Unraveling the Roles of Regulatory Genes during Domestication of Cultivated Camellia: Evidence and Insights from Comparative and Evolutionary Genomics
Abstract
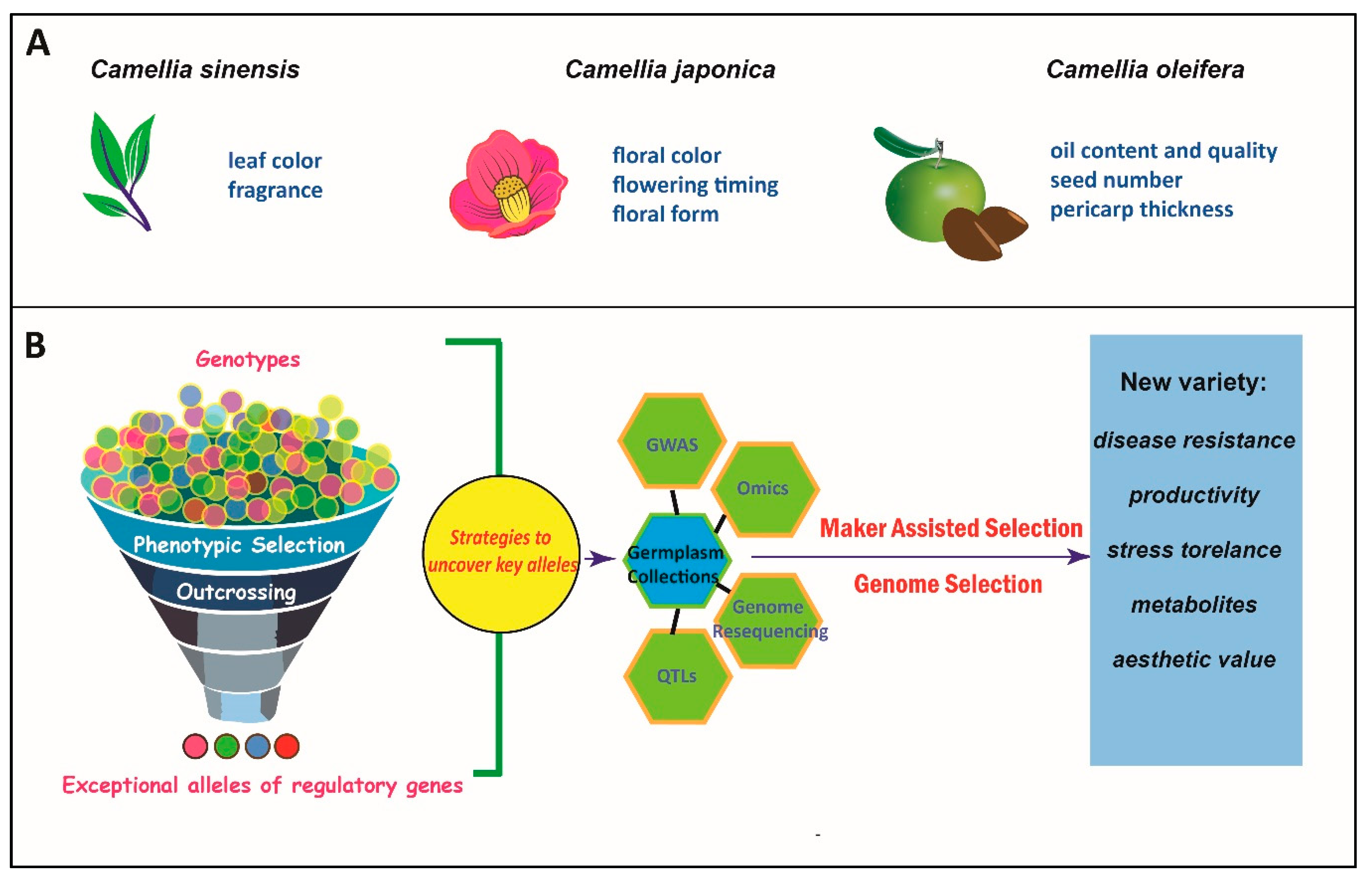
:1. Introduction
2. Genetics and Genomic Resources in Genus Camellia Empowered by High-Throughput Sequencing
2.1. Responses of Biotic and Abiotic Stress in Camellia Plants
2.2. Transcriptomic Analyses in the Control of Secondary Metabolism in Camellia
2.3. Transcriptomics Studies Related to Floral Patterning, Flowering Timing, and Bud Dormancy
2.4. Transcriptomics in Oil Camellia Plants
2.5. Markers Development Based on RNA-Sequencing
3. GWAS and QTL Mapping of Key Traits in Camellia Plants
4. Functional Characterization of Genes Related to Key Pathways of Camellia Plants
5. Future Perspectives: A Roadmap for Camellia
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lenser, T.; Theißen, G. Molecular mechanisms involved in convergent crop domestication. Trends Plant Sci. 2013, 18, 704–714. [Google Scholar] [CrossRef] [PubMed]
- Abbo, S.; Pinhasi van-Oss, R.; Gopher, A.; Saranga, Y.; Ofner, I.; Peleg, Z. Plant domestication versus crop evolution: A conceptual framework for cereals and grain legumes. Trends Plant Sci. 2014, 19, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.; Li, X.; Shannon, L.M.; Yeh, C.T.; Wang, M.L.; Bai, G.; Peng, Z.; Li, J.; Trick, H.N.; Clemente, T.E.; et al. Parallel domestication of the Shattering1 genes in cereals. Nat. Genet. 2012, 44, 720–724. [Google Scholar] [CrossRef] [PubMed]
- Fuller, D.Q.; Denham, T.; Arroyo-Kalin, M.; Lucas, L.; Stevens, C.J.; Qin, L.; Allaby, R.G.; Purugganan, M.D. Convergent evolution and parallelism in plant domestication revealed by an expanding archaeological record. Proc. Natl. Acad. Sci. USA 2014, 111, 6147–6152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Purugganan, M.D.; Fuller, D.Q. The nature of selection during plant domestication. Nature 2009, 457, 843–848. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Chen, L.L.; Ruan, X.; Chen, D.; Zhu, A.; Chen, C.; Bertrand, D.; Jiao, W.B.; Hao, B.H.; Lyon, M.P.; et al. The draft genome of sweet orange (Citrus sinensis). Nat. Genet. 2013, 45, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.A.; Prochnik, S.; Jenkins, J.; Salse, J.; Hellsten, U.; Murat, F.; Perrier, X.; Ruiz, M.; Scalabrin, S.; Terol, J.; et al. Sequencing of diverse mandarin, pummelo and orange genomes reveals complex history of admixture during citrus domestication. Nat. Biotechnol. 2014, 32, 656–662. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- International Peach Genome Initiative; Verde, I.; Abbott, A.G.; Scalabrin, S.; Jung, S.; Shu, S.; Marroni, F.; Zhebentyayeva, T.; Dettori, M.T.; Grimwood, J.; et al. The high-quality draft genome of peach (Prunus persica) identifies unique patterns of genetic diversity, domestication and genome evolution. Nat. Genet. 2013, 45, 487–494. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Chen, W.; Sun, L.; Zhao, F.; Huang, B.; Yang, W.; Tao, Y.; Wang, J.; Yuan, Z.; Fan, G.; et al. The genome of Prunus mume. Nat. Commun. 2012, 3, 1318. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.A.; Terol, J.; Ibanez, V.; Lopez-Garcia, A.; Perez-Roman, E.; Borreda, C.; Domingo, C.; Tadeo, F.R.; Carbonell-Caballero, J.; Alonso, R.; et al. Genomics of the origin and evolution of citrus. Nature 2018, 554, 311–316. [Google Scholar] [CrossRef] [PubMed]
- Gao, J. Collected Species of the Genus Camellia- an Illustrated Outline; Zhejiang Science and Technology Publishing House: Hangzhou, China, 2005; pp. 12–35. ISBN 978-7534125942. [Google Scholar]
- Sealy, J.R. A revision of the Genus Camellia; Royal Horticultural Society: London, UK, 1958; pp. 1–235. [Google Scholar]
- Wei, C.; Yang, H.; Wang, S.; Zhao, J.; Liu, C.; Gao, L.; Xia, E.; Lu, Y.; Tai, Y.; She, G.; et al. Draft genome sequence of Camellia sinensis var. Sinensis provides insights into the evolution of the tea genome and tea quality. Proc. Natl. Acad. Sci. USA 2018, 115, E4151–E4158. [Google Scholar]
- Xia, E.H.; Zhang, H.B.; Sheng, J.; Li, K.; Zhang, Q.J.; Kim, C.; Zhang, Y.; Liu, Y.; Zhu, T.; Li, W.; et al. The tea tree genome provides insights into tea flavor and independent evolution of caffeine biosynthesis. Mol. Plant 2017, 10, 866–877. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.Y.; Yang, H.; Wei, C.L.; Yu, O.; Zhang, Z.Z.; Jiang, C.J.; Sun, J.; Li, Y.Y.; Chen, Q.; Xia, T.; et al. Deep sequencing of the Camellia sinensis transcriptome revealed candidate genes for major metabolic pathways of tea-specific compounds. BMC Genom. 2011, 12. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.C.; Zhao, Q.Y.; Ma, C.L.; Zhang, Z.H.; Cao, H.L.; Kong, Y.M.; Yue, C.; Hao, X.Y.; Chen, L.; Ma, J.Q.; et al. Global transcriptome profiles of Camellia sinensis during cold acclimation. BMC Genom. 2013, 14. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.J.; Li, X.H.; Liu, Z.W.; Li, H.; Wang, Y.X.; Zhuang, J. Transcriptome-based discovery of ap2/erf transcription factors related to temperature stress in tea plant (Camellia sinensis). Funct. Integr. Genom. 2015, 15, 741–752. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.J.; Li, X.H.; Liu, Z.W.; Li, H.; Wang, Y.X.; Zhuang, J. Transcriptome-wide identification of Camellia sinensis WRKY transcription factors in response to temperature stress. Mol. Genet. Genom. 2016, 291, 255–269. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Lei, S.; Du, K.; Li, L.; Pang, X.; Wang, Z.; Wei, M.; Fu, S.; Hu, L.; Xu, L. RNA-seq based transcriptomic analysis uncovers α-linolenic acid and jasmonic acid biosynthesis pathways respond to cold acclimation in Camellia japonica. Sci. Rep. 2016, 6, 36463. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Cai, M.; Yu, X.; Wang, L.; Guo, C.; Ming, R.; Zhang, J. Transcriptome dynamics of Camellia sinensis in response to continuous salinity and drought stress. Tree Genet. Genomes 2017, 13. [Google Scholar] [CrossRef]
- Jin, X.; Liu, D.; Ma, L.; Gong, Z.; Cao, D.; Liu, Y.; Li, Y.; Jiang, C. Transcriptome and expression profiling analysis of recalcitrant tea (Camellia sinensis L.) seeds sensitive to dehydration. Int. J. Genom. 2018, 2018. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Xiang, F.; Zhong, M.; Zhou, L.; Liu, H.; Li, S.; Wang, X. Transcriptome and metabolite analysis identifies nitrogen utilization genes in tea plant (Camellia sinensis). Sci. Rep. 2017, 7, 1693. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Wang, W.; Li, D.; Shu, Z.; Ye, X.; Chang, P.; Wang, Y. Gene expression profile indicates involvement of no in Camellia sinensis pollen tube growth at low temperature. BMC Genom. 2016, 17. [Google Scholar] [CrossRef] [PubMed]
- Jayaswall, K.; Mahajan, P.; Singh, G.; Parmar, R.; Seth, R.; Raina, A.; Swarnkar, M.K.; Singh, A.K.; Shankar, R.; Sharma, R.K. Transcriptome analysis reveals candidate genes involved in Blister Blight defense in tea (Camellia sinensis (L) kuntze). Sci. Rep. 2016, 6, 30412. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.N.; Tang, L.; Hou, Y.; Wang, P.; Yang, H.; Wei, C.L. Differential transcriptome analysis of leaves of tea plant (Camellia sinensis) provides comprehensive insights into the defense responses to Ectropis oblique attack using rna-seq. Funct. Integr. Genom. 2016, 16, 383–398. [Google Scholar] [CrossRef] [PubMed]
- Li, C.F.; Zhu, Y.; Yu, Y.; Zhao, Q.Y.; Wang, S.J.; Wang, X.C.; Yao, M.Z.; Luo, D.; Li, X.; Chen, L.; et al. Global transcriptome and gene regulation network for secondary metabolite biosynthesis of tea plant (Camellia sinensis). BMC Genom. 2015, 16. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.B.; Xia, E.H.; Huang, H.; Jiang, J.J.; Liu, B.Y.; Gao, L.Z. De novo transcriptome assembly of the wild relative of tea tree (Camellia taliensis) and comparative analysis with tea transcriptome identified putative genes associated with tea quality and stress response. BMC Genom. 2015, 16. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhou, Y.; Wu, Y.; Dai, X.; Liu, Y.; Qian, Y.; Li, M.; Jiang, X.; Wang, Y.; Gao, L.; et al. An insight into catechins metabolic pathways of Camellia sinensis based on genome and transcriptome analysis. J. Agric. Food Chem. 2018, 66, 4281–4293. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.J.; Li, X.H.; Liu, Z.W.; Xu, Z.S.; Zhuang, J. De novo assembly and transcriptome characterization: Novel insights into catechins biosynthesis in Camellia sinensis. BMC Plant Biol. 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Lv, X.; Wang, L.; Qiu, Z.; Song, X.; Lin, J.; Chen, W. Transcriptome analysis reveals the accumulation mechanism of anthocyanins in ‘Zijuan’ tea (Camellia sinensis var. asssamica (Masters) kitamura) leaves. Plant Growth Regul. 2017, 81, 51–61. [Google Scholar] [CrossRef]
- Zhou, X.; Li, J.; Zhu, Y.; Ni, S.; Chen, J.; Feng, X.; Zhang, Y.; Li, S.; Zhu, H.; Wen, Y. De novo assembly of the Camellia nitidissima transcriptome reveals key genes of flower pigment biosynthesis. Front Plant Sci. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Fan, Z.; Guo, H.; Ye, N.; Lyu, T.; Yang, W.; Wang, J.; Wang, J.T.; Wu, B.; Li, J.; et al. Comparative genomics analysis reveals gene family expansion and changes of expression patterns associated with natural adaptations of flowering time and secondary metabolism in yellow Camellia. Funct. Integr. Genom. 2018. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, J.; Fan, Z.; Liu, Z.; Tanaka, T.; Yin, H. Global gene expression defines faded whorl specification of double flower domestication in Camellia. Sci. Rep. 2017, 7, 3197. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.; Fan, Z.; Li, X.; Wang, J.; Liu, W.; Wu, B.; Ying, Z.; Liu, L.; Liu, Z.; Li, J. Phylogenetic tree-informed micrornaome analysis uncovers conserved and lineage-specific miRNAs in Camellia during floral organ development. J. Exp. Bot. 2016, 67, 2641–2653. [Google Scholar] [CrossRef] [PubMed]
- Hao, X.; Yang, Y.; Yue, C.; Wang, L.; Horvath, D.P.; Wang, X. Comprehensive transcriptome analyses reveal differential gene expression profiles of Camellia sinensis axillary buds at para-, endo-, ecodormancy, and bud flush stages. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Thirugnanasambantham, K.; Prabu, G.; Palanisamy, S.; Chandrabose, S.R.; Mandal, A.K. Analysis of dormant bud (Banjhi) specific transcriptome of tea (Camellia sinensis (L.) o. Kuntze) from cDNA library revealed dormancy-related genes. Appl. Biochem. Biotechnol. 2013, 169, 1405–1417. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.; Li, J.; Li, X.; Wu, B.; Wang, J.; Liu, Z.; Yin, H. Genome-wide transcriptome profiling provides insights into floral bud development of summer-flowering Camellia azalea. Sci. Rep. 2015, 5, 9729. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Wang, Y.; Ding, Z.; Zhao, L.; Xiao, J.; Wang, L.; Ding, S. Transcriptomic analysis of flower development in tea (Camellia sinensis (L.)). Gene 2017, 631, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Paul, A.; Jha, A.; Bhardwaj, S.; Singh, S.; Shankar, R.; Kumar, S. RNA-seq-mediated transcriptome analysis of actively growing and winter dormant shoots identifies non-deciduous habit of evergreen tree tea during winters. Sci. Rep. 2014, 4, 5932. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Chen, Z.; Sun, W.; Deng, T.; Chen, M. De novo sequencing of the leaf transcriptome reveals complex light-responsive regulatory networks in Camellia sinensis cv. Baijiguan. Front. Plant. Sci. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Wei, K.; Wang, L.; Cheng, H.; Zhang, C.; Ma, C.; Zhang, L.; Gong, W.; Wu, L. Identification of genes involved in indole-3-butyric acid-induced adventitious root formation in nodal cuttings of Camellia sinensis (L.) by suppression subtractive hybridization. Gene 2013, 514, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Wei, K.; Wang, L.Y.; Wu, L.Y.; Zhang, C.C.; Li, H.L.; Tan, L.Q.; Cao, H.L.; Cheng, H. Transcriptome analysis of indole-3-butyric acid-induced adventitious root formation in nodal cuttings of Camellia sinensis (L.). PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Xia, E.H.; Jiang, J.J.; Huang, H.; Zhang, L.P.; Zhang, H.B.; Gao, L.Z. Transcriptome analysis of the oil-rich tea plant, Camellia oleifera, reveals candidate genes related to lipid metabolism. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Dong, B.; Wu, B.; Hong, W.; Li, X.; Li, Z.; Xue, L.; Huang, Y. Transcriptome analysis of the tea oil Camellia (Camellia oleifera) reveals candidate drought stress genes. PLoS ONE 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.; Wang, K.; Zhou, C.; Xie, Y.; Yao, X.; Yin, H. Seed transcriptomics analysis in Camellia oleifera uncovers genes associated with oil content and fatty acid composition. Int. J. Mol. Sci. 2018, 19, 118. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Yang, X.; Huang, X.; Duan, S.; Long, C.; Chen, J.; Rong, J. Leaf transcriptome analysis of a subtropical evergreen broadleaf plant, wild oil-tea camellia (Camellia oleifera), revealing candidate genes for cold acclimation. BMC Genom. 2017, 18. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.W.; Jiang, C.; Wen, Q.; Wang, N.; Tao, Y.Y.; Xu, L.A. Deep sequencing of the Camellia chekiangoleosa transcriptome revealed candidate genes for anthocyanin biosynthesis. Gene 2014, 538, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.L.; Yang, Z.J.; Bai, W.W.; Chen, S.P.; Xu, W.Q.; El-Kassaby, Y.A.; Chen, H. Transcriptome comparative analysis of two Camellia species reveals lipid metabolism during mature seed natural drying. Trees 2017, 31, 1827–1848. [Google Scholar] [CrossRef]
- Jiang, C.; Wen, Q.; Chen, Y.; Xu, L.; Huang, M. Efficient extraction of RNA from various Camellia species rich in secondary metabolites for deep transcriptome sequencing and gene expression analysis. Afr. J. Biotechnol. 2011, 10, 16769–16773. [Google Scholar] [CrossRef]
- Wu, H.; Chen, D.; Li, J.; Yu, B.; Qiao, X.; Huang, H.; He, Y. De novo characterization of leaf transcriptome using 454 sequencing and development of EST-SSR markers in tea (Camellia sinensis). Plant Mol. Biol. Rep. 2013, 31, 524–538. [Google Scholar] [CrossRef]
- Tan, L.Q.; Wang, L.Y.; Wei, K.; Zhang, C.C.; Wu, L.Y.; Qi, G.N.; Cheng, H.; Zhang, Q.; Cui, Q.M.; Liang, J.B. Floral transcriptome sequencing for SSR marker development and linkage map construction in the tea plant (Camellia sinensis). PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Liufu, Y.Q.; Peng, G.Q.; Lu, Y.B.; Ye, Q.Q.; Tang, S.Q. Development and characterization of 38 microsatellite markers for Camellia flavida based on transcriptome sequencing. Conserv. Genet. Resour. 2014, 6, 1007–1010. [Google Scholar] [CrossRef]
- Jia, B.; Lin, Q.; Lin, Z.; Tan, X.; Lei, X.; Hu, X.; Shao, F. Development of 15 genic-SSR markers in oil-tea tree (Camellia oleifera) based on transcriptome sequencing. Genetika 2014, 46, 789–797. [Google Scholar] [CrossRef]
- Arbona, V.; Manzi, M.; Ollas, C.; Gomez-Cadenas, A. Metabolomics as a tool to investigate abiotic stress tolerance in plants. Int. J. Mol. Sci. 2013, 14, 4885–4911. [Google Scholar] [CrossRef] [PubMed]
- Arasimowicz, M.; Floryszak-Wieczorek, J. Nitric oxide as a bioactive signalling molecule in plant stress responses. Plant Sci. 2007, 172, 876–887. [Google Scholar] [CrossRef]
- Guo, Z.; Tan, J.; Zhuo, C.; Wang, C.; Xiang, B.; Wang, Z. Abscisic acid, H2O2 and nitric oxide interactions mediated cold-induced S-adenosylmethionine synthetase in Medicago sativa subsp. Falcata that confers cold tolerance through up-regulating polyamine oxidation. Plant Biotechnol. J. 2014, 12, 601–612. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.G.; Chen, L.; Zhang, L.L.; Zhang, W.H. Nitric reductase-dependent nitric oxide production is involved in cold acclimation and freezing tolerance in Arabidopsis. Plant Physiol. 2009, 151, 755–767. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Gao, X.B.; Lian, H.E.; Zhang, Q.; Meng, Z.H.; Zhao-Yun, L.; Chen, J. Application research progress of biological control of tea diseases and pests. Guangdong Agric. Sci. 2014, 41, 105–109. [Google Scholar]
- Tang, Y.; Zhou, G.; He, L.I.; Zhong, W.; Gong, H.; Wang, L. Identification of a new anthracnose of Camellia oleifera based on multiple-gene phylogeny. Chin. J. Trop. Crops 2015, 36, 972–977. [Google Scholar]
- Chang, M.S.; Deng, Y.; Liao, W.J.; Quan, S.U.; Fang, X.Y.; Yao-Jun, W.U. Study on the relationship between enzyme activity and disease resistance of different Camellia oleifera varieties to Colletotrichum gloeosporioides penz. For. Res. 2018, 31, 141–146. [Google Scholar]
- Korte, A.; Farlow, A. The advantages and limitations of trait analysis with GWAS: A review. Plant Methods 2013, 9. [Google Scholar] [CrossRef] [PubMed]
- Collard, B.C.Y.; Mackill, D.J. Marker-assisted selection: An approach for precision plant breeding in the twenty-first century. Philos. Trans. R. Soc. London B. Biol. Sci. 2008, 363, 557–572. [Google Scholar] [CrossRef] [PubMed]
- Yousef, G.G.; Juvik, J.A. Comparison of phenotypic and marker-assisted selection for quantitative traits in sweet corn. Crop Sci. 2001, 41, 645–655. [Google Scholar] [CrossRef]
- Flachowsky, H.; Roux, P.M.L.; Peil, A.; Patocchi, A.; Richter, K.; Hanke, M.V. Application of a high-speed breeding technology to apple (malus × domestica) based on transgenic early flowering plants and marker-assisted selection. New Phytol. 2011, 192, 364–377. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.K.; Langridge, P.; Mir, R.R. Marker-assisted wheat breeding: Present status and future possibilities. Mol. Breed. 2010, 26, 145–161. [Google Scholar] [CrossRef]
- Castro, A.J.; Capettini, F.; Corey, A.E.; Filichkina, T.; Hayes, P.M.; Kleinhofs, A.; Kudrna, D.; Richardson, K.; Sandoval-Islas, S.; Rossi, C.; et al. Mapping and pyramiding of qualitative and quantitative resistance to stripe rust in barley. Theor. Appl. Genet. 2003, 107, 922–930. [Google Scholar] [CrossRef] [PubMed]
- Bali, S.; Mamgain, A.; Raina, S.N.; Yadava, S.K.; Bhat, V.; Das, S.; Pradhan, A.K.; Goel, S. Construction of a genetic linkage map and mapping of drought tolerance trait in Indian beveragial tea. Mol. Breed. 2015, 35. [Google Scholar] [CrossRef]
- Devi, A.M.; Goel, S.; Misra, A.K. Generation of silver stained TE-AFLP markers in tea (Camellia sinensis) and their assessment in filling gaps with construction of a genetic linkage map. Sci. Horti. 2015, 192, 293–301. [Google Scholar] [CrossRef]
- Hackett, C.A.; Wachira, F.N.; Paul, S.; Powell, W.; Waugh, R. Construction of a genetic linkage map for Camellia sinensis (tea). Heredity 2010, 85, 346–355. [Google Scholar] [CrossRef]
- Hu, C.Y.; Lee, T.C.; Tsai, H.T.; Tsai, Y.Z.; Lin, S.F. Construction of an integrated genetic map based on maternal and paternal lineages of tea (Camellia sinensis). Euphytica 2013, 191, 141–152. [Google Scholar] [CrossRef]
- Huang, F.P.; Liang, Y.R.; Jian-Lian, L.U.; Chen, R.B. Genetic mapping of first generation of backcross in tea by RAPD and ISSR markers. J. Tea Sci. 2006, 26, 171–176. [Google Scholar]
- Huang, J.A.; Jia-Xian, L.I.; Huang, Y.H.; Luo, J.W.; Gong, Z.H.; Liu, Z.H. Construction of AFLP molecular markers linkage map in tea plant. J. Tea Sci. 2005, 25, 7–15. [Google Scholar]
- Kamunya, S.M.; Wachira, F.N.; Pathak, R.S.; Korir, R.; Sharma, V.; Kumar, R.; Bhardwaj, P.; Chalo, R.; Ahuja, P.S.; Sharma, R.K. Genomic mapping and testing for quantitative trait loci in tea (Camellia sinensis (L.) o. Kuntze). Tree Genet. Genomes 2010, 6, 915–929. [Google Scholar] [CrossRef]
- Ma, J.Q.; Huang, L.; Ma, C.L.; Jin, J.Q.; Li, C.F.; Wang, R.K.; Zheng, H.K.; Yao, M.Z.; Chen, L. Large-scale SNP discovery and genotyping for constructing a high-density genetic map of tea plant using specific-locus amplified fragment sequencing (SLAF-seq). PLoS ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.Q.; Yao, M.Z.; Ma, C.L.; Wang, X.C.; Jin, J.Q.; Wang, X.M.; Chen, L. Construction of a SSR-based genetic map and identification of QTLs for catechins content in tea plant (Camellia sinensis). PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Fumiya, T.; Kazumi, F.; Sakura, O.M.; Nobuo, Y.; Tomomi, U.; Izumi, K.; Hiroyuki, F.; Junichi, T. Construction of a high-density reference linkage map of tea (Camellia sinensis). Breed. Sci. 2012, 62, 263–273. [Google Scholar]
- Tan, L.Q.; Wang, L.Y.; Xu, L.Y.; Wu, L.Y.; Peng, M.; Zhang, C.C.; Wei, K.; Bai, P.X.; Li, H.L.; Cheng, H.; et al. SSR-based genetic mapping and QTL analysis for timing of spring bud flush, young shoot color, and mature leaf size in tea plant (Camellia sinensis). Tree Genet. Genomes 2016, 12. [Google Scholar] [CrossRef]
- Borevitz, J.O.; Nordborg, M. Update on genomics and natural variation in Arabidopsis. The impact of genomics on the study of natural variation in Arabidopsis. Plant Physiol. 2003, 132, 718–725. [Google Scholar] [CrossRef] [PubMed]
- Han, B.; Huang, X. Sequencing-based genome-wide association study in rice. Curr. Opin. Plant Biol. 2013, 16, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Feng, Q.; Qian, Q.; Zhao, Q.; Wang, L.; Wang, A.; Guan, J.; Fan, D.; Weng, Q.; Huang, T.; et al. High-throughput genotyping by whole-genome resequencing. Genome Res. 2009, 19. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Feng, Q.; Yu, H.; Huang, X.; Zhao, Q.; Xing, Y.; Yu, S.; Han, B.; Zhang, Q. Parent-independent genotyping for constructing an ultrahigh-density linkage map based on population sequencing. Proc. Natl. Acad. Sci. USA 2010, 107, 10578–10583. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hirschhorn, J.N.; Daly, M.J. Genome-wide association studies for common diseases and complex traits. Nat. Rev. Genet. 2005, 6, 95–108. [Google Scholar] [CrossRef] [PubMed]
- Atwell, S.; Huang, Y.S.; Vilhjálmsson, B.J.; Willems, G.; Horton, M.; Li, Y.; Meng, D.Z.; Platt, A.; Tarone, A.M.; Hu, T.T.; et al. Genome-wide association study of 107 phenotypes in Arabidopsis thaliana inbred lines. Nature 2010, 465, 627–631. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Wei, X.; Sang, T.; Zhao, Q.; Feng, Q.; Zhao, Y.; Li, C.; Zhu, C.; Lu, T.; Zhang, Z.; et al. Genome-wide association studies of 14 agronomic traits in rice landraces. Nat. Genet. 2010, 42, 961–967. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Zhao, Y.; Wei, X.; Li, C.; Wang, A.; Zhao, Q.; Li, W.; Guo, Y.; Deng, L.; Zhu, C.; et al. Genome-wide association study of flowering time and grain yield traits in a worldwide collection of rice germplasm. Nat. Genet. 2011, 44, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Jiang, Y.; Wang, Z.; Gou, Z.; Lyu, J.; Li, W.; Yu, Y.; Shu, L.; Zhao, Y.; Ma, Y.; et al. Resequencing 302 wild and cultivated accessions identifies genes related to domestication and improvement in soybean. Nat. Biotechnol. 2015, 33, 408–414. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jia, G.; Huang, X.; Zhi, H.; Zhao, Y.; Zhao, Q.; Li, W.; Chai, Y.; Yang, L.; Liu, K.; Lu, H.; et al. A haplotype map of genomic variations and genome-wide association studies of agronomic traits in foxtail millet (Setaria italica). Nat. Genet. 2013, 45, 957–961. [Google Scholar] [CrossRef] [PubMed]
- Qi, J.; Liu, X.; Shen, D.; Miao, H.; Xie, B.; Li, X.; Zeng, P.; Wang, S.; Shang, Y.; Gu, X.; et al. A genomic variation map provides insights into the genetic basis of cucumber domestication and diversity. Nat. Genet. 2013, 45, 1510–1515. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.; Zhu, G.; Zhang, J.; Xu, X.; Yu, Q.; Zheng, Z.; Zhang, Z.; Lun, Y.; Li, S.; Wang, X.; et al. Genomic analyses provide insights into the history of tomato breeding. Nat. Genet. 2014, 46, 1220–1226. [Google Scholar] [CrossRef] [PubMed]
- Fang, L.; Wang, Q.; Hu, Y.; Jia, Y.; Chen, J.; Liu, B.; Zhang, Z.; Guan, X.; Chen, S.; Zhou, B.; et al. Genomic analyses in cotton identify signatures of selection and loci associated with fiber quality and yield traits. Nat. Genet. 2017, 49, 1089–1098. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; Huang, G.; He, S.; Yang, Z.; Sun, G.; Ma, X.; Li, N.; Zhang, X.; Sun, J.; Liu, M.; et al. Resequencing of 243 diploid cotton accessions based on an updated A genome identifies the genetic basis of key agronomic traits. Nat. Genet. 2018, 50, 796–802. [Google Scholar] [CrossRef] [PubMed]
- Evans, L.M.; Slavov, G.T.; Rodgers-Melnick, E.; Martin, J.; Ranjan, P.; Muchero, W.; Brunner, A.M.; Schackwitz, W.; Gunter, L.; Chen, J.G.; et al. Population genomics of Populus trichocarpa identifies signatures of selection and adaptive trait associations. Nat. Genet. 2014, 46, 1089–1096. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.M.; Sul, J.H.; Service, S.K.; Zaitlen, N.A.; Kong, S.Y.; Freimer, N.B.; Sabatti, C.; Eskin, E. Variance component model to account for sample structure in genome-wide association studies. Nat, Genet. 2010, 42, 348–354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Korte, A.; Vilhjálmsson, B.J.; Segura, V.; Platt, A.; Long, Q.; Nordborg, M. A mixed-model approach for genome-wide association studies of correlated traits in structured populations. Nat. Genet. 2012, 44, 1066–1071. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Segura, V.; Vilhjálmsson, B.J.; Platt, A.; Korte, A.; Seren, Ü.; Long, Q.; Nordborg, M. An efficient multi-locus mixed-model approach for genome-wide association studies in structured populations. Nat. Genet. 2013, 44, 825–830. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Pressoir, G.; Briggs, W.H.; Vroh, B.I.; Yamasaki, M.; Doebley, J.F.; Mcmullen, M.D.; Gaut, B.S.; Nielsen, D.M.; Holland, J.B.; et al. A unified mixed-model method for association mapping that accounts for multiple levels of relatedness. Nat. Genet. 2006, 38, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Ersoz, E.; Lai, C.Q.; Todhunter, R.J.; Tiwari, H.K.; Gore, M.A.; Bradbury, P.J.; Yu, J.; Arnett, D.K.; Ordovas, J.M.; et al. Mixed linear model approach adapted for genome-wide association studies. Nat. Genet. 2010, 42, 355–360. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cui, L.; Yao, S.; Dai, X.; Yin, Q.; Liu, Y.; Jiang, X.; Wu, Y.; Qian, Y.; Pang, Y.; Gao, L.; et al. Identification of UDP-glycosyltransferases involved in the biosynthesis of astringent taste compounds in tea (Camellia sinensis). J. Exp. Bot. 2016, 67, 2285–2297. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Wang, X.; Guo, L.; Xu, Q.; Zhao, S.; Li, F.; Yan, X.; Liu, S.; Wei, C. Characterization and alternative splicing profiles of lipoxygenase gene family in tea plant (Camellia sinensis). Plant Cell Physiol. 2018, 59, 1765–1781. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Han, B. Differential expression pattern of an acidic 9/13-lipoxygenase in flower opening and senescence and in leaf response to phloem feeders in the tea plant. BMC Plant Biol. 2010, 10. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.K.; Fan, Z.Q.; Li, X.L.; Li, J.Y.; Yin, H.F. The APETALA1 and FRUITFUL homologs in Camellia japonica and their roles in double flower domestication. Mol. Breed. 2014, 33, 821–834. [Google Scholar] [CrossRef]
- Sun, Y.; Fan, Z.; Li, X.; Liu, Z.; Li, J.; Yin, H. Distinct double flower varieties in Camellia japonica exhibit both expansion and contraction of C-class gene expression. BMC Plant Biol. 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Liljegren, S.J.; Ditta, G.S.; Eshed, Y.; Savidge, B.; Bowman, J.L.; Yanofsky, M.F. SHATTERPROOF MADS-box genes control seed dispersal in Arabidopsis. Nature 2000, 404, 766–770. [Google Scholar] [CrossRef] [PubMed]
- Powell, A.L.; Nguyen, C.V.; Hill, T.; Cheng, K.L.; Figueroa-Balderas, R.; Aktas, H.; Ashrafi, H.; Pons, C.; Fernandez-Munoz, R.; Vicente, A.; et al. Uniform ripening encodes a golden 2-like transcription factor regulating tomato fruit chloroplast development. Science 2012, 336, 1711–1715. [Google Scholar] [CrossRef] [PubMed]
- Vrebalov, J.; Ruezinsky, D.; Padmanabhan, V.; White, R.; Medrano, D.; Drake, R.; Schuch, W.; Giovannoni, J. A MADS-box gene necessary for fruit ripening at the tomato Ripening-inhibitor (Rin) locus. Science 2002, 296, 343–346. [Google Scholar] [CrossRef] [PubMed]
- Dardick, C.; Callahan, A.M. Evolution of the fruit endocarp: Molecular mechanisms underlying adaptations in seed protection and dispersal strategies. Front. Plant Sci. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Pabon-Mora, N.; Wong, G.K.; Ambrose, B.A. Evolution of fruit development genes in flowering plants. Front. Plant Sci. 2014, 5. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.H.; Xu, B.Y.; Zhang, J.; Jin, Z.Q. The interaction of MADS-box transcription factors and manipulating fruit development and ripening. Hereditas 2010, 32, 893–902. [Google Scholar] [PubMed]
- Ferrandiz, C.; Fourquin, C. Role of the FUL-SHP network in the evolution of fruit morphology and function. J. Exp. Bot. 2014, 65, 4505–4513. [Google Scholar] [CrossRef] [PubMed]
- Bemer, M.; Karlova, R.; Ballester, A.R.; Tikunov, Y.M.; Bovy, A.G.; Wolters-Arts, M.; Rossetto Pde, B.; Angenent, G.C.; de Maagd, R.A. The tomato FRUITFULL homologs TDR4/FUL1 and MBP7/FUL2 regulate ethylene-independent aspects of fruit ripening. Plant Cell 2012, 24, 4437–4451. [Google Scholar] [CrossRef] [PubMed]
- Albert, N.W.; Lewis, D.H.; Zhang, H.; Irving, L.J.; Jameson, P.E.; Davies, K.M. Light-induced vegetative anthocyanin pigmentation in Petunia. J. Exp. Bot. 2009, 60, 2191–2202. [Google Scholar] [CrossRef] [PubMed]
- Groszmann, M.; Paicu, T.; Alvarez, J.P.; Swain, S.M.; Smyth, D.R. SPATULA and ALCATRAZ, are partially redundant, functionally diverging bHLH genes required for Arabidopsis gynoecium and fruit development. Plant J. 2011, 68, 816–829. [Google Scholar] [CrossRef] [PubMed]
- Rajani, S.; Sundaresan, V. The Arabidopsis myc/bHLH gene ALCATRAZ enables cell separation in fruit dehiscence. Curr. Biol. 2001, 11, 1914–1922. [Google Scholar] [CrossRef]
- Mitsuda, N.; Iwase, A.; Yamamoto, H.; Yoshida, M.; Seki, M.; Shinozaki, K.; Ohme-Takagi, M. NAC transcription factors, NST1 and NST3, are key regulators of the formation of secondary walls in woody tissues of Arabidopsis. Plant Cell 2007, 19, 270–280. [Google Scholar] [CrossRef] [PubMed]
- Shan, W.; Kuang, J.F.; Chen, L.; Xie, H.; Peng, H.H.; Xiao, Y.Y.; Li, X.P.; Chen, W.X.; He, Q.G.; Chen, J.Y.; et al. Molecular characterization of banana NAC transcription factors and their interactions with ethylene signalling component EIL during fruit ripening. J. Exp. Bot. 2012, 63, 5171–5187. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Machemer, K.; Shaiman, O.; Salts, Y.; Shabtai, S.; Sobolev, I.; Belausov, E.; Grotewold, E.; Barg, R. Interplay of MYB factors in differential cell expansion, and consequences for tomato fruit development. Plant J. 2011, 68, 337–350. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vrebalov, J.; Pan, I.L.; Arroyo, A.J.; McQuinn, R.; Chung, M.; Poole, M.; Rose, J.; Seymour, G.; Grandillo, S.; Giovannoni, J.; et al. Fleshy fruit expansion and ripening are regulated by the tomato SHATTERPROOF gene TAGL1. Plant Cell 2009, 21, 3041–3062. [Google Scholar] [CrossRef] [PubMed]
- Garceau, D.C.; Batson, M.K.; Pan, I.L. Variations on a theme in fruit development: The PLE lineage of MADS-box genes in tomato (TAGL1) and other species. Planta 2017, 246, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Pan, I.L.; McQuinn, R.; Giovannoni, J.J.; Irish, V.F. Functional diversification of agamous lineage genes in regulating tomato flower and fruit development. J. Exp. Bot. 2010, 61, 1795–1806. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Huang, W.; Liu, Z.W.; Wang, Y.X.; Zhuang, J. Transcriptome-based analysis of Dof family transcription factors and their responses to abiotic stress in tea plant (Camellia sinensis). Int. J. Genom. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Cao, H.; Qian, W.; Yao, L.; Hao, X.; Li, N.; Yang, Y.; Wang, X. Identification of a novel bZIP transcription factor in Camellia sinensis as a negative regulator of freezing tolerance in transgenic Arabidopsis. Ann. Bot. 2017, 119, 1195–1209. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Zhuang, J.; Zou, Z.; Li, Q.; Xin, H.; Li, X. Overexpression of a Camellia sinensis DREB transcription factor gene (CsDREB) increases salt and drought tolerance in transgenic Arabidopsis thaliana. J. Plant Biol. 2017, 60, 452–461. [Google Scholar] [CrossRef]


| Species | Traits | Methods | Key Pathways & Genes | Reference | Database Accessment (from NCBI) |
|---|---|---|---|---|---|
| C. sinensis | seven tissue types | Transcriptome/Illumina | flavonoid, theanine, and caffeine biosynthesis pathways | [15] | SRX020193, HP701085-HP777243 |
| Responses of Biotic and Abiotic Stress in Camellia Plants | |||||
| C. sinensis | Cold acclimation | 454 GS-FLX | Cold-related genes | [16] | SRA061043, SRX020193 |
| C. sinensis | Same as above | Illumina | AP2/ERF family TFs | [17] | Not found |
| C. sinensis | Leaves with different treatment time of 4 or 38 °C temperature stress | Illumina | WRKY gen family | [18] | Not found |
| C. japonica | mature leaves after 40 d natural low temperature | Illumina | α-linolenic acid and jasmonic acid biosynthesis pathways respond to cold acclimation | [19] | SRP076436 |
| C. sinensis | Drought stress and salt stress young leaves | Illumina | Response to drought stress and salt stress | [20] | PRJEB11522 |
| C. sinensis | Germination seed of different dehydrate treatment | Illumina | Mechanism of seed dehydration sensitivity | [21] | SRP096975 |
| C. sinensis | (NH4)2SO4 treatment buds, leaves and root | Illumina | Nitrogen utilization genes | [22] | SRP077092 |
| C. sinensis | Pollen tubes at 25 °C and 4 °C or with NO treatment | Illumina | Potential mechanisms of the participation of NO in pollen tube responses to low temperature | [23] | SRR3270364, SRR3270376, SRR3270829, SRR3270928, SRR3270974, SRR3270993, SRR3270997, SRR3271001, SRR3271002 |
| C. sinensis | Leaf tissues of blister blight transition | Illumina | Blister Blight defense | [24] | SRP067826, PRJNA306068 |
| C. sinensis | Insect feeding treatment | Illumina | Defense response to insect (Ectropis. oblique) | [25] | SRX998353, SRX1543038 |
| Transcriptomic Analyses in the Control of Secondary Metabolism in Camellia | |||||
| C. sinensis | 13 different tissue samples from various organs and developmental stage | Illumina | Secondary metabolite biosynthesis pathways | [26] | SRR1053623, SRR1051214, SRR1054007, SRR1055110, SRR1055182, SRR1054086, SRR1054152, SRR1055108, SRR1055109, SRR1055932, SRR1055933, SRR1055934, SRR1055944 |
| C. taliensis | Tender shoots, young leaves, flower buds, and flowers | Illumina | Secondary metabolic biosynthesis pathways | [27] | PRJNA274899 |
| C. sinensis | Buds, 2nd leaves, mature leaves and young roots | Illumina | Catechins metabolic pathways | [28] | Not found |
| C. sinensis | Leaf tissues of four tea plant cultivars | Illumina | Catechins biosynthesis pathways | [29] | Not found |
| C. asssamica | Leaf at the purple and green stages | Illumina | Anthocyanin biosynthesis pathway | [30] | Not found |
| C. nitidissima | Floral buds at five different developmental stages | Illumina | Carotenoids and flavonols glucosides biosynthesis pathways | [31] | SRP112181 |
| C.nitidissima, C. chuongtsoensis | Young shoot tip or leaves | Illumina | Floral pigmentation and flowering timing | [32] | PRJNA389977, PRJNA400646 |
| Transcriptomics Studies Related to Floral Patterning, Flowering Timing and Bud Dormancy | |||||
| C. japonica | Double flower development | Illumina | ABCE genes, miR156, and targeted squamosa promoter binding protein-likes (SPLs) | [33] | |
| C. azalea | Floral buds | Illumina | Conserved and lineage-specific miRNA | [34] | PRJNA257896, SRP045386 |
| C. sinensis | Axillary buds | Illumina | Bud dormancy regulation mechanism | [35] | SRR5040773, SRR5040784 |
| C. sinensis | Bud tissues of different developmental stages | ABI PRISM 3730 | Dormancy-related genes | [36] | HM003230–HM003378, GW690681–GW691037 |
| C. azalea | Three stages of floral bud development: | Illumina | Floral dormancy-associated MADS-box genes | [37] | PRJNA257896, SRP045386 |
| C. sinensis | Three opening stages of flowers | Illumina | WRKY, ERF, bHLH, MYB and MADS-box family relate to flower development | [38] | SRR5487532, SRR5487531, SRR5487530, SRR5487529, SRR5487528, SRR5487527, |
| C. sinensis | Two and a buds in July and December | Illumina | Regulatory mechanism of non-deciduous habit in winter | [39] | Not found |
| C. sinensis | Shading leaves (yellow leaf phenotype) | Illumina | Chloroplast development, chlorophyll biosynthesis pathway | [40] | SRX1078570 |
| C. sinensis | Adventitious roots from IBA treatment cuttings | Illumina | Potential mechanisms involved in adventitious root formation | [41,42] | PRJNA240661, JK990996-991074 |
| Transcriptomics in oil Camellia Plants | |||||
| C. oleifera | Four tissues | 454 GS-FLX | Lipid metabolism | [43] | SRR1472854, SRR1472847, SRR1472843, SRR1472842, GBHI00000000 |
| C. oleifera | Drought treatment leaves | Illumina | Drought stress genes | [44] | SRP094080 |
| C. oleifera | Seed | Illumina | Oil content and fatty acid composition | [45] | SRP111395 |
| C. oleifera | Leaves at different elevations of Lu Mountain and Jinggang Mountain | Illumina | Cold acclimation genes | [46] | SRR2146977, SRR2146978, SRR2146979, SRR2146980, SRR2146973, SRR2146974, SRR2146975, SRR2146976 |
| C. chekiangoleosai | Seeds, flowers and leaves | 454 GS FLX | Anthocyanin biosynthesis pathway genes | [47] | Not found |
| C. oleifera, C. meiocarpa | Mature seed of different moisture content | Illumina | Fatty acid biosynthesis and accumulation pathway | [48] | Not found |
| C. oleifera, C. chekiangoleosa, C. brevistyla | Flower buds | 454 GS FLX | Secondary metabolites pathway, CHS gene, FAD2 gene | [49] | HQ704701.1 |
| Markers Development Based on RNA-sequencing (RNA-seq) | |||||
| C. sinensis | Three developmental growth stages leaves | 454 GS FLX | Plant growth, development, secondary metabolite, and (expressed sequence tag–simple sequence repeats (EST-SSR) markers | [50] | SRA052793, KA279444–KA304315, HP701085–HP777243 |
| C. sinensis | Different flower organizations at the big bud stage | Illumina | SSR Markers, SSR-based linkage map | [51] | SRA053025, GAAC01000001–GAAC01052919 |
| C. flavida, C. achrysantha | Flower buds | Illumina | SSR markers | [52] | Not found |
| C. oleifera | Lipid synthesis phase seed | Illumina | SSR markers | [53] | Not found |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yan, C.; Lin, P.; Lyu, T.; Hu, Z.; Fan, Z.; Li, X.; Yao, X.; Li, J.; Yin, H. Unraveling the Roles of Regulatory Genes during Domestication of Cultivated Camellia: Evidence and Insights from Comparative and Evolutionary Genomics. Genes 2018, 9, 488. https://doi.org/10.3390/genes9100488
Yan C, Lin P, Lyu T, Hu Z, Fan Z, Li X, Yao X, Li J, Yin H. Unraveling the Roles of Regulatory Genes during Domestication of Cultivated Camellia: Evidence and Insights from Comparative and Evolutionary Genomics. Genes. 2018; 9(10):488. https://doi.org/10.3390/genes9100488
Chicago/Turabian StyleYan, Chao, Ping Lin, Tao Lyu, Zhikang Hu, Zhengqi Fan, Xinlei Li, Xiaohua Yao, Jiyuan Li, and Hengfu Yin. 2018. "Unraveling the Roles of Regulatory Genes during Domestication of Cultivated Camellia: Evidence and Insights from Comparative and Evolutionary Genomics" Genes 9, no. 10: 488. https://doi.org/10.3390/genes9100488
APA StyleYan, C., Lin, P., Lyu, T., Hu, Z., Fan, Z., Li, X., Yao, X., Li, J., & Yin, H. (2018). Unraveling the Roles of Regulatory Genes during Domestication of Cultivated Camellia: Evidence and Insights from Comparative and Evolutionary Genomics. Genes, 9(10), 488. https://doi.org/10.3390/genes9100488





