Germination and Seedling Development Responses of Sunflower (Helianthus annuus L.) Seeds to Temperature and Different Levels of Water Availability
Abstract
1. Introduction
2. Material and Methods
2.1. Temperature Test
2.2. Water Test
| Amount of Water Based on 1 mL Interval | Amount of Water Based on the TKW % | ||||
|---|---|---|---|---|---|
| 1 TN | 2 WA (mL) | 3 TN | 4 PW % | 5 WA (mL) | 6 RAW (mL) |
| 1 | 0 | 14 | 125 | 0.631 | 0.63075 |
| 2 | 1 | 15 | 250 | 1.262 | 1.2615 |
| 3 | 2 | 16 | 375 | 1.892 | 1.89225 |
| 4 | 3 | 17 | 500 | 2.523 | 2.523 |
| 5 | 4 | 18 | 625 | 3.154 | 3.15375 |
| 6 | 5 | 19 | 750 | 3.785 | 3.7845 |
| 7 | 6 | 20 | 875 | 4.415 | 4.41525 |
| 8 | 7 | 21 | 1000 | 5.046 | 5.046 |
| 9 | 8 | 22 | 1125 | 5.677 | 5.67675 |
| 10 | 9 | 23 | 1250 | 6.308 | 6.3075 |
| 11 | 10 | 24 | 1375 | 6.938 | 6.93825 |
| 12 | 11 | 25 | 1500 | 7.569 | 7.569 |
| 12 | 26 | 1625 | 8.200 | 8.19975 | |
| 27 | 1750 | 8.831 | 8.8305 | ||
| 28 | 1875 | 9.461 | 9.46125 | ||
| 29 | 2000 | 10.092 | 10.092 | ||
| 30 | 2125 | 10.723 | 10.72275 | ||
| 31 | 2250 | 11.354 | 11.3535 | ||
| 32 | 2375 | 11.984 | 11.98425 | ||
| 33 | 2500 | 12.615 | 12.615 | ||
2.3. Seed Number Test
2.4. Antifungal Test
2.5. Statistical Analyses
3. Results
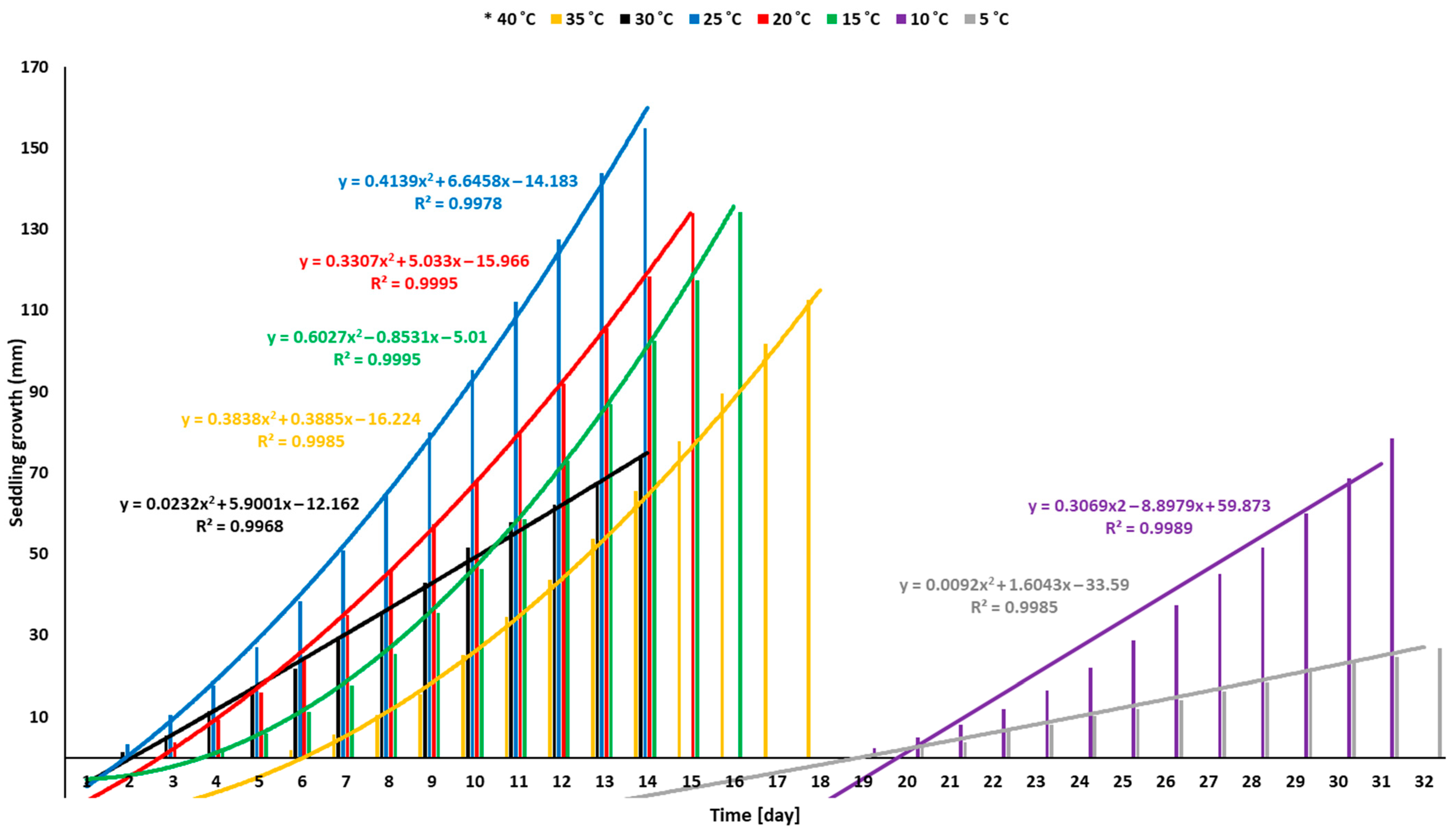
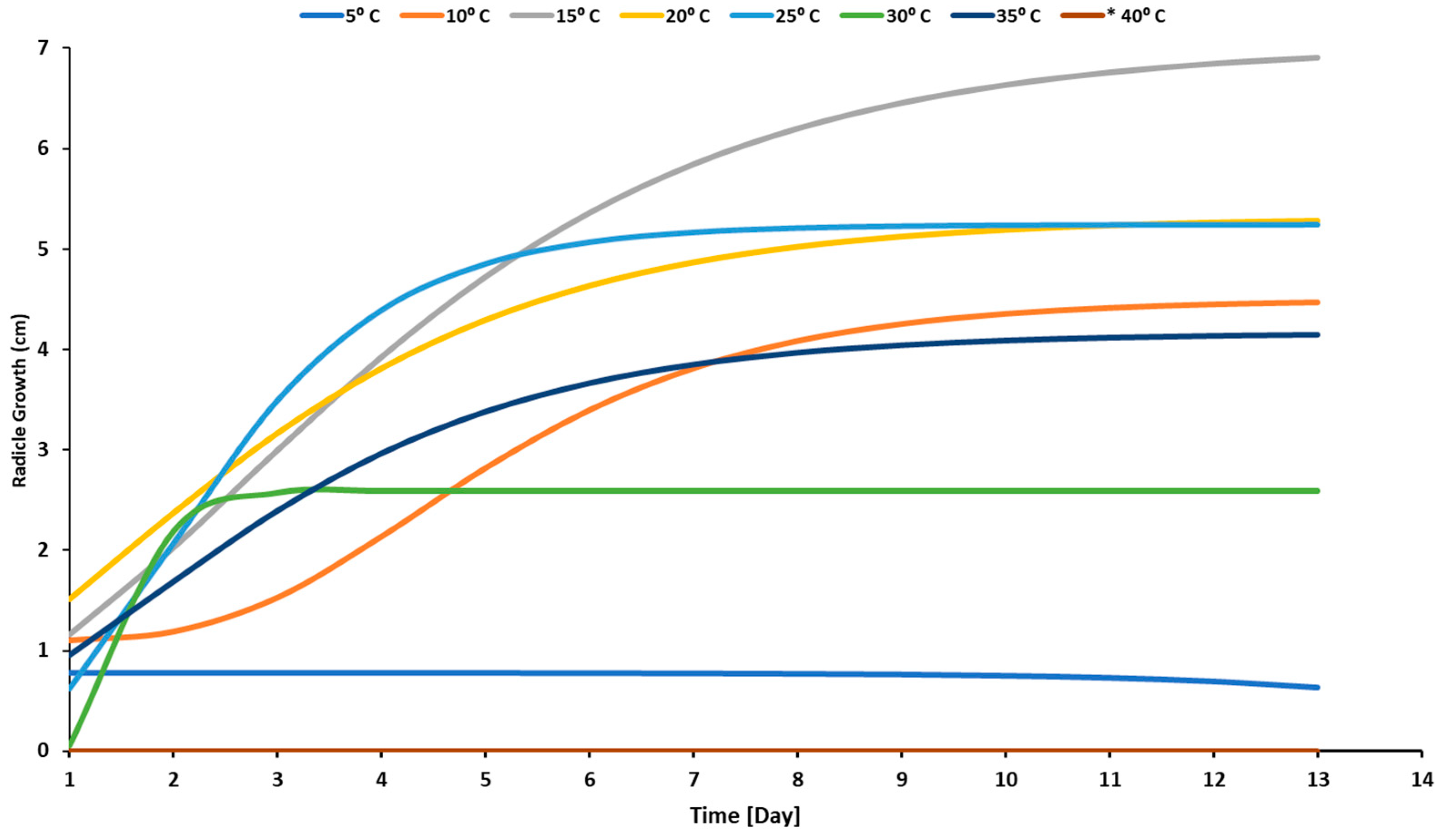
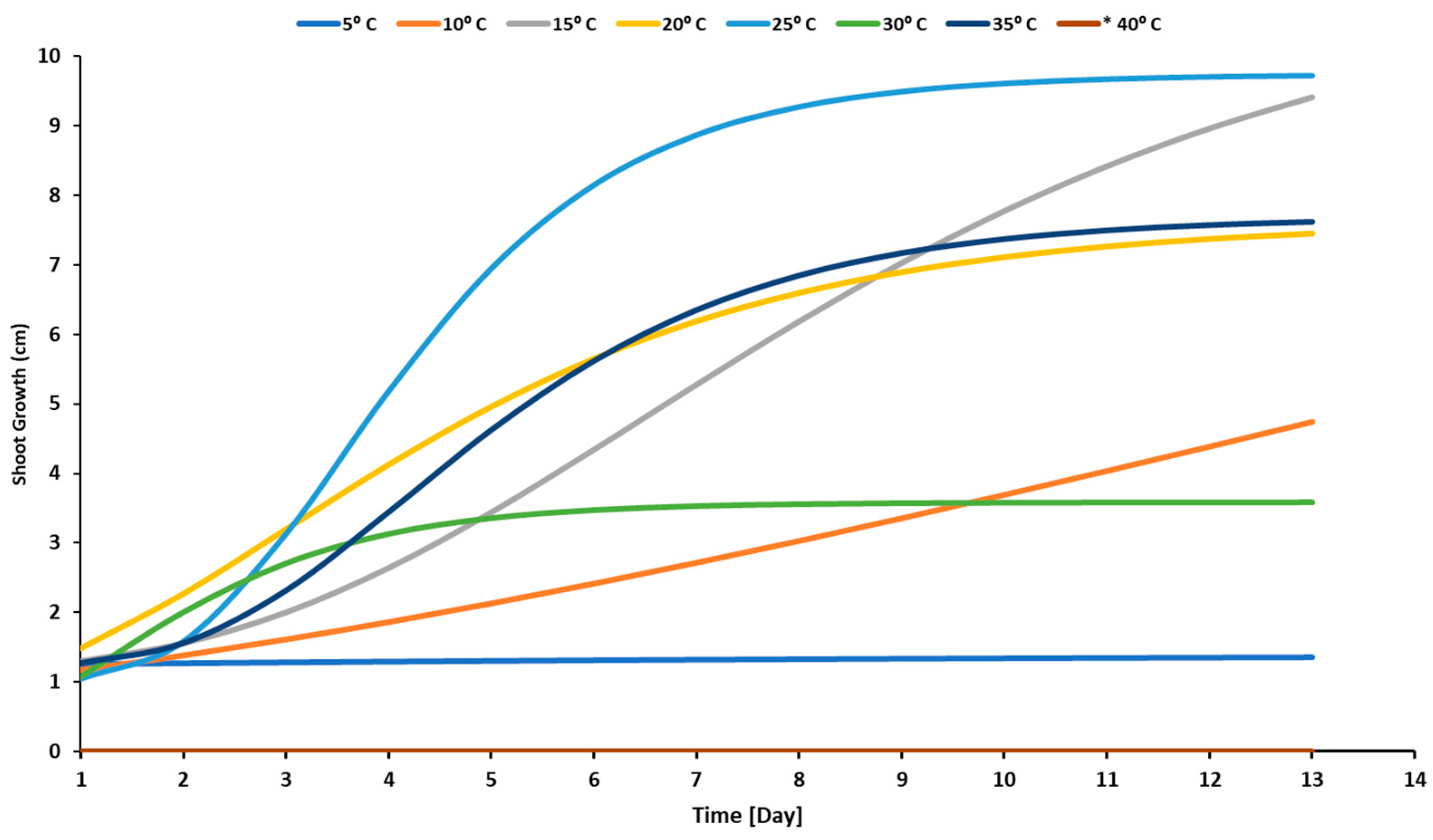
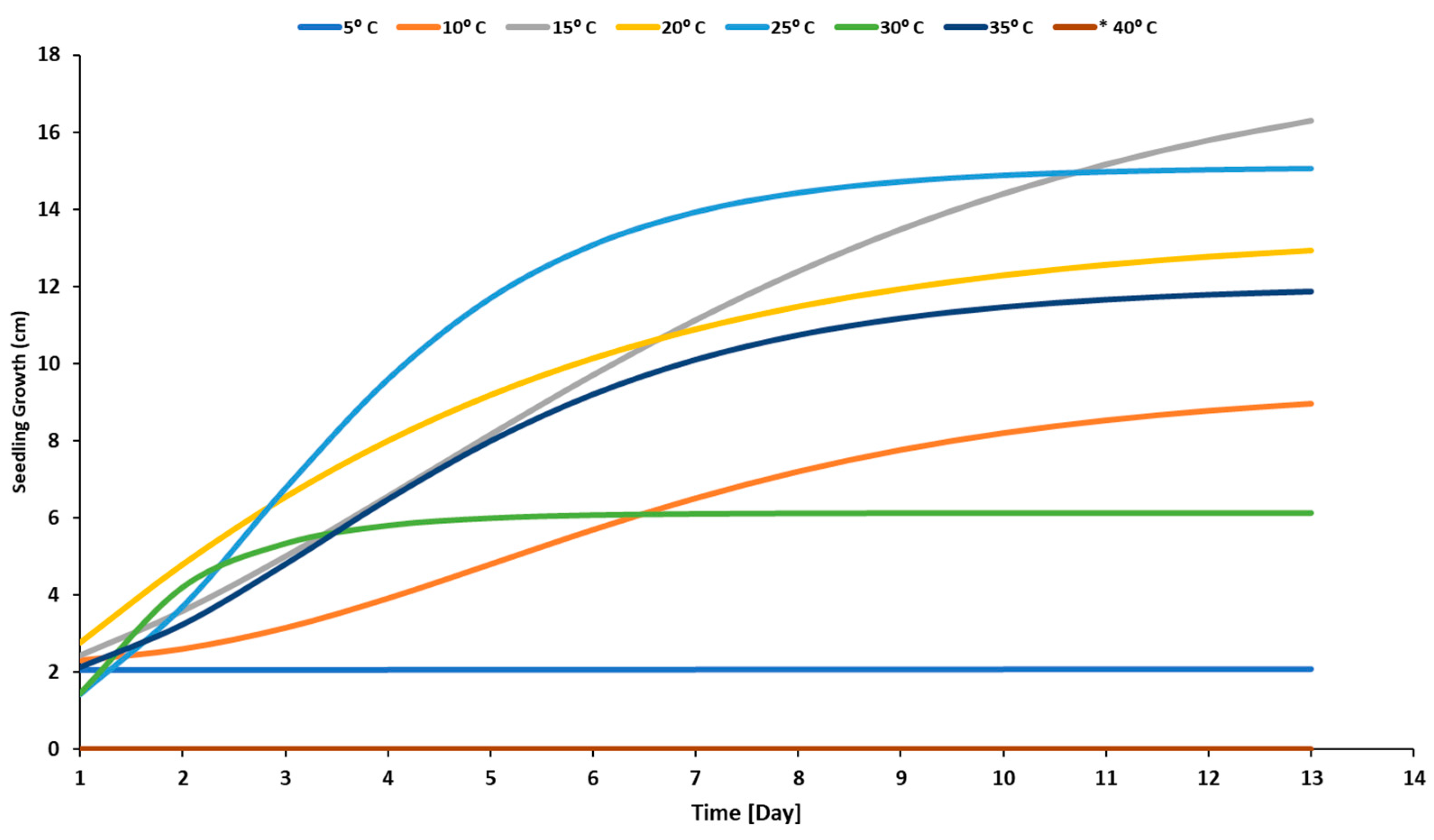
3.1. Temperature Effect
3.2. Water Effect
3.3. Seed Number Effect
3.4. Antifungal Effect
4. Discussion
4.1. Effect of Temperature Levels on a Radicle, Plumule, and Seedling Development of Sunflower
4.2. Effect of Water Levels on the Radicle, Plumule, and Seedling Development of Sunflower
4.3. Effect of Seed Number Density on Seed Germination and Growth Traits
4.4. Effect of Antifungal Experiment on a Radicle, Plumule, and Seedling Development
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Aubertot, J.N.; Deguine, J.P.; Lamichhane, J.R.; Robin, M.H.; Sarthou, J.P.; Steinberg, C. Vers Une Protection Agroécologique Des Cultures En Phase d’implantation. In Réussir l’Implantation Des Cultures. Enjeux Agroécologiques, Itinéraires Techniques; Quae: Paris, France, 2022; pp. 107–134. [Google Scholar]
- Ali, A.S.; Elozeiri, A.A. Metabolic Processes During Seed Germination. In Advances in Seed Biology; InTech: Vienna, Austria, 2017. [Google Scholar]
- Gummerson, R.J. The Effect of Constant Temperatures and Osmotic Potentials on the Germination of Sugar Beet. J. Exp. Bot. 1986, 37, 729–741. [Google Scholar] [CrossRef]
- Maraghni, M.; Gorai, M.; Neffati, M. Seed Germination at Different Temperatures and Water Stress Levels, and Seedling Emergence from Different Depths of Ziziphus lotus. S. Afr. J. Bot. 2010, 76, 453–459. [Google Scholar] [CrossRef]
- de Oliveira, A.K.M.; Ribeiro, J.W.F.; Pereira, K.C.L.; Silva, C.A.A. Efeito Da Temperatura Sobre a Germinação de Sementes de Diptychandra aurantiaca (Fabaceae). Acta Sci. Agron. 2013, 35, 203–208. [Google Scholar] [CrossRef]
- Alvarado, V.; Bradford, K.J. A Hydrothermal Time Model Explains the Cardinal Temperatures for Seed Germination. Plant Cell Environ. 2002, 25, 1061–1069. [Google Scholar] [CrossRef]
- Motsa, M.M.; Slabbert, M.M.; van Averbeke, W.; Morey, L. Effect of Light and Temperature on Seed Germination of Selected African Leafy Vegetables. S. Afr. J. Bot. 2015, 99, 29–35. [Google Scholar] [CrossRef]
- Hawker, J.; Jenner, C. High Temperature Affects the Activity of Enzymes in the Committed Pathway of Starch Synthesis in Developing Wheat Endosperm. Funct. Plant Biol. 1993, 20, 197. [Google Scholar] [CrossRef]
- Nykiforuk, C.L.; Johnson-Flanagan, A.M. Germination and Early Seedling Development under Low Temperature in Canola. Crop Sci. 1994, 34, 1047–1054. [Google Scholar] [CrossRef]
- Hoppe, A.; Theimer, R.R. Degradation of Oil Bodies Isolated from Cotyledons during Germination of Rapeseed Seedlings. J. Plant Physiol. 1997, 151, 471–478. [Google Scholar] [CrossRef]
- Muscolo, A.; Sidari, M.; Anastasi, U.; Santonoceto, C.; Maggio, A. Effect of PEG-Induced Drought Stress on Seed Germination of Four Lentil Genotypes. J. Plant Interact. 2014, 9, 354–363. [Google Scholar] [CrossRef]
- Khaeim, H.M.; Jeber, B.A.; Ali, M.A. Winter Wheat Genotypes Response to Different Water Quality. Int. J. Agric. Stat. Sci. 2019, 15, 669–676. [Google Scholar]
- el Midaoui, M.; Talouizte, A.; Benbella, M.; Serieys, H.; Griveau, Y.; Bervillé, A. Effect of osmotic pressure on germination of sunflower seeds (Helianthus annuus L.). Helia 2001, 24, 129–134. [Google Scholar] [CrossRef]
- Wen, B. Effects of High Temperature and Water Stress on Seed Germination of the Invasive Species Mexican Sunflower. PLoS ONE 2015, 10, e0141567. [Google Scholar] [CrossRef]
- Soleimanzadeh, H. Response of Sunflower (Helianthus annuus L.) to Selenium Application under Water Stress. World Appl. Sci. J. 2012, 17, 1115–1119. [Google Scholar]
- de Morais, M.B.; Barbosa-Neto, A.G.; Willadino, L.; Ulisses, C.; Calsa Junior, T. Salt Stress Induces Increase in Starch Accumulation in Duckweed (Lemna aequinoctialis, Lemnaceae): Biochemical and Physiological Aspects. J. Plant Growth Regul. 2019, 38, 683–700. [Google Scholar] [CrossRef]
- Kovács, Z.; Bedő, J.; Pápai, B.; Tóth-Lencsés, A.K.; Csilléry, G.; Szőke, A.; Bányai-Stefanovits, É.; Kiss, E.; Veres, A. Ripening-Induced Changes in the Nutraceutical Compounds of Differently Coloured Pepper (Capsicum annuum L.) Breeding Lines. Antioxidants 2022, 11, 637. [Google Scholar] [CrossRef] [PubMed]
- Kunos, V.; Cséplő, M.; Seress, D.; Eser, A.; Kende, Z.; Uhrin, A.; Bányai, J.; Bakonyi, J.; Pál, M.; Mészáros, K. The Stimulation of Superoxide Dismutase Enzyme Activity and Its Relation with the Pyrenophora teres f. teres Infection in Different Barley Genotypes. Sustainability 2022, 14, 2597. [Google Scholar] [CrossRef]
- Hoekstra, F.A.; Golovina, E.A.; Buitink, J. Mechanisms of Plant Dessication Tolerance. Trends Plant Sci. 2001, 6, 431–438. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations. Available online: https://www.fao.org/faostat/en/#data/QCL (accessed on 20 February 2022).
- Adeleke, B.S.; Babalola, O.O. Oilseed Crop Sunflower (Helianthus annuus) as a Source of Food: Nutritional and Health Benefits. Food Sci. Nutr. 2020, 8, 4666–4684. [Google Scholar] [CrossRef]
- Forleo, M.B.; Palmieri, N.; Suardi, A.; Coaloa, D.; Pari, L. The Eco-Efficiency of Rapeseed and Sunflower Cultivation in Italy. Joining Environmental and Economic Assessment. J. Clean. Prod. 2018, 172, 3138–3153. [Google Scholar] [CrossRef]
- Ullah Khan, S.; Khan, A.; Raza Gurmani, A.; Jalal-Ud-Din, A. Oil yield, fatty acid profile, achene yield and yield attributes of sunflower (Helianthus annuus L.) as influenced by autumn planting conditions in Islamabad. Pak. J. Bot. 2013, 45 (Suppl. S1), 107–110. [Google Scholar]
- Katsarou, A.I.; Kaliora, A.C.; Papalois, A.; Chiou, A.; Kalogeropoulos, N.; Agrogiannis, G.; Andrikopoulos, N.K. Serum Lipid Profile and Inflammatory Markers in the Aorta of Cholesterol-Fed Rats Supplemented with Extra Virgin Olive Oil, Sunflower Oils and Oil-Products. Int. J. Food Sci. Nutr. 2015, 66, 766–773. [Google Scholar] [CrossRef] [PubMed]
- Oshundiya, F.O.; Olowe, V.I.O.; Sowemimo, F.A.; Odedina, J.N. Seed Yield and Quality of Sunflower (Helianthus annuus L.) as Influenced by Staggered Sowing and Organic Fertilizer Application in the Humid Tropics. Helia 2014, 37, 237–255. [Google Scholar] [CrossRef]
- Hussain, M.; Farooq, S.; Hasan, W.; Ul-Allah, S.; Tanveer, M.; Farooq, M.; Nawaz, A. Drought Stress in Sunflower: Physiological Effects and Its Management through Breeding and Agronomic Alternatives. Agric. Water Manag. 2018, 201, 152–166. [Google Scholar] [CrossRef]
- Khaeim, H.; Kende, Z.; Balla, I.; Gyuricza, C.; Eser, A.; Tarnawa, Á. The Effect of Temperature and Water Stresses on Seed Germination and Seedling Growth of Wheat (Triticum aestivum L.). Sustainability 2022, 14, 3887. [Google Scholar] [CrossRef]
- Khaeim, H.; Kende, Z.; Jolánkai, M.; Kovács, G.P.; Gyuricza, C.; Tarnawa, Á. Impact of Temperature and Water on Seed Germination and Seedling Growth of Maize (Zea mays L.). Agronomy 2022, 12, 397. [Google Scholar] [CrossRef]
- Haj Sghaier, A.; Tarnawa, Á.; Khaeim, H.; Kovács, G.P.; Gyuricza, C.; Kende, Z. The Effects of Temperature and Water on the Seed Germination and Seedling Development of Rapeseed (Brassica napus L.). Plants 2022, 11, 2819. [Google Scholar] [CrossRef]
- Drebee, H.A.; Razak, N.A.A. Measuring the Efficiency of Colleges at the University of Al-Qadisiyah-Iraq: A Data Envelopment Analysis Approach. J. Ekon. Malays. 2018, 52, 163–179. [Google Scholar] [CrossRef]
- Drebee, H.A.; Razak, N.A.A.; Shaybth, R.T. The Impact of Governance Indicators on Corruption in Arab Countries. Contemp. Econ. 2020, 14, 354–365. [Google Scholar] [CrossRef]
- Gay, C.; Corbineau, F.; Come, C. Effects of Temperature and Oxygen on Seed Germination and Seedling Growth in Sunflower (Helianthus annuus L.). Environ. Exp. Bot. 1991, 31, 193–200. [Google Scholar] [CrossRef]
- Khalifa, F.M.; Schneiter, A.A.; Eltayeb, E.I. Temperature-germination responses of sunflower (Helianthus annuus L.) genotypes. Helia 2000, 23, 97–104. [Google Scholar] [CrossRef]
- Filho, M. Fisiologia de Sementes de Plantas Cultivadas; ABRATES: Londrina, Brazil, 2015. [Google Scholar]
- Windauer, L.B.; Martinez, J.; Rapoport, D.; Wassner, D.; Benech-Arnold, R. Germination Responses to Temperature and Water Potential in Jatropha Curcas Seeds: A Hydrotime Model Explains the Difference between Dormancy Expression and Dormancy Induction at Different Incubation Temperatures. Ann. Bot. 2012, 109, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Cassaro-Silva, M. Efeito Da Temperatura Na Germinação de Sementes de Manduirana (Senna macranthera (Collad.) Irwin et Barn.—Caesalpiniaceae). Rev. Bras. De Sementes 2001, 23, 92–99. [Google Scholar] [CrossRef]
- Vierling, E.; Kimpel, J.A. Plant Responses to Environmental Stress. Biotechnology 1992, 3, 164–170. [Google Scholar]
- Ramírez-Tobías, H.M.; Peña-Valdivia, C.B.; Trejo, C.; Aguirre, R.J.R.; Vaquera, H.H. Seed Germination of Agave Species as Influenced by Substrate Water Potential. Biol. Res. 2014, 47, 1–9. [Google Scholar] [CrossRef]
- Campos, H.; Trejo, C.; Peña-Valdivia, C.B.; García-Nava, R.; Víctor Conde-Martínez, F.; Cruz-Ortega, R. Water Availability Effects on Germination, Membrane Stability and Initial Root Growth of Agave lechuguilla and A. salmiana. Flora Morphol. Distrib. Funct. Ecol. Plants 2020, 268, 151606. [Google Scholar] [CrossRef]
- Schütz, W.; Lamont, B.B.; Milberg, P. Germination Requirements and Seedling Responses to Water Availability and Soil Type in Four Eucalypt Species. Acta Oecologica 2002, 23, 23–30. [Google Scholar] [CrossRef]
- Linhart, Y.B. Density-Dependent Seed Germination Strategies in Colonizing Versus Non-Colonizing Plant. J. Ecol. 1976, 64, 375–380. [Google Scholar] [CrossRef]
- Callaway, R.M.; Walker, L.R. Competition and Facilitation: A Synthetic Approach to Interactions in Plant Communities. Ecology 1997, 78, 1958–1965. [Google Scholar] [CrossRef]
- White, T.C.R. Opposing Paradigms: Regulation or Limitation of Populations? Oikos 2001, 93, 148–152. [Google Scholar] [CrossRef]
- Glen, D.M.; Milsom, N.F.; Wiltshire, C.W. Effects of Seed-Bed Conditions on Slug Numbers and Damage to Winter Wheat in a Clay Soil. Ann. Appl. Biol. 1989, 115, 177–190. [Google Scholar] [CrossRef]
- Lortie, C.J.; Turkington, R. The Effect of Initial Seed Density on the Structure of a Desert Annual Plant Community. J. Ecol. 2002, 90, 435–445. [Google Scholar] [CrossRef]
- Bertelsen, J.R.; de Neergaard, E.; Smedegaard-Petersen, V. Fungicidal Effects of Azoxystrobin and Epoxiconazole on Phyllosphere Fungi, Senescence and Yield of Winter Wheat. Plant Pathol. 2001, 50, 190–205. [Google Scholar] [CrossRef]
- Vasanthakumari, M.M.; Shridhar, J.; Madhura, R.J.; Nandhitha, M.; Kasthuri, C.; Janardhana, B.; Nataraja, K.N.; Ravikanth, G.; Uma Shaanker, R. Role of Endophytes in Early Seedling Growth of Plants: A Test Using Systemic Fungicide Seed Treatment. Plant Physiol. Rep. 2019, 24, 86–95. [Google Scholar] [CrossRef]
- Verma, S.K.; Kingsley, K.L.; Bergen, M.S.; Kowalski, K.P.; White, J.F. Fungal Disease Prevention in Seedlings of Rice (Oryza sativa) and Other Grasses by Growth-Promoting Seed-Associated Endophytic Bacteria from Invasive Phragmites Australis. Microorganisms 2018, 6, 21. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.K.; Kingsley, K.; Irizarry, I.; Bergen, M.; Kharwar, R.N.; White, J.F. Seed-Vectored Endophytic Bacteria Modulate Development of Rice Seedlings. J. Appl. Microbiol. 2017, 122, 1680–1691. [Google Scholar] [CrossRef] [PubMed]

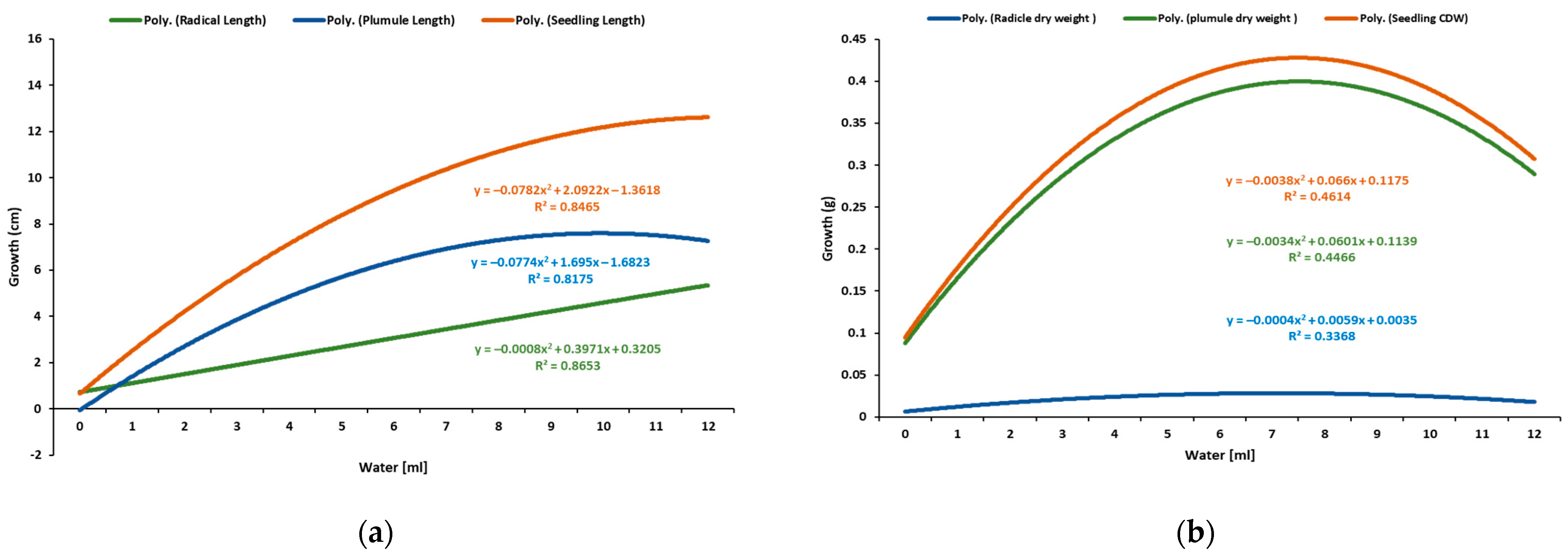
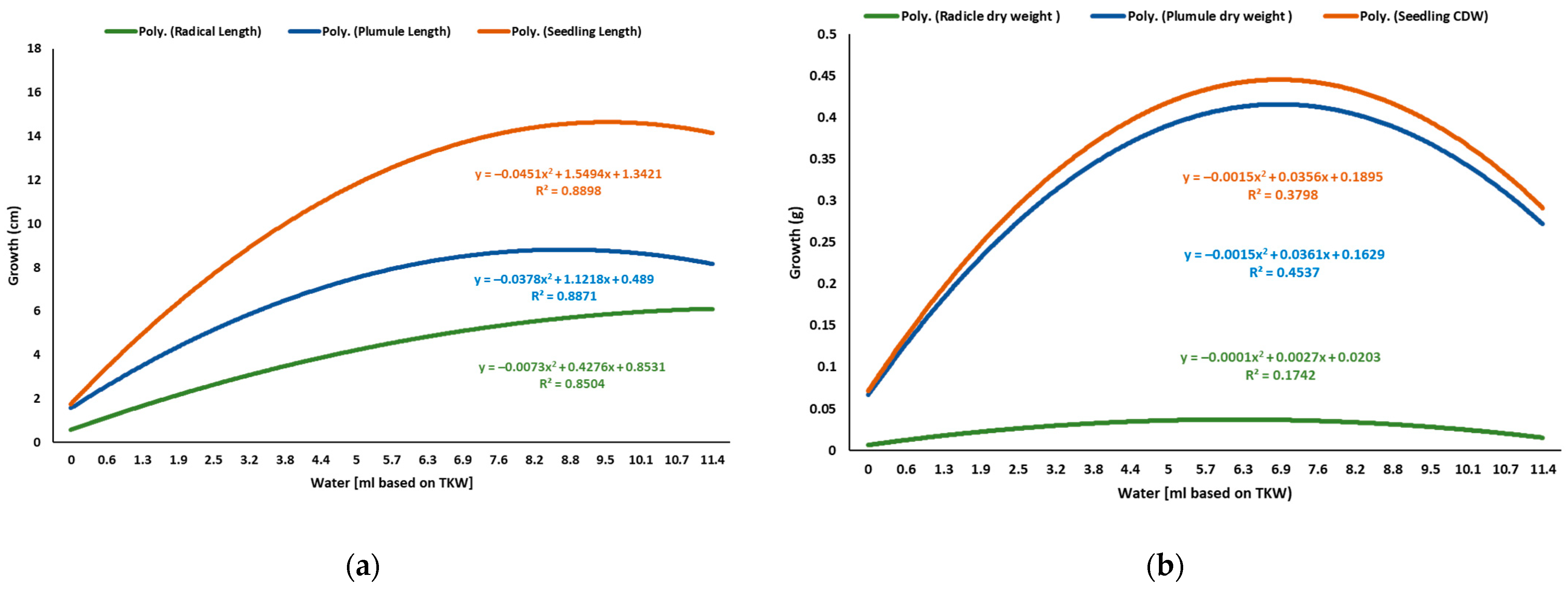
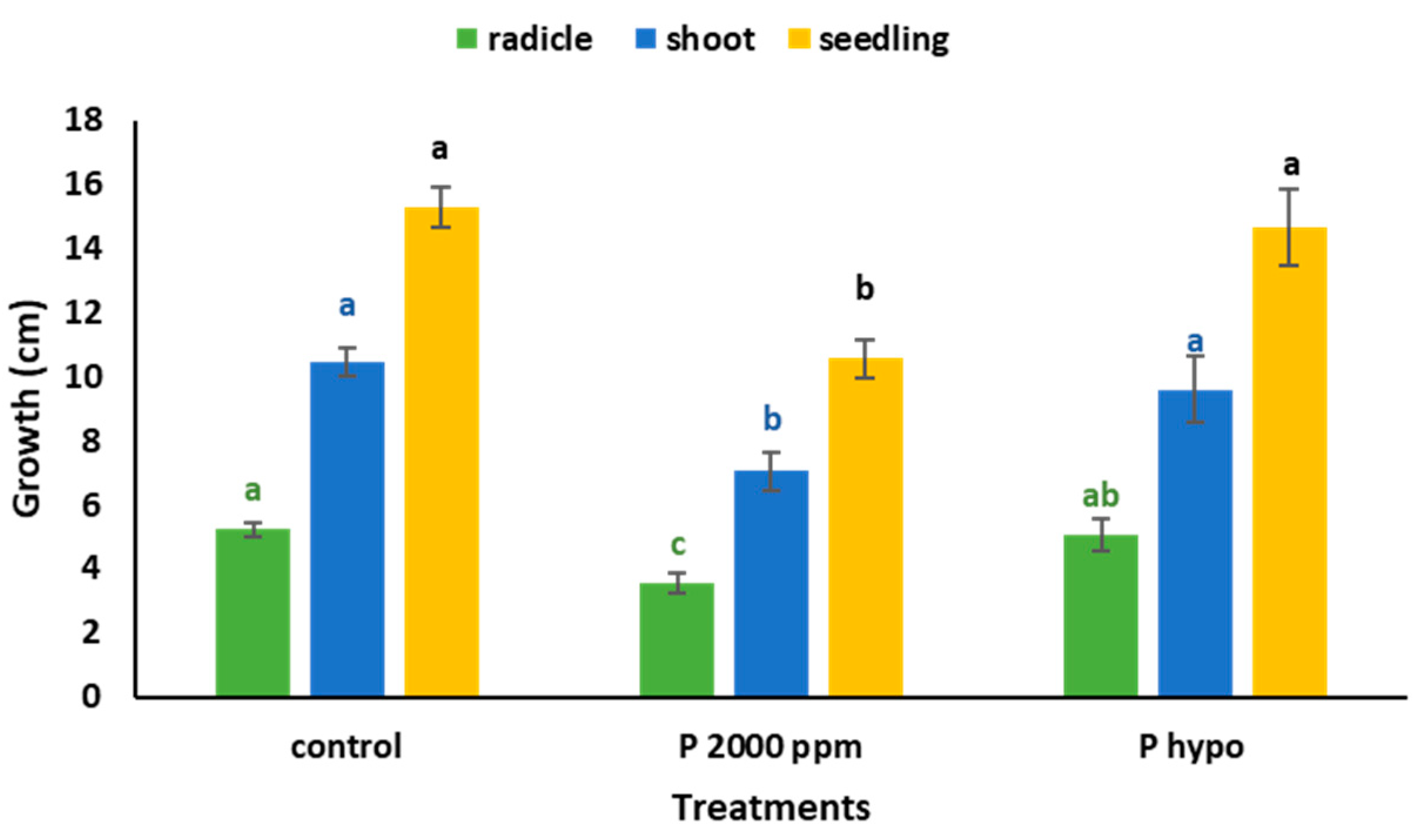
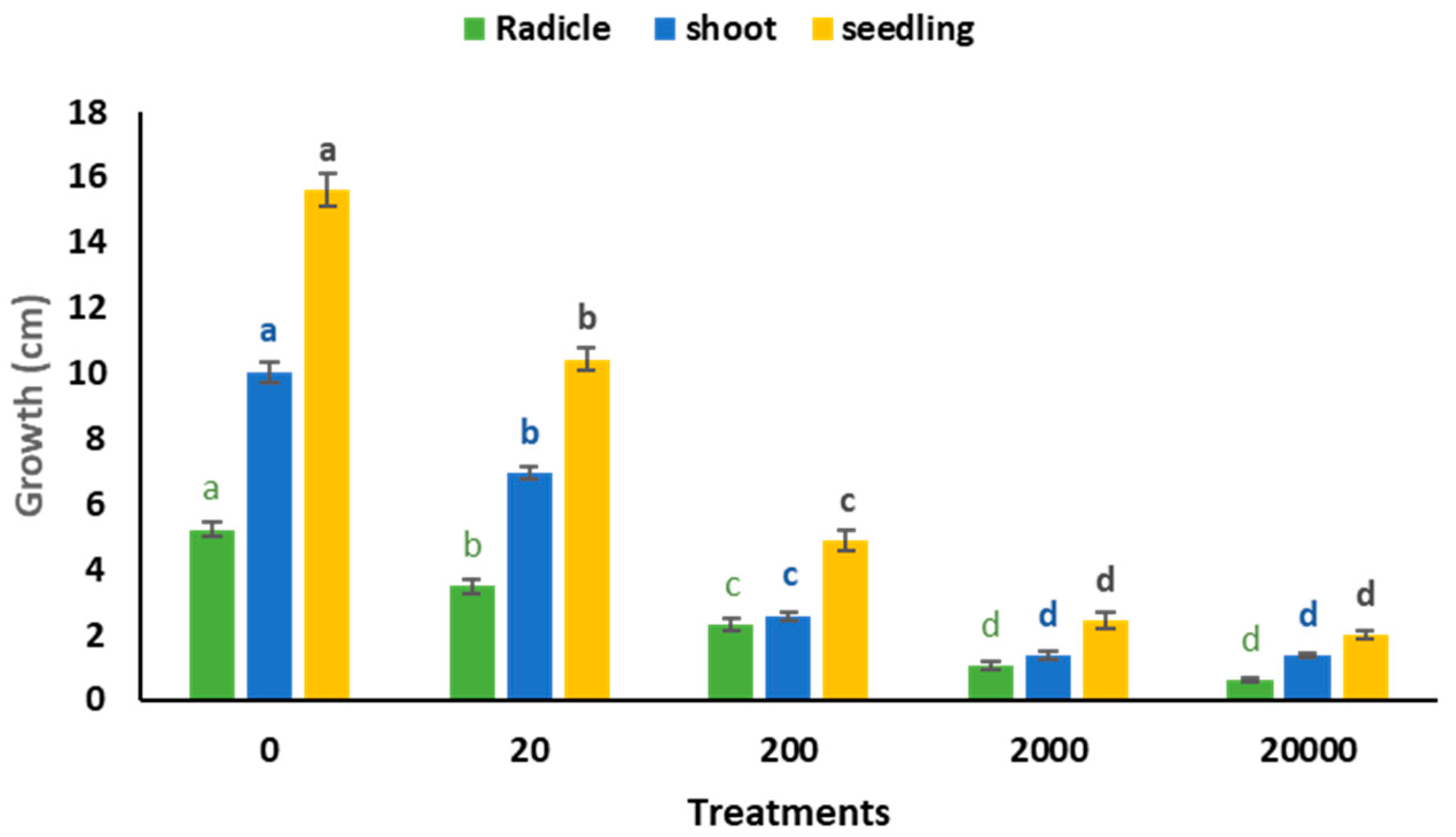
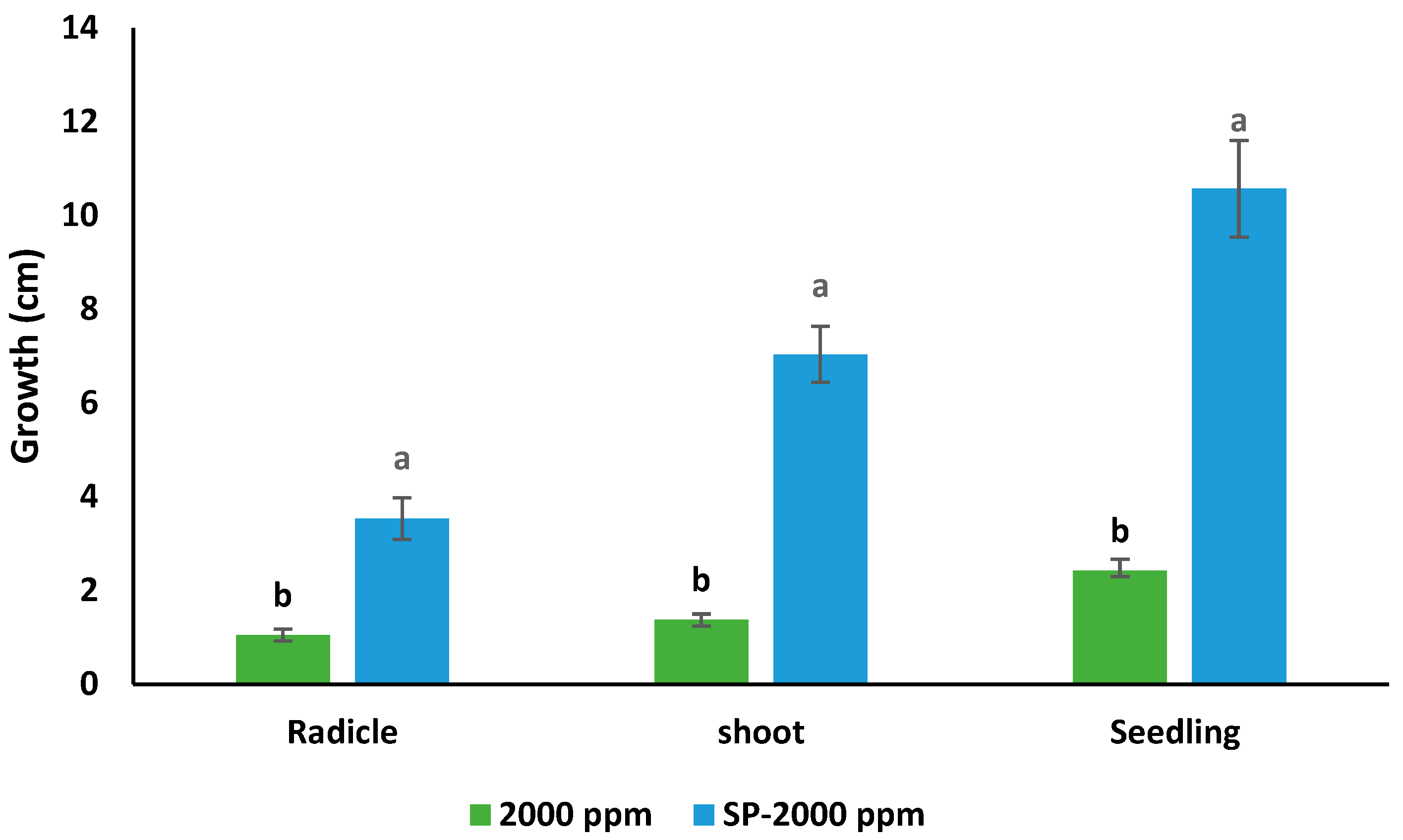
| Water mL | Germinated n | Radicle L (cm) | Shoot S (cm) | Seedling L (cm) | Shoot DW (g) | Radicle DW (g) | Seedling DW (g) |
|---|---|---|---|---|---|---|---|
| 0 | 0.0 ± 0.0 | 0.000 ± 0.00 g | 0.000 ± 0.00 g | 0.000 ± 0.00 f | 0.000 ± 0.00 d | 0.000 ± 0.00 f | 0.000 ± 0.00 d |
| 1 | 10.0 ± 0.0 | 1.860 ± 0.54 f | 1.622 ± 0.05 f | 3.482 ± 0.55 e | 0.351 ± 0.01 abc | 0.016 ± 0.00 de | 0.367 ± 0.01 abc |
| 2 | 10.0 ± 0.0 | 1.844 ± 0.67 f | 2.368 ± 0.55 ef | 4.212 ± 1.14 e | 0.348 ± 0.01 abc | 0.024 ± 0.00 bc | 0.372 ± 0.02 abc |
| 3 | 10.0 ± 0.0 | 1.770 ± 0.67 f | 3.096 ± 0.85 e | 4.866 ± 1.44 e | 0.316 ± 0.07 c | 0.026 ± 0.01 bc | 0.342 ± 0.07 c |
| 4 | 10.0 ± 0.0 | 2.216 ± 0.19 f | 5.462 ± 0.44 cd | 7.678 ± 0.51 d | 0.367 ± 0.02 ab | 0.036 ± 0.00 a | 0.404 ± 0.03 a |
| 5 | 10.0 ± 0.0 | 2.474 ± 0.67 f | 5.834 ± 1.22 cd | 8.308 ± 1.84 d | 0.339 ± 0.03 abc | 0.018 ± 0.01 bcde | 0.358 ± 0.03 bc |
| 6 | 10.0 ± 0.0 | 3.506 ± 0.28 de | 6.962 ± 0.77 b | 10.468 ± 0.94 bc | 0.335 ± 0.03 abc | 0.021 ± 0.01 bcd | 0.356 ± 0.03 bc |
| 7 | 10.0 ± 0.0 | 2.612 ± 0.47 ef | 5.506 ± 1.09 cd | 8.118 ± 1.45 d | 0.341 ± 0.02 abc | 0.017 ± 0.01 cde | 0.359 ± 0.02 bc |
| 8 | 10.0 ± 0.0 | 4.064 ± 0.78 cd | 8.996 ± 0.85 a | 13.060 ± 1.55 a | 0.340 ± 0.03 abc | 0.034 ± 0.01 a | 0.373 ± 0.03 abc |
| 9 | 10.0 ± 0.0 | 5.140 ± 0.83 ab | 8.918 ± 0.68 a | 14.058 ± 0.93 a | 0.328 ± 0.02 bc | 0.034 ±0.01 a | 0.363 ± 0.02 abc |
| 10 | 10.0 ± 0.0 | 3.686 ± 0.65 cd | 5.102 ± 0.98 d | 8.788 ± 1.46 cd | 0.363 ± 0.04 ab | 0.014 ± 0.00 e | 0.377 ± 0.04 abc |
| 11 | 10.0 ± 0.0 | 4.648 ± 1.72 bc | 6.380 ± 1.70 bc | 11.028 ± 3.40 b | 0.372 ± 0.05 a | 0.019 ± 0.01 bcde | 0.391 ± 0.04 ab |
| 12 | 10.0 ± 0.0 | 5.832 ± 1.14 a | 8.706 ± 0.80 a | 14.538 ± 1.42 a | 0.346 ± 0.02 abc | 0.026 ± 0.00 b | 0.372 ± 0.02 abc |
| LSD | NS | 0.994 | 1.222 | 1.906 | 0.040 | 0.0075 | 0.041 |
| Water mL of the TKW | Germinated n | Radicle L (cm) | Shoot S (cm) | Seedling L (cm) | Shoot DW (g) | Radicle DW (g) | Seedling DW (g) |
|---|---|---|---|---|---|---|---|
| 0 | 0.0 ± 0.00 | 0.000 ± 0.00 i | 0.000 ± 0.00 h | 0.000 ± 0.00 j | 0.000 ± 0.00 g | 0.000 ± 0.00 i | 0.000 ± 0.00 f |
| 0.6 | 10.0 ± 0.00 | 2.412 ± 0.62 h | 4.182 ± 0.34 g | 6.594 ± 0.88 i | 0.324 ± 0.02 f | 0.037 ± 0.01 abcd | 0.360 ± 0.03 e |
| 1.3 | 10.0 ± 0.00 | 2.804 ± 0.57 gh | 3.208 ± 0.17 g | 6.012 ± 0.63 i | 0.338 ± 0.01 def | 0.030 ± 0.00 ef | 0.368 ± 0.01 de |
| 1.9 | 10.0 ± 0.00 | 3.102 ± 0.50 gh | 4.388 ± 0.60 g | 7.490 ± 0.19 hi | 0.346 ± 0.02 bcde | 0.039 ± 0.00 ab | 0.384 ± 0.01 abcde |
| 2.5 | 10.0 ± 0.00 | 2.782 ± 0.34 gh | 5.912 ± 0.70 f | 8.694 ± 0.46 gh | 0.333 ± 0.02 ef | 0.040 ± 0.00 a | 0.374 ± 0.02 bcde |
| 3.2 | 10.0 ± 0.00 | 2.448 ± 0.32 h | 6.008 ± 0.31 f | 8.456 ± 0.61 gh | 0.351 ± 0.02 abcde | 0.024 ± 0.00 fgh | 0.374 ± 0.02 bcde |
| 3.8 | 10.0 ± 0.00 | 3.338 ± 0.80 fgh | 6.136 ± 1.21 f | 9.474 ± 1.75 g | 0.348 ± 0.04 def | 0.038 ± 0.01 abc | 0.386 ± 0.04 abcde |
| 4.4 | 10.0 ± 0.00 | 4.508 ± 0.93 de | 7.448 ± 0.90 cde | 11.960 ± 1.81 def | 0.338 ± 0.03 abc | 0.043 ± 0.00 a | 0.381 ± 0.03 abcde |
| 5.0 | 10.0 ± 0.00 | 3.468 ± 1.46 fg | 6.778 ± 2.85 ef | 10.246 ± 4.24 fg | 0.367 ± 0.02 a | 0.032 ± 0.01 cde | 0.399 ± 0.02 ab |
| 5.7 | 10.0 ± 0.00 | 4.852 ± 0.57 cde | 8.59 ± 0.32 abc | 13.438 ± 0.68 cd | 0.373 ± 0.02 ab | 0.031 ± 0.01 cde | 0.405 ± 0.01 a |
| 6.3 | 10.0 ± 0.00 | 4.218 ± 0.19 ef | 8.734 ± 0.84 ab | 12.950 ± 0.93 cde | 0.369 ± 0.02 abcde | 0.026 ± 0.01 efgh | 0.396 ± 0.02 abc |
| 6.9 | 10.0 ± 0.00 | 4.178 ± 0.46 ef | 7.372 ± 1.14 de | 11.550 ± 1.31 ef | 0.349 ± 0.02 def | 0.023 ± 0.01 gh | 0.371 ± 0.01 cde |
| 7.6 | 10.0 ± 0.00 | 5.056 ± 0.55 cde | 8.568 ± 0.51 abc | 13.620 ± 0.76 bcd | 0.340 ± 0.02 abcd | 0.028 ± 0.01 efg | 0.368 ± 0.02 de |
| 8.2 | 10.0 ± 0.00 | 6.400 ± 1.32 ab | 9.326 ± 0.27 a | 15.726 ± 1.46 a | 0.359 ± 0.02 ab | 0.032 ± 0.00 bcde | 0.392 ± 0.02 abcd |
| 8.8 | 10.0 ± 0.00 | 5.358 ± 0.14 cd | 7.86 ± 0.56 bcd | 13.21 ± 0.62 cde | 0.369 ±0.02 abcde | 0.023 ± 0.00 gh | 0.391 ± 0.02 abcd |
| 9.5 | 10.0 ± 0.00 | 6.584 ± 0.80 a | 9.496 ± 0.84 a | 16.08 ± 1.56 a | 0.353 ± 0.01 abcde | 0.030 ± 0.00 ef | 0.383 ± 0.01 abcde |
| 10.1 | 10.0 ± 0.00 | 5.468 ± 0.60 bc | 7.478 ± 1.25 cde | 12.946 ± 1.26 cde | 0.359 ± 0.01 abcd | 0.021 ± 0.01 h | 0.381 ± 0.01 abcde |
| 10.7 | 10.0 ± 0.00 | 5.594 ± 1.47 bc | 9.090 ± 0.44 a | 14.684 ± 1.84 abc | 0.342 ± 0.01 cdef | 0.031 ± 0.00 de | 0.373 ± 0.01 bcde |
| 11.4 | 10.0 ± 0.00 | 6.834 ± 0.33 a | 8.53± 0.42 bcd | 15.360 ± 0.69 ab | 0.354 ± 0.03 abcde | 0.024 ± 0.00 fgh | 0.378 ± 0.02 bcde |
| LSD | NS | 0.947 | 1.189 | 1.830 | 0.025 | 0.006 | 0.026 |
| Seed Number | Radical cm | Shoot (cm) | Seedling (cm) | Radicle DW (g) | Shoot DW (g) | Seedling DW (g) |
|---|---|---|---|---|---|---|
| 6 | 4.277 ± 1.19 ab | 5.673 ± 0.53 ab | 9.950 ± 1.60 | 0.210 ± 0.01 d | 0.036 ± 0.01 c | 0.246 ± 0.02 d |
| 8 | 4.846 ± 0.99 a | 5.438 ± 0.55 b | 10.28 ± 1.34 | 0.275 ± 0.02 c | 0.056 ± 0.01 b | 0.331 ± 0.02 c |
| 10 | 4.420 ± 1.02 ab | 5.6 ± 0.27 b | 10.04 ± 1.12 | 0.344 ± 0.01 b | 0.071 ± 0.01 a | 0.416 ± 0.02 b |
| 12 | 3.640 ± 0.46 b | 6.004 ± 0.37 a | 9.644 ±0.61 | 0.407 ± 0.02 a | 0.076 ± 0.01 a | 0.483 ± 0.02 a |
| LSD | 0.785 | 0.335 | N.S | 0.013 | 0.007 | 0.017 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haj Sghaier, A.; Khaeim, H.; Tarnawa, Á.; Kovács, G.P.; Gyuricza, C.; Kende, Z. Germination and Seedling Development Responses of Sunflower (Helianthus annuus L.) Seeds to Temperature and Different Levels of Water Availability. Agriculture 2023, 13, 608. https://doi.org/10.3390/agriculture13030608
Haj Sghaier A, Khaeim H, Tarnawa Á, Kovács GP, Gyuricza C, Kende Z. Germination and Seedling Development Responses of Sunflower (Helianthus annuus L.) Seeds to Temperature and Different Levels of Water Availability. Agriculture. 2023; 13(3):608. https://doi.org/10.3390/agriculture13030608
Chicago/Turabian StyleHaj Sghaier, Asma, Hussein Khaeim, Ákos Tarnawa, Gergő Péter Kovács, Csaba Gyuricza, and Zoltán Kende. 2023. "Germination and Seedling Development Responses of Sunflower (Helianthus annuus L.) Seeds to Temperature and Different Levels of Water Availability" Agriculture 13, no. 3: 608. https://doi.org/10.3390/agriculture13030608
APA StyleHaj Sghaier, A., Khaeim, H., Tarnawa, Á., Kovács, G. P., Gyuricza, C., & Kende, Z. (2023). Germination and Seedling Development Responses of Sunflower (Helianthus annuus L.) Seeds to Temperature and Different Levels of Water Availability. Agriculture, 13(3), 608. https://doi.org/10.3390/agriculture13030608








