Cell-Based Biosensors: Electrical Sensing in Microfluidic Devices
Abstract
:1. Introduction
2. Device Considerations
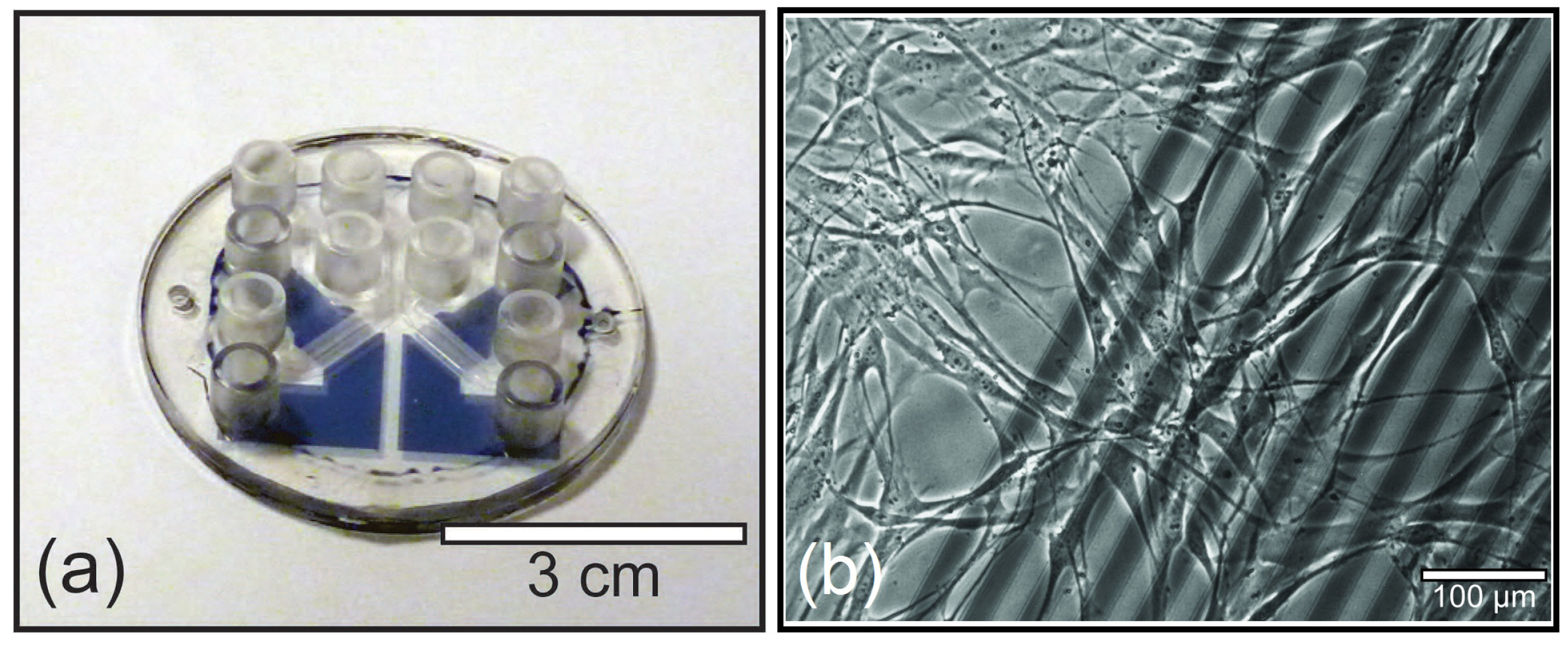
2.1. Microfluidic Systems
2.2. Materials for Fabrication
2.3. Sensing Unit
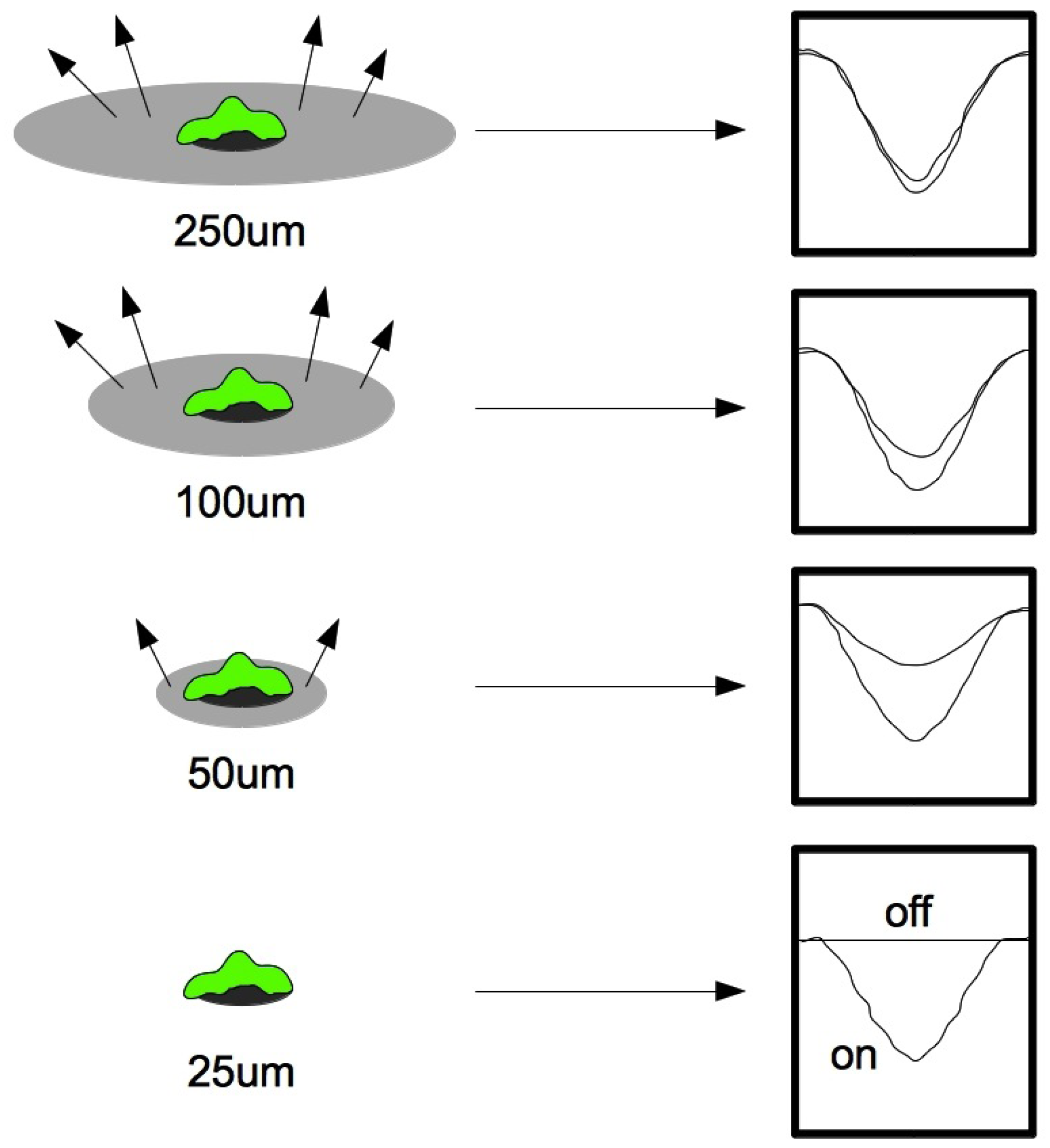
2.4. Microfluidic Design
3. Principles of Transduction
3.1. Electrochemical Methods
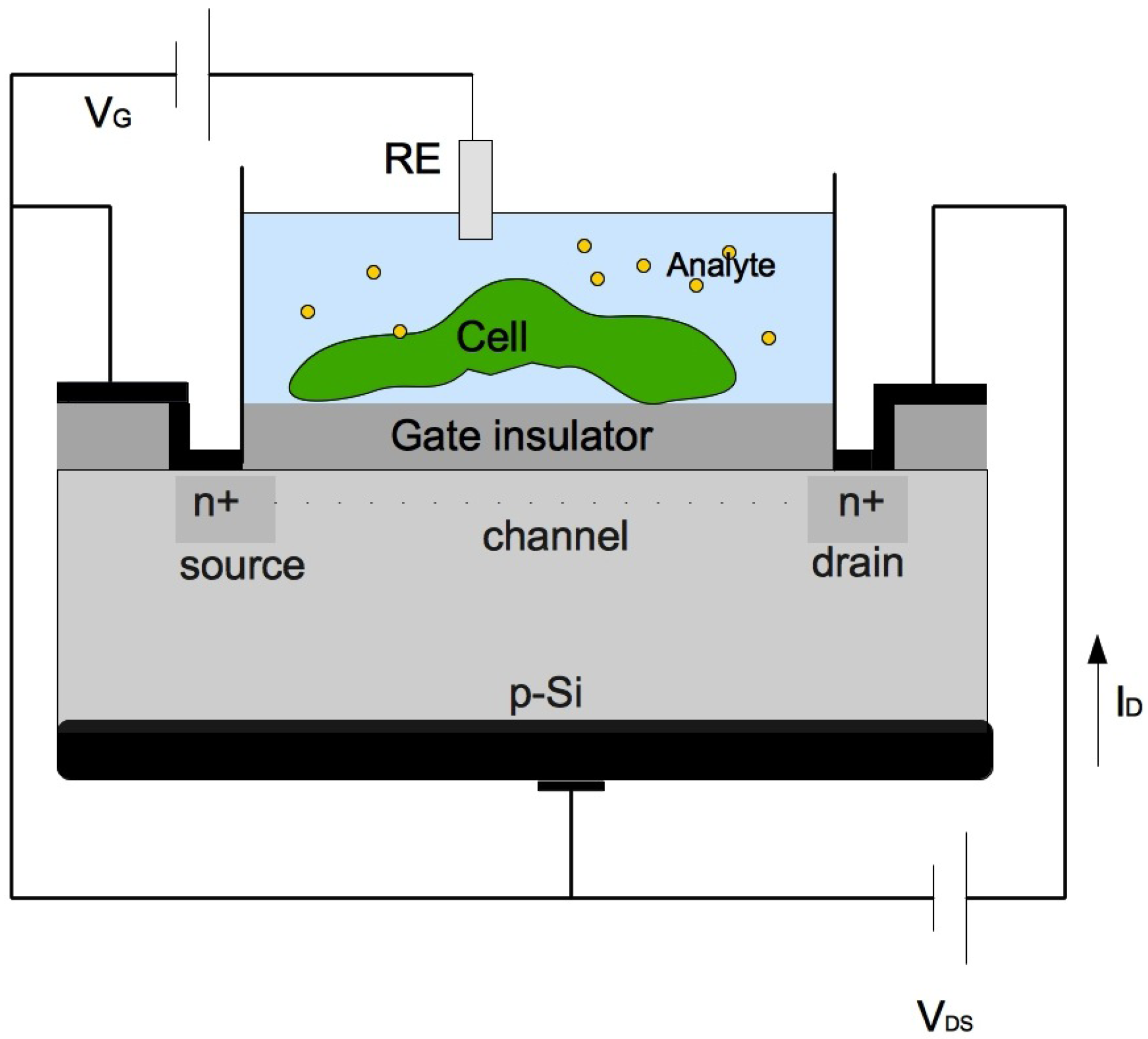
3.2. Field Effect Devices
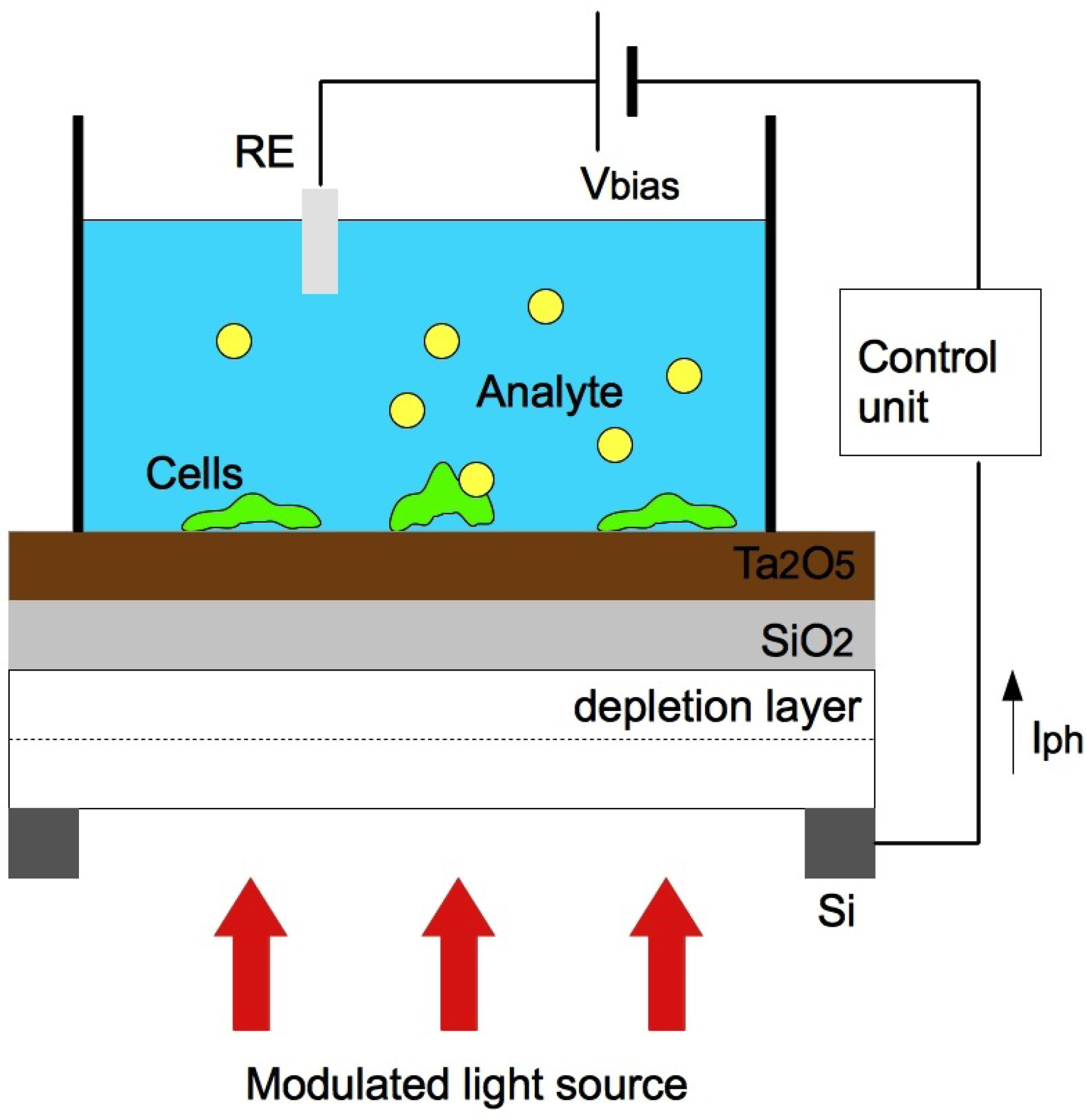
3.3. Light Addressable Potentiometric Sensors
4. Applications
4.1. Pathogens and Toxins
4.2. Single Cell Analysis
4.3. Drug Discovery

4.4. Cancer
5. Conclusion and Outlook
Acknowledgements
References
- Yan, M.; Huang, X.; Jia, Q.; Nadipalli, R.; Wang, T.; Shang, Y.; Yu, H.; Je, M.; Yeo, K. High-speed CMOS image sensor for high-throughput lensless microfluidic imaging system. Proc. SPIE 2012. [Google Scholar] [CrossRef]
- Cheng, X.; Chen, G.; Rodriguez, W.R. Micro- and nanotechnology for viral detection. Anal. Bioanal. Chem. 2009, 393, 487–501. [Google Scholar] [CrossRef]
- Lazcka, O.; Del Campo, F.J.; Munoz, F.X. Pathogen detection: A perspective of traditional methods and biosensors. Biosens. Bioelectron. 2007, 22, 1205–1217. [Google Scholar] [CrossRef]
- Wang, P.; Xu, G.; Qin, L.; Xu, Y.; Li, R. Cell-based biosensors and its application in biomedicine. Sens. Actuator. B Chem. 2005, 108, 576–584. [Google Scholar] [CrossRef]
- Banerjee, P.; Bhunia, A.K. Mammalian cell-based biosensors for pathogens and toxins. Trends Biotech. 2009, 27, 179–188. [Google Scholar] [CrossRef]
- Pancrazio, J.; Whelan, J.; Borkholder, D.; Ma, W.; Stenger, D. Development and application of cell-based biosensors. Ann. Biomed. Eng. 1999, 27, 697–711. [Google Scholar] [CrossRef]
- Cheran, L.E.; Cheung, S.; Wang, X.; Thompson, M. Probing the bioelectrochemistry of living cells. Electrochimi. Acta 2008, 53, 6690–6697. [Google Scholar] [CrossRef]
- Ding, L.; Du, D.; Zhang, X.; Ju, H. Trends in Cell-Based Electrochemical Biosensors. Curr. Med. Chem. 2008, 15, 3160–3170. [Google Scholar] [CrossRef]
- Stott, S.L.; Hsu, C.H.; Tsukrov, D.I.; Yu, M.; Miyamoto, D.T.; Waltman, B.A.; Rothenberg, S.M.; Shah, A.M.; Smas, M.E.; Korir, G.K.; et al. Isolation of circulating tumor cells using a microvortex-generating herringbone-chip. Proc. Natl. Acad. Sci. USA 2010, 107, 18392–18397. [Google Scholar] [CrossRef] [PubMed]
- Rosenthal, A.; Macdonald, A.; Voldman, J. Cell patterning chip for controlling the stem cell microenvironment. Biomaterials 2007, 28, 3208–3216. [Google Scholar] [CrossRef]
- Kim, L.; Toh, Y.C.; Voldman, J.; Yu, H. A practical guide to microfluidic perfusion culture of adherent mammalian cells. Lab Chip 2007, 7, 681–694. [Google Scholar] [CrossRef]
- Svendsen, W.; Castillo-Len, J.; Lange, J.; Sasso, L.; Olsen, M.; Abaddi, M.; Andresen, L.; Levinsen, S.; Shah, P.; Vedarethinam, I.; Dimaki, M. Micro and nano-platforms for biological cell analysis. Sens. Actuator. A Phys. 2011, 172, 54–60. [Google Scholar] [CrossRef]
- Ng, J.; Gitlin, I.; Stroock, A.; Whitesides, G. Components for integrated poly(dimethylsiloxane) microfluidic systems. Electrophoresis 2002, 23, 3461–3473. [Google Scholar] [CrossRef]
- Wright, D.; Rajalingam, B.; Karp, J.M.; Selvarasah, S.; Ling, Y.; Yeh, J.; Langer, R.; Dokmeci, M.R.; Khademhosseini, A. Reusable, reversibly sealable parylene membranes for cell and protein patterning. J. Biomed. Mater. Res. A 2008, 85A, 530–538. [Google Scholar] [CrossRef]
- Becker, H.; Locascio, L. Polymer microfluidic devices. Talanta 2002, 56, 267–287. [Google Scholar] [CrossRef]
- Sia, S.; Whitesides, G. Microfluidic devices fabricated in poly(dimethylsiloxane) for biological studies. Electrophoresis 2003, 24, 3563–3576. [Google Scholar] [CrossRef]
- Utko, P.; Persson, F.; Kristensen, A.; Larsen, N.B. Injection molded nanofluidic chips: Fabrication method and functional tests using single-molecule DNA experiments. Lab Chip 2011, 11, 303–308. [Google Scholar] [CrossRef]
- Hansen, T.S.; Selmeczi, D.; Larsen, N.B. Fast prototyping of injection molded polymer microfluidic chips. J. Micromech. Microeng. 2010, 20. [Google Scholar] [CrossRef]
- He, Q.; Sudibya, H.G.; Yin, Z.; Wu, S.; Li, H.; Boey, F.; Huang, W.; Chen, P.; Zhang, H. Centimeter-long and large-scale micropatterns of reduced graphene oxide films: Fabrication and sensing applications. ACS Nano 2010, 4, 3201–3208. [Google Scholar] [CrossRef]
- Nguyen, P.; Berry, V. Graphene interfaced with biological cells: Opportunities and challenges. J. Phys. Chem. Lett. 2012, 3, 1024–1029. [Google Scholar] [CrossRef]
- Rozlosnik, N. New directions in medical biosensors employing poly(3,4-ethylenedioxy thiophene) derivative-based electrodes. Anal. Bioanal. Chem. 2009, 395, 637–645. [Google Scholar] [CrossRef]
- Balamurugan, A.; Chen, S.M. Poly(3,4-ethylenedioxythiophene-co-(5-amino-2-naphthalenesulfonic acid)) (PEDOT-PANS) film modified glassy carbon electrode for selective detection of dopamine in the presence of ascorbic acid and uric acid. Anal. Chim. Acta 2007, 596, 92–98. [Google Scholar] [CrossRef]
- Vasantha, V.S.; Chen, S.M. Electrocatalysis and simultaneous detection of dopamine and ascorbic acid using poly(3,4-ethylenedioxy)thiophene film modified electrodes. J. Electroanal. Chem. 2006, 592, 77–87. [Google Scholar] [CrossRef]
- Larsen, S.T.; Vreeland, R.F.; Heien, M.L.; Taboryski, R. Characterization of poly(3,4-ethylenedioxythiophene):tosylate conductive polymer microelectrodes for transmitter detection. Analyst 2012, 137, 1831–1836. [Google Scholar] [CrossRef]
- Kiilerich-Pedersen, K.; Poulsen, C.R.; Jain, T.; Rozlosnik, N. Polymer based biosensor for rapid electrochemical detection of virus infection of human cells. Biosens. Bioelectron. 2011, 28, 386–392. [Google Scholar] [CrossRef]
- Thein, M.; Asphahani, F.; Cheng, A.; Buckmaster, R.; Zhang, M.; Xu, J. Response characteristics of single-cell impedance sensors employed with surface-modified microelectrodes. Biosens. Bioelectron. 2010, 25, 1963–1969. [Google Scholar] [CrossRef]
- Wu, Y.L.; Hsu, P.Y.; Hsu, C.P.; Wang, C.C.; Lee, L.W.; Lin, J.J. Electrical characterization of single cells using polysilicon wire ion sensor in an isolation window. Biomed. Microdevices 2011, 13, 939–947. [Google Scholar] [CrossRef]
- Wu, Y.L.; Hsu, P.Y.; Hsu, C.P.; Lin, J.J. Detecting the effect of targeted anti-cancer medicines on single cancer cells using a poly-silicon wire ion sensor integrated with a confined sensitive window. Biomed. Microdevices 2012, 14, 839–848. [Google Scholar] [CrossRef]
- Velve-Casquillas, G.; Berre, M.L.; Piel, M.; Tran, P.T. Microfluidic tools for cell biological research. Nano Today 2010, 5, 28–47. [Google Scholar] [CrossRef]
- Barkefors, I.; Le Jan, S.; Jakobsson, L.; Hejll, E.; Carlson, G.; Johansson, H.; Jarvius, J.; Park, J.W.; Li Jeon, N.; Kreuger, J. Endothelial cell migration in stable gradients of vascular endothelial growth factor A and fibroblast growth factor 2. J. Biol. Chem. 2008, 283, 13905–13912. [Google Scholar] [CrossRef] [PubMed]
- Tang, Z.; Akiyama, Y.; Itoga, K.; Kobayashi, J.; Yamato, M.; Okano, T. Shear stress-dependent cell detachment from temperature-responsive cell culture surfaces in a microfluidic device. Biomaterials 2012, 33, 7405–7411. [Google Scholar] [CrossRef] [PubMed]
- Buchinger, S.; Grill, P.; Morosow, V.; Ben-Yoav, H.; Shacham-Diamand, Y.; Biran, A.; Pedahzur, R.; Belkin, S.; Reifferscheid, G. Evaluation of chrono-amperometric signal detection for the analysis of genotoxicity by a whole cell biosensor. Anal. Chim. Acta 2010, 659, 122–128. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, J.; Yang, H.; Fu, F.; Chen, G. A strategy for development of electrochemical DNA biosensor based on site-specific DNA cleavage of restriction endonuclease. Biosens. Bioelectron. 2010, 26, 144–148. [Google Scholar] [CrossRef]
- Primiceri, E.; Chiriaco, M.S.; Ionescu, R.E.; D’Amone, E.; Cingolani, R.; Rinaldi, R.; Maruccio, G. Development of EIS cell chips and their application for cell analysis. Microelectron. Eng. 2009, 86, 1477–1480. [Google Scholar] [CrossRef]
- Diouani, M.F.; Helali, S.; Hafaid, I.; Hassen, W.M.; Snoussi, M.A.; Ghram, A.; Jaffrezic-Renault, N.; Abdelghani, A. Miniaturized biosensor for avian influenza virus detection. Mater. Sci. Eng. C 2008, 28, 580–583. [Google Scholar] [CrossRef]
- Hnaien, M.; Diouani, M.F.; Helali, S.; Hafaid, I.; Hassen, W.M.; Renault, N.J.; Ghram, A.; Abdelghani, A. Immobilization of specific antibody on SAM functionalized gold electrode for rabies virus detection by electrochemical impedance spectroscopy. Biochem. Eng. J. 2008, 39, 443–449. [Google Scholar] [CrossRef]
- Daniels, J.S.; Pourmand, N. Label-free impedance biosensors: Opportunities and challenges. Electroanalysis 2007, 19, 1239–1257. [Google Scholar] [CrossRef]
- Mathebula, N.S.; Pillay, J.; Toschi, G.; Verschoor, J.A.; Ozoemena, K.I. Recognition of anti-mycolic acid antibody at self-assembled mycolic acid antigens on a gold electrode: A potential impedimetric immunosensing platform for active tuberculosis. Chem. Commun. 2009. [Google Scholar] [CrossRef]
- Arias, L.R.; Perry, C.A.; Yang, L. Real-time electrical impedance detection of cellular activities of oral cancer cells. Biosens. Bioelectron. 2010, 25, 2225–2231. [Google Scholar] [CrossRef]
- Ona, T.; Shibata, J. Advanced dynamic monitoring of cellular status using label-free and non-invasive cell-based sensing technology for the prediction of anticancer drug efficacy. Anal. Bioanal. Chem. 2010, 398, 2505–2533. [Google Scholar] [CrossRef]
- Cheung, K.; Gawad, S.; Renaud, P. Impedance spectroscopy flow cytometry: On-chip label-free cell differentiation. Cytometry Part A 2005, 65A, 124–132. [Google Scholar] [CrossRef]
- Giaever, I.; Keese, C.R. Use of electric-fields to monitor the dynamic aspect of cell behavior in tissue-culture. IEEE Trans. Biomed. Eng. 1986, 33, 242–247. [Google Scholar] [CrossRef]
- Keese, C.; Giaever, I. A biosensor that monitors cell morphology with electrical fields. IEEE Eng. Med. Biol. Mag. 1994, 13, 402–408. [Google Scholar] [CrossRef]
- Offenhausser, A.; Knoll, W. Cell-transistor hybrid systems and their potential applications. Trends Biotech. 2001, 19, 62–66. [Google Scholar] [CrossRef]
- Poghossian, A.; Ingebrandt, S.; Offenhusser, A.; Schning, M. Field-effect devices for detecting cellular signals. Semin. Cell Dev. Biol. 2009, 20, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Schoening, M.J.; Poghossian, A. Bio FEDs (Field-Effect devices): State-of-the-art and new directions. Electroanalysis 2006, 18, 1893–1900. [Google Scholar] [CrossRef]
- McCoy, M.H.; Wang, E. Use of electric cell-substrate impedance sensing as a tool for quantifying cytopathic effect in influenza A virus infected MDCK cells in real-time. J. Virol. Meth. 2005, 130, 157–161. [Google Scholar] [CrossRef]
- Campbell, C.E.; Laane, M.M.; Haugarvoll, E.; Giaever, I. Monitoring viral-induced cell death using electric cell-substrate impedance sensing. Biosens. Bioelectron. 2007, 23, 536–542. [Google Scholar] [CrossRef]
- Arndt, S.; Seebach, J.; Psathaki, K.; Galla, H.J.; Wegener, J. Bioelectrical impedance assay to monitor changes in cell shape during apoptosis. Biosens. Bioelectron. 2004, 19, 583–594. [Google Scholar] [CrossRef]
- Solly, K.; Wang, X.; Xu, X.; Strulovici, B.; Zheng, W. Application of real-time cell electronic sensing (RT-CES) technology to cell-based assays. ASSAY Drug Dev. Technol. 2004, 2, 363–372. [Google Scholar] [CrossRef]
- Pancrazio, J.; Bey, P.; Cuttino, D.; Kusel, J.; Borkholder, D.; Shaffer, K.; Kovacs, G.; Stenger, D. Portable cell-based biosensor system for toxin detection. Sens. Actuator. B Chem. 1998, 53, 179–185. [Google Scholar] [CrossRef]
- Liu, Q.; Cai, H.; Xu, Y.; Xiao, L.; Yang, M.; Wang, P. Detection of heavy metal toxicity using cardiac cell-based biosensor. Biosens. Bioelectron. 2007, 22, 3224–3229. [Google Scholar] [CrossRef]
- Gray, S.A.; Kusel, J.K.; Shaffer, K.M.; Shubin, Y.S.; Stenger, D.A.; Pancrazio, J.J. Design and demonstration of an automated cell-based biosensor. Biosens. Bioelectron. 2001, 16, 535–542. [Google Scholar] [CrossRef]
- Schmidt, C.; Leach, J. Neural tissue engineering: Strategies for repair and regeneration. Annu. Rev. Biomed. Eng. 2003, 5, 293–347. [Google Scholar] [CrossRef]
- Lee, J.Y.; Bashur, C.A.; Milroy, C.A.; Forciniti, L.; Goldstein, A.S.; Schmidt, C.E. Nerve growth factor-immobilized electrically conducting fibrous scaffolds for potential use in neural engineering applications. IEEE Trans. Nanobiosci. 2012, 11, 15–21. [Google Scholar] [CrossRef]
- Larsen, S.; Matteucci, M.; Taboryski, R. Conductive Polymer Microelectrodes for On-Chip Measurement of Transmitter Release from Living Cells. In Proceedings of Nanotech Conference and Expo 2012, Santa Clara, CA, USA, 18–21 June 2012; 2, pp. 302–305.
- Migita, S.; Wada, K.I.; Taniguchi, A. Reproducible fashion of the HSP70B’ promoter-induced cytotoxic response on a live cell-based biosensor by cell cycle synchronization. Biotechnol. Bioeng. 2010, 107, 561–565. [Google Scholar] [CrossRef]
- Nie, F.Q.; Yamada, M.; Kobayashi, J.; Yamato, M.; Kikuchi, A.; Okano, T. On-chip cell migration assay using microfluidic channels. Biomaterials 2007, 28, 4017–4022. [Google Scholar] [CrossRef]
- Liu, Y.; Sai, J.; Richmond, A.; Wikswo, J.P. Microfluidic switching system for analyzing chemotaxis responses of wortmannin-inhibited HL-60 cells. Biomed. Microdevices 2008, 10, 499–507. [Google Scholar] [CrossRef]
- Pihl, J.; Karlsson, M.; Chiu, D. Microfluidic technologies in drug discovery. Drug Discov. Today 2005, 10, 1377–1383. [Google Scholar] [CrossRef]
- Lin, F.; Butcher, E.C. T cell chemotaxis in a simple microfluidic device. Lab Chip 2006, 6, 1462–1469. [Google Scholar] [CrossRef]
- Georgescu, W.; Jourquin, J.; Estrada, L.; Anderson, A.R.A.; Quaranta, V.; Wikswo, J.P. Model-controlled hydrodynamic focusing to generate multiple overlapping gradients of surface-immobilized proteins in microfluidic devices. Lab Chip 2008, 8, 238–244. [Google Scholar] [CrossRef]
- Keenan, T.M.; Folch, A. Biomolecular gradients in cell culture systems. Lab Chip 2008, 8, 34–57. [Google Scholar] [CrossRef]
- Kwasny, D.; Kiilerich-Pedersen, K.; Moresco, J.; Dimaki, M.; Rozlosnik, N.; Svendsen, W.E. Microfluidic device to study cell transmigration under physiological shear stress conditions. Biomed. Microdevices 2011, 13, 899–907. [Google Scholar] [CrossRef]
- Yun, Y.H.; Eteshola, E.; Bhattacharya, A.; Dong, Z.; Shim, J.S.; Conforti, L.; Kim, D.; Schulz, M.J.; Ahn, C.H.; Watts, N. Tiny medicine: Nanomaterial-based biosensors. Sensors 2009, 9, 9275–9299. [Google Scholar] [CrossRef]
- Arya, S.K.; Lee, K.C.; Bin Dah’alan, D.; Daniel; Rahman, A.R.A. Breast tumor cell detection at single cell resolution using an electrochemical impedance technique. Lab Chip 2012, 12, 2362–2368. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wong, C.C.; Pui, T.S.; Nadipalli, R.; Weerasekera, R.; Chandran, J.; Yu, H.; Rahman, A.R. CMOS high density electrical impedance biosensor array for tumor cell detection. Sens. Actuator. B Chem. 2012, 173, 903–907. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kiilerich-Pedersen, K.; Rozlosnik, N. Cell-Based Biosensors: Electrical Sensing in Microfluidic Devices. Diagnostics 2012, 2, 83-96. https://doi.org/10.3390/diagnostics2040083
Kiilerich-Pedersen K, Rozlosnik N. Cell-Based Biosensors: Electrical Sensing in Microfluidic Devices. Diagnostics. 2012; 2(4):83-96. https://doi.org/10.3390/diagnostics2040083
Chicago/Turabian StyleKiilerich-Pedersen, Katrine, and Noemi Rozlosnik. 2012. "Cell-Based Biosensors: Electrical Sensing in Microfluidic Devices" Diagnostics 2, no. 4: 83-96. https://doi.org/10.3390/diagnostics2040083



