Nucleic Acid Based Fluorinated Derivatives: New Tools for Biomedical Applications
Abstract
:1. Introduction

2. Fluorocarbon Nucleoside Amphiphiles (FNs)
2.1. Fluorocarbon Amphiphiles
2.2. Nucleoside Amphiphiles
2.3. Hybrid Fluorocarbon Nucleoside Amphiphiles

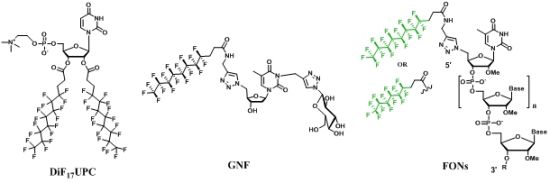
3. Fluorocarbon OligoNucleotide Amphiphiles (FONs) and Fluorinated Oligonucleotide Analogues
3.1. Fluorocarbon OligoNucleotide Amphiphiles (FONs)

3.2. Application of Fluorinated Oligonucleotide Analogues



4. Conclusions
Acknowledgments
Conflict of Interest
References
- Watson, J.D. A structure for deoxyribose nucleic acid. Nature 1953, 171, 737–738. [Google Scholar] [CrossRef]
- Franklin, R.E. Molecular configuration in sodium thymonucleate. Nature 1953, 171, 740–741. [Google Scholar]
- Wilkins, M.H.F. Molecular structure of deoxypentose nucleic acids. Nature 1953, 171, 738–740. [Google Scholar] [CrossRef]
- Sessler, J.L.; Lawrence, C.M.; Jayawickramarajah, J.G.M. Molecular recognition via base-pairing. Chem. Soc. Rev. 2007, 36, 314–325. [Google Scholar]
- Whitesides, G.M.; Mathias, J.P.; Seto, C.T. Molecular self-assembly and nanochemistry: A chemical strategy for the synthesis of nanostructures. Science 1991, 254, 1312–1319. [Google Scholar]
- Bowden, N.; Terfort, A.; Carbeck, J.; Whitesides, G.M. Self-assembly of mesoscale objects into ordered two-dimensional arrays. Science 1997, 276, 233–235. [Google Scholar]
- Rosemeyer, H. Nucleolipids: Natural occurrence, synthesis, molecular recognition, and supramolecular assemblies as potential precursors of life and bioorganic materials. Chem. Biodivers. 2005, 2, 977–1062. [Google Scholar] [CrossRef]
- Gissot, A.; Camplo, M.; Grinstaff, M.W.; Barthélémy, P. Nucleoside, nucleotide and oligonucleotide based amphiphiles: A successful marriage of nucleic acids with lipids. Org. Biomol. Chem. 2008, 6, 1324–1333. [Google Scholar] [CrossRef]
- Ahlers, M.; Ringsdorf, H.; Rosemeyer, H.; Seela, F. Orientation, recognition, and photoreaction of nucleolipids in model membranes. Colloid Polym. Sci. 1990, 268, 132–142. [Google Scholar] [CrossRef]
- Huang, J.; Li, C.; Liang, Y. FT-SERS studies on molecular recognition capabilities of monolayers of novel nucleolipid amphiphiles. Langmuir 2000, 16, 3937–3940. [Google Scholar] [CrossRef]
- Lebeau, L.; Olland, S.; Oudet, P.; Mioskowski, C. Rational design and synthesis of phospholipids for the two-dimensional crystallization of DNA gyrase, a key element in chromosome organization. Chem. Phys. Lipid 1992, 62, 93–103. [Google Scholar] [CrossRef]
- Barthélémy, P.; Lee, S.J.; Grinstaff, M. Supramolecular assemblies with DNA. Pure Appl. Chem. 2005, 77, 2133–2148. [Google Scholar]
- Berti, D.; Franchi, L.; Baglioni, P.; Luisi, P.L. Molecular recognition in monolayers. Complementary base pairing in dioleoylphosphatidyl derivatives of adenosine, uridine, and cytidine. Langmuir 1997, 13, 3438–3444. [Google Scholar] [CrossRef]
- Baglioni, P.; Berti, D. Self assembly in micelles combining stacking and H-bonding. Curr. Opin. Colloid Interface Sci. 2003, 8, 55–61. [Google Scholar] [CrossRef]
- Fortini, M.; Berti, D.; Baglioni, P.; Ninham, B.W. Specific anion effects on the aggregation properties of anionic nucleolipids. Curr. Opin. Colloid Interface Sci. 2004, 9, 168–172. [Google Scholar] [CrossRef]
- Mulet, X.; Kaasgaard, T.; Conn, C.E.; Waddington, L.J.; Kennedy, D.F.; Weerawardena, A.; Drummond, C.J. Nanostructured nonionic thymidine nucleolipid self-assembly materials. Langmuir 2010, 26, 18415–18423. [Google Scholar]
- Murgia, S.; Lampis, S.; Angius, R.; Berti, D.; Monduzzi, M. Orientation and specific interactions of nucleotides and nucleolipids inside monoolein-based liquid crystals. J. Phys. Chem. B 2009, 113, 9205–9215. [Google Scholar] [CrossRef]
- Godeau, G.; Barthélémy, P. Glycosyl-nucleoside lipids as low-molecular-weight gelators. Langmuir 2009, 25, 8447–8450. [Google Scholar] [CrossRef]
- Khiati, S.; Luvino, D.; Oumzil, K.; Chauffert, B.; Camplo, M.; Barthélémy, P. Nucleoside-lipid-based nanoparticles for cisplatin delivery. ACS Nano 2011, 5, 8649–8655. [Google Scholar] [CrossRef]
- Hong, C.I.; Nechaev, A.; Kirisits, A.J.; Vig, R.; West, C.R.; Manouilov, K.K.; Chu, C.K. Synthesis and biological activity of anti-HIV nucleoside conjugates of ether and thioether phospholipids. J. Med. Chem. 1996, 39, 1771–1777. [Google Scholar] [CrossRef]
- Ambike, A.; Rosilio, V.; Stella, B.; Lepétre-Mouelhi, S.; Couvreur, P. Interaction of self-assembled squalenoyl gemcitabine nanoparticles with phospholipid−cholesterol monolayers mimicking a biomembrane. Langmuir 2011, 27, 4891–4899. [Google Scholar]
- Riess, J.G. Highly fluorinated amphiphilic molecules and self-assemblies with biomedical potential. Curr. Opin. Colloid Interface Sci. 2009, 14, 294–304. [Google Scholar] [CrossRef]
- Krafft, M.P. Controlling phospholipid self-assembly and film properties using highly fluorinated components—Fluorinated monolayers, vesicles, emulsions and microbubbles. Biochimie 2012, 94, 11–25. [Google Scholar]
- Krafft, M.P.; Riess, J.G. Highly fluorinated amphiphiles and colloidal systems, and their applications in the biomedical field. A contribution. Biochimie 1998, 80, 489–514. [Google Scholar] [CrossRef]
- Riess, J.G.; Krafft, M.P. Fluorinated phosphocholine-based amphiphiles as components of fluorocarbon emulsions and fluorinated vesicles. Chem. Phys. Lipids 1995, 75, 1–14. [Google Scholar] [CrossRef]
- Held, P.; Lach, F.; Lebeau, L.; Mioskowski, C. Synthesis and preliminary evaluation of a new class of fluorinated amphiphiles designed for in-plane immobilisation of biological macromolecules. Tetrahedron Lett. 1997, 38, 1937–1940. [Google Scholar] [CrossRef]
- Lebeau, L.; Lach, F.; Vénien-Bryan, C.; Renault, A.; Dietrich, J.; Jahn, T.; Palmgren, M.G.; Kühlbrandt, W.; Mioskowski, C. Two-dimensional crystallization of a membrane protein on a detergent-resistant lipid monolayer. J. Mol. Biol. 2001, 308, 639–647. [Google Scholar] [CrossRef]
- Courty, S.; Lebeau, L.; Martel, L.; Lenné, P.-F.; Balavoine, F.; Dischert, W.; Konovalov, O.; Mioskowski, C.; Legrand, J.-F.; Vénien-Bryan, C. Two-dimensional crystallization of a histidine-tagged protein on monolayers of fluidity-enhanced Ni2+-chelating lipids. Langmuir 2002, 18, 9502–9512. [Google Scholar]
- Clary, L.; Gadras, C.; Greiner, J.; Rolland, J.-P.; Santaella, C.; Vierling, P.; Gulik, A. Phase behavior of fluorocarbon and hydrocarbon double-chain hydroxylated and galactosylated amphiphiles and bolaamphiphiles. Long-term shelf-stability of their liposomes. Chem. Phys. Lipids 1999, 99, 125–137. [Google Scholar] [CrossRef]
- Pasc-Banu, A.; Blanzat, M.; Belloni, M.; Perez, E.; Mingotaud, C.; Rico-Lattes, I.; Labrot, T.; Oda, R. Spontaneous vesicles of single-chain sugar-based fluorocarbon surfactants. J. Fluor. Chem. 2005, 126, 33–38. [Google Scholar] [CrossRef]
- Roytman, R.; Adler-Abramovich, L.; Ajish, K.S.; Kuan, T.-C.; Lin, C.-C.; Gazit, E.; Brik, A. Exploring the self-assembly of glycopeptides using a diphenylalanine scaffold. Org. Biomol. Chem. 2011, 9, 5755–5761. [Google Scholar]
- Platen, T.; Schüler, T.; Tremel, W.; Hoffmann-Röder, A. Synthesis and antibody bonding of higly fluorinated amphiphilic MUC1 glycopeptide antigens. Eur. J. Org. Chem. 2011, 3878–3887. [Google Scholar]
- Riess, J.G. Fluorinated vesicles. J. Drug Target. 1994, 2, 455–468. [Google Scholar] [CrossRef]
- Fabio, K.; Di Giorgio, C.; Vierling, P. New perfluorinated polycationic dimerizable detergents for the formulation of monomolecular DNA nanoparticles and their in vitro transfection efficiency. Biochim. Biophys. Acta 1724, 203–214. [Google Scholar]
- Klein, E.; Leborgne, C.; Ciobanu, M.; Klein, J.; Frisch, B.; Pons, F.; Zuber, G.; Scherman, D.; Kichler, A.; Lebeau, L. Nucleic acid transfer with hemifluorinated polycationic lipids. Biomaterials 2010, 31, 4781–4788. [Google Scholar]
- Klein, E.; Ciobanu, M.; Klein, J.; MacHi, V.; Leborgne, C.; Vandamme, T.; Frisch, B.; Pons, F.; Kichler, A.; Zuber, G.; Lebeau, L. "HFP" fluorinated cationic lipids for enhanced lipoplex stability and gene delivery. Bioconjug. Chem. 2010, 21, 360–371. [Google Scholar] [CrossRef]
- Berti, D.; Montis, C.; Baglioni, P. Self-assembly of designer biosurfactants. Soft Matter 2011, 7, 7150–7158. [Google Scholar]
- Wang, Y.; Moreau, L.; Barthélémy, P.; El Maataoui, M.; Grinstaff, M.W. Supramolecular assemblies of nucleoside phosphocholine amphiphiles. J. Am. Chem. Soc. 2004, 126, 7533–7539. [Google Scholar]
- Desbat, B.; Manet, S.; Aimé, C.; Labrot, T.; Oda, R. Aggregation behaviors of gemini nucleotide at the air-water interface and in solutions induced by adenine-uracil interaction. J. Colloid Interface Sci. 2005, 283, 555–564. [Google Scholar] [CrossRef]
- Pincet, F.; Perez, E.; Bryant, G.; Lebeau, L.; Mioskowski, C. Long-range attraction between nucleosides with short-range specificity: Direct measurements. Phys. Rev. Lett. 1994, 73, 2780–2783. [Google Scholar] [CrossRef]
- Pincet, F.; Perez, E.; Lebeau, L.; Mioskowski, C. Long range H-bond specific interactions between nucleosides. J. Chem. Soc. Faraday Trans. 1995, 91, 4329–4330. [Google Scholar] [CrossRef]
- Pincet, F.; Perez, E.; Bryant, G.; Lebeau, L.; Mioskowski, C. Specific forces between DNA bases. Mod. Phys. Lett. B 1996, 10, 81–90. [Google Scholar] [CrossRef]
- Pincet, F.; Rawicz, W.; Perez, E.; Lebeau, L.; Mioskowski, C.; Evans, E. Electrostatic nanotitration of weak biochemical bonds. Phys. Rev. Lett. 1997, 79, 1949–1952. [Google Scholar] [CrossRef]
- Pincet, F.; Lebeau, L.; Cribier, S. Short-range specific forces are able to induce hemifusion. Eur. Biophys. J. 2001, 30, 91–97. [Google Scholar] [CrossRef]
- Perez, E.; Pincet, F.; Goldmann, M.; Mioskowski, C.; Lebeau, L. Translational order in liquid-expanded lipid monolayers functionalized with nucleosides. Eur. Phys. J. B 1998, 6, 1–4. [Google Scholar] [CrossRef]
- Tareste, D.; Pincet, F.; Perez, E.; Rickling, S.; Mioskowski, C.; Lebeau, L. Energy of hydrogen bonds probed by the adhesion of functionalized lipid layers. Biophys. J. 2002, 83, 3675–3681. [Google Scholar] [CrossRef]
- Tareste, D.; Pincet, F.; Lebeau, L.; Perez, É. Hydrophobic forces and hydrogen bonds in the adhesion between retinoid-coated surfaces. Langmuir 2007, 23, 3225–3229. [Google Scholar] [CrossRef]
- Yang, H.W.; Yi, J.W.; Bang, E.-K.; Jeon, E.M.; Kim, B.H. Cationic nucleolipids as efficient siRNA carriers. Org. Biomol. Chem. 2011, 9, 291–296. [Google Scholar]
- Ceballos, C.; Khiati, S.; Prata, C.A.; Zhang, X.X.; Giorgio, S.; Marsal, P.; Grinstaff, M.W.; Barthélémy, P.; Camplo, M. Cationic nucleoside lipids derived from universal bases: A rational approach for siRNA transfection. Bioconjug. Chem. 2010, 21, 1062–1069. [Google Scholar] [CrossRef]
- Moreau, L.; Barthélémy, P.; Li, Y.; Luo, D.; Prata, C.A.H.; Grinstaff, M.W. Nucleoside Phosphocholine Amphiphile for in vitro DNA transfection. Mol. BioSyst. 2005, 1, 260–264. [Google Scholar] [CrossRef]
- Chabaud, P.; Camplo, M.; Payet, D.; Serin, G.; Moreau, L.; Barthélémy, P.; Grinstaff, M.W. Cationic nucleoside lipids for gene delivery. Bioconjug. Chem. 2006, 17, 466–472. [Google Scholar] [CrossRef]
- Céballos, C.; Khiati, S.; Barthélémy, P.; Camplo, M. Acyclic anionic nucleolipids for DNA delivery. J. Nanosci. Lett. 2012, 2, 20. [Google Scholar]
- Khiati, S.; Pierre, N.; Andriamanarivo, S.; Grinstaff, M.W.; Arazam, N.; Nallet, F.; Navailles, L.; Barthélémy, P. Anionic nucleotide-lipids for in vitro DNA transfection. Bioconjug. Chem. 2009, 20, 1765–1772. [Google Scholar] [CrossRef]
- Moreau, L.; Barthélémy, P.; El Maataoui, M.; Grinstaff, M.W. Supramolecular assemblies of nucleoside phosphocholine amphiphiles. J. Am. Chem. Soc. 2004, 126, 7533–7539. [Google Scholar]
- Moreau, L.; Campins, N.; Grinstaff, M.W.; Barthélémy, P. A fluorocarbon nucleoamphiphile for the construction of actinide loaded microspheres. Tetrahedron Lett. 2006, 47, 7117–7120. [Google Scholar]
- Godeau, G.; Barthélémy, P. Glycosyl-nucleoside lipids as low-molecular-weight gelators. Langmuir 2009, 25, 8447–8450. [Google Scholar] [CrossRef]
- Godeau, G.; Bernard, L.; Staedel, C.; Barthélémy, P. Glycosyl-nucleoside-lipid based supramolecular assembly as a nanostructured material with nucleic acid delivery capabilities. Chem. Commun. 2009, 34, 5127–5129. [Google Scholar]
- Godeau, G.; Brun, C.; Arnion, H.; Staedel, C.; Barthélémy, P. Glycosyl-nucleoside fluorinated amphiphiles as compoents of nanostructured hydrogels. Tetrahedron Lett. 2010, 51, 1012–1015. [Google Scholar]
- Studer, A.; Hadida, S.; Ferritto, R.; Kim, S.-Y.; Jeger, P.; Wipf, P.; Curran, D.P. Fluorous synthesis: A fluorous-phase strategy for improving separation efficiency in organic synthesis. Science 1997, 275, 823–826. [Google Scholar] [CrossRef]
- Pongdee, R.; Liu, H.-W. Elucidation of enzyme mechanisms using fluorinated substrate analogues. Bioorg. Chem. 2004, 32, 393–437. [Google Scholar] [CrossRef]
- Müller, K.; Faeh, C.; Diederich, F. Fluorine in pharmaceuticals: Looking beyond intuition. Science 2007, 317, 1881–1886. [Google Scholar]
- Curran, D.P. Chemistry: Fluorous tags unstick messy chemical biology problems. Science 2008, 321, 1645–1646. [Google Scholar] [CrossRef]
- Godeau, G.; Arnion, H.; Brun, C.; Staedel, C.; Barthélémy, P. Fluorocarbon oligonucleotide conjugates for nucleic acids delivery. Med. Chem. Commun. 2010, 1, 76–78. [Google Scholar] [CrossRef]
- Patwa, A.; Gissot, A.; Bestel, I.; Barthélémy, P. Hybrid lipid oligonucleotide conjugates: Synthesis, self-assemblies and biomedical applications. Chem. Soc. Rev. 2011, 40, 5844–5854. [Google Scholar] [CrossRef]
- Godeau, G.; Staedel, C.; Barthélémy, P. Lipid-conjugated oligonucleotides via “click chemistry” efficiently inhibit hepatitis C virus translation. J. Med. Chem. 2008, 51, 4374–4376. [Google Scholar] [CrossRef]
- Huisgen, R. 1,3-Dipolar Cycloaddition Chemistry; Wiley: New York, NY ,USA, 1984; Volume 1, pp. 1–176. [Google Scholar]
- Schmidt, S.; Niemann, A.; Krynetskaya, N.F.; Oretskaya, T.S.; Metelev, V.G.; Suchomlinov, V.V.; Shabarova, Z.A.; Cech, D. The use of oligonucleotide probes containing 2'-deoxy-2'-fluoronucleosides for regiospecific cleavage of RNA by RNase H from Escherichia coli. Biochim. Biophys. Acta 1130, 41–46. [Google Scholar]
- Kawasaki, A.M.; Casper, M.D.; Freier, S.M.; Lesnik, E.A.; Zounes, M.C.; Cummins, L.L.; Gonzalez, C.; Dan Cook, P. Uniformly modified 2′-deoxy-2′-fluoro phosphorothioate oligonucleotides as nuclease-resistant antisense compounds with high affinity and specificity for RNA targets. J. Med. Chem. 1993, 36, 831–841. [Google Scholar] [CrossRef]
- Kois, P.; Tocik, Z.; Spassova, M.; Ren, W.-Y.; Rosenberg, I.; Soler, J.F.; Watanabe, K.A. Synthesis and some properties of modified oligonucleotides. 2. oligonucleotides containing 2'-deoxy-2'-fluoro-β-D-arabinofuranosyl pyrimidine nucleosides. Nucleosides Nucleotides 1993, 12, 1093–1109. [Google Scholar] [CrossRef]
- Rosenberg, I.; Soler, J.F.; Tocik, Z.; Ren, W.-Y.; Ciszewski, L.A.; Kois, P.; Pankiewicz, K.W.; Spassova, M.; Watanabe, K.A. Synthesis of oligodeoxynucleotides containing the C-nucleoside and 2'- deoxy-2'-fluoro-ara-nucleoside moieties by the H-phosphonate method. Nucleosides Nucleotides 1993, 12, 381–401. [Google Scholar] [CrossRef]
- Reif, B.; Wittmann, V.; Schwalbe, H.; Griesinger, C.; Wörner, K.; Jahn-Hofmann, K.; Engels, J.W.; Bermel, W. 132. Structural comparison of oligoribonucleotides and their 2′-deoxy-2′-fluoro analogs by heteronuclear NMR spectroscopy. Helv. Chim. Acta 1997, 80, 1952–1971. [Google Scholar] [CrossRef]
- Damha, M.J.; Wilds, C.J.; Noronha, A.; Brukner, I.; Borkow, G.; Arion, D.; Parniak, M.A. Hybrids of RNA and arabinonucleic acids (ANA and 2'F-ANA) are substrates of ribonuclease H. J. Am. Chem. Soc. 1998, 120, 12976–12977. [Google Scholar]
- Wilds, C.J.; Damha, M.J. Duplex recognition by oligonucleotides containing 2'-deoxy-2'-fluoro-D-arabinose and 2'-deoxy-2'-fluoro-D-ribose. Intermolecular 2'-OH-phosphate contacts versus sugar puckering in the stabilization of triple-helical complexes. Bioconjugate Chem. 1999, 10, 299–305. [Google Scholar] [CrossRef]
- Wilds, C.J.; Damha, M.J. 2'-Deoxy-2'-fluoro-β-D-arabinonucleosides and oligonucleotides (2'F-ANA): Synthesis and physicochemical studies. Nucleic Acids Res. 2000, 28, 3625–3635. [Google Scholar] [CrossRef]
- Damha, M.J.; Noronha, A.M.; Wilds, C.J.; Trempe, J.-F.; Denisov, A.; Pon, R.T.; Gehring, K. Properties of arabinonucleic acids (ANA & 20′F-ANA): Implications for the design of antisense therapeutics that invoke RNase H cleavage of RNA. Nucleosides Nucleotides Nucleic Acids 2001, 20, 429–440. [Google Scholar] [CrossRef]
- Denisov, A.Yu.; Noronha, A.M.; Wilds, C.J.; Trempe, J.-F.; Pon, R.T.; Gehring, K.; Damha, M.J. Solution structure of an arabinonucleic acid (ANA)/RNA duplex in a chimeric hairpin: Comparison with 2′-fluoro-ANA/RNA and DNA/RNA hybrids. Nucleic Acids Res. 2001, 29, 4284–4293. [Google Scholar] [CrossRef]
- Lok, C.-N.; Viazovkina, E.; Min, K.-L.; Nagy, E.; Wilds, C.J.; Damha, M.J.; Parniak, M.A. Potent gene-specific inhibitory properties of mixed-backbone antisense oligonucleotides comprised of 2′-deoxy-2′-fluoro-D-arabinose and 2′-deoxyribose nucleotides. Biochemistry 2002, 41, 3457–3467. [Google Scholar]
- Doi, Y.; Katafuchi, A.; Fujiwara, Y.; Hitomi, K.; Tainer, J.A.; Ide, H.; Iwai, S. Synthesis and characterization of oligonucleotides containing 2′-fluorinated thymidine glycol as inhibitors of the endonuclease III reaction. Nucleic Acids Res. 2006, 34, 1540–1551. [Google Scholar] [CrossRef]
- Dowler, T.; Bergeron, D.; Tedeschi, A.-L.; Paquet, L.; Ferrari, N.; Damha, M.J. Improvements in siRNA properties mediated by 2′-deoxy-2′-fluoro-β-D-arabinonucleic acid (FANA). Nucleic Acids Res. 2006, 34, 1669–1675. [Google Scholar] [CrossRef]
- Watts, J.K.; Choubdar, N.; Sadalapure, K.; Robert, F.; Wahba, A.S.; Pelletier, J.; Pinto, B.M.; Damha, M.J. 2′-Fluoro-4′-thioarabino-modified oligonucleotides: Conformational switches linked to siRNA activity. Nucleic Acids Res. 2007, 35, 1441–1451. [Google Scholar] [CrossRef]
- Peng, C.G.; Damha, M.J. Polymerase-directed synthesis of 2′-deoxy-2′-fluoro-β-D- arabinonucleic acids. J. Am. Chem. Soc. 2007, 129, 5310–5311. [Google Scholar] [CrossRef]
- Peng, C.G.; Damha, M.J. G-quadruplex induced stabilization by 2′-deoxy-2′-fluoro-d-arabinonucleic acids (2′F-ANA). Nucleic Acids Res. 2007, 35, 4977–4988. [Google Scholar] [CrossRef]
- Watts, J.K.; Martín-Pintado, N.; Gómez-Pinto, I.; Schwartzentruber, J.; Portella, G.; Orozco, M.; González, C.; Damha, M.J. Differential stability of 2′F-ANA•RNA and ANA•RNA hybrid duplexes: Roles of structure, pseudohydrogen bonding, hydration, ion uptake and flexibility. Nucleic Acids Res. 2010, 38, 2498–2511. [Google Scholar]
- Deleavey, G.F.; Watts, J.K.; Alain, T.; Robert, F.; Kalota, A.; Aishwarya, V.; Pelletier, J.; Gewirtz, A.M.; Sonenberg, N.; Damha, M.J. Synergistic effects between analogs of DNA and RNA improve the potency of siRNA-mediated gene silencing. Nucleic Acids Res. 2010, 38, 4547–4557. [Google Scholar]
- Erande, N.; Gunjal, A.D.; Fernandes, M.; Kumar, V.A. Probing the furanose conformation in the 2’-5’ strand of isoDNA:RNA duplexes by freezing the nucleoside conformations. Chem. Commun. 2011, 47, 4007–4009. [Google Scholar]
- Parsch, J.; Engels, J.W. Synthesis of fluorobenzene and benzimidazole nucleic-acid analogues and their influence on stability of RNA duplexes. Helv. Chim. Acta 2000, 83, 1791–1808. [Google Scholar] [CrossRef]
- Parsch, J.; Engels, J.W. C–F … H–C hydrogen bonds in ribonucleic acids. J. Am. Chem. Soc. 2002, 124, 5664–5672. [Google Scholar] [CrossRef]
- Rastinejad, F.; Evilia, C.; Lu, P. Studies of nucleic acids and their protein interactions by 19F NMR. Methods Enzymol. 1995, 261, 560–575. [Google Scholar] [CrossRef]
- Rastinejad, F.; Lu, P. Bacteriophage T7 RNA polymerase. 19F-nuclear magnetic resonance observations at 5-fluorouracil-substituted promoter DNA and RNA transcript. J. Mol. Biol. 1993, 232, 105–122. [Google Scholar] [CrossRef]
- Chu, W.-C.; Feiz, V.; Derrick, W.B.; Horowitz, J. Fluorine-19 nuclear magnetic resonance as a probe of the solution structure of mutants of 5-Fluorouracil-substituted Escherichia coli valine tRNA. J. Mol. Biol. 1992, 227, 1164–1172. [Google Scholar] [CrossRef]
- Chu, W.-C.; Horowitz, J. Fluorine-19 NMR studies of the thermal unfolding of 5-fluorouracil-substituted Escherichia coli valine transfer RNA. FEBS Lett. 1991, 295, 159–162. [Google Scholar] [CrossRef]
- Parisot, D.; Malet-Martino, M.C.; Martino, R.; Crasnier, P. 19F nuclear magnetic resonance analysis of 5-fluorouracil metabolism in four differently pigmented strains of Nectria haematococca. Appl. Environ. Microbil. 1991, 57, 3605–3612. [Google Scholar]
- Tanabe, K.; Sugiura, M.; Nishimoto, S.-I. Monitoring of duplex and triplex formation by 19F NMR using oligodeoxynucleotides possessing 5-fluorodeoxyuridine unit as 19F signal transmitter. Bioorg. Med. Chem. 2010, 18, 6690–6694. [Google Scholar] [CrossRef] [Green Version]
- Danielson, M.A.; Falke, J.J. Use of 19F NMR to probe protein structure and conformational changes. Annu. Rev. Biophys. Biomol. Struct. 1996, 25, 163–195. [Google Scholar] [CrossRef]
- Barhate, N.B.; Barhate, R.N.; Cekan, P.; Drobny, G.; Sigurdsson, S.Th. A nonafluoro nucleoside as a sensitive 19F NMR probe of nucleic acid conformation. Org. Lett. 2008, 10, 2745–2747. [Google Scholar] [CrossRef]
- Dollé, F.; Hinnen, F.; Vaufrey, F.; Tavitian, B.; Crouzel, C. A general method for labeling oligodeoxynucleotides with 18F for in vivo PET imaging. J. Label. Compd. Radiopharm. 1997, 39, 319–330. [Google Scholar] [CrossRef]
- Tavitian, B.; Terrazzino, S.; Kühnast, B.; Marzabal, S.; Stettler, O.; Dollé, F.; Deverre, J.-R.; Jobert, A.; Hinnen, F.; Bendriem, B.; et al. In vivo imaging of oligonucleotides with positron emission tomography. Nat. Med. 1998, 4, 467–470. [Google Scholar] [CrossRef]
- Kuhnast, B.; Dollé, F.; Vaufrey, F.; Hinnen, F.; Crouzel, C.; Tavitian, B. Fluorine-18 labeling of oligonucleotides bearing chemically-modified ribose-phosphate backbones. J. Label. Compd. Radiopharm. 2000, 43, 837–848. [Google Scholar] [CrossRef]
- Kuhnast, B.; Hinnen, F.; Boisgard, R.; Tavitian, B.; Dollé, F. Fluorine-18 labelling of oligonucleotides: Prosthetic labelling at the 5′-end using the N-(4-[18F]fluorobenzyl)-2-bromoacetamide reagent. J. Label. Compd. Radiopharm. 2003, 46, 1093–1103. [Google Scholar] [CrossRef]
- Viel, T.; Kuhnast, B.; Hinnen, F.; Boisgard, R.; Tavitian, B.; Dollé, F. Fluorine-18 labelling of small interfering RNAs (siRNAs) for PET imaging. J. Label. Compd. Radiopharm. 2007, 50, 1159–1168. [Google Scholar] [CrossRef]
- Hedberg, E.; Långström, B. 18F-labelling of oligonucleotides using succinimido 4-[18F]fluorobenzoate. Acta Chem. Scand. 1998, 52, 1034–1039. [Google Scholar] [CrossRef]
- Pan, D.; Gambhir, S.S.; Toyokuni, T.; Iyer, M.R.; Acharya, N.; Phelps, M.E.; Barrio, J.R. Rapid synthesis of a 5'-fluorinated oligodeoxy-nucleotide: A model antisense probe for use in imaging with positron emission tomography (PET). Bioorg. Med. Chem. Lett. 1998, 8, 1317–1320. [Google Scholar] [CrossRef]
- Mercier, F.; Paris, J.; Kaisin, G.; Thonon, D.; Flagothier, J.; Teller, N.; Lemaire, C.; Luxen, A. General method for labeling siRNA by click chemistry with fluorine-18 for the purpose of PET imaging. Bioconjug. Chem. 2011, 22, 108–114. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dolain, C.; Patwa, A.; Godeau, G.; Barthélémy, P. Nucleic Acid Based Fluorinated Derivatives: New Tools for Biomedical Applications. Appl. Sci. 2012, 2, 245-259. https://doi.org/10.3390/app2020245
Dolain C, Patwa A, Godeau G, Barthélémy P. Nucleic Acid Based Fluorinated Derivatives: New Tools for Biomedical Applications. Applied Sciences. 2012; 2(2):245-259. https://doi.org/10.3390/app2020245
Chicago/Turabian StyleDolain, Christel, Amit Patwa, Guilhem Godeau, and Philippe Barthélémy. 2012. "Nucleic Acid Based Fluorinated Derivatives: New Tools for Biomedical Applications" Applied Sciences 2, no. 2: 245-259. https://doi.org/10.3390/app2020245