A Theoretical Study of One- and Two-Photon Activity of D-Luciferin
Abstract
:1. Introduction
2. Computational Details
3. Results
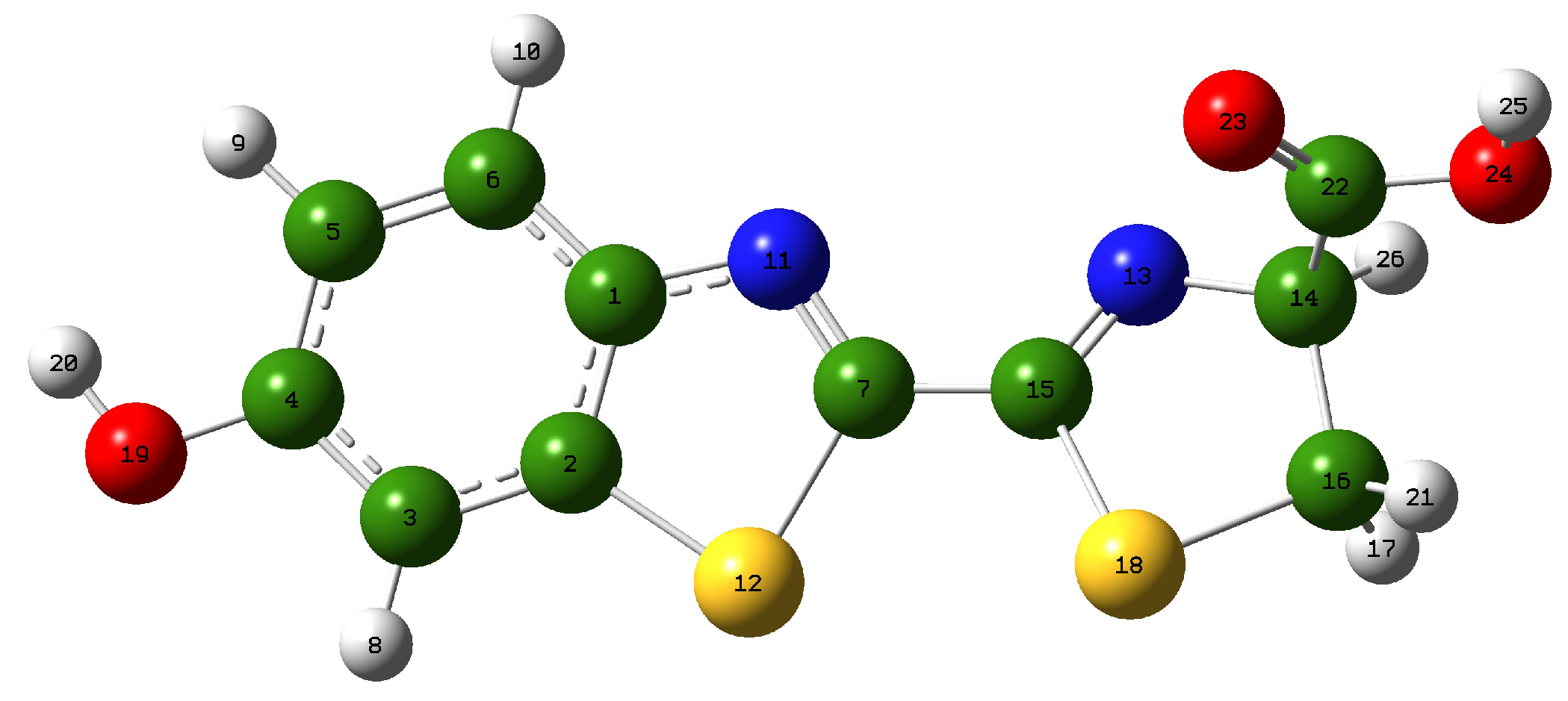
3.1. Geometry of the Studied System
3.2. One-Photon Absorption
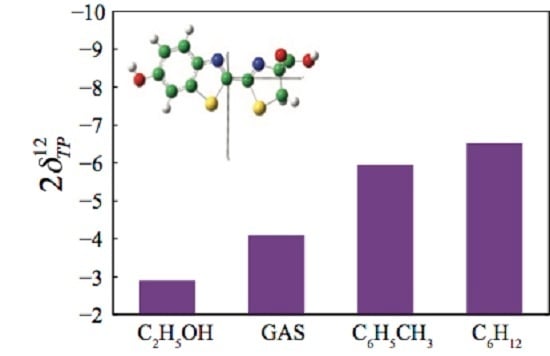
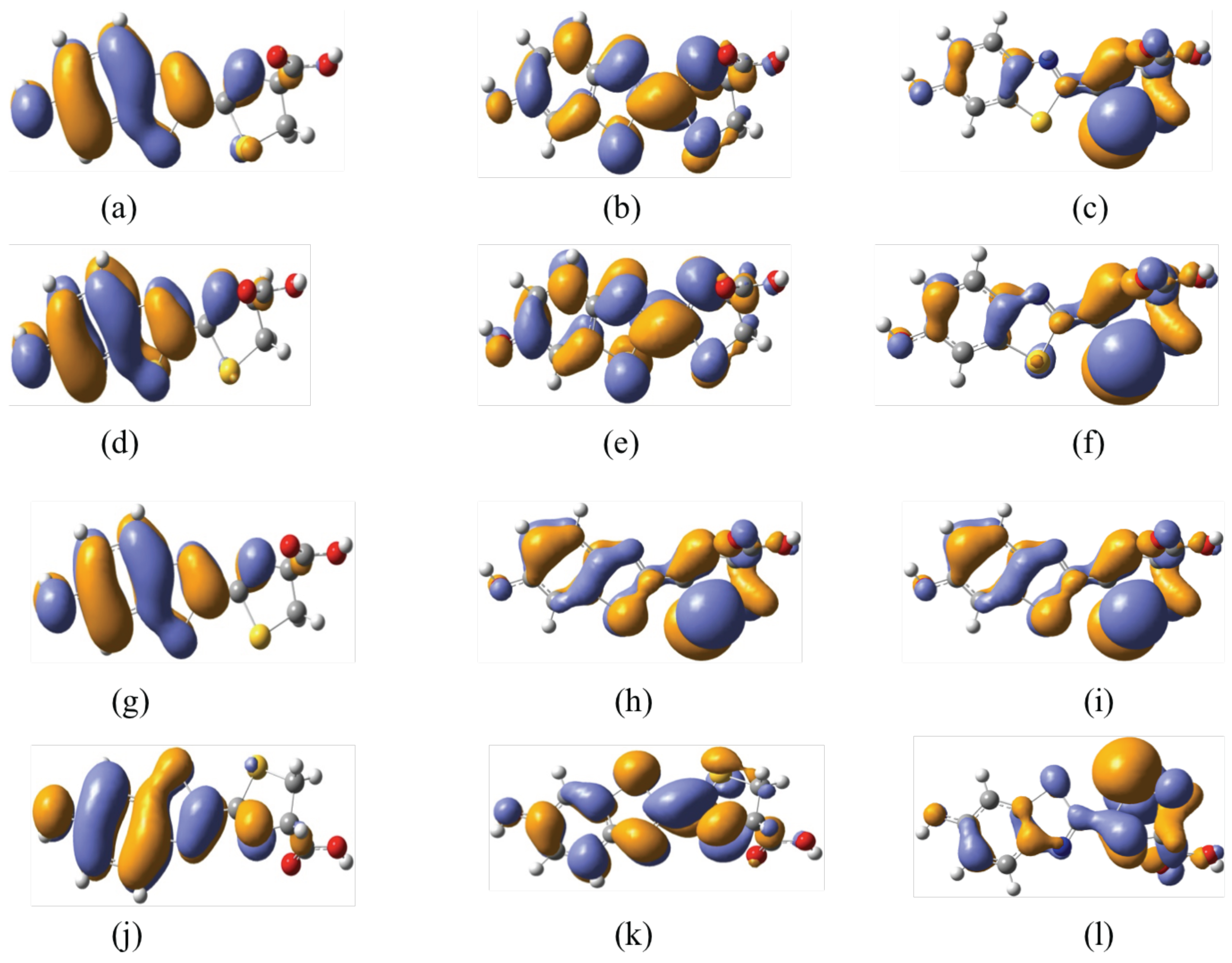
3.3. Two-Photon Absorption
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Göppert-Mayer, M. Über elementarakte mit zwei quantensprüngen. Ann. Phys. 1931, 9, 237–294. [Google Scholar] [CrossRef]
- Barsu, B.; Cheaib, R.; Chambert, S.; Queneau, Y.; Maury, O.; Cottet, D.; Wege, H.; Douady, J.; Bretonniere, Y.; Andraud, C. Neutral push-pull chromophores for nonlinear optical imaging of cell membranes. Org. Biomol. Chem. 2010, 8, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.-C.; Huang, Y.-J.; Chen, Y.-F.; Hu, C.-L. Two-photon absorption and effective broadband optical power limiting properties of a multi-branched chromophore containing 2, 3-diarylquinoxalinyl moieties as the electron-pulling units. Tetrahedron 2010, 66, 1375–1382. [Google Scholar] [CrossRef]
- Hammerer, F.; Garcia, G.; Chen, S.; Poyer, F.; Achelle, S.; Fiorini-Debuisschert, C.; Teulade-Fichou, M.-P.; Maillard, P. Synthesis and characterization of glycoconjugated porphyrin triphenylamine hybrids for targeted two-photon photodynamic therapy. J. Org. Chem. 2014, 79, 1406–1417. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Ohulchanskyy, Y.; Pudavar, H.E.; Pandey, R.K.; Prasad, P.N. Organically modified silica nanoparticles co-encapsulating photosensitizing drug and aggregation-enhanced two-photon absorbing fluorescent dye aggregates for two-photon photodynamic therapy. J. Am. Chem. Soc. 2007, 129, 2669–2675. [Google Scholar] [CrossRef] [PubMed]
- Pawlicki, M.; Collins, H.; Denning, R.; Anderson, H. Two-photon absorption and the design of two-photon dyes. Angew. Chem. Int. Ed. 2009, 48, 3244–3266. [Google Scholar] [CrossRef] [PubMed]
- Kawata, S.; Sun, H.B.; Tanaka, T.; Takada, K. Finer features for functional microdevices. Nature 2001, 412, 697. [Google Scholar] [CrossRef] [PubMed]
- Dvornikov, A.S.; Walker, E.P.; Rentzepis, P.M. Two-photon three-dimensional optical storage memory. J. Phys. Chem. A 2009, 113, 13633–13644. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.M.; Chattopadhyaya, M.; Chakrabarti, S. On the origin of large two-photon activity of DANS molecule. J. Phys. Chem. A 2012, 116, 11034–11040. [Google Scholar] [CrossRef] [PubMed]
- Badaeva, E.A.; Timofeeva, T.V.; Masunov, A.; Tretiak, S. Role of donor-acceptor strengths and separation on the two-photon absorption response of cytotoxic dyes: A TD-DFT study. J. Phys. Chem. A 2005, 109, 7276–7284. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.; Li, Q.; Zhu, L.; Shuai, Z. Effects of donor/acceptor strengths on the multiphoton absorption: An EOM-CCSD correction vector study. J. Phys. Chem. A 2007, 111, 9291–9298. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Pons, Ò.; Luo, Y.; Ågren, H. Very large infrared two-photon absorption cross section of asymmetric zinc porphyrin aggregates: Role of intermolecular interaction and donor-acceptor strengths. J. Chem. Phys. A 2006, 110, 12342–12347. [Google Scholar]
- Wang, C.K.; Macak, P.; Luo, Y.; Ågren, H. Effects of π centers and symmetry on two-photon absorption cross sections of organic chromophores. J. Chem. Phys. 2001, 114, 9813–9820. [Google Scholar] [CrossRef]
- Chakrabarti, S.; Ruud, K. Large two-photon absorption cross section: Molecular tweezer as a new promising class of compounds for nonlinear optics. Phys. Chem. Chem. Phys. 2009, 11, 2592–2596. [Google Scholar] [CrossRef] [PubMed]
- Norman, P.; Luo, Y.; Ågren, H. Large two-photon absorption cross sections in two-dimensional, charge-transfer, cumulene-containing aromatic molecules. J. Chem. Phys. 1999, 111, 7758–7765. [Google Scholar] [CrossRef]
- Jha, P.C.; Rinkevicius, Z.; Ågren, H. Modeling two photon absorption cross sections of open-shell systems. J. Chem. Phys. 2009, 130, 014103. [Google Scholar] [CrossRef] [PubMed]
- He, G.S.; Tan, L.-S.; Zheng, Q.; Prasad, P.N. Multiphoton absorbing materials: molecular designs, characterizations, and applications. Chem. Rev. 2008, 108, 1245–1330. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyaya, M.; Alam, M.M.; Chakrabarti, S. New design strategy for the two-photon active material based on push–pull substituted bisanthene molecule. J. Phys. Chem. A 2011, 115, 2607–2614. [Google Scholar] [CrossRef] [PubMed]
- Cronstand, P.; Luo, P.; Ågren, H. Effects of dipole alignment and channel interference on two-photon absorption cross sections of two-dimensional charge-transfer systems. J. Chem. Phys. 2002, 117, 11102–11106. [Google Scholar] [CrossRef]
- Cronstand, P.; Luo, P.; Ågren, H. Generalized few-state models for two-photon absorption of conjugated molecules. Chem. Phys. Lett. 2002, 352, 262–269. [Google Scholar] [CrossRef]
- Alam, M.M.; Chattopadhyaya, M.; Chakrabarti, S. Solvent induced channel interference in the two-photon absorption process—A theoretical study with a generalized few-state-model in three dimensions. Phys. Chem. Chem. Phys. 2012, 14, 1156–1165. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.M.; Chattopadhyaya, M.; Chakrabarti, S.; Ruud, K. Chemical control of channel interference in two-photon absorption processes. Acc. Chem. Res. 2014, 47, 1604–1612. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Norman, P.; Macak, P.; Ågren, H. Solvent-induced two-photon absorption of a push-pull molecule. J. Phys. Chem. A 2000, 104, 4718–4722. [Google Scholar] [CrossRef]
- Alam, M.M.; Kundi, V.; Thankachan, P.P. Solvent effects on static polarizability, static first hyperpolarizability and one-and two-photon absorption properties of functionalized triply twisted Möbius annulenes: A DFT study. Phys. Chem. Chem. Phys. 2016, 18, 21833–21842. [Google Scholar] [CrossRef] [PubMed]
- Ferrighi, L.; Frediani, L.; Fossgaard, E.; Ruud, K. Two-photon absorption of [2.2] paracyclophane derivatives in solution: A theoretical investigation. J. Chem. Phys. 2007, 127, 244103. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.M.; Chattopadhyaya, M.; Chakrabarti, S.; Ruud, K. High-polarity solvents decreasing the two-photon transition probability of through-space charge-transfer systems–A surprising in silico observation. J. Phys. Chem. Lett. 2012, 3, 961–966. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.M.; Chattopadhyaya, M.; Chakrabarti, S. A critical theoretical study on the two-photon absorption properties of some selective triaryl borane-1-naphthylphenyl amine based charge transfer molecules. Phys. Chem. Chem. Phys. 2011, 13, 9285–9292. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.M. Donor’s position-specific channel interference in substituted biphenyl molecules. Phys. Chem. Chem. Phys. 2015, 17, 17571–17576. [Google Scholar] [CrossRef] [PubMed]
- Miertus, S.; Scrocco, E.; Tomasi, J. Electrostatic interaction of a solute with a continuum. A direct utilizaion of AB initio molecular potentials for the prevision of solvent effects. J. Chem. Phys. 1981, 55, 117–129. [Google Scholar] [CrossRef]
- Tomasi, J.; Mennucci, B.; Cammi, R. Quantum mechanical continuum solvation models. Chem. Rev. 2005, 105, 2999–3094. [Google Scholar] [CrossRef] [PubMed]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian-09; Revision A.02; Gaussian Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Mikkelsen, K.V.; Ågren, H.; Jensen, H.J.A. A multiconfigurational self-consistent reaction-field method. J. Chem. Phys. 1988, 89, 3086–3095. [Google Scholar] [CrossRef]
- Mikkelsen, K.V.; Cesar, A.; Ågren, H.; Jensen, H.J.A. Multiconfigurational self-consistent reaction field theory for nonequilibrium solvation. J. Chem. Phys. 1995, 103, 9010–9023. [Google Scholar] [CrossRef]
- Aidas, K.; Angeli, C.; Bak, K.L.; Bakken, V.; Bast, R.; Boman, L.; Christiansen, O.; Cimiraglia, R.; Coriani, S.; Dahle, P.; et al. The Dalton quantum chemistry program system. Wiley Interdiscip. Rev. Comput. Mol. Sci. 2014, 4, 269–284. [Google Scholar] [CrossRef] [PubMed]
- Dalton, a Molecular Electronic Structure Program, Release Dalton, 2013.4, 2013. Available online: http://daltonprogram.org (accessed on 12 November 2016).
- Conley, N.R.; Dragulescu-Andrasi, A.; Rao, J.; Moerner, W.E. A selenium analogue of firefly D-luciferin with red-shifted bioluminescence emission. Angew. Chem. Int. Ed. 2012, 51, 3350–3353. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.R. The Principle of Nonlinear Optics; Wiley: New York, NY, USA, 1984; pp. 23–25. [Google Scholar]
- McClain, W.M. Excited state symmetry assignment through polarized wwo-photon absorption studies of fluids. J. Chem. Phys. 1971, 55, 2789–2796. [Google Scholar] [CrossRef]
- Murugan, N.A.; Kongsted, J.; Rinkevicius, Z.; Aidas, K.; Mikkelsen, K.V.; Ågren, H. Hybrid density functional theory/molecular mechanics calculations of two-photon absorption of dimethylamino nitro stilbene in solution. Phys. Chem. Chem. Phys. 2011, 13, 12506–12516. [Google Scholar] [CrossRef] [PubMed]
| Dihedral Angle | Gas | CH | CHOH | CHCH |
|---|---|---|---|---|
| S-C-C-N | 158.34 | 158.67 | 158.33 | 158.70 |
| S-C-C-S | −21.83 | −21.54 | −22.28 | −21.52 |
| N-C-C-N | −21.31 | −20.95 | −21.48 | −20.92 |
| Bond Length | Gas | CH | CHOH | CHCH |
|---|---|---|---|---|
| C-O | 1.359 | 1.354 | 1.346 | 1.353 |
| C-O | 1.198 | 1.200 | 1.205 | 1.201 |
| C-O | 1.366 | 1.365 | 1.363 | 1.365 |
| C-S | 1.834 | 1.835 | 1.829 | 1.834 |
| C-S | 1.851 | 1.848 | 1.842 | 1.848 |
| C-N | 1.284 | 1.285 | 1.288 | 1.285 |
| C-N | 1.265 | 1.266 | 1.269 | 1.267 |
| C-C | 1.467 | 1.467 | 1.467 | 1.467 |
| C-C | 1.534 | 1.534 | 1.54 | 1.534 |
| Excited State | Solvent | Excitation Energy (eV) | Oscillator Strength (a.u.) | Transition Moment | Λ | Orbital Contributions | |||
|---|---|---|---|---|---|---|---|---|---|
| X | Y | Z | Total | ||||||
| 1 | Gas | 3.85 | 0.247 | −1.618 | −0.209 | −0.119 | 1.636 | 0.674 | H-L |
| CHOH | 3.73 | 0.439 | −2.191 | −0.434 | −0.102 | 2.236 | 0.686 | H-L | |
| CHCH | 3.77 | 0.447 | −2.200 | −0.414 | −0.110 | 2.241 | 0.698 | H-L | |
| CH | 3.78 | 0.431 | 2.157 | 0.398 | 0.110 | 2.916 | 0.699 | H-L | |
| 2 | Gas | 3.93 | 0.112 | 1.072 | 0.479 | −0.057 | 1.175 | 0.530 | H-1-L |
| CHOH | 3.92 | 0.017 | 0.345 | 0.366 | −0.117 | 0.517 | 0.501 | H-1-L | |
| CHCH | 3.91 | 0.030 | −0.518 | −0.413 | 0.106 | 0.672 | 0.501 | H-1-L | |
| CH | 3.91 | 0.031 | −0.566 | −0.420 | 0.102 | 0.712 | 0.501 | H-1-L | |
| Solvents | Excited States | ( a.u.) | ||||||
|---|---|---|---|---|---|---|---|---|
| Gas | 1 | 96.3 | 2.1 | 0.9 | 27.1 | 1.6 | −0.3 | 6.27 |
| 2 | −112.3 | 10.0 | 0.5 | −1.0 | −8.7 | −4.1 | 7.23 | |
| CHOH | 1 | 176.6 | −2.8 | 0.7 | 31.4 | 7.1 | 1.9 | 19.41 |
| 2 | −74.2 | 12.1 | 1.0 | 13.7 | −9.5 | −4.6 | 3.24 | |
| CHCH | 1 | 173.2 | −1.8 | 0.8 | 33.8 | 6.4 | 1.6 | 18.87 |
| 2 | 91.0 | −13.0 | −1.0 | −11.3 | 10.1 | 4.9 | 4.77 | |
| CH | 1 | −166.0 | 1.5 | −0.8 | −33.1 | −5.9 | −1.4 | 17.40 |
| 2 | 93.3 | −12.7 | −0.9 | −10.1 | 9.9 | 4.8 | 4.98 |
| Solvents | Excited | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| States | ||||||||||||
| Gas | 1 | 0.95 | 5.26 | −4.07 | 2.13 | 6.27 | 160.97 | 14.92 | 161.97 | 22.92 | 175.02 | 37.23 |
| 2 | 11.80 | 1.05 | −6.49 | 6.42 | 7.23 | 5.07 | 170.39 | 161.97 | 23.00 | 27.02 | 165.17 | |
| CHOH | 1 | 24.30 | 0.11 | −2.88 | 21.5 | 19.41 | 172.28 | 42.39 | 141.53 | 3.77 | 174.83 | 45.94 |
| 2 | 3.92 | 1.16 | −3.68 | 1.39 | 3.24 | 5.26 | 149.65 | 141.53 | 12.02 | 9.21 | 137.70 | |
| CHCH | 1 | 21.0 | 0.49 | −5.94 | 15.60 | 18.87 | 172.71 | 32.13 | 149.73 | 3.82 | 176.18 | 37.26 |
| 2 | 8.24 | 2.01 | −7.41 | 2.84 | 4.77 | 3.74 | 159.52 | 149.73 | 11.71 | 10.77 | 147.95 | |
| CH | 1 | 18.3 | 0.66 | −6.50 | 12.50 | 17.40 | 172.58 | 29.62 | 151.76 | 5.98 | 176.38 | 35.38 |
| 2 | 9.12 | 2.14 | −8.12 | 3.14 | 4.98 | 3.54 | 161.91 | 151.76 | 11.79 | 11.17 | 150.3 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chattopadhyaya, M.; Alam, M.M. A Theoretical Study of One- and Two-Photon Activity of D-Luciferin. Computation 2016, 4, 43. https://doi.org/10.3390/computation4040043
Chattopadhyaya M, Alam MM. A Theoretical Study of One- and Two-Photon Activity of D-Luciferin. Computation. 2016; 4(4):43. https://doi.org/10.3390/computation4040043
Chicago/Turabian StyleChattopadhyaya, Mausumi, and Md. Mehboob Alam. 2016. "A Theoretical Study of One- and Two-Photon Activity of D-Luciferin" Computation 4, no. 4: 43. https://doi.org/10.3390/computation4040043