Long-Term Monitoring of the Antibody Response to a SARS-CoV-2 Infection
Abstract
:1. Introduction
2. Material and Methods
2.1. Group of Patients
2.2. Serum Samples
2.3. Immunoassays
3. Statistical Methods
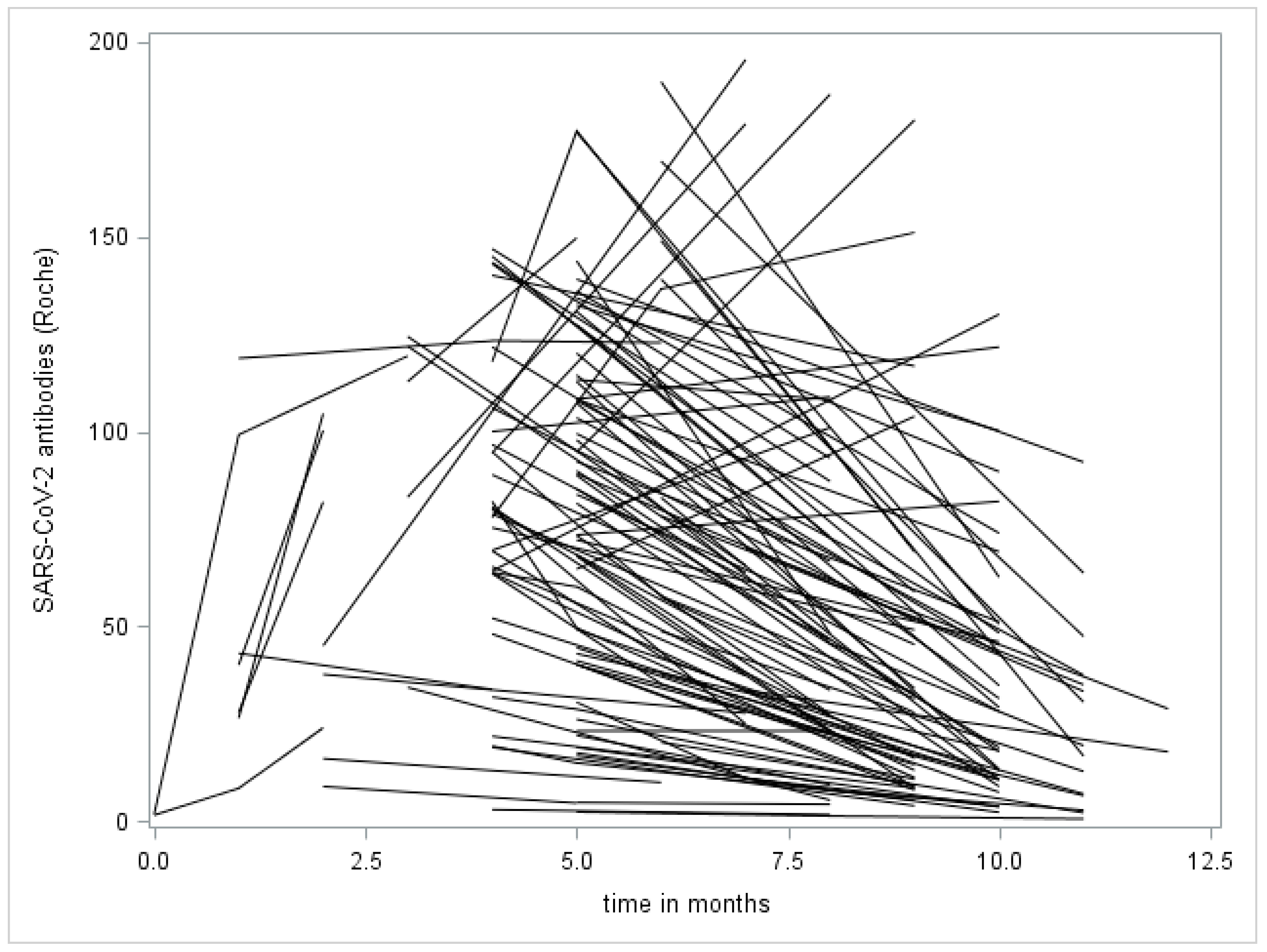
4. Results
5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gozalbo-Rovira, R.; Gimenez, E.; Latorre, V.; Francés-Gómez, C.; Albert, E.; Buesa, J.; Marina, A.; Blasco, M.L.; Signes-Costa, J.; Rodríguez-Díaz, J.; et al. SARS-CoV-2 Antibodies, Serum Inflammatory Biomarkers and Clinical Severity of Hospitalized COVID-19 Patients. J. Clin. Virol. 2020, 131, 104611. [Google Scholar] [CrossRef]
- Gaebler, C.; Wang, Z.; Lorenzi, J.C.C.; Muecksch, F.; Finkin, S.; Tokuyama, M.; Ladinsky, M.; Cho, A.; Jankovic, M.; Schaefer, D.; et al. Evolution of Antibody Immunity to SARS-CoV-2. Nature 2021, 59, 639–644. [Google Scholar] [CrossRef]
- Sekine, T.; Perez-Potti, A.; Rivera-Ballesteros, O.; Strålin, K.; Gorin, J.-B.; Olsson, A.; Llewellyn-Lacey, S.; Kamal, H.; Bogdanovic, G.; Muschiol, S.; et al. Robust T Cell Immunity in Convalescent Individuals with Asymptomatic or Mild COVID-19. Cell 2020, 183, 158–168.e14. [Google Scholar] [CrossRef]
- Kellam, P.; Barclay, W. The Dynamics of Humoral Immune Responses Following SARS-CoV-2 Infection and the Potential for Reinfection. J. Gen. Virol. 2020, 101, 791–797. [Google Scholar] [CrossRef] [PubMed]
- Lega, S.; Naviglio, S.; Volpi, S.; Tommasini, A. Recent Insight into SARS-CoV2 Immunopathology and Rationale for Potential Treatment and Preventive Strategies in COVID-19. Vaccines 2020, 8, 224. [Google Scholar] [CrossRef] [PubMed]
- Tolone, S.; Gambardella, C.; Brusciano, L.; del Genio, G.; Lucido, F.S.; Docimo, L. Telephonic Triage before Surgical Ward Admission and Telemedicine during COVID-19 Outbreak in Italy. Effective and Easy Procedures to Reduce In-Hospital Positivity. Int. J. Surg. 2020, 78, 123–125. [Google Scholar] [CrossRef]
- Post, N.; Eddy, D.; Huntley, C.; van Schalkwyk, M.C.I.; Shrotri, M.; Leeman, D.; Rigby, S.; Williams, S.V.; Bermingham, W.H.; Kellam, P.; et al. Antibody Response to SARS-CoV-2 Infection in Humans: A Systematic Review. PLoS ONE 2020, 15, e0244126. [Google Scholar] [CrossRef] [PubMed]
- Chaudhury, S.; Hutter, J.; Bolton, J.S.; Hakre, S.; Mose, E.; Wooten, A.; O’Connell, W.; Hudak, J.; Krebs, S.J.; Darden, J.M.; et al. Serological Profiles of Pan-Coronavirus-Specific Responses in COVID-19 Patients Using a Multiplexed Electro-Chemiluminescence-Based Testing Platform. PLoS ONE 2021, 16, e0252628. [Google Scholar] [CrossRef]
- Xiao, A.T.; Gao, C.; Zhang, S. Profile of Specific Antibodies to SARS-CoV-2: The First Report. J. Infect. 2020, 81, 147–178. [Google Scholar] [CrossRef]
- Li, K.; Huang, B.; Wu, M.; Zhong, A.; Li, L.; Cai, Y.; Wang, Z.; Wu, L.; Zhu, M.; Li, J.; et al. Dynamic Changes in Anti-SARS-CoV-2 Antibodies during SARS-CoV-2 Infection and Recovery from COVID-19. Nat. Commun. 2020, 11, 6044. [Google Scholar] [CrossRef] [PubMed]
- Isho, B.; Abe, K.T.; Zuo, M.; Jamal, A.J.; Rathod, B.; Wang, J.H.; Li, Z.; Chao, G.; Rojas, O.L.; Bang, Y.M.; et al. Persistence of Serum and Saliva Antibody Responses to SARS-CoV-2 Spike Antigens in COVID-19 Patients. Sci. Immunol. 2020, 5, eabe5511. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Feng, Y.; Mo, X.; Zheng, P.; Wang, Q.; Li, P.; Peng, P.; Liu, X.; Chen, Z.; Huang, H.; et al. Kinetics of SARS-CoV-2 Specific IgM and IgG Responses in COVID-19 Patients. Emerg. Microbes Infect. 2020, 9, 940–948. [Google Scholar] [CrossRef]
- Adams, E.; Ainsworth, M.; Anand, R.; Andersson, M.I.; Auckland, K.; Baillie, J.K.; Barnes, E.; Beer, S.; Bell, J.I.; Berry, T.; et al. Evaluation of Antibody Testing for SARS-CoV-2 Using ELISA and Lateral Flow Immunoassays. MedRxiv 2020. [Google Scholar]
- Huang, J.; Mao, T.; Li, S.; Wu, L.; Xu, X.; Li, H.; Xu, C.; Su, F.; Dai, J.; Shi, J.; et al. Long Period Dynamics of Viral Load and Antibodies for SARS-CoV-2 Infection: An. Observational Cohort Study. MedRxiv 2020. [Google Scholar]
- Khoury, D.S.; Cromer, D.; Reynaldi, A.; Schlub, T.E.; Wheatley, A.K.; Juno, J.A.; Subbarao, K.; Kent, S.J.; Triccas, J.A.; Davenport, M.P. Neutralizing Antibody Levels Are Highly Predictive of Immune Protection from Symptomatic SARS-CoV-2 Infection. Nat. Med. 2021, 27, 1205–1211. [Google Scholar] [CrossRef]
- Carsetti, R.; Zaffina, S.; Piano Mortari, E.; Terreri, S.; Corrente, F.; Capponi, C.; Palomba, P.; Mirabella, M.; Cascioli, S.; Palange, P.; et al. Different Innate and Adaptive Immune Responses to SARS-CoV-2 Infection of Asymptomatic, Mild, and Severe Cases. Front. Immunol. 2020, 11, 610300. [Google Scholar] [CrossRef]
- Marcos-Jiménez, A.; Sánchez-Alonso, S.; Alcaraz-Serna, A.; Esparcia, L.; López-Sanz, C.; Sampedro-Núñez, M.; Mateu-Albero, T.; Sánchez-Cerrillo, I.; Martínez-Fleta, P.; Gabrie, L.; et al. Deregulated Cellular Circuits Driving Immunoglobulins and Complement Consumption Associate with the Severity of COVID-19 Patients. Eur. J. Immunol. 2021, 51, 634–647. [Google Scholar] [CrossRef]
- Dembrovszky, F.; Váncsa, S.; Farkas, N.; Erőss, B.; Szakó, L.; Teutsch, B.; Bunduc, S.; Nagy, R.; Dohos, D.; Kiss, S.; et al. Immunoglobulin Response and Prognostic Factors in Repeated SARS-CoV-2 Positive Patients: A Systematic Review and Meta-Analysis. Viruses 2021, 13, 809. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Zeng, W.; He, H.; Zhao, D.; Jiang, D.; Zhou, P.; Cheng, L.; Li, Y.; Ma, X.; Jin, T. Serum IgA, IgM, and IgG Responses in COVID-19. Cell Mol. Immunol. 2020, 17, 773–775. [Google Scholar] [CrossRef] [PubMed]
- Madariaga, M.L.L.; Guthmiller, J.J.; Schrantz, S.; Jansen, M.O.; Christensen, C.; Kumar, M.; Prochaska, M.; Wool, G.; Durkin-Celauro, A.; Oh, W.H.; et al. Clinical Predictors of Donor Antibody Titre and Correlation with Recipient Antibody Response in a COVID-19 Convalescent Plasma Clinical Trial. J. Intern. Med. 2021, 289, 559–573. [Google Scholar] [CrossRef]
- Luchsinger, L.L.; Ransegnola, B.; Jin, D.; Muecksch, F.; Weisblum, Y.; Bao, W.; George, P.J.; Rodriguez, M.; Tricoche, N.; Schmidt, F.; et al. Serological Analysis of New York City COVID19 Convalescent Plasma Donors. medRxiv 2020. [Google Scholar]
- Brochot, E.; Demey, B.; Touzé, A.; Belouzard, S.; Dubuisson, J.; Schmit, J.-L.; Duverlie, G.; Francois, C.; Castelain, S.; Helle, F. Anti-Spike, Anti-Nucleocapsid and Neutralizing Antibodies in SARS-CoV-2 Inpatients and Asymptomatic Individuals. Front. Microbiol. 2020, 11, 584251. [Google Scholar] [CrossRef] [PubMed]
- Gambardella, C.; Pagliuca, R.; Pomilla, G.; Gambardella, A. COVID-19 Risk Contagion: Organization and Procedures in a South Italy Geriatric Oncology Ward. J. Geriatr. Oncol. 2020, 11, 1187–1188. [Google Scholar] [CrossRef] [PubMed]
- Šimánek, V.; Pecen, L.; Krátká, Z.; Fürst, T.; Řezáčková, H.; Topolčan, O.; Fajfrlík, K.; Sedláček, D.; Šín, R.; Pazdiora, P.; et al. Five Commercial Immunoassays for SARS-CoV-2 Antibody Determination and Their Comparison and Correlation with the Virus Neutralization Test. Diagnostics 2021, 11, 593. [Google Scholar] [CrossRef]
- Zeng, F.; Dai, C.; Cai, P.; Wang, J.; Xu, L.; Li, J.; Hu, G.; Wang, Z.; Zheng, F.; Wang, L. A Comparison Study of SARS-CoV-2 IgG Antibody between Male and Female COVID-19 Patients: A Possible Reason Underlying Different Outcome between Sex. J. Med. Virol. 2020, 92, 2050–2054. [Google Scholar] [CrossRef]
- Fafi-Kremer, S.; Bruel, T.; Madec, Y.; Grant, R.; Tondeur, L.; Grzelak, L.; Staropoli, I.; Anna, F.; Souque, P.; Fernandes-Pellerin, S.; et al. Serologic Responses to SARS-CoV-2 Infection among Hospital Staff with Mild Disease in Eastern France. EBioMedicine 2020, 59, 102915. [Google Scholar] [CrossRef]

| Characteristic | Category | N (%) |
|---|---|---|
| Gender | Female | 59 (53.6) |
| Male | 51 (46.4) | |
| Total | 110 | |
| Age | Median [range] | 49 (4–73) |
| Clinical symptoms | fever ≥37.5 °C | 56 (50.9) |
| dry cough | 32 (29.1) | |
| difficulty breathing or shortness of breath | 28 (25.5) | |
| loss of taste or smell | 47 (42.7) | |
| headache | 54 (49.1) | |
| tiredness | 78 (70.9) | |
| Risk factors | obesity | 16 (14.5) |
| smoking (last 10 years) | 15 (13.6) | |
| diabetes | 9 (8.2) | |
| cardiovascular disease | 10 (9.1) | |
| Flu vaccination | for season 2019 | 7 (6.4) |
| Month After PCR Test | 1st Blood Draw | 2nd Blood Draw | 3rd Blood Draw | Total |
|---|---|---|---|---|
| 0 | 2 | 0 | 0 | 2 |
| 1 | 5 | 2 | 0 | 7 |
| 2 | 4 | 3 | 1 | 8 |
| 3 | 5 | 0 | 1 | 6 |
| 4 | 34 | 2 | 0 | 36 |
| 5 | 48 | 7 | 0 | 55 |
| 6 | 10 | 7 | 1 | 18 |
| 7 | 2 | 10 | 0 | 12 |
| 8 | 0 | 22 | 1 | 23 |
| 9 | 0 | 16 | 10 | 26 |
| 10 | 0 | 29 | 3 | 32 |
| 11 | 0 | 10 | 5 | 15 |
| 12 | 0 | 2 | 0 | 2 |
| Total | 110 | 110 | 22 | 242 |
| Manufacturer | Methodology | Antigen Used | Cut-Off Index (COI) | Manufacturer’s Catalog Number | |
|---|---|---|---|---|---|
| Negative | Positive | ||||
| Roche | CLIA | Total Ig NP | <1.0 | ≥1.0 | 09 203 095 190 |
| Month After PCR Test | Count of Samples | Mean | Standard Deviation | Minimum | Lower Quartile | Median | Upper Quartile | Maximum |
|---|---|---|---|---|---|---|---|---|
| 0 | 2 | 1.6 | 0.1 | 1.5 | 1.5 | 1.6 | 1.7 | 1.7 |
| 1 | 7 | 52.0 | 15.5 | 8.4 | 26.4 | 40.1 | 99.2 | 118.8 |
| 2 | 8 | 52.3 | 13.5 | 8.8 | 19.9 | 41.3 | 91.1 | 104.6 |
| 3 | 6 | 99.3 | 14.4 | 34.3 | 83.1 | 116.1 | 121.9 | 124.4 |
| 4 | 36 | 80.7 | 6.4 | 2.9 | 63.6 | 79.4 | 103.1 | 146.9 |
| 5 | 55 | 79.0 | 6.2 | 2.3 | 40.2 | 83.8 | 112.8 | 177.2 |
| 6 | 18 | 81.3 | 13.2 | 9.9 | 37.6 | 63.6 | 136.6 | 189.8 |
| 7 | 12 | 73.2 | 16.9 | 10.9 | 30.6 | 64.6 | 84.8 | 195.5 |
| 8 | 23 | 50.0 | 9.7 | 1.3 | 9.3 | 33.6 | 87.2 | 186.6 |
| 9 | 26 | 41.7 | 9.2 | 3.9 | 9.1 | 22.2 | 51.4 | 180.0 |
| 10 | 32 | 40.2 | 6.2 | 2.2 | 11.2 | 30.3 | 56.8 | 130.1 |
| 11 | 15 | 27.1 | 6.7 | 0.6 | 6.5 | 19.3 | 37.4 | 92.1 |
| 12 | 2 | 23.3 | 5.5 | 17.8 | 17.8 | 23.3 | 28.8 | 28.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Šimánek, V.; Pecen, L.; Řezáčková, H.; Topolčan, O.; Fajfrlík, K.; Sedláček, D.; Šín, R.; Bludovská, M.; Pazdiora, P.; Slouka, D.; et al. Long-Term Monitoring of the Antibody Response to a SARS-CoV-2 Infection. Diagnostics 2021, 11, 1915. https://doi.org/10.3390/diagnostics11101915
Šimánek V, Pecen L, Řezáčková H, Topolčan O, Fajfrlík K, Sedláček D, Šín R, Bludovská M, Pazdiora P, Slouka D, et al. Long-Term Monitoring of the Antibody Response to a SARS-CoV-2 Infection. Diagnostics. 2021; 11(10):1915. https://doi.org/10.3390/diagnostics11101915
Chicago/Turabian StyleŠimánek, Václav, Ladislav Pecen, Hana Řezáčková, Ondřej Topolčan, Karel Fajfrlík, Dalibor Sedláček, Robin Šín, Monika Bludovská, Petr Pazdiora, David Slouka, and et al. 2021. "Long-Term Monitoring of the Antibody Response to a SARS-CoV-2 Infection" Diagnostics 11, no. 10: 1915. https://doi.org/10.3390/diagnostics11101915






