Ultrastructural Alterations of the Human Pathogen Giardia intestinalis after Drug Treatment
Abstract
1. Introduction
Giardia Intestinalis and Its Biological Cycle
2. Morphology of the Two Stages in the Life Cycle of Giardia intestinalis as Observed Using Light and Electron Microscopy
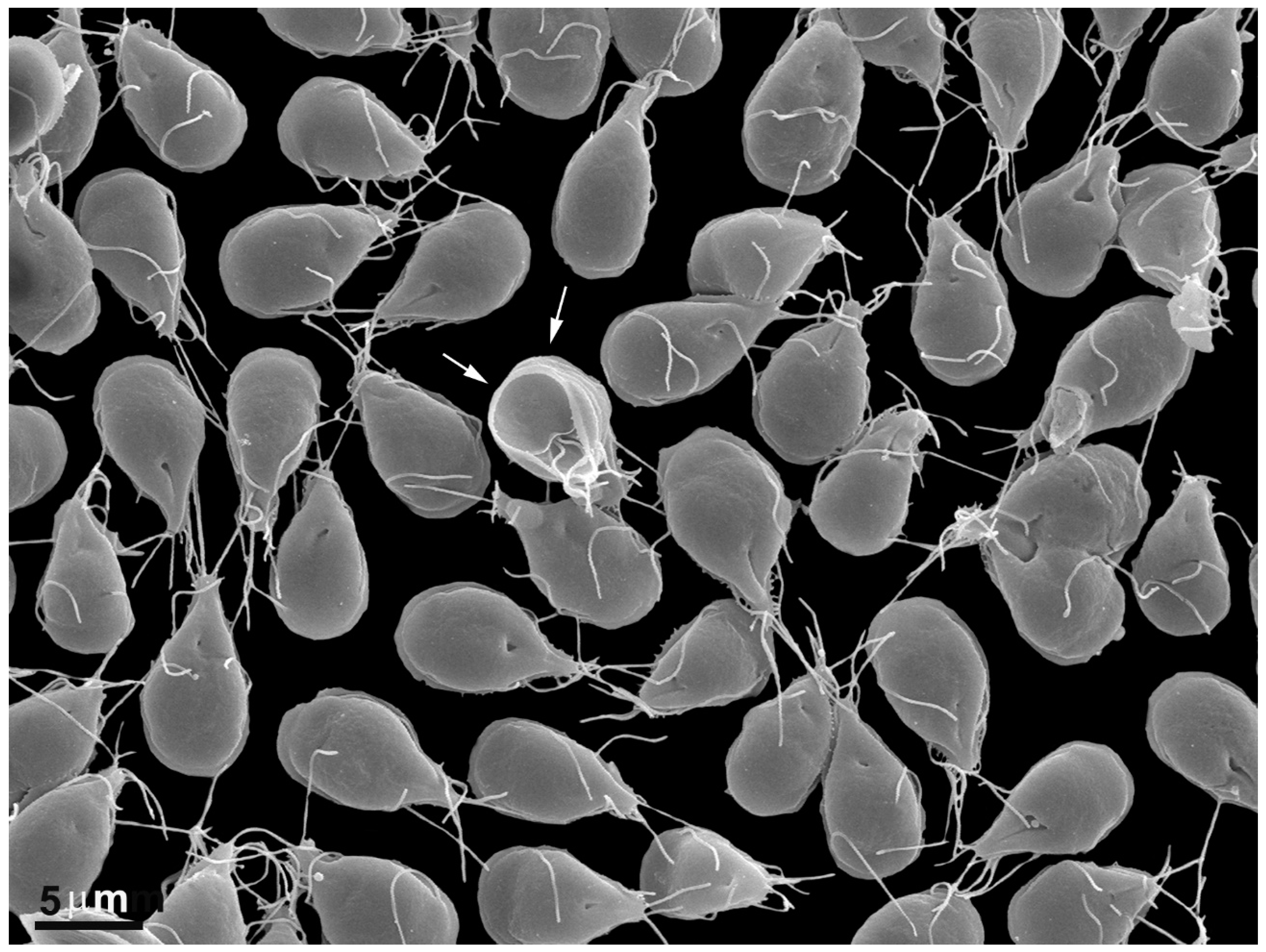
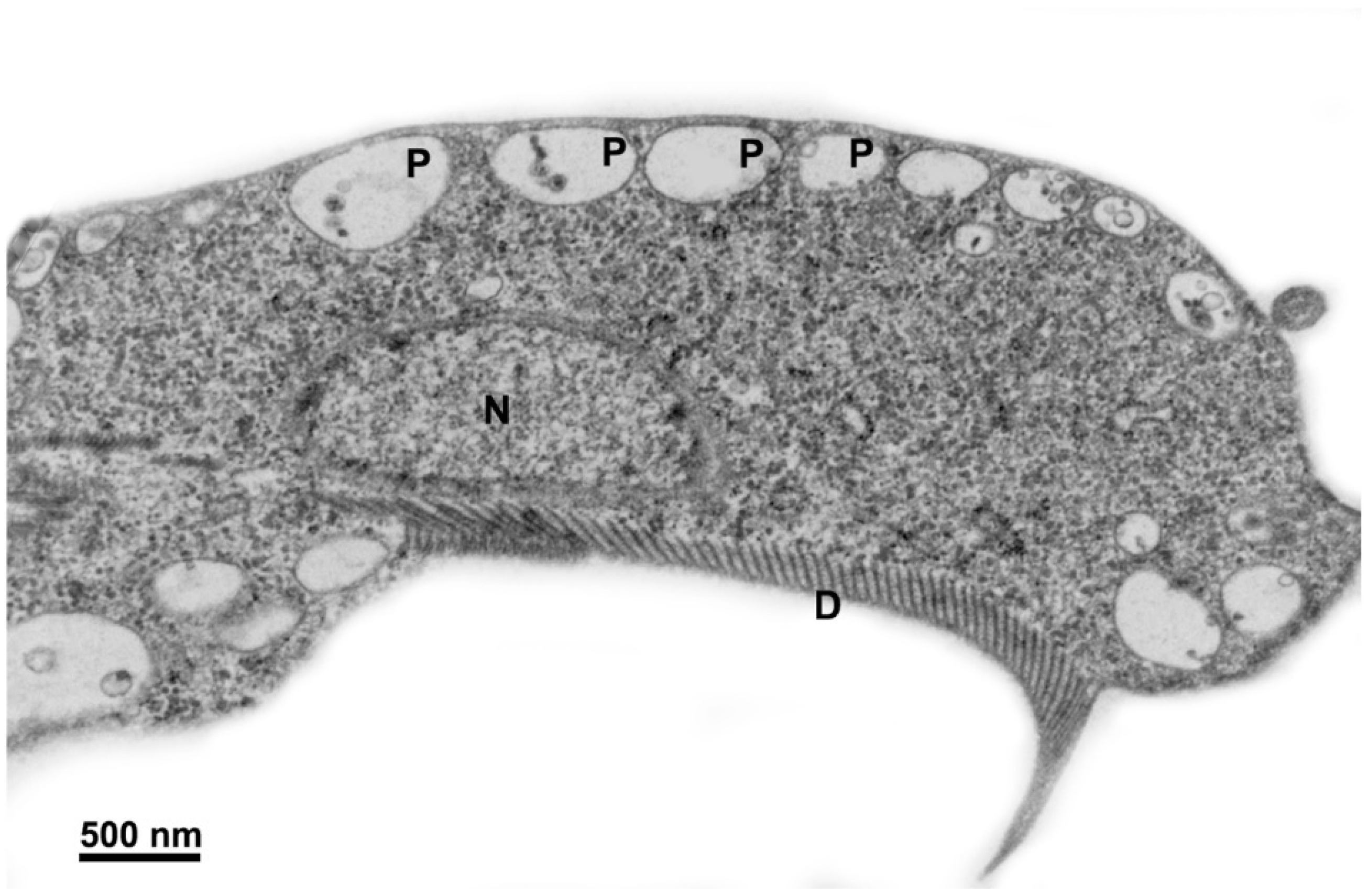
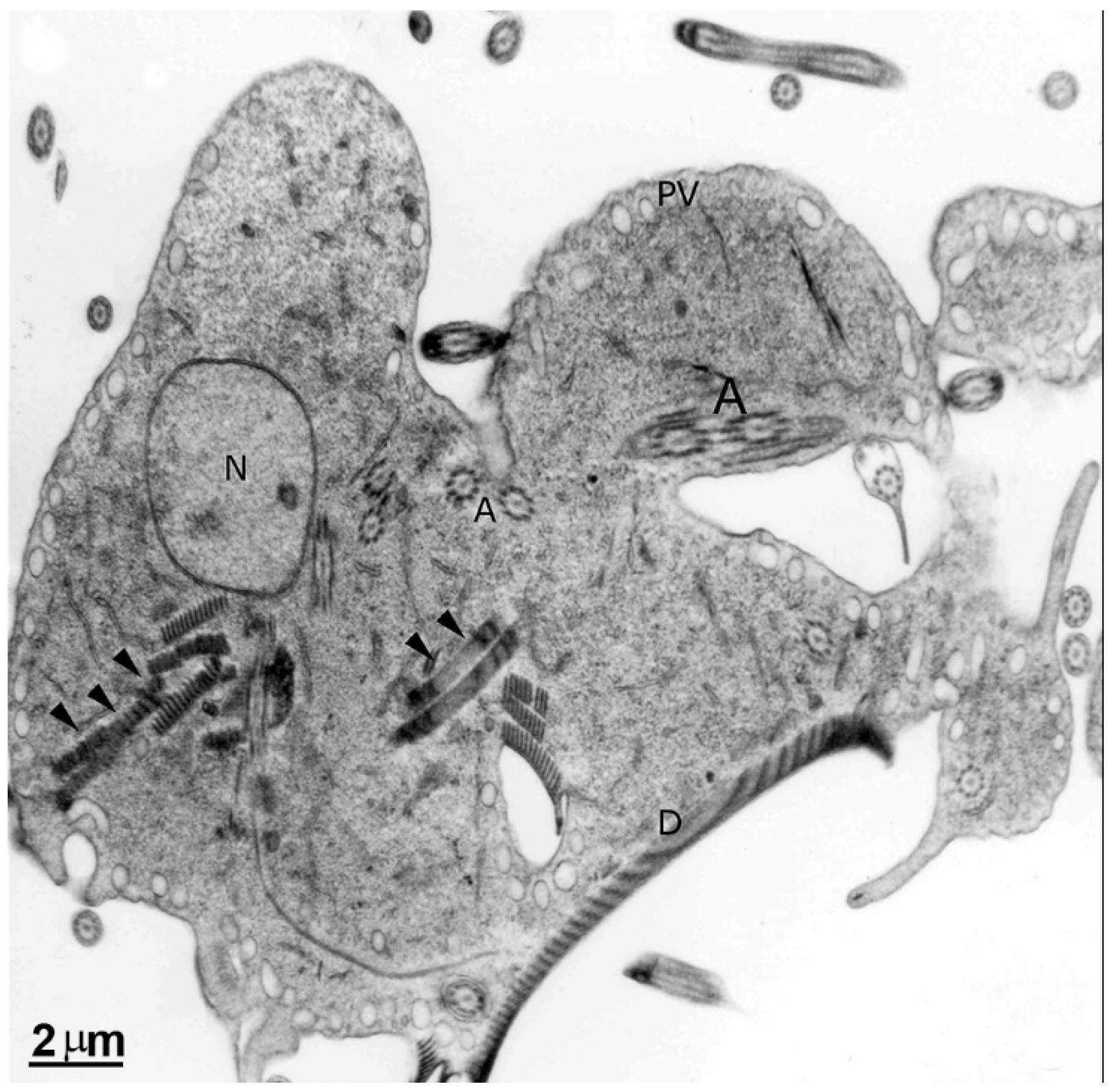
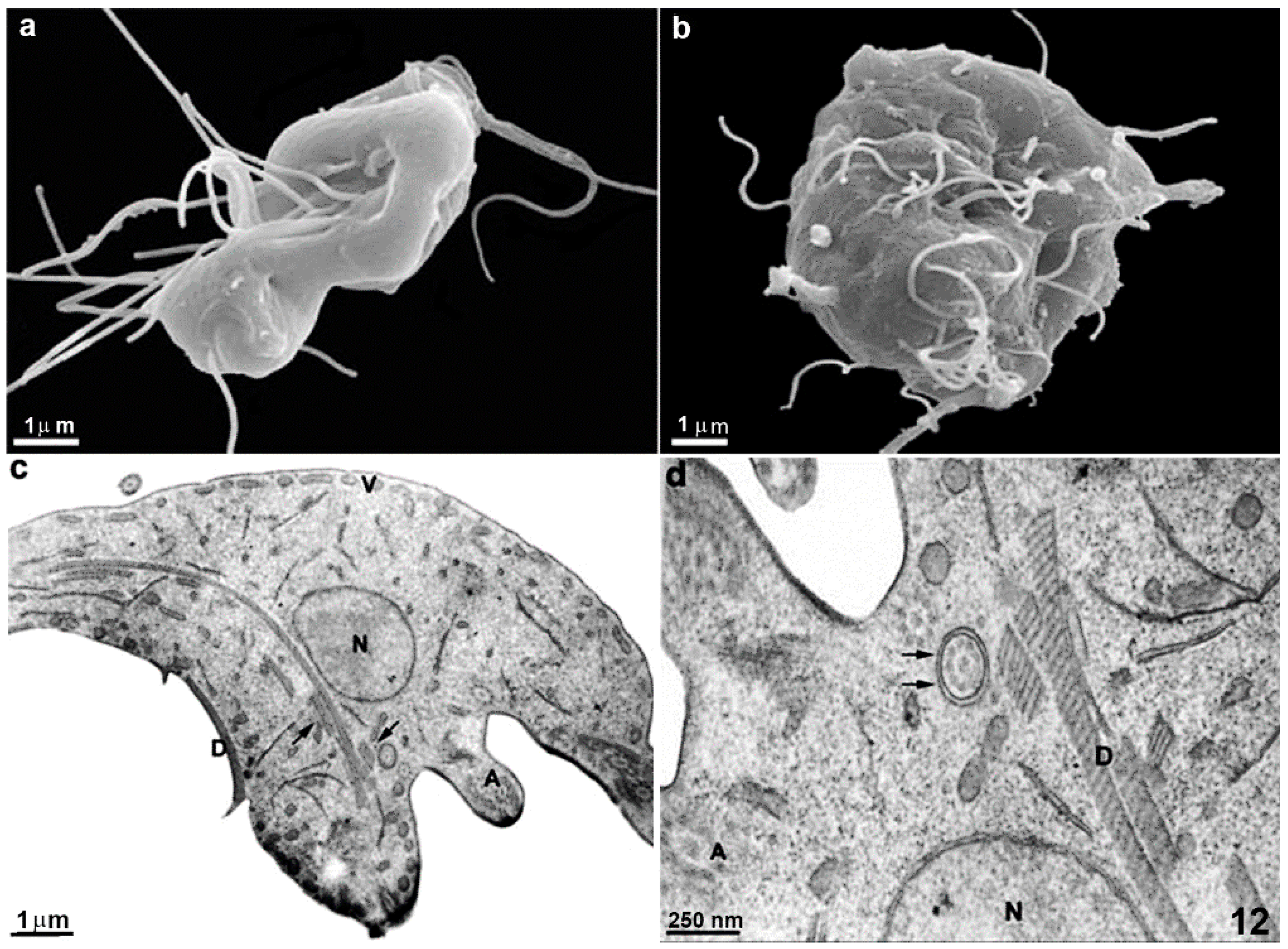
2.1. Trophozoite
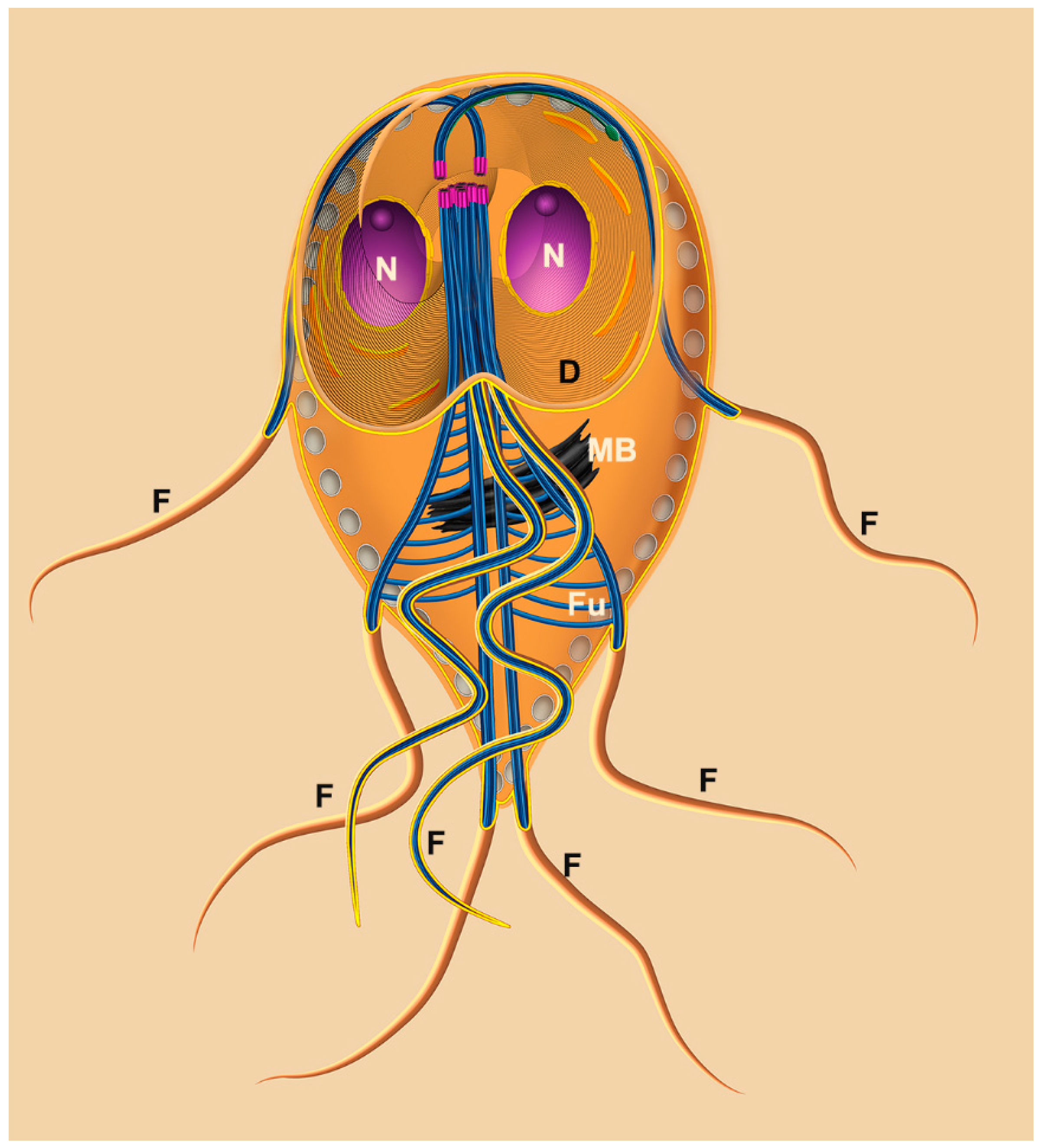
2.1.1. Nuclei
2.1.2. Flagella
2.1.3. Ventral Disc
2.1.4. Median Body
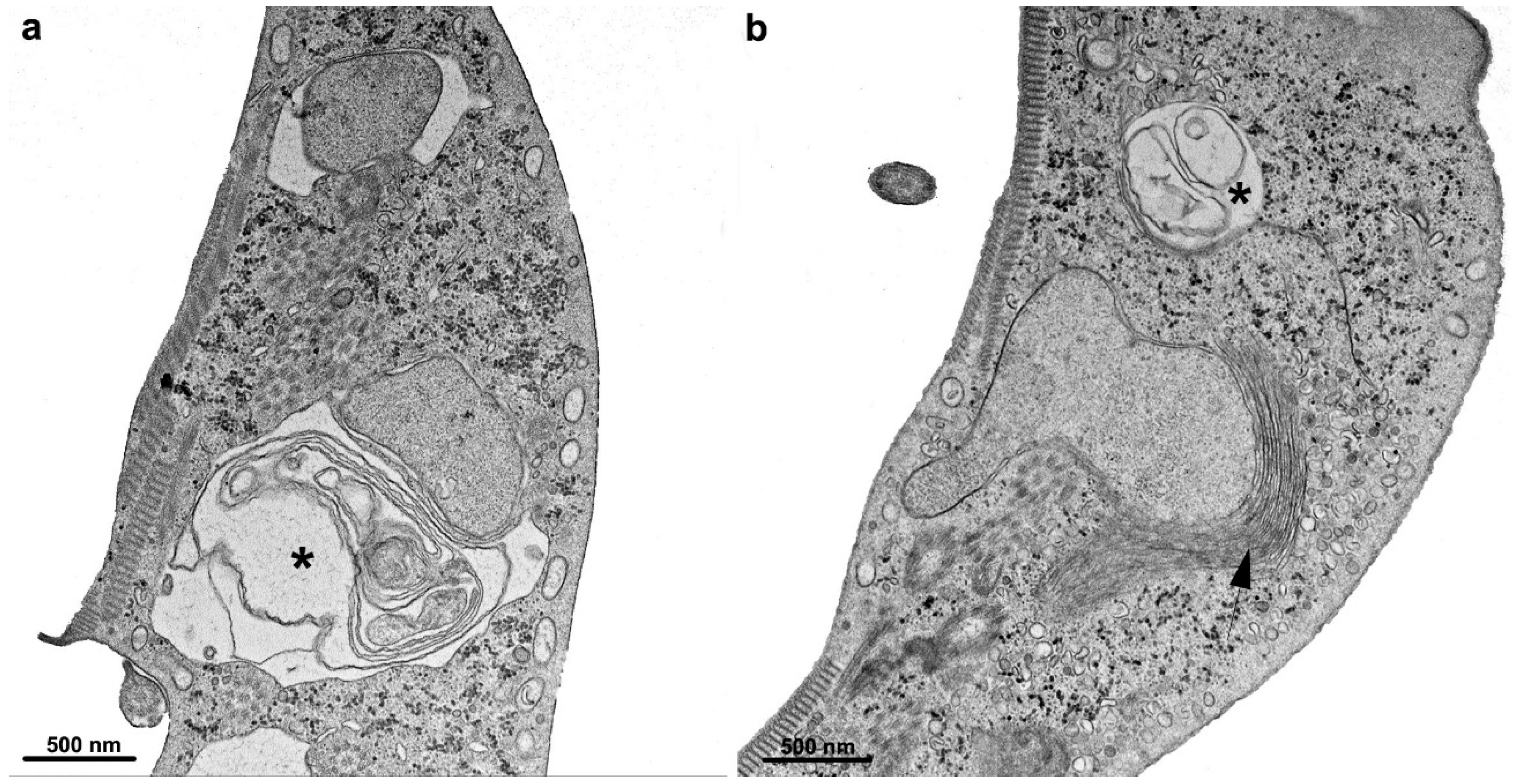
2.1.5. Peripheral Vesicles and the Endocytic Pathway
2.1.6. Endoplasmic Reticulum and Golgi Complex
2.1.7. Mitosomes
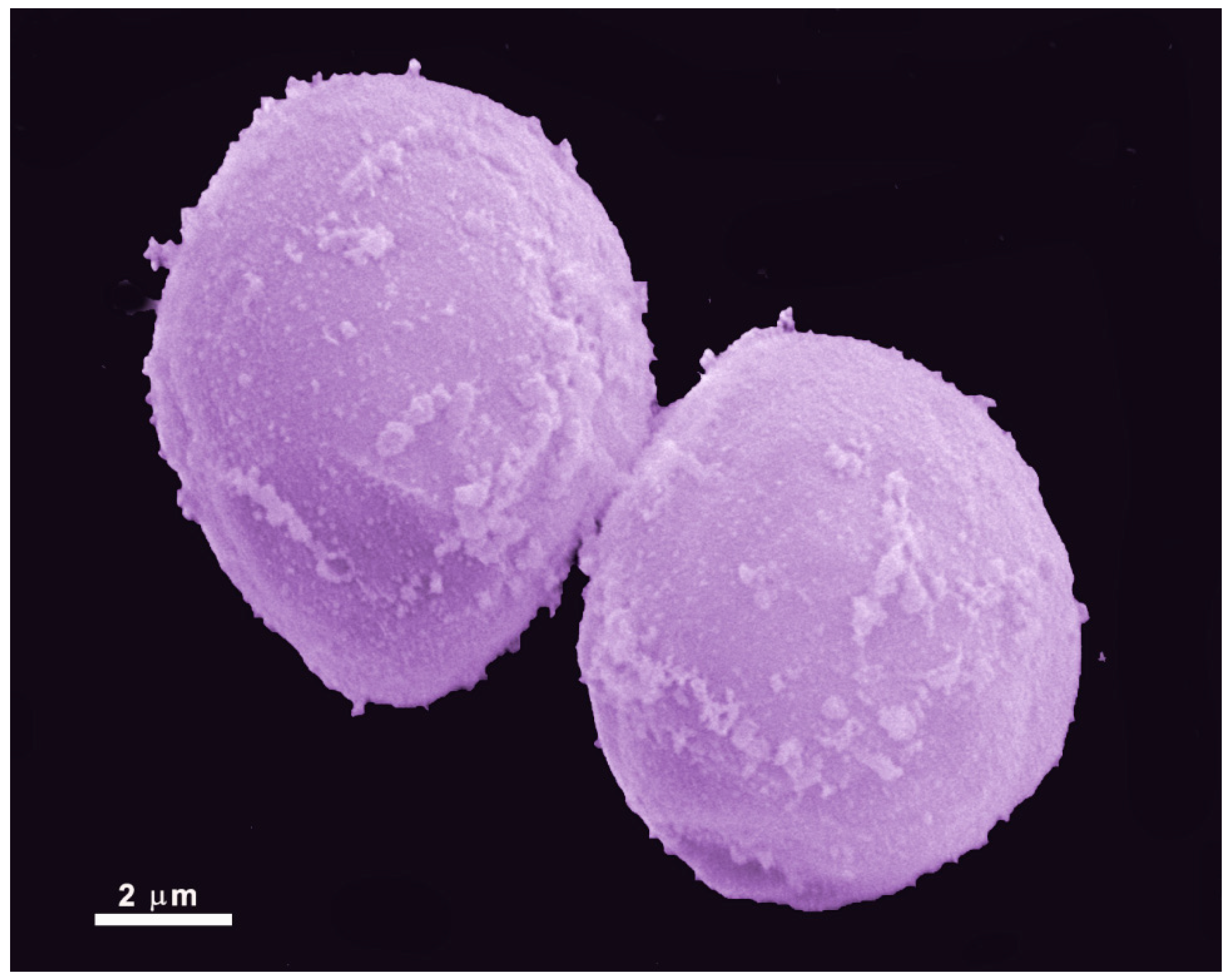
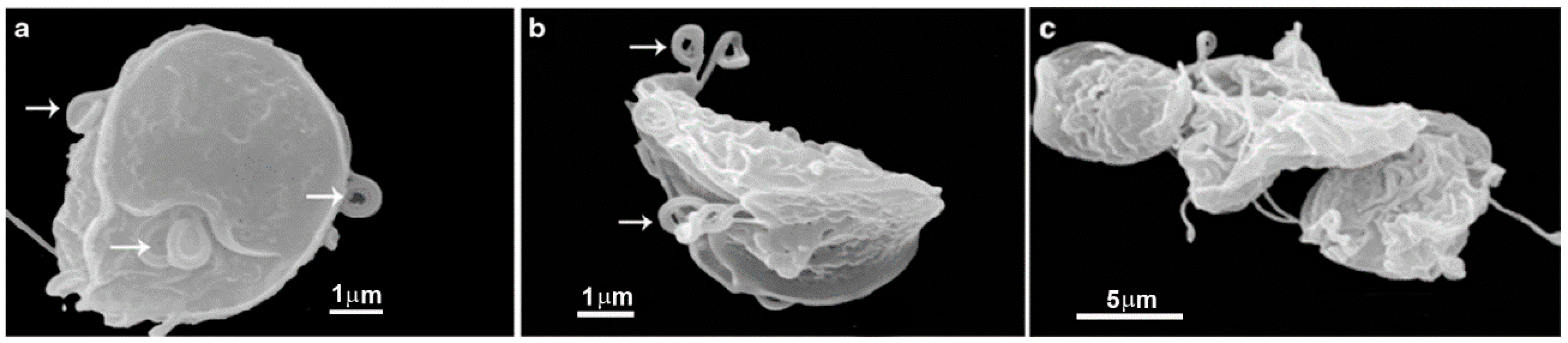
2.2. Cyst
3. Compounds Used in the Giardiasis Treatment and Their Effects on the Parasite (Table 1)
3.1. Metronidazole
3.2. Albendazole
3.3. Other Compounds
| Drug | Mechanism | In Vitro Effects on the Parasite | Reference |
|---|---|---|---|
Metronidazole | Reduced nitro group; forms toxic intermediate |
| [33,34] |
Albendazole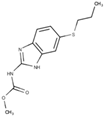 | Bind to the beta-tubulin forming a “cap” at the plus end of microtubules |
| [44] |
Nitazoxanide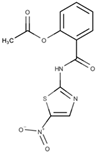 | Inhibition of enzyme activity (PFOR, nitroreductase) |
| [48] |
Furazolidone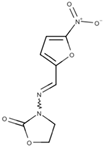 | Reduced furazolidone; forms a toxic intermediate |
| [34] |
Paromomycin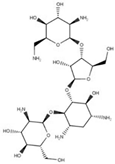 | Inhibit protein synthesis |
| [52] |
Quinacrine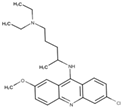 | Inhibit DNA synthesis |
| [49] |
4. Potential Drug Target
4.1. Giardia Cytoskeleton—Drugs Affecting Microtubules: Nocodazole, Colchicine, Taxol, Vinblastine, and Sulfonamide Oryzalin
4.2. Giardia Cytoskeleton—Drugs Affecting Actin: Cytochalasin D, Latrunculin A, Jasplakinolide
4.3. Giardia Organelles Affected by Drugs (Table 2)
4.4. Drugs Inducing Cell Death
4.5. Drugs Affecting Carbohydrates and Lipids Metabolism
4.6. G. intestinalis Kinases as Target Drug
4.7. Histone Acetylation as a Targeted Drug
4.8. G. intestinalis Redox Metabolism
| Potential Target | Drug | In Vitro Effects on the Parasite | Reference |
|---|---|---|---|
| Cytoskeleton—Microtubules |
|
| [58,59,60,61] |
| Cytoskeleton—Actin |
|
| [62,63] |
| Organelles |
|
| [34,64] |
| Cell death |
|
| [66,67] |
| Carbohydrate metabolism |
|
| [70,71] |
| Lipid metabolism |
|
| [73,74] |
| Kinases |
|
| [76,77,78,79,80] |
| Histone acetylation |
|
| [81,82,83,85,86] |
| Redox metabolism |
|
| [34,90,91] |
5. New Compounds
5.1. Repositioning Compounds—Drugs Used for Other Diseases
- Robenidine is an anticoccidial drug first developed in the 1970s and now used regularly. In addition, it is added to the diet of various economically important animals, such as chickens and rabbits. Chemically, it is classified as a guanidine and has demonstrated inhibitory activity against the growth of G. intestinalis, with an IC50 in the range of 1 µm. Several analogs have been synthesized, and a library of 275 compounds has been developed and patented. Some of these derivatives, specifically compounds 45, 47, 48, and 49, have exhibited IC50 values below 10 µM. Additionally, certain derivatives have shown an ability to inhibit the adhesion of the trophozoite to the substrate. This action is particularly interesting as the pathogenicity of the protozoan largely relies on its capacity to adhere to the surface of intestinal epithelial cells [92,93].
- Secnidazole has been reported by Cheung et al. [94] as a treatment option for Giardiasis.
- As mentioned in the previous section, azidothymidine (AZT), an antiretroviral drug, has activity against Giardia, including strains resistant to metronidazole. In addition, AZT has shown inhibitory effects on cyst formation and, in experimental trials conducted on infected gerbils, reduced the number of trophozoites and cysts [80].
- As mentioned in the previous section, Auranofin, an antirheumatic compound, showed promising results against G. intestinalis [90].
- Mavelertinib exhibited substantial growth inhibition at a concentration of 5 µM. Furthermore, it demonstrated a significant effect in experimental Giardiasis, resulting in a notable reduction in parasite presence. This effect was evaluated through non-invasive imaging of whole mice infected with a Giardia green beetle strain [79].
- Steroid hormone 20-hydroxyecdysone showed efficacy against human Giardia infection. Furthermore, this drug exhibited superior effectiveness to metronidazole, as 4% of patients resisted the classical compound [99].
- Fumagillin, an anti-microsporidiosis drug, showed effects in a giardiasis mouse model. [100].
- Orlistat, a pancreatic lipase inhibitor used for treating obesity, demonstrated in vitro activity against G. intestinalis [101].
5.2. Repositioning Compounds—Compound Screening Library and Other Synthetic Drugs
- A library of 130 quinoxaline 1,4-di-N-oxides and certain derivatives within this library has demonstrated growth inhibitory activity with IC50 values below 10 µM. Notably, compounds 50, 51, 52, 53, and 54 were subjected to experimental infections in mice, and they exhibited superior activities compared to metronidazole and nitazoxanide [102,103].
- Triazoxins, a class of novel nucleoside analogs, have exhibited noteworthy activity against Giardia. Certain compounds within this group have demonstrated the ability to inhibit trophozoite growth with an IC50 of 5 µM. Additionally, other analogs have shown the capacity to inhibit trophozoite–cyst transformation in vitro [104].
- A screening of a substantial number (2451) of compounds from the Australian Scaffolds Library identified 40 hits against Giardia, exhibiting an IC50 of approximately 10 µM after 48 h of cultivation. Among these hits, three compounds demonstrated an IC50 of 1 µM, while CL9406 exhibited the lowest IC50 at 180 nM. Notably, the compound SN00797640 displayed potent activity against assemblages A, B, and metronidazole-resistant parasites [105]. However, further analysis of these compounds is required.
- Recently, Zheng et al. [106] reported the remarkable potency of a 3-nitroimidazo[1,2-b]pyridazine compound with an IC50 in the nanomolar (nM) range. This finding highlights the significance of this particular compound and warrants special attention.
- Due to the high glycolytic activity of Giardia, researchers have explored the testing of triose phosphate isomerase inhibitors. Compounds including benzothiazole, benzoxazole, benzimidazole, and sulphydryl derivatives have demonstrated meaningful activity in this regard [107].
- Interestingly, an inhibitor targeting the fused proteins glucose-6-phosphate dehydrogenase and 6-phosphogluconolactonase was observed with an IC50 of 10 µM [108].
- Benzopyrrolizidines have been successfully tested in Giardia cultures. Seventy-four compounds were evaluated, with several demonstrating an IC50 of 11 µM. These compounds induced notable morphological alterations in the parasite, including the loss of nuclei. Therefore, further detailed analysis of these c-mercapto benzimidazole compounds is warranted [109].
- Fernandez-Lainez et al. [110] reported the efficacy of compounds targeting arginine deiminase, an enzyme involved in a metabolic pathway associated with ATP synthesis, against Giardia.
- Gold nanoparticles, known for their broad-spectrum microbicidal activity, have demonstrated effectiveness against experimental Giardiasis in rats. These nanoparticles effectively inhibit the proliferation of trophozoites in the small intestine and the formation of cysts. The effects were evaluated using light and electron microscopy, revealing intestinal cell lesions’ recovery [111].
- The association of chitosan nanoparticles with metronidazole also exhibited significant efficacy in treating experimental Giardiasis in hamsters [112].
- In a recent study, Zoghroban et al. [113] demonstrated the significant efficacy of L-citrulline in controlling experimental Giardiasis in rats. The treatment resulted in reduced trophozoites in the intestinal mucosa and the complete elimination of cysts in the stool.
5.3. Natural Compounds
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dixon, B.R. Giardia duodenalis in humans and animals—Transmission and Disease. Res. Vet. Sci. 2021, 135, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Benchimol, M.; de Souza, W. Observation of Giardia sp. in the Termite Gut of Heterotermes tenuis. Parasitol. Res. 2021, 120, 1131–1135. [Google Scholar]
- Thompson, R.C.; Hopkins, R.M.; Homan, W.L. Nomenclature and genetic groupings of Giardia infecting mammals. Parasitol. Today 2000, 16, 210–213. [Google Scholar] [CrossRef]
- Ryan, U.; Zahedi, A. Molecular epidemiology of Giardiasis from a veterinary perspective. Adv. Parasitol. 2019, 106, 209–254. [Google Scholar]
- Cai, W.; Ryan, U.; Xiao, L.; Feng, Y. Zoonotic giardiasis: An update. Parasitol. Res. 2021, 120, 4199–4218. [Google Scholar] [CrossRef]
- Benchimol, M.; Gadelha, A.P.R.; de Souza, W. Cell biology of the life cycle of Giardia intestinalis. In Lifecycles of Pathogenic Protists in Humans; De Souza, W., Ed.; Microbiol. Monographs; Springer: Cham, Switzerland, 2022; Volume 35. [Google Scholar]
- Elmendorf, H.G.; Dawson, S.C.; McCaffery, J.M. The cytoskeleton of Giardia lamblia. Int. J. Parasitol. 2003, 33, 3–28. [Google Scholar] [CrossRef]
- Campanati, L.; Holloshi, A.; Troester, H.; Spring, H.; Souza, W.; Monteiro-Leal, L.H. Video-Microscopy observations of fast dynamic process in the protozoon Giardia lamblia. Cell Motil. Cytoskelet. 2002, 51, 213–214. [Google Scholar] [CrossRef] [PubMed]
- Lenaghan, S.C.; Davis, C.A.; Henson, W.R.; Zhang, Z.; Zhang, M. High-speed microscopic imaging of flagella motility and swimming in Giardia lamblia trophozoites. Proc. Natl. Acad. Sci. USA 2011, 108, 550–558. [Google Scholar] [CrossRef]
- Holberton, D.V. Attachment of Giardia: Hydrodynamic model based on flagellar activity. J. Exp. Biol. 1974, 60, 207–221. [Google Scholar] [CrossRef]
- Woessner, D.J.; Dawson, S.C. The Giardia median body protein is a ventral disc protein that is critical for maintaining a domed disc conformation during attachment. Eukaryot. Cell 2012, 11, 292–301. [Google Scholar] [CrossRef]
- Holberton, D.V. Fine structure of the ventral disc apparatus and the mechanism of attachment in the flagellate Giardia muris. J. Cell Sci. 1973, 13, 11–41. [Google Scholar] [CrossRef]
- Schwartz, C.L.; Heumann, J.M.; Dawson, S.C.; Hoenger, A. A detailed, hierarchical study of Giardia lamblia’s ventral disc reveals novel microtubule-associated protein complexes. PLoS ONE 2012, 7, e43783. [Google Scholar]
- Feely, D.E.; Schollmeyer, J.V.; Erlandsen, S.L. Giardia spp.: Distribution of contractile proteins in the attachment organelle. Exp. Parasitol. 1982, 53, 145–154. [Google Scholar] [CrossRef]
- Piva, B.; Benchimol, M. The median body of Giardia lamblia: An ultrastructural study. Biol. Cell 2004, 96, 735–746. [Google Scholar] [CrossRef]
- Lanfredi-Rangel, A.; Attias, M.; Carvalho, T.M.U.; Kattenbach, W.M.; de Souza, W. The peripheral vesicles of trophozoites of the primitive protozoan Giardia lamblia may correspond to early and late endosomes and to lysosomes. J. Struct. Biol. 1998, 123, 225–235. [Google Scholar] [CrossRef]
- Midlej, V.; de Souza, W.; Benchimol, M. The peripheral vesicles gather multivesicular bodies with different behavior during the Giardia intestinalis life cycle. J. Struct. Biol. 2019, 207, 301–311. [Google Scholar] [CrossRef] [PubMed]
- Abodeely, M.; DuBois, K.N.; Hehl, A.; Stefanic, S.; Sajid, M.; De Souza, W.; Attias, M.; Engel, J.C.; Hsieh, I.; Fetter, R.D.; et al. A contiguous compartment functions as endoplasmic reticulum and endosome/lysosome in Giardia lamblia. Eukaryot. Cell 2009, 8, 1665–1676. [Google Scholar] [CrossRef]
- McCaffery, J.M.; Gillin, F.D. Giardia lamblia: Ultrastructural basis of protein transport during growth and encystation. Exp. Parasitol. 1994, 29, 220–235. [Google Scholar] [CrossRef]
- Tovar, J.; León-Avila, G.; Sánchez, L.B.; Sutak, R.; Tachezy, J.; van der Giezen, M.; Hernández, M.; Müller, M.; Lucocq, J.M. Mitochondrial remnant organelles of Giardia function in iron-sulphur protein maturation. Nature 2003, 426, 172–176. [Google Scholar] [CrossRef]
- Tachezy, J.; Sanchez, L.B.; Muller, M. Mitochondrial type iron-sulfur cluster assembly in the amitochondriate eukaryotes Trichomonas vaginalis and Giardia intestinalis, as indicated by the phylogeny of IscS. Mol. Biol. Evol. 2001, 18, 1919–1928. [Google Scholar] [CrossRef] [PubMed]
- Jedelský, P.L.; Doležal, P.; Rada, P.; Pyrih, J.; Smíd, O.; Hrdý, I.; Sedinová, M.; Marcinčiková, M.; Voleman, L.; Perry, A.J.; et al. The minimal proteome in the reduced mitochondrion of the parasitic protist Giardia intestinalis. PLoS ONE 2011, 6, e17285. [Google Scholar] [CrossRef] [PubMed]
- Sheffield, H.G.; Bjorvat, B. Ultrastructure of the cyst of Giardia lamblia. Am. J. Trop. Med. Hyg. 1977, 26, 23–30. [Google Scholar] [CrossRef]
- Lujan, H.D.; Mowatt, M.R.; Nash, T.E. Mechanisms of Giardia lamblia differentiation into cysts. Microbiol. Mol. Biol. Rev. 1997, 61, 294–304. [Google Scholar]
- Midlej, V.; Benchimol, M. Giardia lamblia behavior during encystment: How morphological changes in shape occur. Parasitol. Int. 2009, 58, 72–80. [Google Scholar] [CrossRef]
- Reiner, D.S.; McCaffery, M.; Gillin, F.D. Sorting of cyst wall proteins to a regulated secretory pathway during differentiation of the primitive eukaryote, Giardia lamblia. Eur. J. Cell Biol. 1990, 53, 142–153. [Google Scholar]
- Buchel, L.A.; Gorenflot, A.; Chochillon, C.; Savel, J.; Gobert, J.G. In vitro excystation of Giardia from humans: A scanning electron microscopy study. J. Parasitol. 1987, 73, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Cosar, C.; Julou, L. Activity of 1-(2-hydroxyethyl)-2-methyl-5-nitroimidazole (8823 RP) against experimental Trichomonas vaginalis infection. Ann. Inst. Pasteur 1959, 96, 238–241. [Google Scholar]
- Darbon, A.; Portal, A.; GIrier, L.; Pantim, J.; Leclaire, C. Traitement de la giardiase (lambliase) par le métronidazole. Presse Med. 1962, 70, 15–16. [Google Scholar]
- Jiménez, J.C.; Pinon, A.; Dive, D.; Capron, M.; Dei-Cas, E.; Convit, J. Antibody response in children infected with Giardia intestinalis before and after Treatment with Secnidazole. Am. J. Trop. Med. Hyg. 2009, 80, 11–15. [Google Scholar] [CrossRef]
- Ordonez-Mena, J.M.; McCarthy, N.D.; Fanshawe, T.R. Comparative efficacy of drugs for treating Giardiasis: A systematic update of the literature and network meta-analysis of randomized clinical trials. J. Antimicrob. Chemother. 2018, 73, 596–606. [Google Scholar] [CrossRef]
- Leitsch, D.; Burgess, A.G.; Dunn, L.A.; Krauer, K.G.; Tan, K.; Duchêne, M.; Upcroft, P.; Eckmann, L.; Upcroft, J.A. Pyruvate:ferredoxin oxidoreductase and thioredoxin reductase are involved in 5-nitroimidazole activation while flavin metabolism is linked to 5-nitroimidazole resistance in Giardia lamblia. J. Antimicrob. Chemother. 2011, 66, 1756–1765. [Google Scholar] [CrossRef]
- Uzlikova, M.; Nohynkova, E. The effect of metronidazole on the cell cycle and DNA in Metronidazole-susceptible and -resistant Giardia cell lines. Mol. Biochem. Parasitol. 2014, 198, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Campanati, L.; Monteiro-Leal, L.H. The effects of the antiprotozoal drugs metronidazole and furazolidone on trophozoites of Giardia lamblia (P1 strain). Parasitol. Res. 2002, 88, 80–85. [Google Scholar] [CrossRef]
- Leitsch, D.; Schlosser, S.; Burgess, A.; Duchêne, M. Nitroimidazole drugs vary in their mode of action in the human parasite Giardia lamblia. Int. J. Parasitol. Drugs Drug Resist. 2012, 2, 166–170. [Google Scholar] [CrossRef]
- Bernal-Redondo, R.; Martínez-Méndez, L.G.; Mendoza-Chavez, A.; Velasco-Perales, D.; Chavez-Munguia, B. Evaluation of the in vitro effect of albendazole, metronidazole and nitazoxanide on viability and structure of Giardia lamblia cysts. J. Submicrosc. Cytol. Pathol. 2004, 36, 241–245. [Google Scholar] [PubMed]
- Saghaug, C.S.; Gamlem, A.L.; Hauge, K.B.; Vahokoski, J.; Klotz, C.; Aebischer, T.; Langeland, N.; Hanevik, K. Genetic diversity in the Metronidazole metabolism genes nitroreductases and pyruvate ferredoxin oxidoreductases in susceptible and refractory clinical samples of Giardia lamblia. Int. J. Parasitol. Drugs Drug Resist. 2022, 21, 51–60. [Google Scholar] [CrossRef]
- Adam, R.D. Giardia duodenalis: Biology and Pathogenesis. Clin. Microbiol. Rev. 2021, 34, e0002419. [Google Scholar] [CrossRef] [PubMed]
- Juliano, C.; Martinotti, M.G.; Cappuccinelli, P. “In vitro” effect of microtubule inhibitors on Trichomonas vaginalis. Microbiologica 1985, 8, 31–42. [Google Scholar]
- Chavez, B.; Cedillo-Rivera, R.; Martinez-Palomo, A. Giardia lamblia: Ultrastructural study of the in vitro effect of benzimidazoles. J. Protozool. 1992, 39, 510–515. [Google Scholar] [CrossRef]
- Katiyar, S.K.; Gordon, V.R.; McLaughlin, G.L.; Edlind, T.D. Antiprotozoal activities of benzimidazoles and correlations with beta-tubulin sequence. Antimicrob. Agents Chemother. 1994, 38, 2086–2090. [Google Scholar] [CrossRef]
- MacDonald, L.M.; Armson, A.; Thompson, A.R.; Reynoldson, J.A. Characterisation of benzimidazole binding with recombinant tubulin from Giardia duodenalis, Encephalitozoon intestinalis, and Cryptosporidium parvum. Mol. Biochem. Parasitol. 2004, 138, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Pech-Santiago, E.O.; Argüello-García, R.; Vázquez, C.; Saavedra, E.; González-Hernández, I.; Jung-Cook, H.; Rafferty, S.P.; Ortega-Pierres, M.G. Giardia duodenalis: Flavohemoglobin is involved in drug biotransformation and resistance to albendazole. PLoS Pathog. 2022, 18, e1010840. [Google Scholar] [CrossRef] [PubMed]
- Oxberry, M.E.; Thompson, R.C.; Reynoldson, J.A. Evaluation of the effects of Albendazole and Metronidazole on the ultrastructure of Giardia duodenalis, Trichomonas vaginalis and Spironucleus muris using transmission electron microscopy. Int. J. Parasitol. 1994, 24, 695–703. [Google Scholar] [CrossRef]
- Emery-Corbin, S.J.; Su, Q.; Tichkule, S.; Baker, L.; Lacey, E.; Jex, A.R. In vitro selection of Giardia duodenalis for Albendazole resistance identifies a β-tubulin mutation at amino acid E198K. Int. J. Parasitol. Drugs Drug Resist. 2021, 16, 162–173. [Google Scholar] [CrossRef] [PubMed]
- Argüello-García, R.; Cruz-Soto, M.; González-Trejo, R.; Paz-Maldonado, L.M.; Bazán-Tejeda, M.L.; Mendoza-Hernández, G.; Ortega-Pierres, G. An antioxidant response is involved in resistance of Giardia duodenalis to albendazole. Front. Microbiol. 2015, 10, 286. [Google Scholar] [CrossRef]
- Muller, J.; Wastling, J.; Sanderson, S.; Muller, N.; Hemphill, A. A novel Giardia lamblia nitroreductase, GlNR1, interacts with nitazoxanide and other thiazolides. Antimicrob. Agents Chemother. 2007, 51, 1979–1986. [Google Scholar] [CrossRef]
- Cedillo-Rivera, R.; Chávez, B.; González-Robles, A.; Tapia, A.; Yépez-Mulia, L. In vitro effect of nitazoxanide against Entamoeba histolytica, Giardia intestinalis and Trichomonas vaginalis trophozoites. J. Eukaryot. Microbiol. 2002, 49, 201–208. [Google Scholar] [CrossRef]
- Riches, A.; Hart, C.J.S.; Trenholme, K.R.; Skinner-Adams, T.S. Anti-Giardia Drug Discovery: Current Status and Gut Feelings. J. Med. Chem. 2020, 63, 13330–13354. [Google Scholar] [CrossRef]
- Brown, D.M.; Upcroft, J.A.; Upcroft, P. A H2O-producing NADH oxidase from the protozoan parasite Giardia duodenalis. Eur. J. Biochem. 1996, 241, 155–161. [Google Scholar] [CrossRef]
- Smith, N.C.; Bryant, C.; Boreham, P.F. Possible roles for pyruvate:ferredoxin oxidoreductase and thiol-dependent peroxidase and reductase activities in resistance to nitroheterocyclic drugs in Giardia intestinalis. Int. J. Parasitol. 1988, 18, 991–997. [Google Scholar] [CrossRef]
- Edlind, T.D. Susceptibility of Giardia lamblia to aminoglycoside protein synthesis inhibitors: Correlation with rRNA structure. Antimicrob. Agents Chemother. 1989, 33, 484–488. [Google Scholar] [CrossRef] [PubMed]
- Adam, R.D. Biology of Giardia lamblia. Clin. Microbiol. Rev. 2001, 14, 447–475. [Google Scholar] [CrossRef]
- Genel, F.; Erermis, S.; Aksu, G.; Ozturk, C.; Kutukculer, N. Quinacrine-induced psychiatric disturbances in a child with common variable immunodeficiency and chronic Giardiasis. Hum. Psychopharmacol. 2002, 17, 357–359. [Google Scholar] [CrossRef]
- Argüello-García, R.; Cruz-Soto, M.; Romero-Montoya, L.; Ortega-Pierres, G. In vitro resistance to nitroimidazoles and benzimidazoles in Giardia duodenalis: Variability and variation in gene expression. Infect. Genet. Evol. 2009, 9, 1057–1064. [Google Scholar] [CrossRef] [PubMed]
- Food & Drug Administration. Metronidazole. 2018. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/018890s052lbl.pdf (accessed on 1 October 2021).
- Maia-Brigagão, C.; Gadelha, A.P.; de Souza, W. New associated structures of the anterior flagella of Giardia duodenalis. Microsc. Microanal. 2013, 19, 1374–1376. [Google Scholar] [CrossRef] [PubMed]
- Mariante, R.M.; Vancini, R.G.; Melo, A.L.; Benchimol, M. Giardia lamblia: Evaluation of the in vitro effects of nocodazole and colchicine on trophozoites. Exp. Parasitol. 2005, 11, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Dawson, S.C.; Sagolla, M.S.; Mancuso, J.J.; Woessner, D.J.; House, S.A.; Fritz-Laylin, L.; Cande, W.Z. Kinesin-13 regulates flagellar, interphase, and mitotic microtubule dynamics in Giardia intestinalis. Eukaryot. Cell 2007, 6, 2354–2364. [Google Scholar] [CrossRef]
- Edlind, T.D.; Hang, T.L.; Chakraborty, P.R. Activity of the anthelmintic benzimidazoles against Giardia lamblia in vitro. J. Infect. Dis. 1990, 162, 1408–1411. [Google Scholar] [CrossRef]
- Terra, L.L.; Campanati, L.; De Souza, W. Heterogeneity in the sensitivity of microtubules of Giardia lamblia to the herbicide oryzalin. Parasitol. Res. 2010, 107, 47–54. [Google Scholar] [CrossRef]
- Castillo-Romero, A.; Leon-Avila, G.; Perez Rangel, A.; Cortes Zarate, R.; Garcia Tovar, C.; Hernandez, J.M. Participation of actin on Giardia lamblia growth and encystation. PLoS ONE 2009, 4, e7156. [Google Scholar] [CrossRef]
- Corrêa, G.; Benchimol, M. Giardia lamblia behavior under cytochalasins treatment. Parasitol. Res. 2006, 98, 250–256. [Google Scholar] [CrossRef]
- Campanati, L.; Gadelha, A.P.; Monteiro-Leal, L.H. Electron and video-light microscopy analysis of the in vitro effects of pyrantel pamoate on Giardia lamblia. Exp. Parasitol. 1999, 97, 9–14. [Google Scholar] [CrossRef]
- Hernandez, Y.; Castillo, C.; Roychowdhury, S.; Hehl, A.; Aley, S.B.; Das, S. Clathrin-dependent pathways and the cytoskeleton network are involved in ceramide endocytosis by a parasitic protozoan, Giardia lamblia. Int. J. Parasitol. 2007, 37, 21–32. [Google Scholar] [CrossRef]
- Corrêa, G.; Vilela, R.; Menna-Barreto, R.F.; Midlej, V.; Benchimol, M. Cell death induction in Giardia lamblia: Effect of beta-lapachone and starvation. Parasitol. Int. 2009, 58, 424–437. [Google Scholar] [CrossRef] [PubMed]
- Gadelha, A.P.R.; Bravim, B.; Vidal, J.; Reignault, L.C.; Cosme, B.; Huber, K.; Bracher, F.; de Souza, W. Alterations on growth and cell organization of Giardia intestinalis trophozoites after Treatment with KH-TFMDI, a novel class III histone deacetylase inhibitor. Int. J. Med. Microbiol. 2019, 309, 130–142. [Google Scholar] [CrossRef] [PubMed]
- Bruchhaus, I.; Roeder, T.; Rennenberg, A.; Heussler, V.T. Protozoan parasites: Programmed cell death as a mechanism of parasitism. Trends Parasitol. 2007, 23, 376–383. [Google Scholar] [CrossRef]
- Han, J.; Collins, L.J. Reconstruction of sugar metabolic pathways of Giardia lamblia. Int. J. Proteom. 2012, 2012, 980829. [Google Scholar] [CrossRef] [PubMed]
- Castillo-Villanueva, A.; Rufino-González, Y.; Méndez, S.T.; Torres-Arroyo, A.; Ponce-Macotela, M.; Martínez-Gordillo, M.N.; Reyes-Vivas, H.; Oria-Hernández, J. Disulfiram as a novel inactivator of Giardia lamblia triosephosphate isomerase with antigiardial potential. Int. J. Parasitol. Drugs Drug Resist. 2017, 7, 425–432. [Google Scholar] [CrossRef]
- López-Velázquez, G.; Fernández-Lainez, C.; de la Mora-de la Mora, J.I.; Caudillo de la Portilla, D.; Reynoso-Robles, R.; González-Maciel, A.; Ridaura, C.; García-Torres, I.; Gutiérrez-Castrellón, P.; Olivos-García, A.; et al. On the molecular and cellular effects of omeprazole to further support its effectiveness as an antigiardial drug. Sci. Rep. 2019, 9, 8922. [Google Scholar] [CrossRef]
- Yichoy, M.; Nakayasu, E.S.; Shpak, M.; Aguilar, C.; Aley, S.B.; Almeida, I.C.; Das, S. Lipidomic analysis reveals that phosphatidylglycerol and phosphatidylethanolamine are newly generated phospholipids in an early-divergent protozoan, Giardia lamblia. Mol. Biochem. Parasitol. 2009, 165, 67–78. [Google Scholar] [CrossRef]
- Maia, C.; Attias, M.; Urbina, J.; Gilbert, I.; Magaraci, F.; de Souza, W. Azasterols impair Giardia lamblia proliferation and induces encystation. Biochem. Biophys. Res. Commun. 2007, 363, 310–316. [Google Scholar] [CrossRef]
- Gadelha, A.P.R.; Brigadão, C.M.; da Silva, M.B.; Rodrigues, A.B.M.; Guimarães, A.C.R.; Paiva, F.; de Souza, W.; Henriques, C. Insights about the structure of farnesyl diphosphate synthase (FPPS) and the activity of bisphosphonates on the proliferation and ultrastructure of Leishmania and Giardia. Parasites Vectors 2020, 13, 168. [Google Scholar] [CrossRef]
- Manning, G.; Reiner, D.S.; Lauwaet, T.; Dacre, M.; Smith, A.; Zhai, Y.; Svard, S.; Gillin, F.D. The minimal kinome of Giardia lamblia illuminates early kinase evolution and unique parasite biology. Genome Biol. 2011, 12, R66. [Google Scholar] [CrossRef]
- Davids, B.J.; Williams, S.; Lauwaet, T.; Palanca, T.; Gillin, F.D. Giardia lamblia aurora kinase: A regulator of mitosis in a binucleate parasite. Int. J. Parasitol. 2008, 38, 353–369. [Google Scholar] [CrossRef]
- Hennessey, K.M.; Smith, T.R.; Xu, J.W.; Alas, G.C.; Ojo, K.K.; Merritt, E.A.; Paredez, A.R. Identification and Validation of Small-Gatekeeper Kinases as Drug Targets in Giardia lamblia. PLoS Negl. Trop. Dis. 2016, 10, e0005107. [Google Scholar] [CrossRef] [PubMed]
- Park, E.A.; Kim, J.; Shin, M.Y.; Park, S.J. A polo-like kinase modulates cytokinesis and flagella biogenesis in Giardia lamblia. Parasites Vectors. 2021, 14, 182. [Google Scholar] [CrossRef] [PubMed]
- Michaels, S.A.; Hulverson, M.A.; Whitman, G.R.; Tran, L.T.; Choi, R.; Fan, E.; McNamara, C.W.; Love, M.S.; Ojo, K.K. Repurposing the kinase inhibitor Mavelertinib for Giardiasis therapy. Antimicrob. Agents Chemother. 2022, 66, e0001722. [Google Scholar] [CrossRef]
- Krakovka, S.; Ranjbarian, F.; LKrakovka, S.; Ranjbarian, F.; Luján, L.A.; Saura, A.; Larsen, N.B.; Jiménez-González, A.; Reggenti, A.; Luján, H.D.; et al. Giardia intestinalis thymidine kinase is a high-affinity enzyme crucial for DNA synthesis and an exploitable target for drug discovery. J. Biol. Chem. 2022, 298, 102028. [Google Scholar] [CrossRef] [PubMed]
- Sonda, S.; Morf, L.; Bottova, I.; Baetschmann, H.; Rehrauer, H.; Caflisch, A.; Hehl, A.B. Epigenetic mechanisms regulate stage differentiation in the minimized protozoan Giardia lamblia. Mol. Microbiol. 2010, 76, 48–67. [Google Scholar] [CrossRef]
- Carranza, P.G.; Gargantini, P.R.; Prucca, C.G.; Torri, A.; Saura, A.; Svärd, S.; Lujan, H.D. Specific histone modifications play critical roles in the control of encystation and antigenic variation in the early-branching eukaryote Giardia lamblia. Int. J. Biochem. Cell Biol. 2016, 81, 32–43. [Google Scholar] [CrossRef]
- Orozco, D.R.; Garlapati, S. Histone deacetylase inhibitors induce expression of chromosomally tagged variant-specific surface protein genes in Giardia lamblia. BMC Res. Notes 2020, 13, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Bondarev, A.D.; Attwood, M.M.; Jonsson, J.; Chubarev, V.N.; Tarasov, V.V.; Schiöth, H.B. Recent developments of HDAC inhibitors: Emerging indications and novel molecules. Br. J. Clin. Pharmacol. 2021, 87, 4577–4597. [Google Scholar] [CrossRef]
- Lagunas-Rangel, F.A.; Bazán-Tejeda, M.L.; García-Villa, E.; Bermúdez-Cruz, R.M. Nicotinamide induces G2 cell cycle arrest in Giardia duodenalis trophozoites and promotes changes in sirtuins transcriptional expression. Exp. Parasitol. 2020, 209, 107822. [Google Scholar] [CrossRef]
- Oliveira, R.V.F.; de Souza, W.; Vögerl, K.; Bracher, F.; Benchimol, M.; Gadelha, A.P.R. In vitro effects of the 4-[(10H-phenothiazin-10-yl)methyl]-N-hydroxybenzamide on Giardia intestinalis trophozoites. Acta Trop. 2022, 232, 106484. [Google Scholar] [CrossRef]
- Mastronicola, D.; Falabella, M.; Testa, F.; Pucillo, L.P.; Teixeira, M.; Sarti, P.; Saraiva, L.M.; Giuffrè, A. Functional characterization of peroxiredoxins from the human protozoan parasite Giardia intestinalis. PLoS Negl. Trop. Dis. 2014, 8, e2631. [Google Scholar] [CrossRef] [PubMed]
- Testa, F.; Mastronicola, D.; Cabell, D.E.; Bordi, E.; Pucillo, L.P.; Sarti, P.; Saraiva, L.M.; Giuffrè, A.; Teixeira, M. The superoxide reductase from the early diverging eukaryote Giardia intestinalis. Free Radic. Biol. Med. 2011, 51, 1567–1574. [Google Scholar] [CrossRef] [PubMed]
- Leitsch, D.; Williams, C.F.; Hrdý, I. Redox Pathways as Drug Targets in Microaerophilic Parasites. Trends Parasitol. 2018, 34, 576–589. [Google Scholar] [CrossRef]
- Capparelli, E.V.; Bricker-Ford, R.; Rogers, M.J.; McKerrow, J.H.; Reed, S.L. Phase I Clinical Trial Results of Auranofin, a Novel Antiparasitic Agent. Antimicrob. Agents Chemother. 2016, 61, e01947-16. [Google Scholar] [CrossRef]
- Brogi, S.; Fiorillo, A.; Chemi, G.; Butini, S.; Lalle, M.; Ilari, A.; Gemma, S.; Campiani, G. Structural characterization of Giardia duodenalis thioredoxin reductase (gTrxR) and computational analysis of its interaction with NBDHEX. Eur. J. Med. Chem. 2017, 135, 479–490. [Google Scholar] [CrossRef]
- Stevens, A.J.; Abraham, R.; Young, K.A.; Russel, A.J.; McCluskey, S.N.; Baker, J.R.; Rusdi, B.; Page, S.W.; O’Handley, R.; O’Dea, M.; et al. Antigiardial activity of novel guanidine compounds. ChemMedChem 2022, 17, e202200341. [Google Scholar] [CrossRef]
- Ogunniyi, A.D.; Khazandi, M.; Stevens, A.J.; Sims, S.K.; Page, S.W.; Venter, G.H.; Powell, A.; White, K.; Petrovski, K.R.; Laven-Law, G.; et al. Evaluation of robenidine analog NCL195 as a novel broad-spectrum antibacterial agent. PLoS ONE 2017, 12, e0183457. [Google Scholar] [CrossRef]
- Cheung, W.; Russo, C.; Maher, S.; Malik, R.; Slapeta, J. Successful use of secnidazole to manage a Giardiasis outbreak in a shelter. Vet. Parasitol. 2019, 274, 108911. [Google Scholar] [CrossRef] [PubMed]
- Bourque, D.L.; Neumayr, A.; Libman, M.; Chen, L.H. Treatment strategies for nitroimidazole-refractory Giardiasis: A systematic review. J. Travel Med. 2022, 29, taab120. [Google Scholar] [CrossRef] [PubMed]
- Ydsten, K.A.; Hellgren, U.; Asgeirsson, H. Quinacrine treatment of nitroimidazole-refractory Giardiasis. J. Infect. Dis. 2022, 225, 1773–1776. [Google Scholar] [CrossRef]
- Custodio, M.M.; Sparks, J.; Long, T.E. Disulfiram: A repurposed drug in preclinical and clinical development for the Treatment of infectious diseases. Antiinfect. Agents 2022, 20, e040122199856. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Ochoa, B.; Gomez-Manzo, S.; Sanchez-Carrillo, A.; Marcial-Quino, J.; Rocha-Ramirez, L.M.; Santos-Segura, A.; Ramirez-Nava, E.; Arreguin-Espinosa, R.; Cuevas-Cruz, M.; Mendez-Tenoria, A.; et al. Enhanced anti-Giardia effect of omeprazole analog benzimidazole compounds. Molecules 2020, 25, 3979. [Google Scholar] [CrossRef] [PubMed]
- Toychiev, A.; Kerimov, F.; Islamova, J.; Shaislamova, M.; Mirzakhmedov, M.; Davis, N.; Syrov, V.; Osipova, S. Antiprotozoal effect of steroid hormone 20-hydroxyecdysone in giardiosis patients. Ann. Parasitol. 2022, 68, 605–615. [Google Scholar]
- Kulakova, L.; Galkin, A.; Chen, C.Z.; Southall, N.; Marugan, J.J.; Zheng, W.; Herzberg, O. Discovery of novel antigiardiasis drug candidates. Antimicrob. Agents Chemother. 2014, 58, 7303–7311. [Google Scholar] [CrossRef]
- Hahn, J.; Seeber, F.; Kolodziej, H.; Ignatius, R.; Laue, M.; Aebischer, T.; Klotz, C. High sensitivity of Giardia duodenalis to tetrahydrolipstatin (orlistat) in vitro. PLoS ONE 2013, 8, e71597. [Google Scholar] [CrossRef]
- Barbosa-Cabrera, E.; Moo-Puc, R.; Monge, A.; Paz-González, A.D.; Bocanegra-Garcia, V.; Rivera, G. In vitro and in vivo evaluation of quinoxaline 1,4-di-N-oxide against Giardia lamblia. Lett. Drug Des. Discov. 2019, 17, 428–433. [Google Scholar] [CrossRef]
- Instituto Politécnico Nacional. Use of quinoxaline 1,4-di-Npoxide ester derivatives for the Treatment of Giardiasis as anti-Giardia lamblia agents. MX2017012677A, 2019. [Google Scholar]
- Pogula, P.K.; de Chatterjee, A.; Chi, M.; VanKoten, H.W.; Das, S. Triazoxins: Novel nucleosides with anti-Giardia activity. Bioorganic Med. Chem. Lett. 2020, 30, 127175. [Google Scholar] [CrossRef]
- Hart, C.J.S.; Riches, A.G.; Tiashi, S.; Clapper, E.; Ramu, S.; Zuegg, J.; Ryan, J.H.; Skinner-Adams, T.S. A subset screen of the compounds Australia Scaffold Library identifies 7-acrylaminodibenzoxazepinone as potent and selective hits for anti-Giardia drug discovery. Biomedicines 2022, 10, 3182. [Google Scholar] [CrossRef]
- Zheng, Y.; Müller, J.; Kunz, S.; Siderius, M.; Maes, L.; Caljon, G.; Müller, N.; Hemphill, A.; Sterk, G.J.; Leurs, R. 3-nitroimidazo[1,2-b]pyridazine as a novel scaffold for antiparasitics with sub-nanomolar anti-Giardia lamblia activity. Int. J. Parasitol. Drugs Drug Resist. 2022, 19, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Vasquez-Jimenez, L.K.; Moreno-Herrera, A.; Juarez-Saldivar, A.; Gonzalez-Gonzalez, A.; Ortiz-Pérez, E.; Paz-Gonzalez, A.D.; Palos, I.; Ramirez-Moreno, E.; Rivera, G. Recent advances in the development of triose phosphate isomerase inhibitors as antiprotozoal agents. Curr. Med. Chem. 2022, 29, 2504–2529. [Google Scholar] [CrossRef]
- Morales-Luna, L.; Hernandez-Ochoa, B.; Martinez-Rosas, V.; Navarrete-Vazquez, G.; Ortega-Cuellar, D.; Rufino-Gonzalez, Y.; González-Valdez, A.; Arreguin-Espinosa, R.; Franco-Vasquez, A.M.; de la Cruz, V.P.; et al. Giardia lamblia 6PGL fused protein inhibitors decrease trophozoite viability: A new alternative against Giardiasis. Int. Mol. Sci. 2022, 23, 14358. [Google Scholar] [CrossRef]
- Auriostigue-Bautista, J.C.; Hernández-Vázquez, E.; González-Calderón, D.; Figueroa-Romero, J.L.; Castillo-Villanueva, A.; Torres-Arroyo, A.; Ponce-Macotela, M.; Rufino-González, Y.; Martínez-Gordillo, M.; Miranda, L.D.; et al. Discovery of Benzopyrrolizidines as Promising Antigiardiasic Agents. Front. Cell. Infect. Microbiol. 2022, 12, 828100. [Google Scholar] [CrossRef]
- Fernández-Lainez, C.; de la Mora-de la Mora, I.; Enríquez-Flores, S.; García-Torres, I.; Flores-López, L.A.; Gutiérrez-Castrellón, P.; de Vos, P.; López-Velázquez, G. The Giardial Arginine Deiminase Participates in Giardia-Host Immunomodulation in a Structure-Dependent Fashion via Toll-like Receptors. Int. J. Mol. Sci. 2022, 23, 11552. [Google Scholar] [CrossRef]
- Baz, M.G.; Elmarhoumy, S.M.; Saied, E.M.; Zoghroban, H.S. Evaluation of the efficacy of gold nanoparticles on Giardia lamblia infection in experimental animals. Exp. Parasitol. 2022, 238, 108277. [Google Scholar] [CrossRef] [PubMed]
- El-Gendy, A.M.L.; Mohammed, M.A.A.; Ghallab, M.M.I.; Abdel Aziz, M.O.; Ibrahim, S.M. Therapeutic Effect of Chitosan Nanoparticles and Metronidazole in Treatment of Experimentally Giardiasis Infected Hamsters. Iran. J. Parasitol. 2021, 16, 32–42. [Google Scholar] [CrossRef]
- Zoghroban, H.S.; Ibrahim, F.M.; Nassef, N.A.; Saad, A. The impact of L-citrulline on murine intestinal cell integrity, immune response, and arginine metabolism in the face of Giardia lamblia infection. Acta Trop. 2022, 237, 106748. [Google Scholar] [CrossRef] [PubMed]
- Palomo-Ligas, L.; Estrada-Camacho, J.; Garza-Ontiveros, M.; Vargas-Villanueva, J.R.; Gutiérrez-Gutiérrez, F.; Nery-Flores, S.D.; Cañas Montoya, J.A.; Ascacio-Valdés, J.; Campos-Muzquiz, L.G.; Rodriguez-Herrera, R. Polyphenolic extract from Punica granatum peel causes cytoskeleton-related damage on Giardia lamblia trophozoites in vitro. PeerJ 2022, 10, e13350. [Google Scholar] [CrossRef] [PubMed]
- Pryshliak, O.Y.; Protsyk, A.L.; Semaniv, M.V.; Boichuk, O.P.; Gerych, P.R. Effect of probiotics on the intestinal microbiota of patients with Giardiasis and ascariasis. J. Med. Life 2022, 15, 1278–1282. [Google Scholar] [CrossRef]
- Ribeiro, M.R.S.; Oliveira, D.R.; Oliveira, F.M.S.; Callari, M.V.; Martins, F.S.; Nicoli, J.R.; Torres, M.F.; Andrade, M.E.R.; Cardoso, V.N.; Gomes, M.A. Effect of probiotic Saccharomyces boulardii in experimental Giardiasis. Benef. Microbes 2018, 18, 789–797. [Google Scholar] [CrossRef]
- Fahmy, A.; Abuelenain, G.L.; Rasheed, N.; Abdou, A. “de novo” repurposing of Daflon as anti-intestinal parasitic drug in experimental Giardiasis. Exp. Parasitol. 2021, 226–227, 108124. [Google Scholar] [CrossRef]
- Drinić, M.; Raninger, A.; Zraunig, A.; Astelbauer, F.; Leitsch, D.; Obwaller, A.; Walochnik, J.; Greger, H.; Duchene, M. Activity of methylgerambullin from Glycosmis species (Rutaceae) against Entamoeba histolytica and Giardia duodenalis in vitro. Int. J. Parasitol. Drugs Drug Resist. 2019, 10, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Peckova, R.; Sak, B.; Kvetonova, D.; Kvac, M.; Koritakva, E.; Foitova, I. The course of experimental Giardiasis in Mongolian gerbil. Parasitol. Res. 2018, 117, 2437–2443. [Google Scholar] [CrossRef] [PubMed]









Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benchimol, M.; Gadelha, A.P.; de Souza, W. Ultrastructural Alterations of the Human Pathogen Giardia intestinalis after Drug Treatment. Pathogens 2023, 12, 810. https://doi.org/10.3390/pathogens12060810
Benchimol M, Gadelha AP, de Souza W. Ultrastructural Alterations of the Human Pathogen Giardia intestinalis after Drug Treatment. Pathogens. 2023; 12(6):810. https://doi.org/10.3390/pathogens12060810
Chicago/Turabian StyleBenchimol, Marlene, Ana Paula Gadelha, and Wanderley de Souza. 2023. "Ultrastructural Alterations of the Human Pathogen Giardia intestinalis after Drug Treatment" Pathogens 12, no. 6: 810. https://doi.org/10.3390/pathogens12060810
APA StyleBenchimol, M., Gadelha, A. P., & de Souza, W. (2023). Ultrastructural Alterations of the Human Pathogen Giardia intestinalis after Drug Treatment. Pathogens, 12(6), 810. https://doi.org/10.3390/pathogens12060810







