Heterotrophic Protists in Hypersaline Microbial Mats and Deep Hypersaline Basin Water Columns
Abstract
:1. Introduction
2. Views of Eukaryotic Diversity in Selected Hypersaline Habitats
2.1. Microbialites in Hypersaline Shark Bay, Australia

| Sample | Coordinates | Water Depth (m) | Total Salinity (PSU) | Oxygen (mL/L) | |
|---|---|---|---|---|---|
| Discovery Interface 2 | 35°19'N | 21°41'E | 3,580 | 70 1 | 0.50 |
| Thetis Interface 3 | 34°40'N | 22°08'E | 3,259 | 80 | 0.68 |
| Thetis Brine 3 | 34°40'N | 22°08'E | 3,415 | 340 | 0 |
| Bannock Brine 2 | 34°17'N | 20°00'E | 3,790 | 280 | 0 |
| Bannock Interface 2 | 34°17'N | 20°00'E | 3,300 | 246 | 0.50 |
| Atalante Upper Interface 5 | 35°18’N | 21°23'E | 3,499 | 39 | 0.44 |
| Atalante Lower Interface 5 | 35°18'N | 21°23'E | 3,501 | 365 | 0 |
| Urania Interface 6 | 35°13'N | 21°28'E | 3,467 | 63 | 1.22 |
| Hamelin Pool microbialites 7 | 26°15'S | 114°14'E | 0–3 | 66–72 | supersaturated at 0–1 cm, 0 at 1–2 cm |
| Guerrero Negro saltern mats 4 | 27°41'N | 113°55'W | 1–2 | 90 | na |
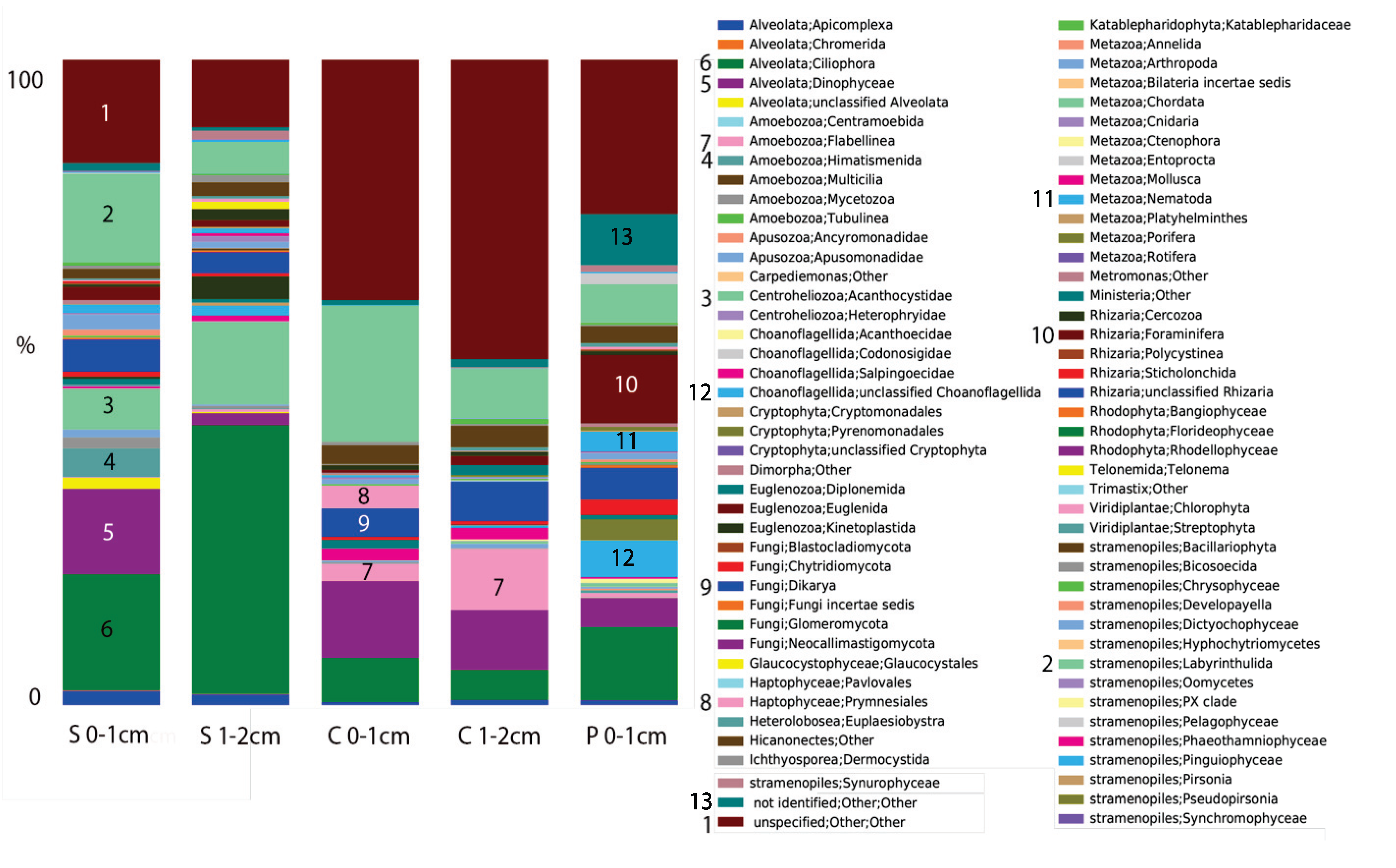
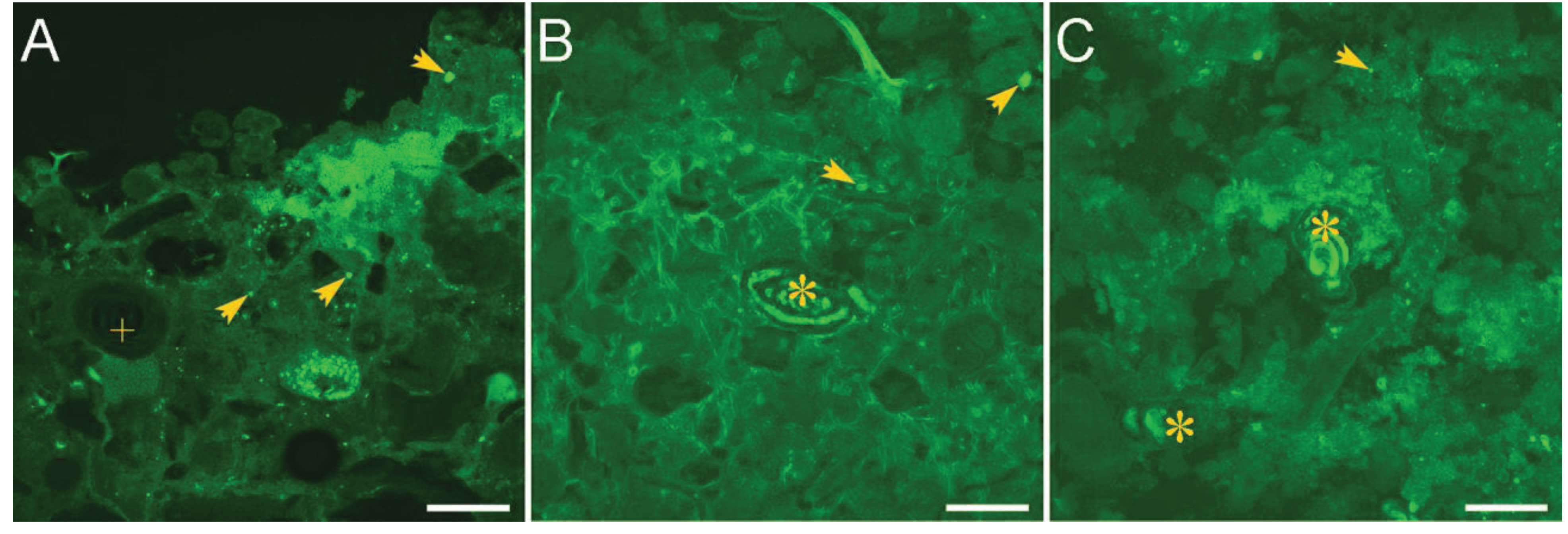
2.2. Microbial Mats in Hypersaline Solar Salterns
2.3. Deep Hypersaline Anoxic Basins in the Eastern Mediterranean Sea (DHABs)

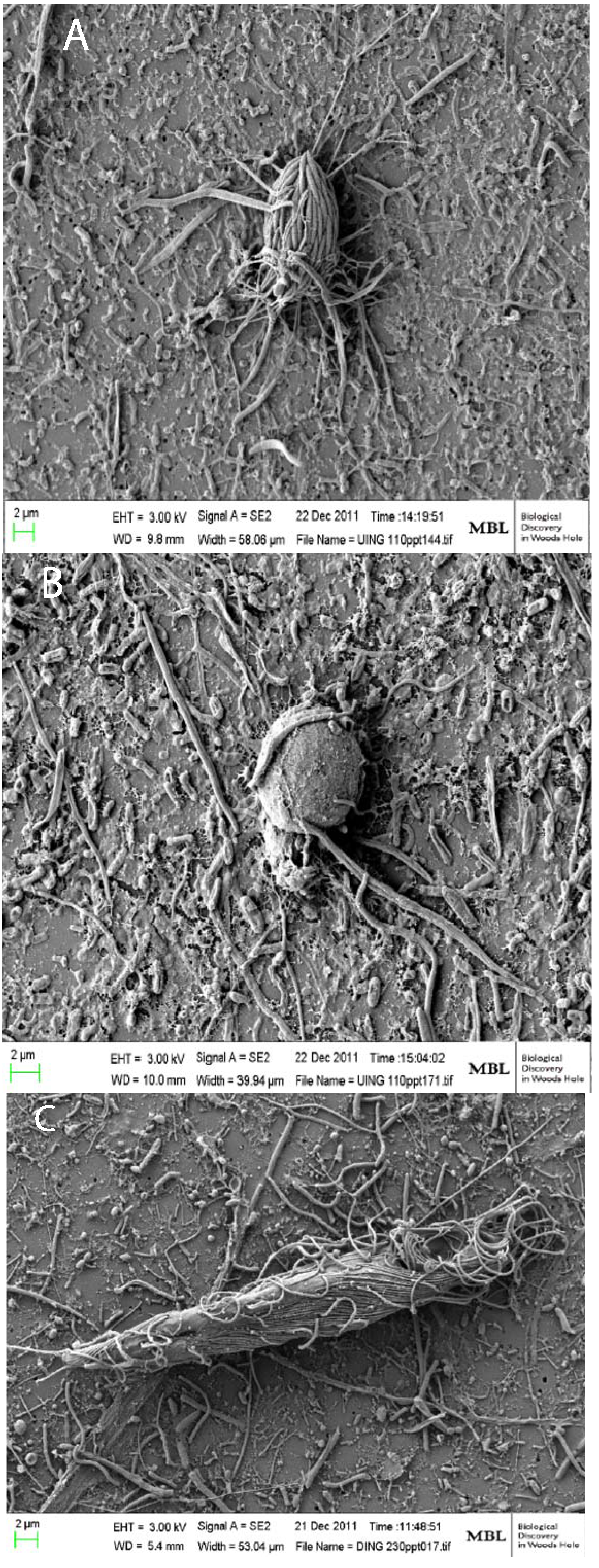
3. Commonalities between Protist Communities in DHABs and Hypersaline Shallow Water Mat Communities (Hamelin Pool and Guerrero Negro)
Acknowledgments
References
- Litchfield, C.D. Survival strategies for microorganisms in hypersaline environments and their relevance to life on early Mars. Meteorites Planet Sci. 1998, 33, 813–819. [Google Scholar] [CrossRef]
- Baxter, B.K.; Litchfield, C.D.; Sowers, K.; Griffith, J.D.; DasSarma, P.A.; DasSarma, S. Microbial Diversity of Great Salt Lake. In Adaptation to Life in High Salt Concentrations in Archaea, Bacteria, and Eukarya; Gunde-Cimerman, N., Oren, A., Plemenitas, A., Eds.; Springer: Dordrecht, The Netherlands, 2005; Volume 9, pp. 9–25. [Google Scholar]
- Brock, T.D.; Madigan, M.T.; Martinko, J.M. Biology of Microorganisms, 7th ed.; Benjamin Cummings: San Francisco, CA, USA, 1994; p. 909. [Google Scholar]
- Galinsky, E.A. Compatible solutes of halophilic bacteria: Molecular principles, water-soluble interactions, stress protection. Experientia 1993, 49, 487–496. [Google Scholar] [CrossRef]
- Oren, A. Halophilic Microorganisms and Their Environments; Kluwer Academic: Dordrecht, The Netherlands, 2003; p. 575. [Google Scholar]
- Ruinen, J.; Raas Becking, L.G.M. Rhizopods living in unusual environments. Arch. Néerl Zool. 1938, 3, 183–198. [Google Scholar]
- Volcani, B.E. The Microorganisms of the Dead Sea. In Papers Collected to Commemorate the 70th Anniversary of Dr. Chaim Weizmann; Daniel Sieff Research Institute: Rehovoth, Israel, 1944; pp. 71–85. [Google Scholar]
- Namyslowski, B. Adaptation of zooflagellates to higher salinity. Biol. Vnutr. Vod. Inform Bull. 1913, 61, 21–24. [Google Scholar]
- Kirby, H. Two protozoa from brine. Trans. Am. Microsc. Soc. 1932, 51, 8–15. [Google Scholar] [CrossRef]
- Ruinen, J. Notizen über Salzflagellaten. II Über die Verbereitung der Salzflagellaten. Arch. Protistenkd. 1938, 90, 210–258. [Google Scholar]
- Patterson, D.J. ; Simpson, A.G. Heterotrophic flagellates from coastal marine and hypersaline sediments in Western Australia. Eur. J. Protistol. 1996, 32, 423–448. [Google Scholar] [CrossRef]
- Post, F.J.; Borowitzka, L.J.; Borowitzka, M.A.; Mackay, B.; Moulton, T. The protozoa of a Western Australian hypersaline lagoon. Hydrobiologia 1983, 105, 95–113. [Google Scholar] [CrossRef]
- Hauer, G.; Rogerson, A. Heterotrophic Protozoa from Hypersaline Environments. In Adaptation to Life at High Salt Concentrations in Archaea, Bacteria, and Eukarya; Gunde-Cimerman, N., Oren, A., Plemenitaš, A., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 519–540. [Google Scholar]
- Por, F. A classification of hypersaline waters, based on trophic criteria. Mar. Ecol. 1980, 1, 121–131. [Google Scholar] [CrossRef]
- Ramos-Cormenzana, A. Halophilic Organisms and Their Environment. In General and Applied Aspects of Halophilic Microorganisms; Rodriguez-Valera, F., Ed.; Plenum Press: New York, NY, USA, 1991; pp. 15–24. [Google Scholar]
- Oren, A. Diversity of halophilic microorganisms: Environments, phylogeny, physiology, and applications. J. Indust. Microbiol. Biotechnol. 2002, 28, 56–63. [Google Scholar]
- Pedros-Alió, C.; Calderón-Paz, J.I.; MacLean, M.H.; Medina, G.; Marrasé, C.; Gasol, J.M.; Guixa-Boixereu, N. The microbial food web along salinity gradients. FEMS Microbiol. Ecol. 2000, 32, 143–155. [Google Scholar]
- Elloumi, J.; Carrias, J.-F.; Ayadi, H.; Sime-Ngando, T.; Boukhris, M.; Bouain, A. Composition and distribution of planktonic ciliates from ponds of different salinity in the solar saltwork of Sfax, Tunisia. Estuar. Coast. Shelf Sci. 2006, 67, 21–29. [Google Scholar] [CrossRef]
- Finlay, B.J. Physiological ecology of free-living protozoa. Adv. Microbiol. Ecol. 1990, 11, 1–34. [Google Scholar] [CrossRef]
- McLachlan, J. The culture of Dunaliella tertiolecta Butcher—A euryhaline organism. Can. J. Microbiol. 1960, 6, 367–379. [Google Scholar] [CrossRef]
- Al-Qassab, S.; Lee, W.J.; Muray, S.; Simpson, A.G.B.; Patterson, D.J. Flagellates from stramatolites and surrounding sediments in Shark Bay, Western Australia. Acta Protozool. 2002, 41, 91–144. [Google Scholar]
- Park, J.S.; Simpson, A.G.B. Characterization of halotolerant Bicosoecida and Placididea (Stramenopila) that are distinct from marine forms, and the phylogenetic pattern of salinity preference in hetertrophic stramenopiles. Environ. Microbiol. 2010, 12, 1173–1184. [Google Scholar] [CrossRef]
- Park, J.S. Effects of different ion compositions on growth of obligately halophillic protozoan Halocafeteria seosinensis. Extremophiles 2012, 16, 161–164. [Google Scholar] [CrossRef]
- Fenchel, T. Ecology of heterotrophic microflagellates. IV. Quantitative occurrence and importance as bacterial consumers. Mar. Ecol. Prog. Ser. 1982, 9, 35–42. [Google Scholar] [CrossRef]
- Alexander, E.; Stock, A.; Breiner, H.W.; Behnke, A.; Bunge, J.; Yakimov, M.M.; Stoeck, T. Microbial eukaryotes in the hypersaline anoxic L'Atalante deep-sea basin. Environ. Microbiol. 2009, 11, 360–381. [Google Scholar] [CrossRef]
- Ayadi, H.; Toumi, N.; Abid, O.; Medhioub, K.; Hammami, M.; Sime-Ngando, T.; Amblard, C.; Sargos, D. Qualitative and quantitative study of phyto- and zooplankton communities in the saline ponds of Sfax, Tunisia. Revue Des. Sci. L’Eau 2002, 15, 123–135. [Google Scholar]
- Laybourn-Parry, J.; Quayle, W.; Henshaw, T. The biology and evolution of Antarctic saline lakes in relation to salinity and trophy. Polar. Biol. 2002, 25, 542–552. [Google Scholar] [CrossRef]
- Feazel, L.M.; Spear, J.R.; Berger, A.B.; Harris, J.K.; Frank, D.N.; Ley, R.E.; Pace, N.R. Eucaryotic diversity in a hypersaline microbial mat. Appl. Environ. Microbiol. 2007, 74, 329–332. [Google Scholar]
- Cho, B.C.; Park, J.S.; Xu, K.; Choi, J.K. Morphology and molecular phylogeny of Trimyema koreanum n. sp., a ciliate from the hypersaline water of a solar saltern. J. Eukaryot. Microbiol. 2008, 55, 417–426. [Google Scholar] [CrossRef]
- Park, J.S.; Cho, B.C.; Simpson, A.G.B. Halocafeteria seoinensis gen. et sp. nov. (Bicosoecida), a halophilic bacterivorous nanoflagellate isolated from a solar saltern. Extremophiles 10, 493–504. [CrossRef]
- Park, J.S.; Simpson, A.G.B.; Brown, S.; Cho, B.C. Ultrastructure and molecular phylogeny of two heterolobosean amoebae, Euplaesiobystra hypersalinica gen. et sp. nov. and Tulamoeba peronaphora gen. et sp. nov., isolated from an extremely hypersaline habitat. Protist 2009, 160, 265–283. [Google Scholar] [CrossRef]
- Park, J.S.; Simpson, A.G.B.; Lee, W.J.; Cho, B.C. Ultrastructure and phylogenetic placement within Heterolobosea of the previously unclassified, extremely halophilic heterotrophic flagellate Pleurostomum flabellatum (Ruinen 1938). Protist 2007, 158, 397–413. [Google Scholar] [CrossRef]
- Edgcomb, V.P.; Orsi, W.; Breiner, H.-W.; Stock, A.; Filker, S.; Yakimov, M.M.; Stoeck, T. Novel kinetoplastids associated with hypersaline anoxic lakes in the Eastern Mediterranean deep-sea. Deep Sea Res. 2011b, 58, 1040–1048. [Google Scholar] [CrossRef]
- Edgcomb, V.P.; Orsi, W.; Taylor, G.T.; Vdacny, P.; Taylor, C.; Suarez, P.; Epstein, S. Accessing marine protists from the anoxic Cariaco Basin. ISME J. 2011, 5, 1237–1241. [Google Scholar] [CrossRef]
- Jahnert, R.L.; Collins, L.B. Significance of subtidal microbial deposits in Shark Bay, Australia. Mar. Geol. 2011, 286, 106–111. [Google Scholar] [CrossRef]
- Dupraz, C.; Visscher, P.T. Microbial lithification in marine stromatolites and hypersaline mats. Trends Microbiol. 2005, 13, 429–438. [Google Scholar] [CrossRef]
- Allwood, A.C.; Walter, M.R.; Kamber, B.S.; Marshall, C.P.; Burch, I.W. Stromatolite reef from the Early Archaean era of Australia. Nature 2006, 441, 714–718. [Google Scholar] [CrossRef]
- Grotzinger, J.P.; Knoll, A.H. Stromatolites in Precambrian carbonates: Evolutionary mileposts or environmental dipsticks? Ann. Rev. Earth Planet Sci. 1999, 27, 313–358. [Google Scholar] [CrossRef]
- Tong, S.M. Heterotrophic flagellates from the water column in Shark Bay, Western Australia. Mar. Biol. 1997, 128, 517–536. [Google Scholar] [CrossRef]
- Papineau, D.; Walker, J.J.; Mojzsis, S.J.; Pace, N.R. Composition and structure of microbial communities from stromatolites of Hamelin Pool in Shark Bay, Western Australia. Appl. Environ. Microbiol. 2005, 71, 4822–4832. [Google Scholar] [CrossRef]
- Bernhard, J.M.; Edgcomb, V.P.; Visscher, P.T.; McIntyre-Wressnig, A.; Summons, R.E.; Bouxsein, M.; Louis, L.; Jeglinski, M. Microbialites at Highborne Cay, Bahamas: insights on foraminiferal inhabitants and influence on their microfabric. Proc. Natl. Acad. Sci USA 2013, in press. [Google Scholar]
- Logan, B.W. Cryptozoon and associate stromatolites from the Recent, Shark Bay, Western Australia. J. Geol. 1961, 69, 517–533. [Google Scholar]
- Stoeck, T.; Bass, D.; Nebel, M.; Christen, R.; Jones, M.D.; Breiner, H.W.; Richards, T.A. Multiple marker parallel tag environmental DNA sequencing reveals a highly complex eukaryotic community in marine anoxic water. Mol. Ecol. 2010, 19, 21–31. [Google Scholar] [CrossRef]
- Pawlowski, J. Introduction to the molecular systematics of foraminifera. Micropaleontology 2000, 46, 1–12. [Google Scholar]
- Caporaso, J.G.; Kuczynski, J.; Strombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Gonzalez Pena, A.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Meth. 2010, 7, 335–336. [Google Scholar] [CrossRef]
- Nebel, M.E.; Wild, S.; Holzhauser, M.; Reitzig, R.; Sperber, M.; Stoeck, T. Jaguc—A software package for environmental diversity analyses. J. Bioinf. Comp. Biol. 2011, 9, 749–773. [Google Scholar] [CrossRef]
- Edgcomb, V.; Orsi, W.; Leslin, C.; Epstein, S.S.; Bunge, J.; Jeon, S.; Yakimov, M.M.; Behnke, A.; Stoeck, T. Protistan community patterns within the brine and halocline of deep hypersaline anoxic basins in the eastern Mediterranean Sea. Extremophiles 2009, 13, 151–167. [Google Scholar] [CrossRef]
- Stock, A.; Breiner, H.-W.; Pachiadaki, M.; Edgcomb, V.; Filker, S.; LaCono, V.; Yakimov, M.M.; Stoeck, T. Microbial eukaryote life in the new hypersaline deep-sea basin Thetis. Extremophiles 2011, 16, 21–34. [Google Scholar]
- Bebout, B.M.; Carpenter, S.P.; Des Marais, D.J.; Discipulo, M.; Embaye, T.; Garcia-Pichel, F.; Hoehler, T.M.; Hogan, M.; Jahnke, L.L.; Keller, R.M.; et al. Long-term manipulations of intact microbial mat communities in a greenhouse collaboratory: Simulating earth’s present and past field environments. Astrobiology 2002, 2, 383–402. [Google Scholar] [CrossRef]
- Orsi, W.; Charvet, S.; Bernhard, J.; Edgcomb, V.P. Prevalence of partnerships between bacteria and ciliates in oxygen-depleted marine water columns. Front. Ext. Microbiol. 2012, 3, 341. [Google Scholar]
- Bosak, T.; Lahr, D.J.G.; Pruss, S.B.; Macdonald, F.A.; Gooday, A.J.; Dalton, L.; Matys, E.D. Possible early foraminiferans in post-Sturtian (716–635 Ma) cap carbonates. Geology 2012, 40, 67–70. [Google Scholar] [CrossRef]
- Bernhard, J.M.; Visscher, P.T.; Bowser, S.S. Submillimeter life positions of bacteria, protists, and position="float" in laminated sediments of the Santa Barbara Basin. Limnol. Oceanogr. 2003, 48, 813–828. [Google Scholar] [CrossRef]
- Cita, M.B. Exhumation of Messinian evaporites in the deep-sea and creation of deep anoxic brine filled collapsed basins. Sed. Geol 2006, 188–189, 357–378. [Google Scholar] [CrossRef]
- Danovaro, R.; Dell'Anno, A.; Pusceddu, A.; Gambi, C.; Heiner, I.; Kristensen, R.M. The first metazoa living in permanently anoxic conditions. BMC Biol. 2010, 8, 30. [Google Scholar] [CrossRef] [Green Version]
- Filker, S.; Stock, A.; Breiner, H.W.; Edgcomb, V.P.; Orsi, W.; Yakimov, M.M.; Stoeck, T. Environmental selection of protistan communities in hypersaline anoxic deep-sea basins, Eastern Mediterranean Sea. Microbiologyopen 2013, 2, 54–63. [Google Scholar] [CrossRef]
- Van der Wielen, P.W.; Bolhuis, H.; Borin, S.; Daffonchio, D.; Corselli, C.; Giuliano, L.; D’Auria, G.; de Lange, G.J.; Huebner, A.; Varnavas, S.P.; et al. The enigma of prokaryotic life in deep hypersaline anoxic basins. Science 2005, 307, 121–123. [Google Scholar] [CrossRef]
- Daffonchio, D.; Borin, S.; Brusa, T.; Brusetti, L.; van der Wielen, P.W.; Bolhuis, H.; Yakimov, M.M.; D’Auria, G.; Giuliano, L.; Marty, D.; et al. Stratified prokaryote network in the oxic-anoxic transition of a deep-sea halocline. Nature 2006, 440, 203–207. [Google Scholar] [CrossRef]
- Hallsworth, J.E.; Yakimov, M.M.; Golyshin, P.N.; Gillion, J.L.; D’Auria, G.; de Lima Alves, F.; La Cono, V.; Genovese, M.; McKew, B.A.; Hayes, S.L.; et al. Limits of life in MgCl2-containing environments: Chaotropicity defines the window. Environ. Microbiol. 2007, 9, 801–813. [Google Scholar] [CrossRef]
- Van der Wielen, P.W.; Heijs, S.K. Sulfate-reducing prokaryotic communities in two deep hypersaline anoxic basins in the Eastern Mediterranean deep sea. Environ. Microbiol. 2007, 9, 1335–1340. [Google Scholar] [CrossRef]
- Yakimov, M.M.; Giuliano, L.; Cappello, S.; Denaro, R.; Golyshin, P.N. Microbial community of a hydrothermal mud vent underneath the deep-sea anoxic brine lake Urania (eastern Mediterranean). Orig. Life Evol. Biosph. 2007, 37, 177–188. [Google Scholar] [CrossRef]
- Bernhard, J.M.; Buck, K.R.; Farmer, M.A.; Bowser, S.S. The Santa Barbara Basin is a symbiosis oasis. Nature 2000, 403, 77–80. [Google Scholar] [CrossRef]
- Taylor, G.T.; Scranton, M.L.; Iabichella, M.; Ho, T.-Y.; Thunell, R.C.; Muller-Karger, F.; Varela, R. Chemoautotrophy in the redox transition zone of the Cariaco Basin: A significant midwater source of organic carbon production. Limnol. Oceanogr. 2001, 46, 148–163. [Google Scholar] [CrossRef]
- Edgcomb, V.; Orsi, W.; Bunge, J.; Jeon, S.O.; Christen, R.; Leslin, C.; Holder, M.; Taylor, G.T.; Suarez, P.; Varela, R.; et al. Protistan microbial observatory in the Cariaco Basin, Caribbean. I. Pyrosequencing vs. Sanger insights into species richness. ISME J. 2011, 5, 1344–1356. [Google Scholar] [CrossRef]
- Countway, P.D.; Gast, R.J.; Dennett, M.R.; Savai, P.; Rose, J.M.; Caron, D.A. Distinct protistan assemblages characterize the euphotic zone and deep sea (2,500 m) of the western North Atlantic (Sargasso Sea and Gulf Stream). Environ. Microbiol. 2007, 9, 1219–1232. [Google Scholar] [CrossRef]
- Massana, R.; Castresana, J.; Balagué, V.; Guillou, L.; Romari, K.; Groisillier, A.; Valentin, K.; Pedrós-Alió, C. Phylogenetic and ecological analysis of novel marine stramenopiles. Appl. Environ. Microbiol. 2004, 70, 3528–3534. [Google Scholar] [CrossRef]
- Not, F.; Gausling, R.; Azam, F.; Heidelberg, J.F.; Worden, A.Z. Vertical distribution of picoeukaryotic diversity in the Sargasso Sea. Environ. Microbiol. 2007, 9, 1233–1252. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Edgcomb, V.P.; Bernhard, J.M. Heterotrophic Protists in Hypersaline Microbial Mats and Deep Hypersaline Basin Water Columns. Life 2013, 3, 346-362. https://doi.org/10.3390/life3020346
Edgcomb VP, Bernhard JM. Heterotrophic Protists in Hypersaline Microbial Mats and Deep Hypersaline Basin Water Columns. Life. 2013; 3(2):346-362. https://doi.org/10.3390/life3020346
Chicago/Turabian StyleEdgcomb, Virginia P., and Joan M. Bernhard. 2013. "Heterotrophic Protists in Hypersaline Microbial Mats and Deep Hypersaline Basin Water Columns" Life 3, no. 2: 346-362. https://doi.org/10.3390/life3020346




