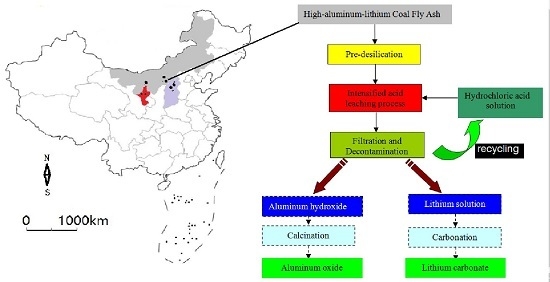
An Efficient Approach for Lithium and Aluminum Recovery from Coal Fly Ash by Pre-Desilication and Intensified Acid Leaching Processes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.2. Pre-Desilication Experiments
2.3. Intensified Acid Leaching Processes
3. Results and Discussion
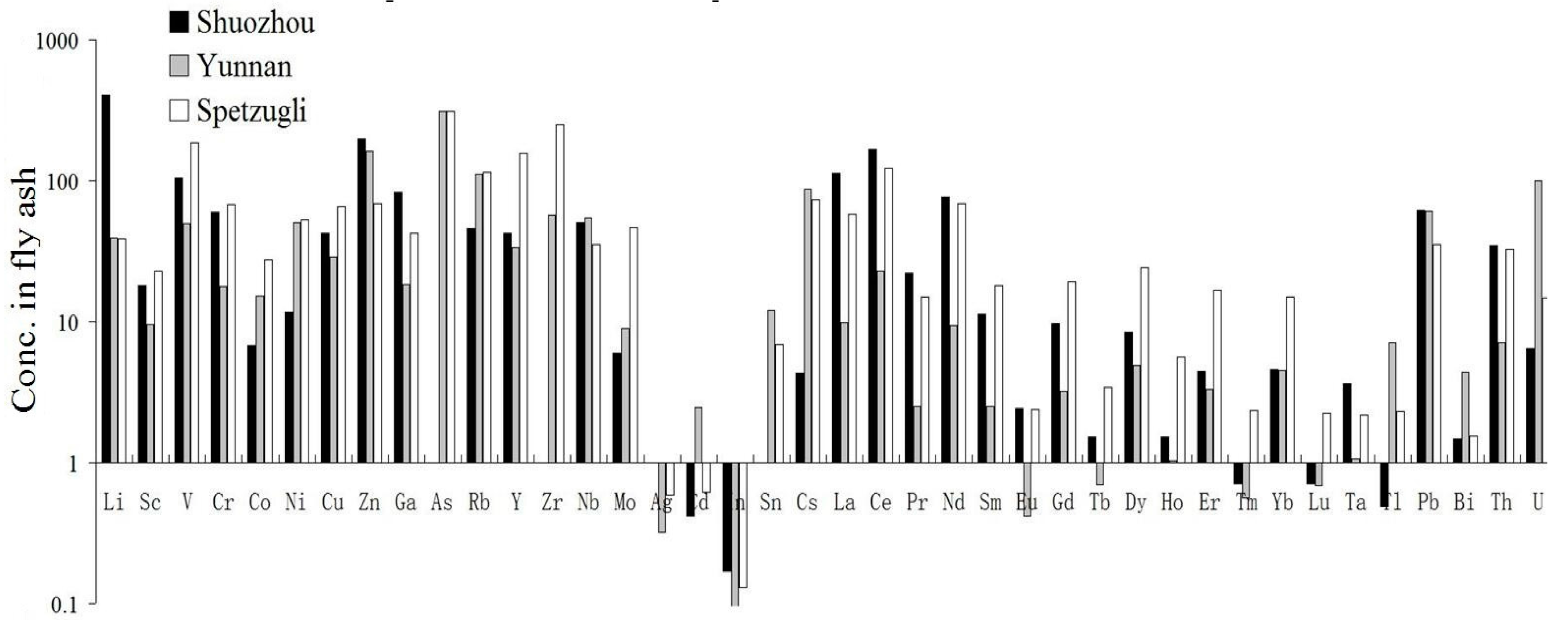
3.1. CFA Analysis
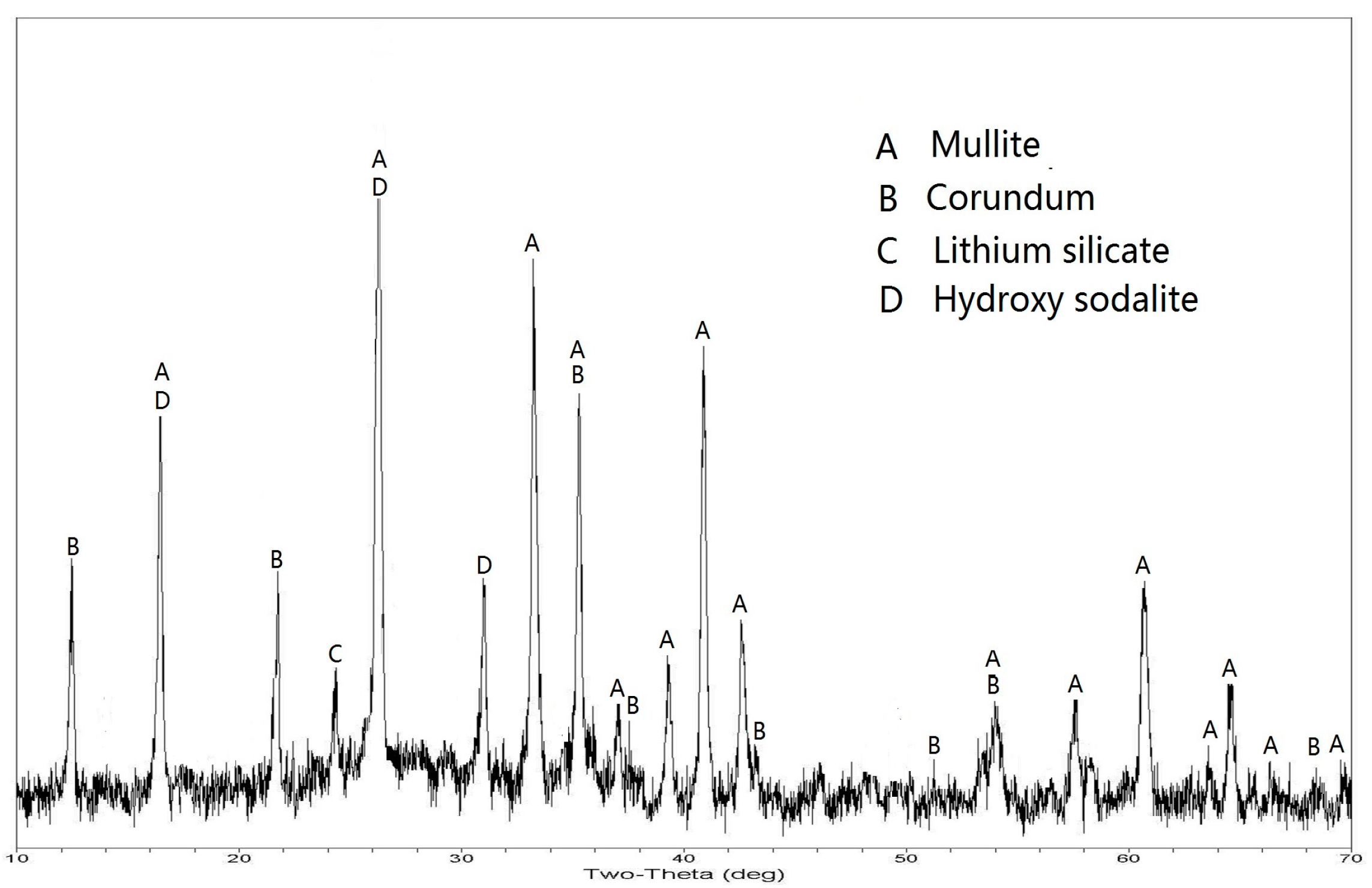
3.2. Pre-Desilication of the CFA
3.3. Intensified Acid Leaching Processes
3.3.1. Effect of Acid Species
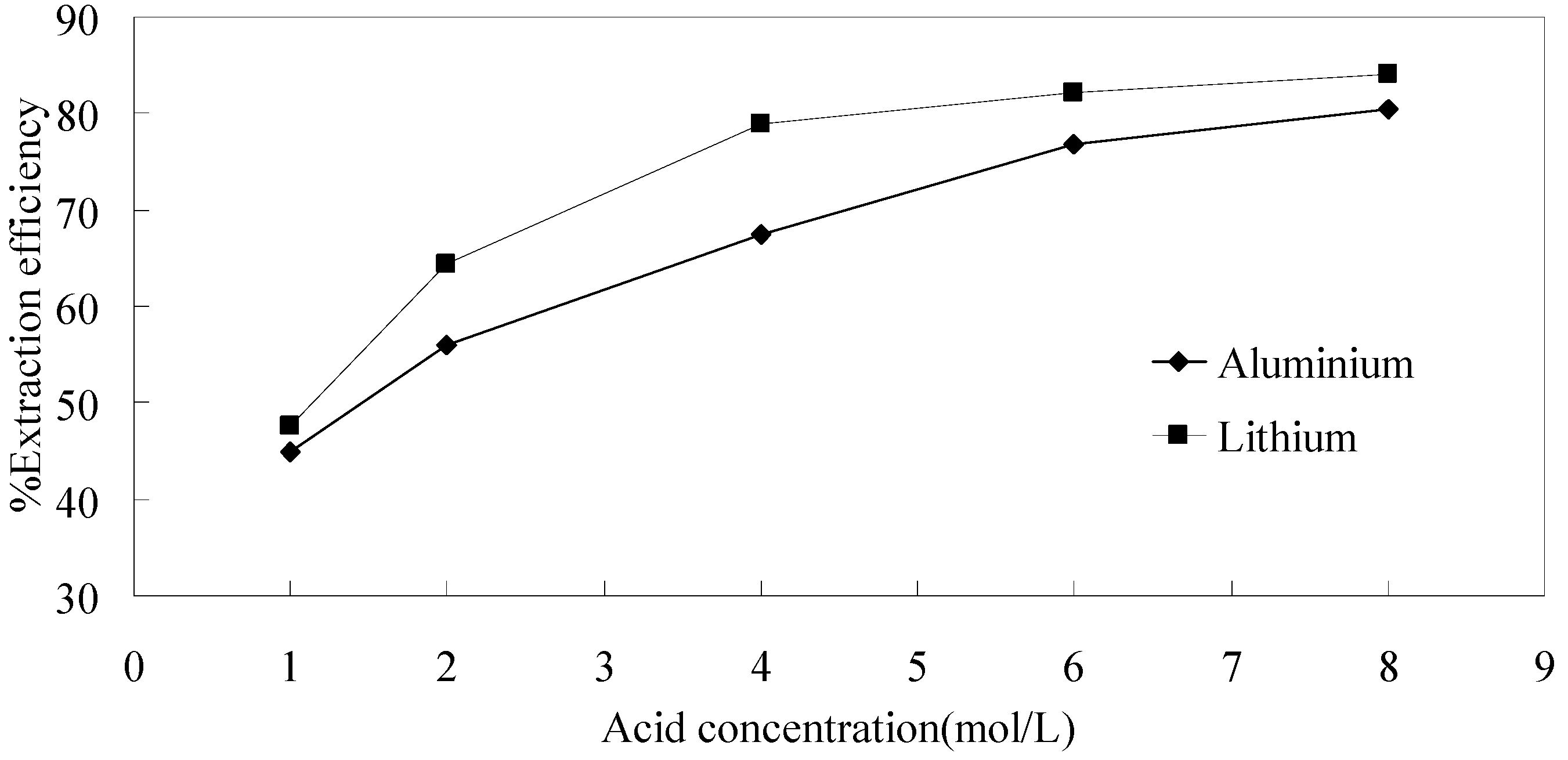
3.3.2. Effect of Hydrochloric Acid Concentration
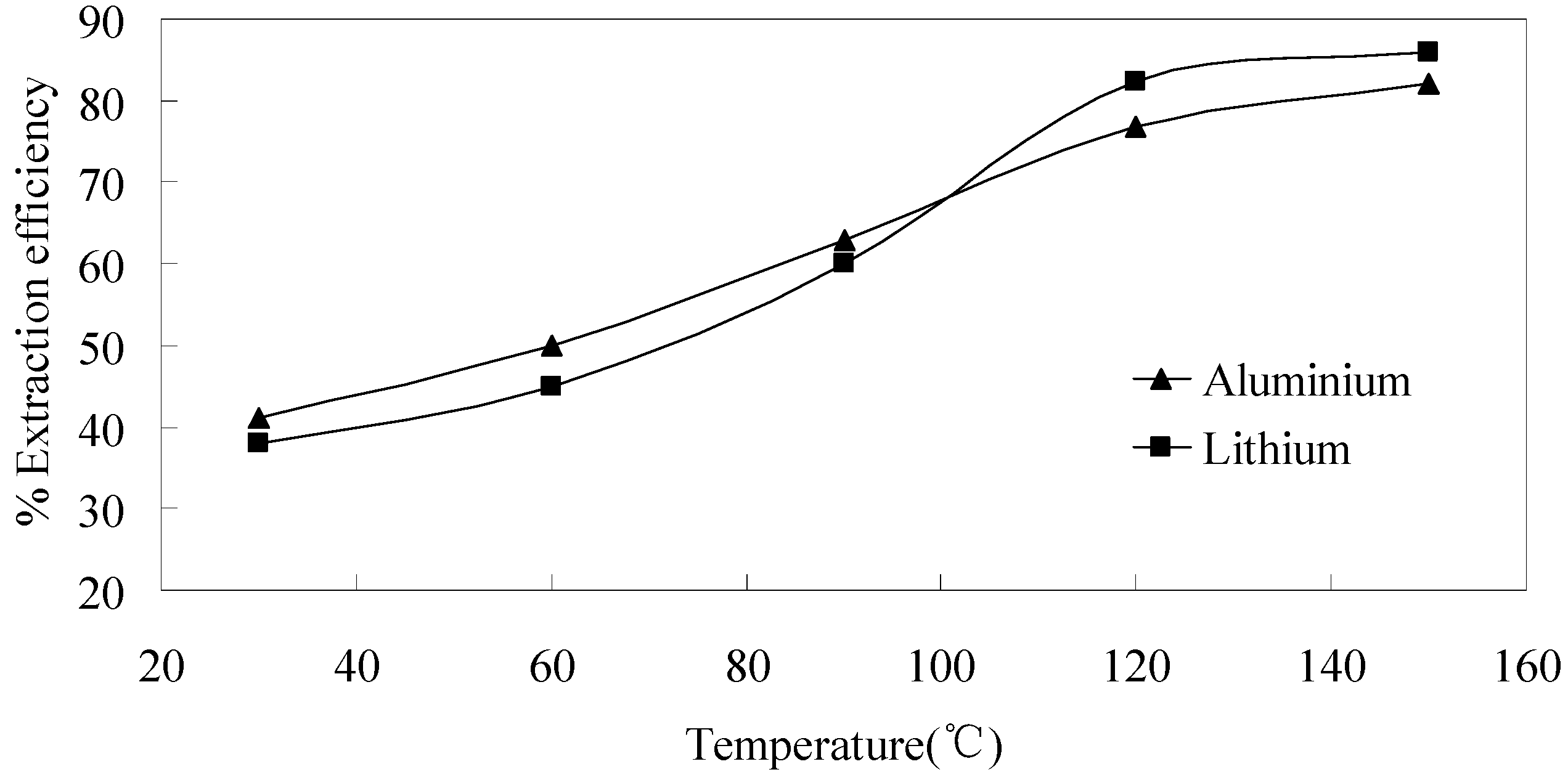
3.3.3. Effect of Temperature
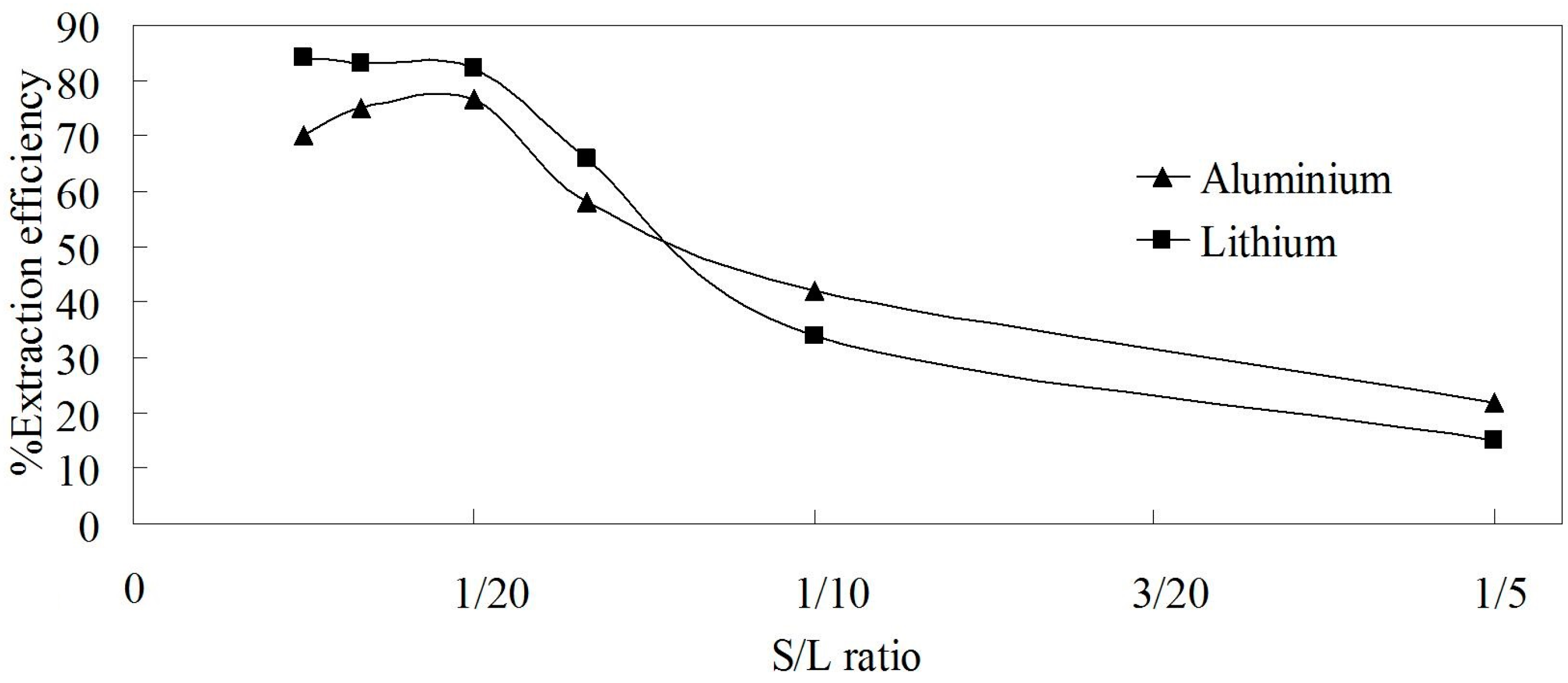
3.3.4. Effect of S/L Ratio
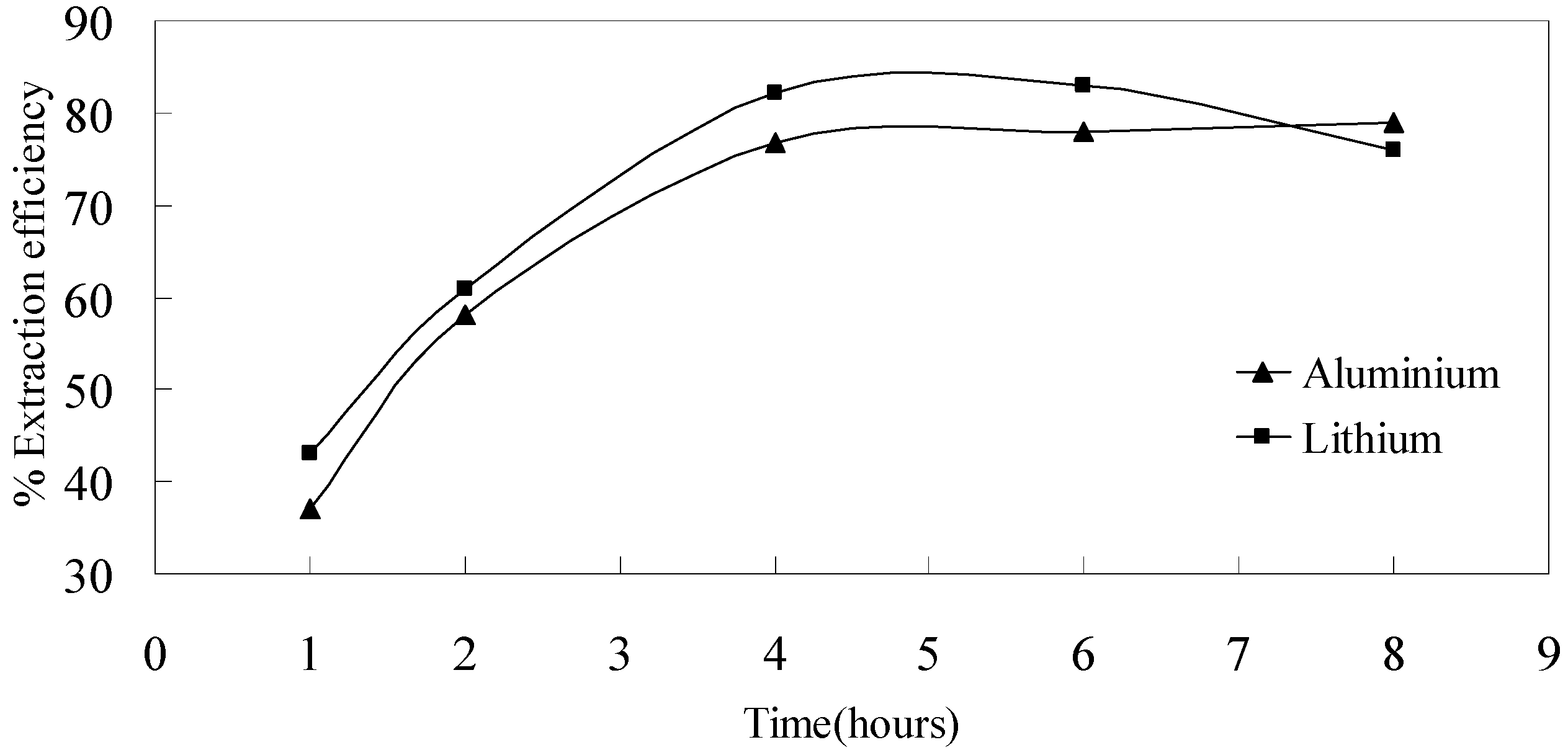
3.3.5. Effect of Time
3.4. Characterization of Lithium and Aluminum Extraction under Optimum Conditions
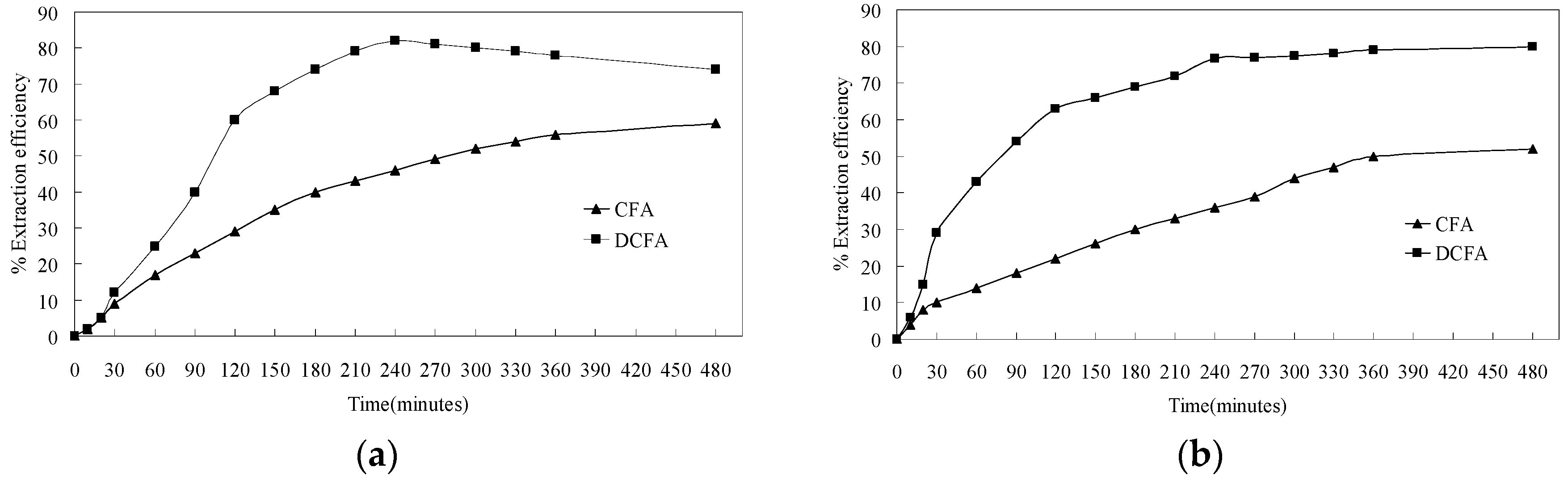
3.4.1. Extraction Efficiency of Lithium and Aluminum from CFA and DCFA
3.4.2. Morphological Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Li, J.F.; Dong, H.; Sun, J.; Nie, J.H.; Zhang, S.Y.; Tang, J.S.; Chen, Z.H. Composition profiles and health risk of PCDD/F in outdoor air and fly ash from municipal solid waste incineration and adjacent villages in East China. Sci. Total Environ. 2016, 571, 876–882. [Google Scholar] [CrossRef] [PubMed]
- Meawad, A.S.; Bojinova, D.Y.; Pelovski, Y.G. Review: An overview of metals recovery from thermal power plant solid wastes. Waste Manag. 2010, 30, 2548–2559. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.F.; DaBoit, K.; Sampaio, C.H.; Jasper, A.; Andrade, M.L.; Kostova, I.J.; Waanders, F.B.; Henke, K.R.; Hower, J.C. The occurrence of hazardous volatile elements and nanoparticles in Bulgarian coal fly ashes and the effect on human health exposure. Sci. Total Environ. 2012, 416, 513–526. [Google Scholar] [CrossRef] [PubMed]
- Komonweeraket, K.; Cetin, B.; Benson, C.H.; Aydilek, A.H.; Edil, T.B. Leaching characteristics of toxic constituents from coal fly ash mixed soils under the influence of pH. Waste Manag. 2015, 38, 174–184. [Google Scholar] [CrossRef] [PubMed]
- Qin, S.J.; Sun, Y.Z.; Li, Y.H.; Wang, J.X.; Zhao, C.L.; Gao, K. Coal deposits as promising alternative sources for gallium. Earth-Sci. Rev. 2015, 150, 95–101. [Google Scholar] [CrossRef]
- Sun, J.M.; Zhang, Z.J.; Chen, G.; Yan, S.Y.; Huo, Q.Z.; Wu, L.C.; Xu, H.L.; Qin, L.; Chen, X.X. Method for Co-Producing Alumina and Activated Calcium Silicate from High-Alumina Fly Ash. U.S. Patent 9,139,445, 22 September 2015. [Google Scholar]
- Dai, S.F.; Zhao, L.; Peng, S.P.; Chou, C.L.; Wang, X.B.; Zhang, Y.; Li, D.; Sun, Y.Y. Abundances and distribution of minerals and elements in high-aluminum coal fly ash from the Jungar Power Plant, Inner Mongolia, China. Int. J. Coal Geol. 2010, 81, 320–332. [Google Scholar] [CrossRef]
- Sun, Y.Z.; Zhao, C.L.; Qin, S.J.; Xiao, L.; Li, Z.S.; Lin, M.Y. Occurrence of some valuable elements in the unique ‘high-aluminium coals’ from the Jungar coalfield, China. Ore Geol. Rev. 2016, 72, 659–668. [Google Scholar] [CrossRef]
- Dai, S.F.; Zhao, L.; Hower, J.C.; Johnston, M.N.; Song, W.J.; Wang, P.P.; Zhang, S.F. Petrology, mineralogy, and chemistry of size-fractioned fly ash from the Jungar power plant, Inner Mongolia, China, with emphasis on the distribution of rare earth elements. Energy Fuels 2014, 28, 1502–1514. [Google Scholar] [CrossRef]
- Wang, J.X.; Wang, Q.; Tian, L. Characteristics of trace elements of the No. 9 coal seam from the Anjialing Mine, Ningwu coalfield, China. Chin. J. Geochem. 2015, 34, 391–400. [Google Scholar]
- Dai, S.F.; Seredin, V.V.; Ward, C.R.; Jiang, J.H.; Hower, J.C.; Song, X.L.; Jiang, Y.F.; Wang, X.B.; Gornostaeva, T.; Li, X.; et al. Composition and modes of occurrence of minerals and elements in coal combustion products derived from high-Ge coals. Int. J. Coal Geol. 2014, 121, 79–97. [Google Scholar] [CrossRef]
- Qin, S.J.; Zhao, C.L.; Li, Y.H.; Zhang, Y. Review of coal as a promising source of lithium. Int. J. Oil Gas Coal Technol. 2015, 9, 215–229. [Google Scholar] [CrossRef]
- Shemi, A.; Mpana, R.N.; Ndlovu, S.; Dyk, L.D.; Sibanda, V.; Seepe, L. Alternative techniques for extracting aluminum from coal fly ash. Miner. Eng. 2012, 34, 30–37. [Google Scholar] [CrossRef]
- Xu, D.H.; Li, H.Q.; Bao, W.J.; Wang, C.Y. A new process of extracting aluminum from high-aluminum coal fly ash in NH4HSO4 + H2SO4 mixed solution. Hydrometallurgy 2016, 165, 336–344. [Google Scholar] [CrossRef]
- Shemi, A.; Ndlovu, S.; Sibanda, V.; Dyk, L.D. Extraction of aluminum from coal fly ash using an acid leach-sinter-acid leach technique. Hydrometallurgy 2015, 157, 348–355. [Google Scholar] [CrossRef]
- Izquierdo, M.; Koukouzas, N.; Touliou, S.; Panopoulos, K.D.; Querol, X.; Itskos, G. Geochemical controls on leaching of lignite-fired combustion by-products from Greece. Appl. Geochem. 2011, 26, 1599–1606. [Google Scholar] [CrossRef]
- Li, G.H.; You, Z.X.; Sun, H.; Sun, R.; Peng, Z.W.; Zhang, Y.B.; Jiang, T. Separation of rhenium from lead-rich molybdenite concentrate via hydrochloric acid leaching followed by oxidative roasting. Metals 2016, 6, 282. [Google Scholar] [CrossRef]
- Tripathy, A.K.; Sarangi, C.K.; Tripathy, B.C.; Sanjay, K.; Bhattacharya, I.N.; Mahapatra, B.K.; Behera, P.K.; Satpathy, B.K. Aluminum recovery from NALCO fly ash by acid digestion in the presence of fluoride ion. Int. J. Miner. Process. 2015, 138, 44–48. [Google Scholar] [CrossRef]
- Zhang, R.; Zheng, S.L.; Ma, S.H.; Zhang, Y. Recovery of alumina and alkali in Bayer red mud by the formation of andradite-grossular hydrogarnet in hydrothermal process. J. Hazard. Mater. 2011, 189, 827–835. [Google Scholar] [CrossRef] [PubMed]
- Li, H.Q.; Hui, J.B.; Wang, C.Y.; Bao, W.J.; Sun, Z.H. Extracion of alumina from coal fly ash by mixed-alkaline hydrothermal method. Hydrometallurgy 2014, 147–148, 183–187. [Google Scholar] [CrossRef]
- Ding, J.; Ma, S.H.; Zheng, S.L.; Zhang, Y.; Xie, Z.L.; Shen, S.; Liu, Z.K. Study of extracting alumina from high-alumina PC fly ash by a hydro-chemical process. Hydrometallurgy 2016, 161, 58–64. [Google Scholar] [CrossRef]
- Matjie, R.H.; Bunt, J.R.; Heerden, J.H.P. Extraction of aluminum from coal fly ash generated from a selected low rank bituminous South African coal. Miner. Eng. 2005, 18, 299–310. [Google Scholar] [CrossRef]
- Bai, G.H.; Teng, W.; Wang, X.G.; Qin, J.G.; Xu, P.; Li, P.C. Alkali desilicated coal fly ash as substitute of bauxite in lime-soda sintering process for aluminum production. Trans. Nonferr. Met. Soc. China 2010, 20, 169–175. [Google Scholar] [CrossRef]
- Li, L.S.; Liao, X.Q.; Wu, Y.S.; Liu, Y.Y. Extracting aluminum from coal fly ash with ammonium sulfate sintering process. In Light Metals; Springer: New York, NY, USA, 2012; Volume 510, pp. 213–217. [Google Scholar]
- Vu, H.; Bernardi, J.; Jandová, J.; Vaculíková, L.; Goliáš, V. Lithium and rubidium extraction from zinnwaldite by alkali digestion process: Sintering mechanism and leaching kinetics. Int. J. Miner. Process. 2013, 123, 9–17. [Google Scholar] [CrossRef]
- Yao, Z.T.; Xia, M.S.; Sarker, P.K.; Chen, T. A review of the aluminum recovery from coal fly ash, with a focus in China. Fuel 2014, 120, 74–85. [Google Scholar] [CrossRef]




| Content/% | SiO2 | Al2O3 | CaO | Fe2O3 | TiO2 | P2O5 | MgO | Li2O | Na2O | MnO | L.O.I |
|---|---|---|---|---|---|---|---|---|---|---|---|
| CFA | 44.12 | 42.17 | 2.44 | 2.43 | 1.67 | 0.69 | 0.68 | 0.20 | 0.14 | 0.02 | 1.41 |
| DCFA | 34.30 | 49.88 | 1.98 | 3.07 | 1.81 | 0.08 | 0.81 | 0.22 | 8.74 | 0.02 | 0.22 |
| Factors | Leaching Condition | |||
|---|---|---|---|---|
| Acid Concentration (mol/L) | Leaching Temperature (°C) | S/L Ratio | Leaching Time (h) | |
| Acid species | 6 | 120 | 1:20 | 4 |
| Acid concentration | 1, 2, 4, 6, 8 | 120 | 1:20 | 4 |
| Temperature | 6 | 30, 60, 90, 120, 150 | 1:20 | 4 |
| S/L ratio | 6 | 120 | 1:5, 1:10, 1:15, 1:20, 1:30, 1:40 | 4 |
| Time | 6 | 120 | 1:20 | 0.5, 1, 2, 4, 6, 8 |
| Phase | Mullite | Quartz | Corundum | Lithium Silicate | Glass Phase | Hydroxy Sodalite | Amphodelite | Calcite |
|---|---|---|---|---|---|---|---|---|
| CFA | 65.3 | 3.7 | 13.2 | 0.9 | 12.4 | - | - | 1.2 |
| DCFA | 63.9 | 2.4 | 12.1 | 0.8 | - | 9.1 | 2.6 | 1.6 |
| Content/% | SiO2 | Al2O3 | CaO | Fe2O3 | TiO2 | MgO | Li2O |
|---|---|---|---|---|---|---|---|
| CFA | 44.12 | 42.17 | 2.44 | 2.43 | 1.67 | 0.68 | 0.20 |
| DCFA | 34.30 | 49.88 | 1.98 | 3.07 | 1.81 | 0.81 | 0.22 |
| Leaching residue | 76.81 | 9.84 | 0.31 | 0.26 | 1.31 | 0.12 | 0.02 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, S.; Qin, S.; Kang, L.; Liu, J.; Wang, J.; Li, Y. An Efficient Approach for Lithium and Aluminum Recovery from Coal Fly Ash by Pre-Desilication and Intensified Acid Leaching Processes. Metals 2017, 7, 272. https://doi.org/10.3390/met7070272
Li S, Qin S, Kang L, Liu J, Wang J, Li Y. An Efficient Approach for Lithium and Aluminum Recovery from Coal Fly Ash by Pre-Desilication and Intensified Acid Leaching Processes. Metals. 2017; 7(7):272. https://doi.org/10.3390/met7070272
Chicago/Turabian StyleLi, Shenyong, Shenjun Qin, Lianwei Kang, Jianjun Liu, Jing Wang, and Yanheng Li. 2017. "An Efficient Approach for Lithium and Aluminum Recovery from Coal Fly Ash by Pre-Desilication and Intensified Acid Leaching Processes" Metals 7, no. 7: 272. https://doi.org/10.3390/met7070272