1. Introduction
Severe trauma, especially when brain injury is involved, is recognized as one of the main reasons for complications and mortality. Multiple-trauma patients either die as a direct consequence of their injuries, or, more commonly, days later, after the development of secondary infectious complications [
1,
2,
3,
4,
5]. Despite the progress in ICU therapeutic measures, the incidence of mortality remains practically unaffected: in 2004, Osborn et al. reported a 23% mortality rate [
6], and almost 20 years later, in 2021, Sy et al. presented a similar rate of 24.3% [
7].
Today, the gut continues to be “the motor” of critical illness, although issues present in a more sophisticated manner: severe trauma, stressful surgical interventions, or sepsis, for example, trigger the disruption of the homeostasis of the microbiota, leading within hours to the loss of bacterial heterogeneity, a reduction in the number of commensal phyla, and thus the prevalence of pathogens. This further triggers and promotes harm to the already-compromised host in a positive feedback spiral [
8].
Following severe injury and the consequent systemic inflammatory response, the host’s archetypal response is an increase in neutrophils and a decrease in lymphocytes, with it being well accepted that the neutrophil is the predominant cellular component of host defense against infectious injury. At the same time, the lymphocytes are known as the predominant cellular components of the adaptive immune system [
9]. Zahorec [
10] has noted a connection between disease severity and the presence of lymphopenia and neutrophilia, since the failure of the host to revert lymphopenia to a normal state has been linked to increased mortality, regardless of the type of leukocytosis [
11].
The complete blood count test is one of the cheapest and easiest analyses to implement in a clinical laboratory examination and provides useful information on all blood cell types, and more particularly on neutrophils and lymphocytes—the most implicated in the progression of many diseases. Thus, the ratio of neutrophils to lymphocytes (NLR) remains a reliable inflammatory indicator of a high prognostication. Additionally, it is easy to calculate, and can be determined at almost zero cost [
9,
10,
12].
Probiotics, defined by the FAO [Food & Agricultural Organization] and the WHO [World Health Organization] as “live microorganisms, which, when consumed in adequate quantities, confer a health benefit on the host” [
13], were introduced early into the armamentarium used against critical illness to stimulate and augment the immunological response of the host, to produce antimicrobial compounds, and to competitively eliminate pathogens [
14,
15,
16,
17]. Recently, it became evident that they are able to promote the healing process after tissue injury, both through the restoration of the compromised local and systemic microbiome and the enhancement of anti-inflammatory, healing, and angiogenetic factors [
18,
19,
20].
Given that the “upon intubation” initiation of probiotic treatment in mechanically ventilated multi-trauma individuals—including cases of brain injury—was found to significantly decrease the prevalence of severe infectious complications [
14,
21,
22], we decided to test the applicability of the NLR as a prognostic index of ventilator-associated pneumonia [VAP] induction in such patients, allocated to probiotic treatment or placebo groups. We aimed to assess whether there was a difference in the NLR critical value [cut-off point] between placebo and probiotic-treated patients in the prognostication of VAP infection, and to interpret the difference, if any, through the immunomodulatory properties of the probiotics.
4. Discussion
In this analysis, we tried to investigate the applicability of the NLR, calculated on the 7th post-trauma day, as a prognostic index of VAP infection in patients that were treated with either a placebo or a four-probiotic regime. Given that [i] the ProVAP multi-center study [
22] revealed that immediate probiotic administration significantly reduced the frequency of VAP, and becaise [ii] probiotics are well documented to exert immunomodulatory properties [
26,
27], we anticipated that the NLR, as the proportion of neutrophils to lymphocytes, would be reduced and may thus thought to be in doubt as a prognostic index of VAP manifestation.
The two groups of patients included in the ProVAP study on which the post hoc analysis is based exhibited an outstanding homogeneity in their characteristics regarding age—being under 55 years old—gender and APACHE II, SAPS II, SOFA, CCI, and GCS scores. They also had the same comorbidities and received the same antibiotics and enteral nutritional support [
22]. All participants had experienced multi-traumas, which were required to involve severe brain trauma and—at minimum—one more organ system, and thus the necessity of immediate intubation either in the pre-hospital setting or the emergency department upon arrival [
28]. The severity of trauma, as a consequence, was required to induce prolonged intubation and mechanical ventilation—at least for the next 10 days—in order to have put the patients at a significant risk of developing VAP.
Brain trauma in general is well known to represent a complex biochemical cascade related with numerous patho-physiological functions, affecting not only the central nervous system but the function of multiple distant organs/systems [
29,
30,
31]. Furthermore, acute brain trauma was found to significantly deteriorate gut microbiota diversity—starting within hours of injury [
32]. Feces, recovered by means of rectal swabs from 101 patients suffering moderate-to-severe traumatic brain injuries, were found to be extensively colonized by
Proteobacteria phyla, with the
Enterobacteriaceae being the predominant group [
33]. Other authors reported a reduction in
Bacteroidetes,
Firmicutes, Verrucomicrobia and
Faecalibacterium and an increase in pathogens, such as
Enterococcus,
Parabacteroides, and
Lachnoclostridium, associated with chronic inflammatory conditions [
34,
35]. Furthermore, brain trauma is linked with gut barrier dysfunction, previously known as “leaky” gut [
36,
37]. Alterations in microbiota diversity are recognized as contributing to infections, and mainly to pneumonia, through the gut–brain–lung axis [
38,
39,
40,
41,
42], while the disruption of the intestinal barrier, as the result of pathogen overgrowth and loss of the beneficial role of the commensal bacteria, contributes to long-term complications [
43,
44,
45,
46] due to the deterioration of host immunity [
47].
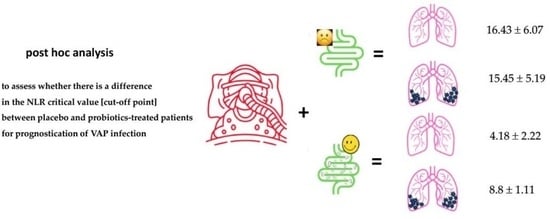
In the present analysis, the number of probiotic-treated patients presented VAP during their ICU stay was significantly small [n = 7 out of 59 patients, rate 11.9%] in relation to the placebo-treated group [n = 15 out of 53 patients, rate 28.3%, p = 0.034]. This post hoc analysis showed a significant reduction in the NLR index in this patient group, compared to the corresponding group of placebo/VAT patients [8.8 ± 1.11 vs. 15.45 ± 5.19, p < 0.001]. Also of interest were the differences within a group on day 7: although there was no significant difference between placebo/no-VAP and placebo/VAP sub-groups, there was a highly significant increase in NLR between the probiotic/no-VAP and probiotic/VAP sub-groups [p < 0.001]. This result may easily be related to the 3-fold value of lymphocytes [involved as denominator in the neutrophil/lymphocyte formula] compared to placebo sub-groups due to the known issue of immune paralysis.
NLR is one of the most accepted prognostic indices and has a positive correlation to disease severity, progress, and mortality, reflecting the immune system’s sufficiency and its ability to “fight” inflammation. It is thus applicable to a variety of disease conditions, from carcinomas to inflammatory bowel and cardiovascular diseases [
48,
49,
50,
51,
52,
53]. In a recent study of ours on cardiac surgery patients, the NLR values on post-operative days 5 and 7 demonstrated significant discriminatory efficacy for predicting mortality within 90 days [
54]. In a meta-analysis, an NLR value greater than 12.65 was found to be associated with bacteremia and was predictive of bacterial sepsis, whereas lower NLR values (cut-off point below 2.06) showed an association with influenza virus infection in patients with lung infections [
55]. In the same manner, the NLR index was demonstrated to be the only biomarker able to discriminate between survivors and non-survivors among major trauma patients [
56]. Among the 1694 patients retrospectively evaluated, the authors found that patients with a preoperative NLR value greater than 3.23 experienced increased mortality and longer ICU stays [
57]. Similarly, Larmann et al. [
58] confirmed a preoperative predictive value of NLR greater than 3.1 for patients facing major adverse events. In our study, we found greater values of around 5 on day 0. However, this finding was easily explainable by the inflammatory response elicited by both the major trauma itself and the psychological stress of the motor vehicle crash, leading to the activation of the neuroendocrine system and thus the release of cytokines and stress hormones [
59]. We knew this because the blood sample was obtained a few hours after these events.
As a common reaction to an overwhelming inflammatory response, the critically ill patient generally experiences reduced lymphocyte numbers and activity as a result of B- and T-cell apoptosis [
60,
61]. Besides lymphocytopenia, the increase in the number of neutrophils, as well as “wrong” systemic neutrophil activation and migration towards the microvasculature, also promotes further tissue injury and multiple organ failure [
62]. A recent study positively correlates the severity of brain trauma with the number of neutrophils in peripheral blood and brain tissue [
63]. Seventy-two critically ill multi-trauma patients were allocated to a 15-day treatment with a formula of four probiotics plus prebiotics [Synbiotic 2000Forte; Medipharm, Sweden, containing
Pediococcus pentosaceus 5-33:3, Leuconostoc mesenteroides 32-77:1,
L. paracasei ssp.
paracasei 19; and
L. plantarum 2362 at a concentration of 10
11 cfu each one] or maltodextrin as a placebo. VAP occurred in 5 and 12 patients (13.9% vs. 33.3%,
p = 0.047] and sepsis in 5 patients and 13 patients (13.9% vs. 36.1%,
p = 0.028) treated with synbiotics compared to the placebo. White blood cell counts were significantly lower in synbiotics-treated patients on post-trauma days in patients who did or did not develop VAP or sepsis [
14]. In an observational study of 68 participants diagnosed with sepsis within the first 24h due to VAP or other nosocomial infections, the absolute count of CD3(+)/CD4(+) lymphocytes was found to be significantly lower (
p = 0.034), while the apoptosis of isolated monocytes significantly increased (
p = 0.007), in sepsis patients due to VAP in comparison to other infections [
64]. Similar findings were also reported in a group of 40 patients suffering from COVID-19 infection: the flow cytometry results of peripheral blood samples examined for lymphocyte subsets revealed significant and sustained reductions in lymphocytes, but increases in neutrophils, directly associated with disease severity, as also occurred with the NLR index [
65].
These alterations in neutrophil and even greater changes in lymphocyte numbers directly mirror a valuable increase in the NLR, which occurs due to a significant increase in the nominator [neutrophils] and also to a decrease in the denominator [lymphocytes], as also seen in our cases. However, in the situation of severe multi-trauma, for individuals receiving probiotics as a part of their intensive care treatment, a differentiation in NLR components—that is neutrophils and lymphocytes—is to be expected, since, in practice, NLR expresses the immune profile of the patient and consequently his/her inflammatory reaction, which is greatly improved by probiotics [
66,
67,
68].
It is also well known that probiotics can potentially suppress various inflammatory states in the intestines by mitigating the inflammatory process, enhancing the mucosal and epithelial barriers, and counteracting colonization by pathogens. However, a lot of these properties are strain-specific since some strains are potent inducers of regulatory cells, while others are not [
69,
70]. This is only shown in a few studies: in severely ill patients who received synbiotics [
L. casei,
L. rhamnosus,
L. acidophilus,
L. bulgaricus,
Bifidobacterium breve,
Bifidobacterium longum,
Streptococcus thermophilus, and fructo-oligo-saccharides as prebiotics], a statistically significant decrease in the NLR index [from 7.83 to 6.01,
p = 0.04] was evident, in parallel with a statistically significant reduction in endotoxin levels and inflammation markers in relation to the placebo the group [
71]. There were similar findings in relation to other diseases:
Bifidobacterium and
Lactobacillus spp. were shown to ameliorate oxidative stress as well as inflammatory markers and thus significantly improve the NLR index in patients suffering from diversion colitis [
72]. A probiotic mixture of
Bifidobacterium longum Bar33 and
Lactobacillus helveticus Bar13 improved immune function in the gut and the periphery in elderly adults [
27], while the probiotic
Bifidobacterium longum combined with an extract from the mushroom Lentinula edodes mycelia was found to control the T-regulatory and dendritic cell phenotypes to produce anti-inflammatory responses after azithromycin treatment for 5 days [
73]. In a model of acute respiratory distress syndrome,
Lactocaseibacillus rhamnosus treatment was found to exert an anti-inflammatory effect by decreasing the proinflammatory cytokine-associated Th1 and Th17 responses, thus inhibiting neutrophil infiltration into lung cells and finally reducing the NLR index [
74]. In the same model receiving broad-spectrum antibiotics for 8 weeks, peroral treatment with the VSL#3 compound, consisting of
Streptococcus thermophilus,
Bifidobacterium breve,
B. longum,
B. infantis,
Lactobacillus acidophilus,
L. plantarum,
L. paracasei, and
L. delbrueckii subsp, produced similar results.
Bulgaricus exhibited extensive control of mucosal, peripheral, and systemic innate and adaptive immunity [
26].
In the present study, a combo regime of the well-studied probiotics
L. acidophilus LA-5,
L. plantarum UBLP-40,
B. animalis subsp.
lactis BB-12 and
S. boulardii Unique-28 was used, with these species all having documented anti-inflammatory and immunomodulatory properties [
19,
20,
75,
76]. This is why, after the generation of the ROC curve for probiotic-treated patients we found—in addition to the number of patients experiencing VAP being small—that a NLR7 cut-off value of 7.20 was a prognostic factor of VAP (AUC: 78.6%,
p = 0.015, 95% CI: 62.6–94.5%), and has a high specificity of 90.2% and a sensitivity of 42.9%. The high specificity of this cut-off value is clearly seen in the difference of NLR7 values in the two sub-groups of probiotic-treated patients: those [
n = 52] showing NLR7 4.18 ± 2.22 did not experience VAP, while those [
n = 7] with NLR7 8.8 ± 1.11 finally presented VAP, despite probiotic treatment. In other words, in patients treated with probiotics—known for having immune-regulatory and anti-inflammatory properties, and finally helping severely ills patient to not exert lymphopenia—a NLR7 value equal or higher than 7.20 may be considered as an alert or as a specified index for an upcoming VAP.
Although the findings of this post hoc analysis impressively documented the immunological status of each of the 4 sub-groups of the ProVAP trial, and underlined the beneficial defense effects exerted by these probiotics, there were some limitations. Firstly, because it was a post hoc analysis and not primary research, the sample size estimation was based on the principal outcomes of the ProVAP study. The absence of power analysis was the reason for the limited VAP events in the group of probiotic-treated patients—our precise analysis related only to information obtained from the ProVap trial versus the 15 cases in the placebo. Conversely, this significant difference reflected the beneficial effects of probiotic use. The second point was the arbitrary decision to compare white blood cell counts on day 0 with those of day 7; this was mainly based on the post-trauma day on which VAP first manifested, although this decision may have contributed to interventional bias. And the third point, directly related to the first, was that our analysis did not allow for conclusive results to be drawn, but only the recognition of significant differences and a clear cut-off value as a prognostic factor of VAP for patients treated with probiotics only. We hope that this clear cut-off value offers a valuable literature background which explains the findings.