Combined Sulfidation and Vacuum Distillation for the Directional Removal of Hazardous Mercury from Crude Selenium
Abstract
:1. Introduction
2. Experimental Section
2.1. Experimental Materials
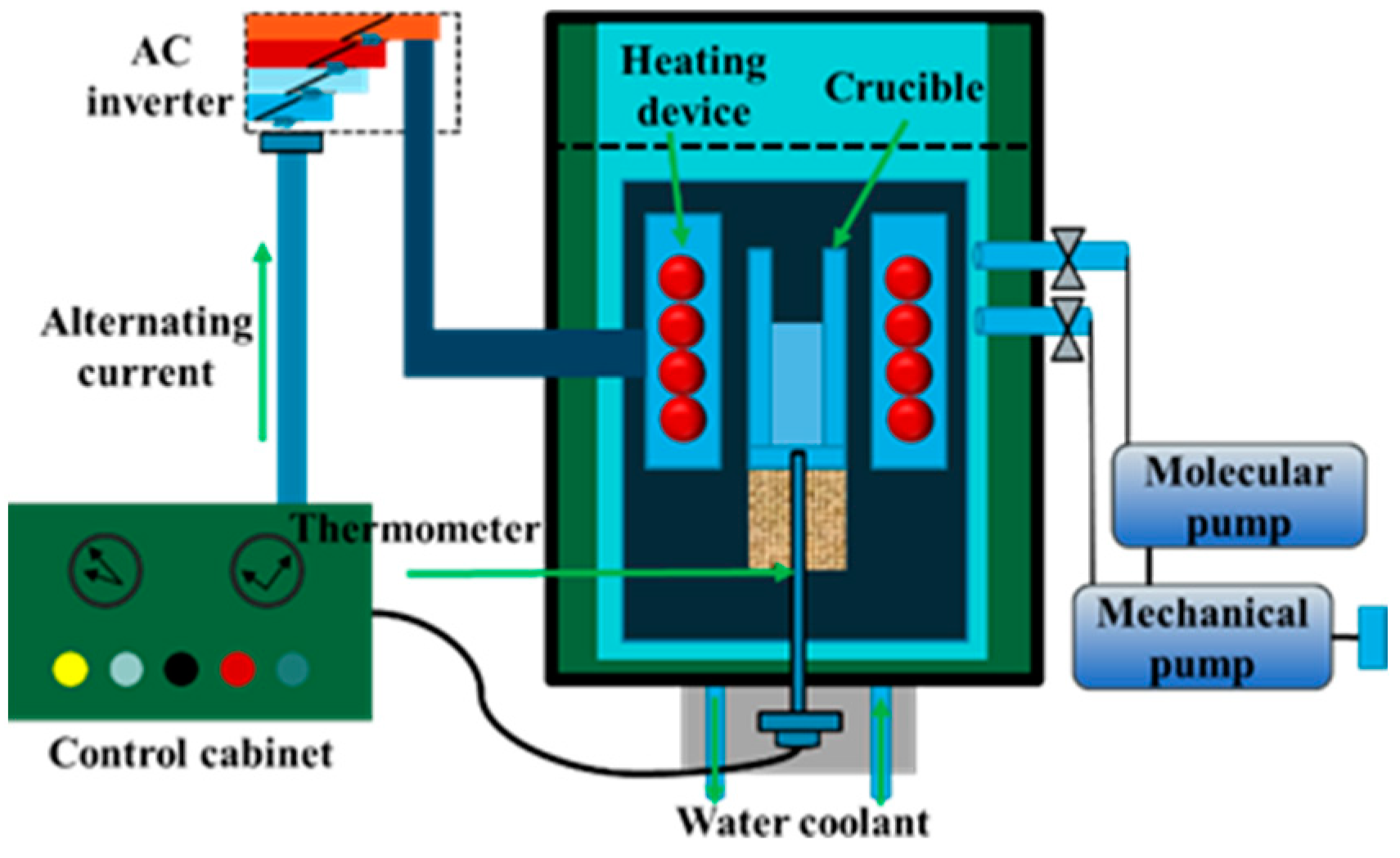
2.2. Experimental Procedure
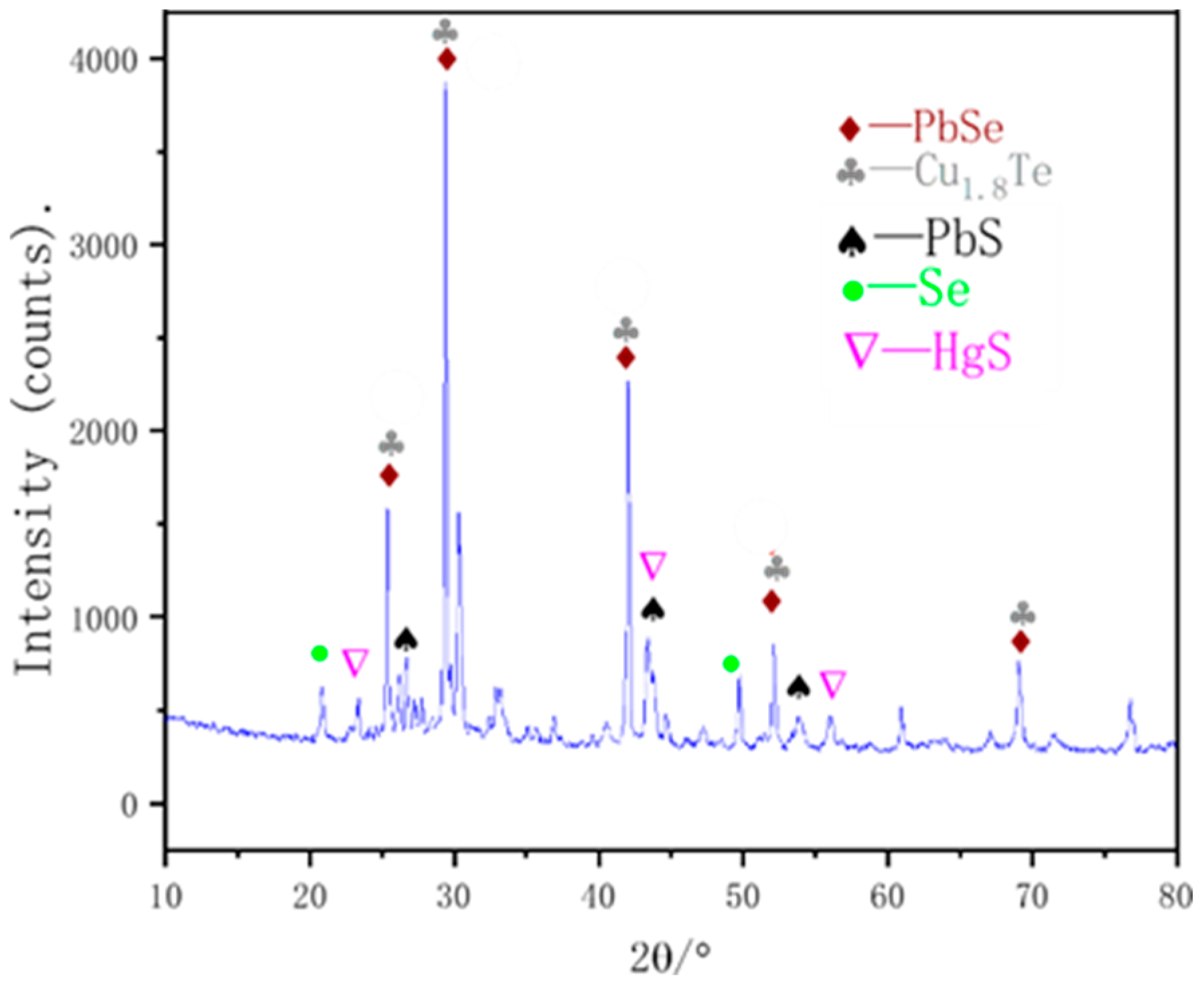
2.3. Characterization
3. Results and Discussion
3.1. Theoretical Analysis
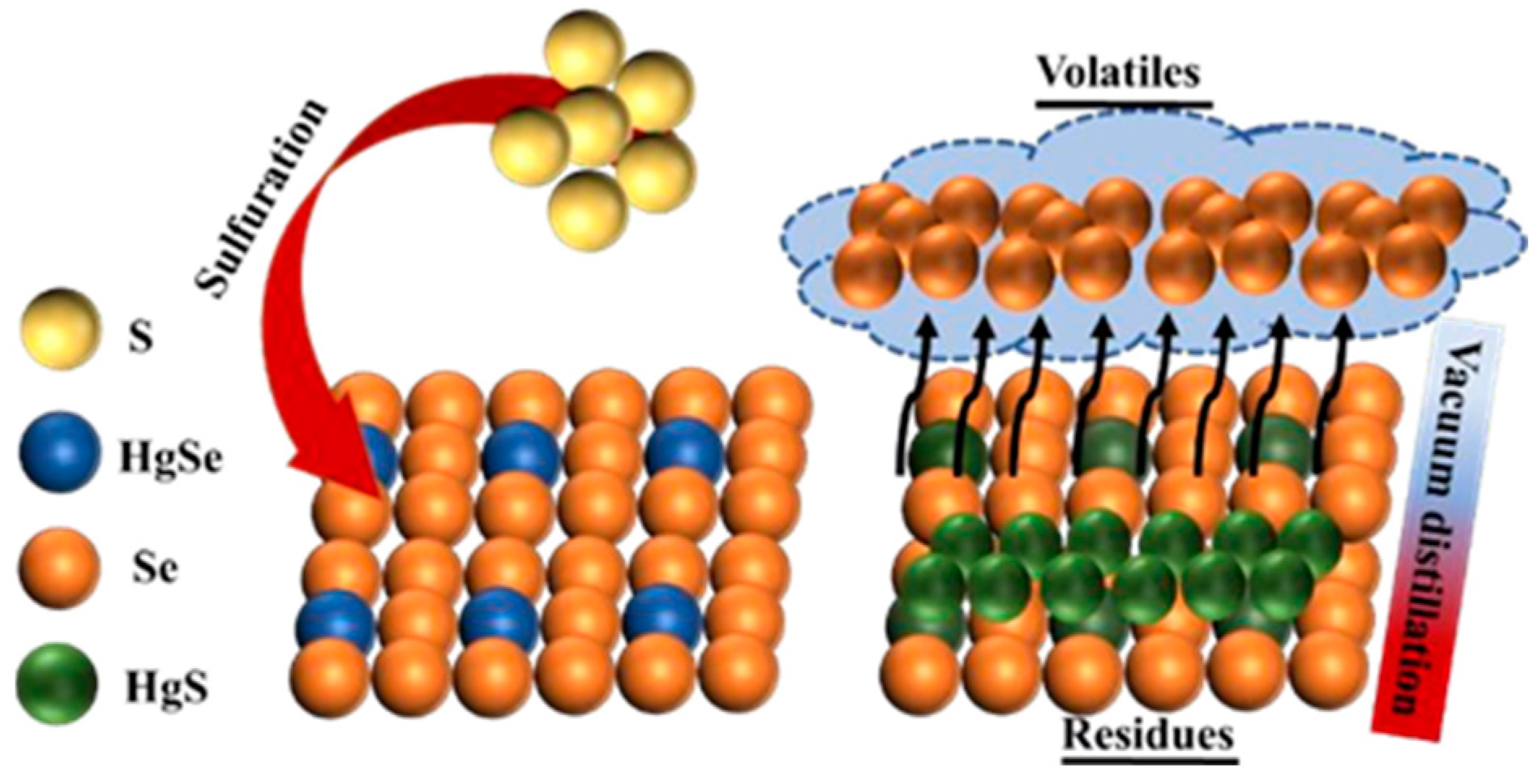
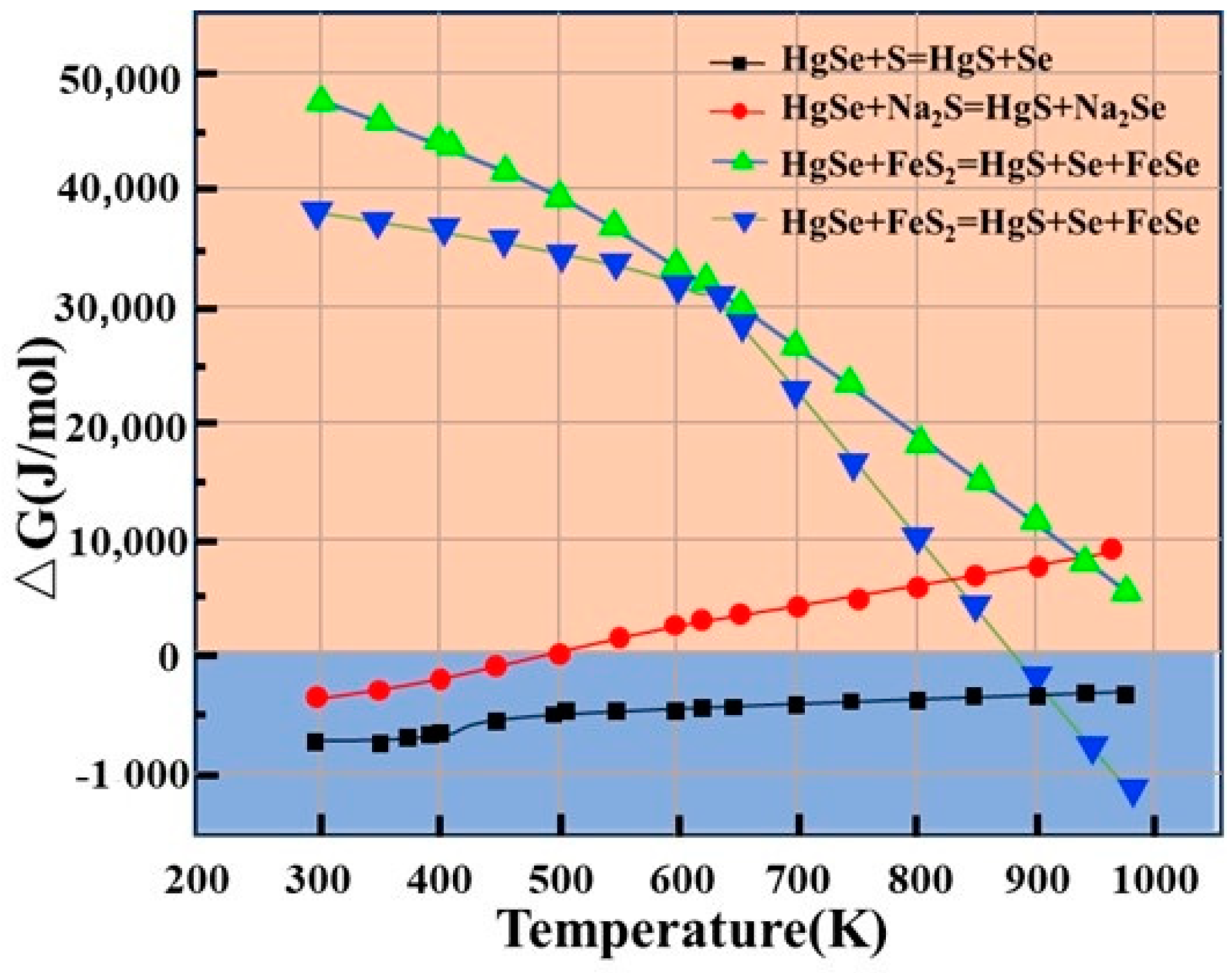
3.1.1. Sulfidation Process
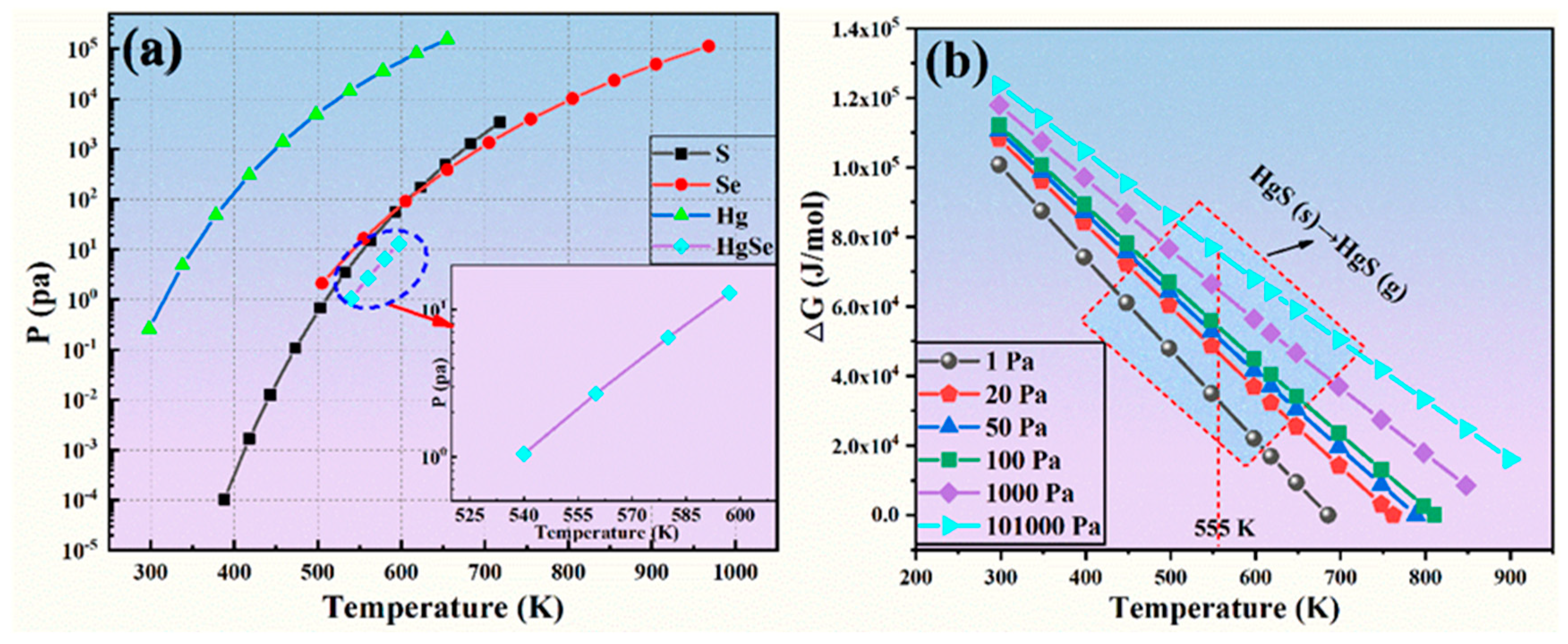
3.1.2. Vacuum Distillation Process
3.2. Experimental Analysis
3.2.1. Selection of Sulfidizing Agent
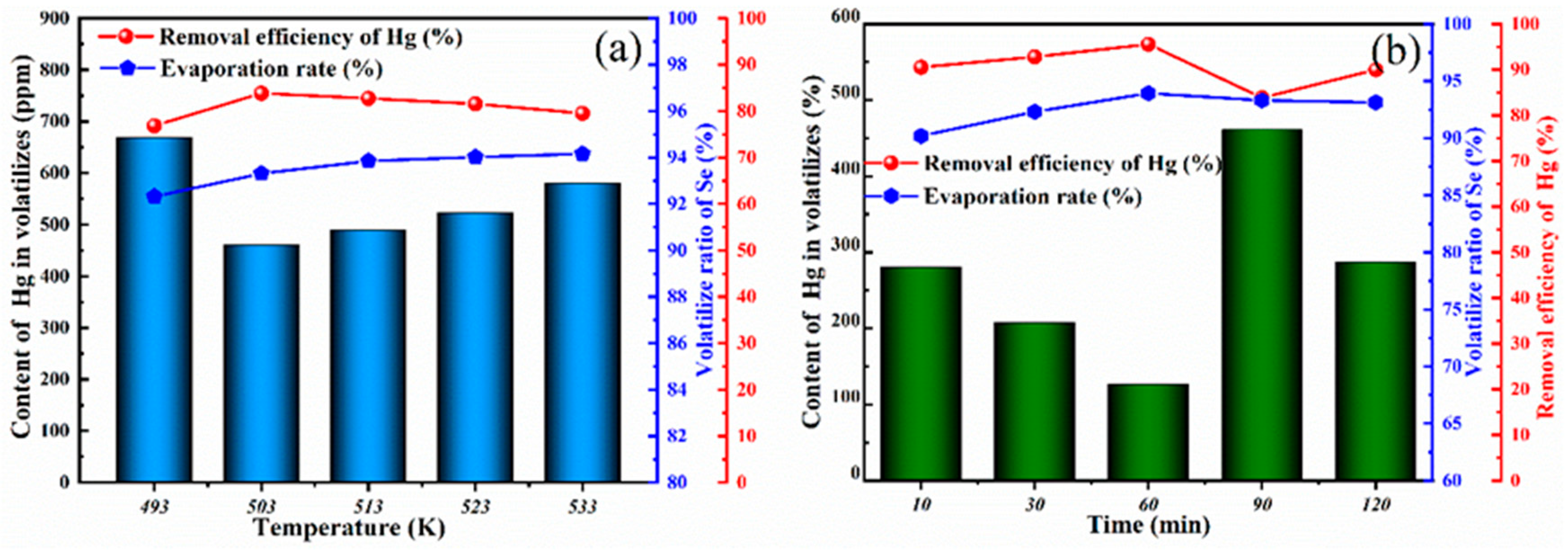
3.2.2. Analysis of Hg Removal Efficiency
3.2.3. Secondary Vacuum Distillation Purification
4. Conclusions
- (1)
- Theoretical analysis shows that the sulfuration reaction potential energy of S, Na2S, and FeS2 for mercury (HgSe) in crude selenium is in the order S > Na2S > FeS2, and the Hg removal after sulfuration that is used to change the phase of mercury is far greater than only vacuum distillation.
- (2)
- Through the experiments, it was found that S was the best curing agent, and vacuum distillation of the curing product could effectively reduce the mercury content of Se to 523 ppm.
- (3)
- A comprehensive analysis shows that the maximum removal rate of mercury was 97.49%, 0.32% of the mercury in the crude selenium was sulfurized, and secondary distillation resulted in a final amount of 88.52 ppm mercury in the volatile matter. The research results have practical value for the separation and purification of selenium and mercury from hazardous wastes.
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liu, D.; Wang, D.; Hong, T.; Wang, Z.; Wang, Y.; Qin, Y.; Su, L.; Yang, T.; Gao, X.; Ge, Z.; et al. Lattice plainification advances highly effective SnSe crystalline thermoelectrics. Science 2023, 380, 841–846. [Google Scholar] [CrossRef]
- Zhao-Karger, Z.; Lin, X.-M.; Minella, C.B.; Wang, D.; Diemant, T.; Behm, R.J.; Fichtner, M. Selenium and selenium-sulfur cathode materials for high-energy rechargeable magnesium batteries. J. Power Sources 2016, 323, 213–219. [Google Scholar] [CrossRef]
- Chaudhary, S.; Umar, A.; Mehta, S. Selenium nanomaterials: An overview of recent developments in synthesis, properties and potential applications. Prog. Mater. Sci. 2016, 83, 270–329. [Google Scholar] [CrossRef]
- Staicu, L.C.; Van Hullebusch, E.D.; Lens, P.N. Lens, Production, recovery and reuse of biogenic elemental selenium. Environ. Chem. Lett. 2015, 13, 89–96. [Google Scholar] [CrossRef]
- Yu, J.G.; Yue, B.Y.; Wu, X.W.; Liu, Q.; Jiao, F.P.; Jiang, X.Y.; Chen, X.Q. Removal of mercury by adsorption: A review. Environ. Sci. Pollut. Res. 2016, 23, 5056–5076. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.R.; Eom, Y.; Lee, T.G. Mercury recovery from mercury-containing wastes using a vacuum thermal desorption system. Waste Manag. 2017, 60, 546–551. [Google Scholar] [CrossRef] [PubMed]
- Gavrilescu, M. Microbial recovery of critical metals from secondary sources. Bioresour. Technol. 2022, 344, 126208. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Wu, Y.; Tang, A.; Pan, D.; Li, B. Recovery of scattered and precious metals from copper anode slime by hydrometallurgy: A review. Hydrometallurgy 2020, 197, 105460. [Google Scholar] [CrossRef]
- Feng, Q.; Li, A.J.; An, Y.F.; Zhang, B.; Zhao, Y.Q. Research Development on Separation and Extraction of Selenium and Mercury. Solid State Phenom. 2022, 330, 161–168. [Google Scholar] [CrossRef]
- Xing, P.; Ma, B.; Wang, C.; Chen, Y. Cleaning of lead smelting flue gas scrubber sludge and recovery of lead, selenium and mercury by the hydrometallurgical route. Environ. Technol. 2018, 39, 1461–1469. [Google Scholar] [CrossRef] [PubMed]
- Fu, M.; Bai, C.; Chen, G. Research on acidic Se-Hg slime treatment with POX alkaline leaching. Chin. Nonferr. Metall. 2022, 5, 51. (In Chinese) [Google Scholar]
- Yang, W.; Dai, L.; Yang, K. Research on Microwave Enhancement of Zinc Smelting Acid Sludge by Calcium Addition and Selenium Fixing. Yunnan Metall. 2021, 5, 50. (In Chinese) [Google Scholar]
- Vereecken, J.; D’Hondt, D.; Gérain, N.; Van Lierde, A. Selective recovery by precipitation of selenium and mercury from acid leaching solutions. In EMC’91: Non-Ferrous Metallurgy—Present and Future; Springer: Berlin/Heidelberg, Germany, 1991; pp. 89–95. [Google Scholar]
- He, B.; Wang, W.; Jiang, W.; Xu, B.; Yang, H. Removing of Fe, Pb and Hg from Crude Selenium by Fractional Crystallization. Metals 2023, 13, 739. [Google Scholar] [CrossRef]
- Luo, H.; Jiang, W.; Zha, G.; Liu, L.; Zhen, T.; Yang, B.; Xu, B. Removal of impurity Pb during crude selenium purification by controlling potential oxidation and vacuum distillation. Vacuum 2022, 195, 110674. [Google Scholar] [CrossRef]
- Li, Z.; Deng, J.; Liu, D.; Jiang, W.; Zha, G.; Huang, D. Waste-free separation and recovery of copper telluride slag by directional sulfidation-vacuum distillation. J. Clean. Prod. 2022, 335, 130356. [Google Scholar] [CrossRef]
- Stinn, C.; Allanore, A. Selective sulfidation of metal compounds. Nature 2022, 602, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Yongnian, D.; Bin, Y. Vacuum Metallurgy of Nonferrous Metal Materials; Metallurgical Industry Press: Beijing, China, 2000. (In Chinese) [Google Scholar]

| Element | Se | Hg | Pb | Cu | Fe | S | Au | Ag |
|---|---|---|---|---|---|---|---|---|
| Content/% | 96.50 | 0.32 | 2.02 | 0.042 | 0.05 | 0.2 | 0.0027 | 0.068 |
| Number | Reaction (298–1000 K) |
|---|---|
| R-1 | HgSe(s) + S(s,l,g) = HgS(s) + Se(s,l,g) |
| R-2 | HgSe(s) + Na2S(s) = HgS(s) + Na2Se(s) |
| R-3 | HgSe(s) + FeS2() = HgS(s) + Se(s,l,g) + FeS(s) |
| R-4 | HgSe(s) = Se(s,l,g) + Hg(l,g) |
| Sulfidizing Agents | Free | S | FeS2 | Na2S | |
|---|---|---|---|---|---|
| Input material | Crude selenium (mass/g) | 20.05 | 20.07 | 20.01 | 20.17 |
| Sulfidizing agent (mass/g) | / | 4.03 | 4.06 | 4.01 | |
| Output material | Residues (mass/g) | 1.48 | 1.44 | 5.61 | 3.67 |
| Volatiles (mass/g) | 18.57 | 22.66 | 18.60 | 20.51 | |
| Experimental results | Content of Hg in volatiles (ppm) | 1226 | 523 | 2136 | 1939 |
| Evaporation rate (%) | 92.62 | 94.02 | 77.27 | 84.82 | |
| Hg removal efficiency (%) | 64.52 | 81.55 | 37.95 | 38.38 |
| Experimental Parameters | Final Result | ||
|---|---|---|---|
| Mass of volatile matter obtained from input/g | 18.41 | Evaporation rate/% | 80.17 |
| Temperature/K | 503 | ||
| Time/min | 60 | Secondary removal efficiency of Hg/% | 43.64 |
| Mass of secondary volatiles/g | 14.76 | ||
| Mass of secondary residues/g | 0.20 | Total removal efficiency of Hg/% | 97.49 |
| Content of Hg in secondary volatiles/ppm | 88.52 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zha, G. Combined Sulfidation and Vacuum Distillation for the Directional Removal of Hazardous Mercury from Crude Selenium. Metals 2023, 13, 1795. https://doi.org/10.3390/met13111795
Zha G. Combined Sulfidation and Vacuum Distillation for the Directional Removal of Hazardous Mercury from Crude Selenium. Metals. 2023; 13(11):1795. https://doi.org/10.3390/met13111795
Chicago/Turabian StyleZha, Guozheng. 2023. "Combined Sulfidation and Vacuum Distillation for the Directional Removal of Hazardous Mercury from Crude Selenium" Metals 13, no. 11: 1795. https://doi.org/10.3390/met13111795





