCode_ and Genetic Background | Genetic Background | GCA_Parent 1 (tha-1) | GCA_Parent 2 (tha-1) | SCA (tha-1) | Grain Yield (tha-1) | Anthesis Date (Days) |
|---|---|---|---|---|---|---|---|---|
| G2 | P10 × P6 | 2{LXS} | LXS | 0.446 ** | 0.643 *** | 0.765 | 9.26 | 75.169 |
| G4 | P10 × P1 | 4{LXT} | LXT | 0.446 ** | 0.390 ** | 1.606 ** | 9.921 | 79.885 |
| G18 | P13 × P12 | 18{LXL} | LXL | −0.577 *** | −0.293 * | 1.87 ** | 8.43 | 72.608 |
| G26 | P6xP7 | 26{SXS} | SXS | 0.643 *** | 0.468 *** | −0.48 | 8.092 | 77.088 |
| G40 | P7xP5 | 40{SXE} | EXS | 0.468 *** | −0.066 | −0.018 | 7.83 | 79.443 |
| G44 | P7xP2 | 44{TXS} | TXS | 0.468 *** | 0.789 *** | 1.814 ** | 10.52 | 76.425 |
| G53 | P1xP2 | 53{TXT} | TXT | 0.390 ** | 0.789 *** | −0.13 | 8.454 | 74.931 |
| G72 | P5xP8 | 72{EXE} | EXE | −0.066 | −0.505 ** | −1.046 | 5.858 | 72.953 |
| G74 | P5xP2 | 74{TXE} | TXE | −0.066 | 0.789 *** | 1.546 * | 9.74 | 78.813 |
| G85 | P8xP14 | 85{LXE} | LXE | −0.505 ** | −0.288 * | 2.559 *** | 9.278 | 76.862 |
| G100 | Check | 100{CHECK} | CHECK | 9.975 | 80.468 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nyoni, R.S.; Magorokosho, C.; Kamutando, C.N. Potential of Temperate, Tropical, and Sub-Tropical Exotic Maize Germplasm for Increased Gains in Yield Performance in Sub-Tropical Breeding Programs. Agronomy 2023, 13, 1605. https://doi.org/10.3390/agronomy13061605
Nyoni RS, Magorokosho C, Kamutando CN. Potential of Temperate, Tropical, and Sub-Tropical Exotic Maize Germplasm for Increased Gains in Yield Performance in Sub-Tropical Breeding Programs. Agronomy. 2023; 13(6):1605. https://doi.org/10.3390/agronomy13061605
Chicago/Turabian StyleNyoni, Rejoice Shumirai, Cosmos Magorokosho, and Casper Nyaradzai Kamutando. 2023. "Potential of Temperate, Tropical, and Sub-Tropical Exotic Maize Germplasm for Increased Gains in Yield Performance in Sub-Tropical Breeding Programs" Agronomy 13, no. 6: 1605. https://doi.org/10.3390/agronomy13061605
APA StyleNyoni, R. S., Magorokosho, C., & Kamutando, C. N. (2023). Potential of Temperate, Tropical, and Sub-Tropical Exotic Maize Germplasm for Increased Gains in Yield Performance in Sub-Tropical Breeding Programs. Agronomy, 13(6), 1605. https://doi.org/10.3390/agronomy13061605







