Metal-Based Nanoparticles as Antimicrobial Agents: An Overview
Abstract
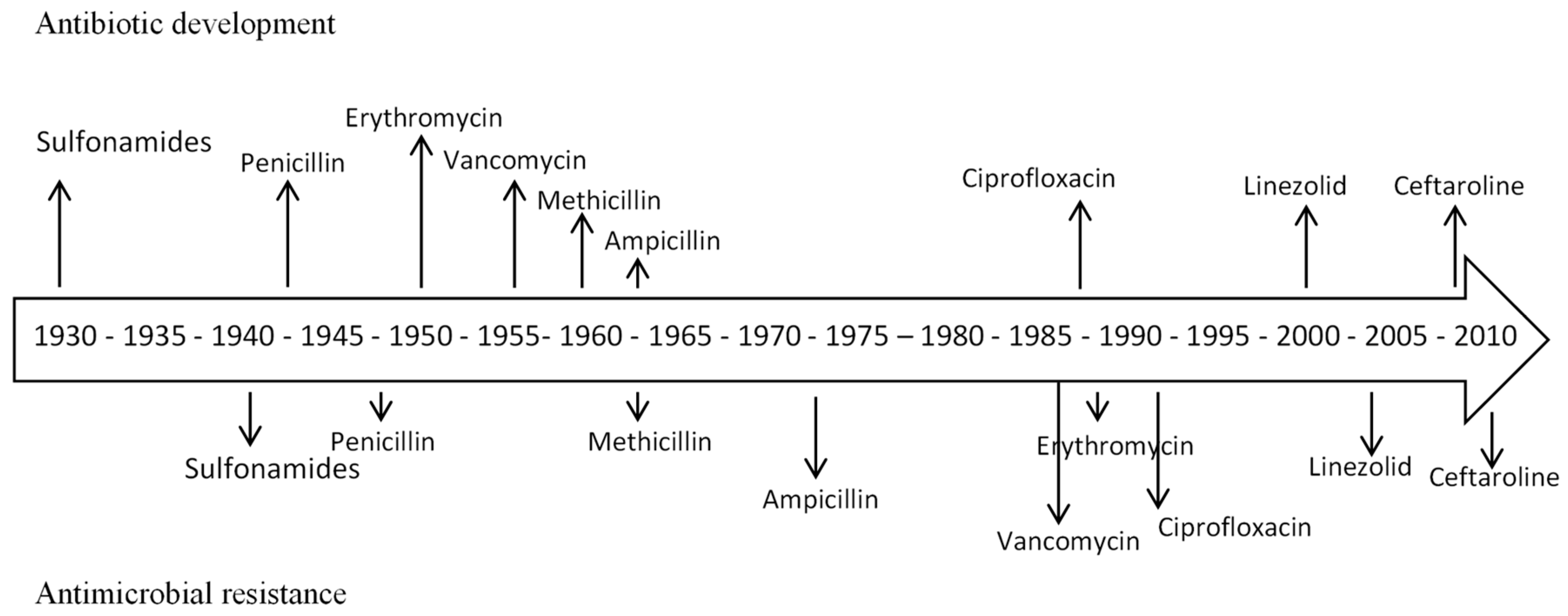
:1. Introduction
2. Metal Nanoparticles: Overview
2.1. Metal-Based Nanoparticle General Mechanisms
2.2. Synthesis of Metal and Metal Oxide Nanoparticles
2.2.1. Thermolysis Methods
2.2.2. Chemical Reduction Methods
2.2.3. Biochemical Methods
2.2.4. Electrochemical Methods
2.2.5. Wave-Assisted Chemical Methods
2.2.6. Cementation Methods
2.2.7. Biological Methods
3. Silver Nanoparticles (AgNPs)
3.1. Synthesis
3.1.1. Conventional Chemistry
3.1.2. Green Chemistry
 Ag+(aq) + NO3−(aq)
Ag+(aq) + NO3−(aq)
3.1.3. Physical Methods
3.2. Characterization of AgNPs
3.3. Pharmacokinetics
3.4. Absorption
3.4.1. Gastrointestinal Absorption
3.4.2. Pulmonary Absorption
3.4.3. Cutaneous Absorption
3.5. Distribution
3.6. Metabolism and Excretion
3.7. Antimicrobial
3.8. Other Pharmaceutical Properties
3.9. Toxicity Assessment
4. Copper and Copper Oxide Nanoparticles (CuNPs, Cu2ONPs and CuONPs)
4.1. Synthesis
4.2. Pharmacokinetics
4.3. Pharmacodynamics
4.4. Pharmaceutical Properties
4.5. Toxicity Assessment
5. Gold Nanoparticles (AuNPs)
5.1. Synthesis
5.2. Pharmacokinetics
5.3. Pharmacodynamics
5.4. Other Pharmaceutical Properties
5.5. Toxicity Assessment
6. Zinc Oxide Nanoparticles (ZnONPs)
6.1. Synthesis and Production Methods
6.2. Pharmacokinetics
6.3. Antibacterial Properties
6.4. Other Pharmaceutical Properties
6.5. Toxicity Assessments
6.5.1. Pulmonary Toxicity
6.5.2. Hepatotoxicity
6.5.3. Nephrotoxicity
6.5.4. Neurotoxicity
6.6. Other Side Effects
7. Comparative Overview
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Antibacterial Agents in Clinical Development: An Analysis of the Antibacterial Clinical Development Pipeline, Including Tuberculosis; Organ. GWH (Ed.) WHO/EMP/IAU/2017.122017; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- Coates, A.R.; Halls, G.; Hu, Y. Novel classes of antibiotics or more of the same? Br. J. Pharmacol. 2011, 163, 184–194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aslam, B.; Wang, W.; Arshad, M.I.; Khurshid, M.; Muzammil, S.; Rasool, M.H.; Nisar, M.A.; Alvi, R.F.; Aslam, M.A.; Qamar, M.U.; et al. Antibiotic resistance: A rundown of a global crisis. Infect. Drug Resist. 2018, 11, 1645–1658. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- IACG. No time to wait: Infections from drug-resistant securing the future. Artforum. Int. 2016, 54, 113–114. [Google Scholar]
- Munita, J.M.; Arias, C.A. Mechanisms of Antibiotic Resistance. Microbiol. Spectr. 2016, 2, 1–37. [Google Scholar]
- Dugassa, J.; Shukuri, N. Antibiotic resistance and its mechanism of Development. J. Heal. Med. Nurs. 2017, 1, 1–17. [Google Scholar]
- Power, E. Impact of antibiotic restrictions: The pharmaceutical perspective. Clin. Microbiol. Infect. 2006, 12, 25–34. [Google Scholar] [CrossRef] [Green Version]
- Fernandes, P.; Martens, E. Antibiotics in late clinical development. Biochem. Pharmacol. 2017, 133, 152–163. [Google Scholar] [CrossRef] [Green Version]
- Global Priority List of Antibiotic Resistant Bacteria to Guide Research, Discovery, and Development of New Antibiotics; WHO: Geneva, Switzerland, 2017.
- Ardal, C.; Findlay, D.; Savic, M.; Carmeli, Y.; Gyssens, I.; Laxminarayan, R.; Outterson, K.; Rex, J. Revitalizing the antibiotic pipeline: Stimulating innovation while driving sustainable use and global access. Drive-Ab Rep. 2018. in production. [Google Scholar]
- Slavin, Y.N.; Asnis, J.; Häfeli, U.O.; Bach, H. Metal nanoparticles: Understanding the mechanisms behind antibacterial activity. J. Nanobiotechnol. 2017, 15, 1–20. [Google Scholar] [CrossRef]
- Wang, L.; Hu, C.; Shao, L. The antimicrobial activity of nanoparticles: Present situation and prospects for the future. Int. J. Nanomed. 2017, 12, 1227–1249. [Google Scholar] [CrossRef] [Green Version]
- Bruslind, L. Bacteria: Internal components. In Microbiology; Open Oregon Stat: Corvallis, OR, USA, 2017. [Google Scholar]
- Stensberg, M.C.; Wei, Q.; McLamore, E.S.; Porterfield, D.M.; Wei, A.; Sepúlveda, M.S. Toxicological studies on si Sepúlveda, Toxicological studies on silver nanoparticles: Challenges and opportunities in assessment, monitoring and imaging. Nanomedicine 2011, 6, 879–898. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuan, P.; Ding, X.; Yang, Y.Y.; Xu, Q.H. Metal Nanoparticles for Diagnosis and Therapy of Bacterial Infection. Adv. Healthc. Mater. 2018, 7, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Heiligtag, F.; Niederberger, M. The fascinating world of nanoparticle research. Mater. Today 2013, 16, 262–271. [Google Scholar] [CrossRef]
- Kandi, V.; Kandi, S. Antimicrobial properties of nanomolecules: Potential candidates as antibiotics in the era of multi-drug resistance. Epidemiol. Health 2015, 37, e2015020. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iravani, S.; Korbekandi, H.; Vahid, M.S.; Zolfaghari, B. Synthesis of silver nanoparticles: Chemical, physical and biological methods. Res. Pharm. Sci. 2014, 9, 385–406. [Google Scholar]
- Wang, Y.; Xia, Y. Bottom-Up and Top-Down Approaches to the Synthesis of Monodispersed Spherical Colloids of Low Melting-Point Metals. Nano Lett. 2004, 4, 2047–2050. [Google Scholar] [CrossRef]
- Watt, J.; Cheong, S.; Tilley, R.D. How to control the shape of metal nanostructures in organic solution phase synthesis for plasmonics and catalysis. Nano Today 2013, 8, 198–215. [Google Scholar] [CrossRef]
- Kashiwagi, Y.; Yamamoto, M.; Nakamoto, M. Facile size-regulated synthesis of silver nanoparticles by controlled thermolysis of silver alkylcarboxylates in the presence of alkylamines with different chain lengths. J. Colloid Interface Sci. 2006, 300, 169–175. [Google Scholar] [CrossRef]
- Tan, K.S.; Cheong, K.Y. Advances of Ag, Cu, and Ag-Cu alloy nanoparticles synthesized via chemical reduction route. J. Nanopart. Res. 2013, 15, 1537. [Google Scholar] [CrossRef]
- Liu, M.S.; Lin, M.C.C.; Tsai, C.Y.; Wang, C.C. Enhancement of thermal conductivity with Cu for nanofluids using chemical reduction method. Int. J. Heat Mass Transf. 2006, 49, 3028–3033. [Google Scholar] [CrossRef]
- Kobayashi, M.; Yamashita, I.; Uraoka, Y.; Shiba, K.; Tomita, S. Gold nanostructures using tobacco mosaic viruses for optical metamaterials. Proc. SPIE Int. Soc. Opt. Eng. 2011, 8070, 80700C. [Google Scholar]
- Kannan, N.; Subbalaxmi, S. Biogenesis of nanoparticles—A current perspective. Rev. Adv. Mater. Sci. 2011, 27, 99–114. [Google Scholar]
- Ma, H.; Yin, B.; Wang, S.; Jiao, Y.; Pan, W.; Huang, S.; Chen, S.; Meng, F. Synthesis of Silver and Gold Nanoparticles by a Novel Electrochemical Method. ChemPhysChem 2004, 5, 68–75. [Google Scholar] [CrossRef]
- Ambrusi, R.E.; Staikov, G.; Garcia, S.G. Electrochemical synthesis of Cd–Ag bimetallic particles and the involved alloy formation. J. Electroanal. Chem. 2014, 728, 130–133. [Google Scholar] [CrossRef]
- Ilias, S.H.; Kok, K.Y.; Ng, I.K.; Saidin, N.U. Electrochemical synthesis and characterization of palladium nanostructures. J. Phys. Conf. Ser. 2013, 431, 012003. [Google Scholar] [CrossRef] [Green Version]
- Okitsu, K. Sonochemical synthesis of metal nanoparticles. Theor. Exp. Sonochem. Involv. Inorg. Syst. 2001, 131–150. [Google Scholar]
- Chadha, R.; Maiti, N.; Kapoor, S. Reduction and aggregation of silver ions in aqueous citrate solutions. Mater. Sci. Eng. C. 2014, 38, 92–196. [Google Scholar] [CrossRef]
- Yamamoto, H.; Kozawa, T.; Tagawa, S.; Naito, M.; Marignier, J.-L.; Mostafavi, M.; Belloni, J. Radiation-induced synthesis of metal nanoparticles in ethers THF and PGMEA. Radiat. Phys. Chem. 2013, 91, 148–155. [Google Scholar] [CrossRef]
- Tung, H.T.; Chen, I.G.; Kempson, I.M.; Song, J.M.; Liu, Y.F.; Chen, P.W.; Hwang, W.S.; Hwu, Y. Shape-controlled synthesis of silver nanocrystals by X-ray irradiation for inkjet printing. ACS Appl. Mater. Interfaces 2012, 4, 5930–5935. [Google Scholar] [CrossRef]
- Cuba, V.; Nemec, M.; Gbur, T.; John, J.; Pospíšil, P.; Múcka, M. Radiation formation of colloidal silver particles in aqueous systems. Appl. Radiat. Isot. 2010, 68, 676–678. [Google Scholar] [CrossRef]
- Wani, I.A.; Ganguly, A.; Ahmed, J.; Ahmad, T. Silver nanoparticles: Ultrasonic wave assisted synthesis, optical characterization and surface area studies. Mater. Lett. 2011, 65, 520–522. [Google Scholar] [CrossRef]
- Nadagouda, M.N.; Speth, T.F.; Varma, R.S. Microwave-assisted green synthesis of silver nanostructures. Acc. Chem. Res. 2011, 44, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Larosa, C.; Salerno, M.; Nanni, P.; Reverberi, A.P. Cobalt cementation in an ethanol–water system: Kinetics and morphology of metal aggregates. Ind. Eng. Chem. Res. 2012, 51, 16564–16572. [Google Scholar] [CrossRef]
- Reverberi, A.P.; Kuznetsov, N.T.; Meshalkin, V.P.; Salerno, M.; Fabiano, B. Systematical analysis of chemical methods in metal nanoparticles synthesis. Theor. Found. Chem. Eng. 2016, 50, 59–66. [Google Scholar] [CrossRef]
- Sharma, A.K.; Kumar, A.; Taneja, G.; Nagaich, U.; Deep, A.; Rajput, S.K. Synthesis and preliminary therapeutic evaluation of copper nanoparticles against diabetes mellitus and -induced micro- (renal) and macro-vascular (vascular endothelial and cardiovascular) abnormalities in rats. RSC Adv. 2016, 6, 36870–36880. [Google Scholar] [CrossRef]
- Shah, M.; Fawcett, D.; Sharma, S.; Tripathy, S.K.; Poinern, G.E.J. Green Synthesis of Metallic Nanoparticles via Biological Entities. Materials 2015, 8, 7278–7308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abdel-Kareem, M.M.; Zohri, A.A. Extracellular mycosynthesis of gold nanoparticles using Trichoderma hamatum: Optimization, characterization and antimicrobial activity. Lett. Appl. Microbiol. 2018, 67, 465–475. [Google Scholar] [CrossRef]
- Nagalingam, M.; Kalpana, V.N.; Devi Rajeswari, V.; Panneerselvam, A. Biosynthesis, characterization, and evaluation of bioactivities of leaf extract-mediated biocompatible gold nanoparticles from Alternanthera bettzickiana. Biotechnol. Rep. 2018, 19, e00268. [Google Scholar]
- Li, J.; Li, Q.; Ma, X.; Tian, B.; Li, T.; Yu, J.; Dai, S.; Weng, Y.; Hua, Y. Biosynthesis of gold nanoparticles by the extreme bacterium Deinococcus radiodurans and an evaluation of their antibacterial properties. Int. J. Nanomed. 2016, 11, 5931–5944. [Google Scholar] [CrossRef] [Green Version]
- Baker, S.; Satish, S. Biosynthesis of gold nanoparticles by Pseudomonas veronii AS41G inhabiting Annona squamosa L. Spectrochim. Acta 2015, 150, 691–695. [Google Scholar] [CrossRef]
- Singh, S.; Vidyarthi, A.S.; Nigam, V.K.; Dev, A. Extracellular facile biosynthesis, characterization and stability of gold nanoparticles by Bacillus licheniformis. Artif. Cells Nanomed. Biotechnol. 2014, 42, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Balagurunathan, R.; Radhakrishnan, M.; Babu Rajendran, R.; Velmurugan, D. Biosynthesis of gold nanoparticles by actinomycete streptomyces viridogens strain HM10. Indian J. Biochem. Biophys. 2011, 48, 331–335. [Google Scholar] [PubMed]
- Arockiya Aarthi Rajathi, F.; Parthiban, C.; Ganesh Kumar, V.; Anantharaman, P. Biosynthesis of antibacterial gold nanoparticles using brown alga, Stoechospermum marginatum (kützing). Spectrochim. Acta 2012, 99, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Thakker, J.N.; Dalwadi, P.; Dhandhukia, P.C. Biosynthesis of Gold Nanoparticles Using Fusarium oxysporum f. sp. cubense JT1, a Plant Pathogenic Fungus. ISRN Biotechnol. 2013, 2013, 515091. [Google Scholar] [CrossRef] [PubMed]
- Lv, Q.; Zhang, B.; Xing, X.; Zhao, Y.; Cai, R.; Wang, W.; Gu, Q. Biosynthesis of copper nanoparticles using Shewanella loihica PV-4 with antibacterial activity: Novel approach and mechanisms investigation. J. Hazard. Mater. 2018, 347, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Shoeibi, S.; Mashreghi, M. Biosynthesis of selenium nanoparticles using Enterococcus faecalis and evaluation of their antibacterial activities. J. Trace Elem. Med. Biol. 2017, 39, 135–139. [Google Scholar] [CrossRef]
- Vijayakumar, S.; Krishnakumar, C.; Arulmozhi, P.; Mahadevan, S.; Parameswari, N. Biosynthesis, characterization and antimicrobial activities of zinc oxide nanoparticles from leaf extract of Glycosmis pentaphylla (Retz.). Microb. Pathog. 2018, 116, 44–48. [Google Scholar] [CrossRef]
- Rajabi, H.R.; Naghiha, R.; Kheirizadeh, M.; Sadatfaraji, H.; Mirzaei, A.; Alvand, Z.M. Microwave assisted extraction as an efficient approach for biosynthesis of zinc oxide nanoparticles: Synthesis, characterization, and biological properties. Mater. Sci. Eng. C 2017, 78, 1109–1118. [Google Scholar] [CrossRef]
- Moghaddam, A.B.; Moniri, M.; Azizi, S.; Rahim, R.A.; Ariff, A.B.; Saad, W.Z.; Namvar, F.; Navaderi, M. Biosynthesis of ZnO nanoparticles by a new Pichia kudriavzevii yeast strain and evaluation of their antimicrobial and antioxidant activities. Molecules 2017, 22, 6. [Google Scholar] [CrossRef] [Green Version]
- Sharma, D.; Myalowenkosi, I.S.; Suvardhan, K.; Phumlane, S.M.; Gulshan, S.; Thor, A.S.; Krishna, B. Biosynthesis of ZnO nanoparticles using Jacaranda mimosifolia flowers extract: Synergistic antibacterial activity and molecular simulated facet specific adsorption studies. J. Photochem. Photobiol. B Biol. 2016, 162, 199–207. [Google Scholar] [CrossRef] [Green Version]
- Gu, H.; Chen, X.; Chen, F.; Zhou, X.; Parsaee, Z. Ultrasound-assisted biosynthesis of CuO-NPs using brown alga Cystoseira trinodis: Characterization, photocatalytic AOP, DPPH scavenging and antibacterial investigations. Ultrason. Sonochem. 2018, 41, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Basiuk, V.A.; Basiuk, E.V. Green Processes for Nanotechnology; From Inorganic to Bioinspiring Nanomaterials; Springer International Publishing: Cham, Switzerland, 2015; pp. 35–73. [Google Scholar]
- Castro, L.; Blázquez, M.L.; Muñoz, J.; González, F.G.; Ballester, A. Process, mechanism and applications of metal nanoparticles prepared by bio-mediated process. Rev. Adv. Sci. Eng. 2014, 3, 199–216. [Google Scholar] [CrossRef]
- Chen, X.; Schluesener, H.J. Nanosilver: A nanoproduct in medical application. Toxicol. Lett. 2008, 176, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Aderibigbe, B.A. Metal-Based Nanoparticles for the Treatment of Infectious Diseases. Molecules 2017, 22, 1370. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.F.; Liu, Z.G.; Shen, W.; Gurunathan, S. Silver nanoparticles: Synthesis, characterization, properties, applications, and therapeutic approaches. Int. J. Mol. Sci. 2016, 17, 9. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Kaurav, N.; Choudhary, K.K.; Okram, G.S. Synthesis and optical properties of silver nanoparticles and arrays. AIP Conf. Proc. 2015, 1670, 1221–1231. [Google Scholar]
- Hamouda, R.A.; Hussein, M.H.; Abo-elmagd, R.A.; Bawazir, S.S. Synthesis and biological characterization of silver nanoparticles derived from the cyanobacterium Oscillatoria limnetica. Sci. Rep. 2019, 9, 1–17. [Google Scholar] [CrossRef]
- Dakal, T.C.; Kumar, A.; Majumdar, R.S.; Yadav, V. Mechanistic basis of antimicrobial actions of silver nanoparticles. Front. Microbiol. 2016, 7, 1831. [Google Scholar] [CrossRef] [Green Version]
- Solomon, S.D.; Bahadory, M.; Jeyarajasingam, A.V.; Rutkowsky, S.A.; Boritz, C.; Mulfinger, L. Synthesis and study of silver nanoparticles. J. Chem. Educ. 2007, 84, 322–325. [Google Scholar]
- Kumar, S.V.; Bafana, A.P.; Pawar, P.; Rahman, A.; Dahoumane, S.A.; Jeffryes, C.S. High conversion synthesis of <10 nm starch-stabilized silver nanoparticles using microwave technology. Sci. Rep. 2018, 8, 1–10. [Google Scholar]
- Ho, C.H.; Thiel, M.; Celik, S.; Odermatt, E.K.; Berndt, I.; Thomann, R.; Tiller, J.C. Conventional and microwave-assisted synthesis of hyperbranched and highly branched polylysine towards amphiphilic core-shell nanocontainers for metal nanoparticles. Polymer 2012, 53, 4623–4630. [Google Scholar] [CrossRef]
- Cao, H. Silver Nanoparticles for Antibacterial Devices: Biocompatibility and Toxicity; CRC Press: Shanghai, China, 2017. [Google Scholar]
- Javaid, A.; Oloketuyi, S.; Khan, M.M.; Khan, F. Diversity of Bacterial Synthesis of Silver Nanoparticles. Bionanoscience 2018, 8, 43–58. [Google Scholar] [CrossRef]
- Haefeli, C.; Franklin, C.; Hardy, K. Plasmid-determined silver resistance in Pseudomonas stutzeri isolated from a silver mine. J. Bacteriol. 1984, 158, 389–392. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gan, S.-L.; Zhang, L.; Zhang, S.; He, Y.; Tian, S.; Biosynthesis, Y.; Gan, L.; Zhang, S.; Zhang, Y.; He, S.; et al. Characterization and antimicrobial activity of silver nanoparticles by a halotolerant B. endophyticus. Prep. Biochem. Biotechnol. 2018, 48, 582–588. [Google Scholar] [CrossRef]
- Jang, E.Y.; Son, Y.J.; Park, S.Y.; Yoo, J.Y.; Cho, Y.N.; Jeong, S.Y.; Liu, S.M.; Son, H.J. Improved biosynthesis of silver nanoparticles using keratinase from Stenotrophomonas maltophilia R13: Reaction optimization, structural characterization, and biomedical activity. Bioprocess Biosyst. Eng. 2018, 41, 381–393. [Google Scholar] [CrossRef]
- Elsayed, M.A.; Othman, A.M.; Hassan, M.M.; Elshafei, A.M. Optimization of silver nanoparticles biosynthesis mediated by Aspergillus niger NRC1731 through application of statistical methods: Enhancement and characterization. 3 Biotech. 2018, 8, 132. [Google Scholar] [CrossRef]
- Zhao, X.; Zhou, L.; Rajoka Riaz, M.S.; Yan, L.; Jiang, C.; Shao, D.; Zhu, J.; Shi, J.; Huang, Q.; Yang, H.; et al. Fungal silver nanoparticles: Synthesis, application and challenges. Crit. Rev. Biotechnol. 2018, 38, 817–835. [Google Scholar] [CrossRef]
- Ahmed, S.; Ahmad, M.; Swami, B.L.; Ikram, S. A review on plants extract mediated synthesis of silver nanoparticles for antimicrobial applications: A green expertise. J. Adv. Res. 2016, 7, 17–28. [Google Scholar] [CrossRef] [Green Version]
- Logeswari, P.; Silambarasan, S.; Abraham, J. Synthesis of silver nanoparticles using plants extract and analysis of their antimicrobial property. J. Saudi Chem. Soc. 2015, 19, 311–317. [Google Scholar] [CrossRef] [Green Version]
- Amendola, V.; Bakr, O.M.; Stellacci, F. A study of the surface plasmon resonance of silver nanoparticles by the discrete dipole approximation method: Effect of shape, size, structure, and assembly. Plasmonics 2010, 5, 85–97. [Google Scholar] [CrossRef]
- Shivakumar, M.; Nagashree, K.L.; Yallappa, S.; Manjappa, S.; Manjunath, K.S.; Dharmaprakash, M.S. Biosynthesis of silver nanoparticles using pre-hydrolysis liquor of Eucalyptus wood and its effective antimicrobial activity. Enzyme Microb. Technol. 2017, 97, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Markus, J.; Wang, D.; Kim, Y.-J.; Ahn, S.; Mathiyalagan, R.; Wang, C.; Yang, D.C. Biosynthesis, characterization, and bioactivities evaluation of silver and gold nanoparticles mediated by the roots of chinese herbal angelica pubescens maxim. Nanoscale Res. Lett. 2017, 12, 46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roy, S.; Mukherjee, T.; Chakraborty, S.; Das, T.K. Biosynthesis, characterisation & antifungal activity of silver nanoparticles synthesized by the fungus Aspergillus foetidus MTCC8876. Dig. J. Nanomater. Biostruct. 2012, 8, 197–205. [Google Scholar]
- Balakrishnan, S.; Sivaji, I.; Kandasamy, S.; Duraisamy, S.; Kumar, N.S.; Gurusubramanian, G. Biosynthesis of silver nanoparticles using Myristica fragrans seed (nutmeg) extract and its antibacterial activity against multidrug-resistant (MDR) Salmonella enterica serovar Typhi isolates. Environ. Sci. Pollut. Res. 2017, 24, 14758–14769. [Google Scholar] [CrossRef] [PubMed]
- Farhadi, S.; Ajerloo, B.; Mohammadi, A. Green biosynthesis of spherical silver nanoparticles by using date palm (Phoenix Dactylifera) fruit extract and study of their antibacterial and catalytic activities. Acta Chim. Slov. 2017, 64, 129–143. [Google Scholar] [CrossRef] [PubMed]
- Umoren, S.A.; Obot, I.B.; Gasem, Z.M. Green synthesis and characterization of silver nanoparticles using red apple (malus domestica) fruit extract at room temperature. J. Mater. Environ. Sci. 2014, 5, 907–914. [Google Scholar]
- Samberg, M.E.; Lin, Z.; Monteiro Riviere, N.A. In Vitro and In Vivo Toxicity and Pharmacokinetics of Silver Nanoparticles. In Bhushan B, Encyclopedia of Nanotechnology; Springer: Dordrecht, The Netherlands, 2016; pp. 1554–1567. [Google Scholar]
- Bachler, G.; von Goetz, N.; Hungerbühler, K. A physiologically based pharmacokinetic model for ionic silver and silver nanoparticles. Int. J. Nanomed. 2013, 8, 3365–3382. [Google Scholar]
- Walczak, A.P.; Fokkink, R.; Peters, R.; Tromp, P.; Herrera Rivera, Z.E.; Rietjens, I.M.; Hendriksen, P.J.; Bouwmeester, H. Behavior of silver nanoparticles and silver ions in an in vitro human gastrointestinal digestion model. Nanotoxicology 2013, 7, 1198–1210. [Google Scholar] [CrossRef]
- Loeschner, K.; Hadrup, N.; Qvortrup, K.; Larsen, A.; Gao, X.; Vogel, U.; Mortensen, A.; Lam, H.R.; Larsen, E.H. Distribution of silver in rats following 28 days of repeated oral exposure to silver nanoparticles or silver acetate. Part. Fibre Toxicol. 2011, 8, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Takenaka, S.; Karg, E.; Roth, C.; Schulz, H.; Ziesenis, A.; Heinzmann, U.; Schramel, P.; Heyder, J. Pulmonary and systemic distribution of inhaled ultrafine silver particles in rats. Environ. Health Perspect. 2001, 109, 547–551. [Google Scholar]
- Souto, E.B.; Ribeiro, A.F.; Ferreira, M.I.; Teixeira, M.C.; Shimojo, A.A.M.; Soriano, J.L.; Naveros, B.C.; Durazzo, A.; Lucarini, M.M.; Souto, S.B.; et al. New nanotechnologies for the treatment and repair of skin burns infections. Int. J. Mol. Sci. 2020, 21, 393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- George, R.; Merten, S.; Wang, T.T.; Kennedy, P.; Maitz, P. In vivo analysis of dermal and systemic absorption of silver nanoparticles through healthy human skin. Australas. J. Dermatol. 2014, 55, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Larese, F.F.; D’Agostin, F.; Crosera, M.; Adami, G.; Renzi, N.; Bovenzi, M.; Maina, G. Human skin penetration of silver nanoparticles through intact and damaged skin. Toxicology 2009, 255, 33–37. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Kim, J.S.; Cho, H.S.; Rha, D.S.; Kim, J.M.; Park, J.D.; Choi, B.S.; Lim, R.; Chang, H.K.; Chung, Y.H.; et al. Twenty-eight-day oral toxicity, genotoxicity, and gender-related tissue distribution of silver nanoparticles in Sprague-Dawley rats. Inhal. Toxicol. 2008, 20, 575–583. [Google Scholar] [CrossRef]
- Dziendzikowska, K.; Gromadzka-Ostrowska, J.; Lankoff, A.; Oczkowski, M.; Krawczyńska, A.; Chwastowska, J.; Sadowska-Bratek, M.; Chajduk, E.; Wojewódzka, M.; Dušinská, M.; et al. Time-dependent biodistribution and excretion of silver nanoparticles in male Wistar rats. J. Appl. Toxicol. 2012, 32, 920–928. [Google Scholar] [CrossRef]
- Jiménez-Lamana, J.; Laborda, F.; Bolea, E.; Abad-Álvaro, I.; Castillo, J.R.; Bianga, J.; He, M.; Bierla, K.; Mounicou, S.; Ouerdane, L. An insight into silver nanoparticles bioavailability in rats. Metallomics 2014, 6, 2242–2249. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.H.; Kim, Y.S.; Song, K.S.; Ryu, H.R.; Sung, J.H.; Park, J.D.; Park, H.M.; Song, N.W.; Shin, B.S.; Marshak, D.; et al. Biopersistence of silver nanoparticles in tissues from Sprague-Dawley rats. Part. Fibre Toxicol. 2013, 10, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Abbaszadegan, A.; Ghahramani, Y.; Gholami, A.; Hemmateenejad, B.; Dorostkar, S.; Nabavizadeh, M.; Sharghi, H. The effect of charge at the surface of silver nanoparticles on antimicrobial activity against Gram-positive and Gram-negative bacteria: A preliminary study. J. Nanomater. 2015, 2015, 720654. [Google Scholar] [CrossRef] [Green Version]
- Losasso, C.; Belluco, S.; Cibin, V.; Zavagnin, P.; Mičetić, I.; Gallocchio, F.; Zanella, M.; Bregoli, L.; Biancotto, G.; Ricci, A. Antibacterial activity of silver nanoparticles: Sensitivity of different Salmonella serovars. Front. Microbiol. 2014, 5, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Qing, Y.; Cheng, L.; Li, R.; Liu, G.; Zhang, Y.; Tang, X.; Wang, J.; Liu, H.; Qin, Y. Potential antibacterial mechanism of silver nanoparticles and the optimization of orthopedic implants by advanced modification technologies. Int. J. Nanomed. 2018, 13, 3311–3327. [Google Scholar] [CrossRef] [Green Version]
- Gordon, O.; Slenters, T.V.; Brunetto, P.S.; Villaruz, A.E.; Sturdevant, D.E.; Otto, M.; Landmann, R.; Fromm, K.M. Silver coordination polymers for prevention of implant infection: Thiol interaction, impact on respiratory chain enzymes, and hydroxyl radical induction. Antimicrob. Agents Chemother. 2010, 54, 4208–4218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Panáček, A.; Kolár, M.; Vecerová, R.; Prucek, R.; Soukupová, J.; Krystof, V.; Hamal, P.; Zboril, R.; Kvítek, L. Antifungal activity of silver nanoparticles against Candida. Biomaterials 2009, 30, 6333–6340. [Google Scholar] [CrossRef] [PubMed]
- Lara, H.H.; Ayala-Nuñez, N.V.; Ixtepan-Turrent, L.; Rodriguez-Padilla, C. Mode of antiviral action of silver nanoparticles against HIV-1. J. Nanobiotechnol. 2010, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, K.; Anwar, A.; Khan, N.A.; Siddiqui, R. Brain-Eating Amoebae: Silver Nanoparticle Conjugation Enhanced Efficacy of Anti-Amoebic Drugs against Naegleria fowleri. ACS Chem. Neurosci. 2017, 8, 2626–2630. [Google Scholar] [CrossRef]
- Faedmaleki, F.; Shirazi, F.H.; Salarian, A.A.; Ashtiani, H.A.; Rastegar, H. Toxicity effect of silver nanoparticles on mice liver primary cell culture and HepG2 cell line. Iran. J. Pharm. Res. 2014, 13, 235–242. [Google Scholar]
- Kedi, P.B.E.; Meva, F.E.; Kotsedi, L.; Nguemfo, E.L.; Zangueu, C.B.; Ntoumba, A.A.; Mohamed, H.E.A.; Dongmo, A.B.; Maaza, M. Eco-friendly synthesis, characterization, in vitro and in vivo anti-inflammatory activity of silver nanoparticle-mediated Selaginella myosurus aqueous extract. Int. J. Nanomed. 2018, 13, 8537–8548. [Google Scholar] [CrossRef] [Green Version]
- Ávalos, A.; Haza, A.; Mateao, D.; Morales, P. Nanopartículas de plata: Aplicaciones y riesgos tóxicos para la salud y el medio ambiente. Rev. Complut. Ciencias Vet. 2013, 7, 1–23. [Google Scholar] [CrossRef] [Green Version]
- El Badawy, A.M.; Silva, R.G.; Morris, B.; Scheckel, K.G.; Suidan, M.T.; Tolaymat, T.M. Surface charge-dependent toxicity of silver nanoparticles. Environ. Sci. Technol. 2011, 45, 283–287. [Google Scholar] [CrossRef]
- De Matteis, V.; Rizzello, L.; Ingrosso, C.; Liatsi-Douvitsa, E.; De Giorgi, M.L.; De Matteis, G.; Rinaldi, R. Cultivar-dependent anticancer and antibacterial properties of silver nanoparticles synthesized using leaves of different olea europaea trees. Nanomaterials 2019, 9, 1544. [Google Scholar] [CrossRef] [Green Version]
- Tanase, C.; Berta, L.; Coman, N.A.; Roșca, I.; Man, A.; Toma, F.; Mocan, A.; Nicolescu, A.; Jakab-Farkas, L.; Biró, D.; et al. Antibacterial and antioxidant potential of silver nanoparticles biosynthesized using the spruce bark extract. Nanomaterials 2019, 9, 11. [Google Scholar] [CrossRef] [Green Version]
- Patil, S.P.; Kumbhar, S.T. Antioxidant, antibacterial and cytotoxic potential of silver nanoparticles synthesized using terpenes rich extract of Lantana camara L. leaves. Biochem. Biophys. Rep. 2017, 10, 76–81. [Google Scholar]
- Dada, A.O.; Adekola, F.A.; Dada, F.E.; Adelani-Akande, A.T.; Bello, M.O.; Okonkwo, C.R.; Inyinbor, A.A.; Oluyori, A.P.; Olayanju, A.; Ajanaku, K.O.; et al. Silver nanoparticle synthesis by Acalypha wilkesiana extract: Phytochemical screening, characterization, influence of operational parameters, and preliminary antibacterial testing. Heliyon 2019, 5, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Figueiredo, E.P.; Ribeiro, J.M.; Nishio, E.K.; Scandorieiro, S.; Costa, A.F.; Cardozo, V.F.; Oliveira, A.G.; Durán, N.; Panagio, L.A.; Kobayashi, R.; et al. New approach for simvastatin as an antibacterial: Synergistic effect with bio-synthesized silver nanoparticles against multidrug-resistant bacteria. Int. J. Nanomed. 2019, 14, 7975–7985. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hossain, M.M.; Polash, S.A.; Takikawa, M.; Shubhra, R.D.; Saha, T.; Islam, Z.; Hossain, S.; Hasan, M.A.; Takeoka, S.; Sarker, S.R. Investigation of the Antibacterial Activity and in vivo Cytotoxicity of Biogenic Silver Nanoparticles as Potent Therapeutics. Front. Bioeng. Biotechnol. 2019, 7, 239. [Google Scholar] [CrossRef]
- Wypij, M.; Golinska, P.; Dahm, H.; Rai, M. Actinobacterial-mediated synthesis of silver nanoparticles and their activity against pathogenic bacteria. IET Nanobiotechnol. 2017, 11, 336–342. [Google Scholar] [CrossRef]
- Shameli, K.; Ahmad, M.B.; Zargar, M.; Yunus, W.M.; Ibrahim, N.A.; Shabanzadeh, P.; Moghaddam, M.G. Synthesis and characterization of silver/montmorillonite/chitosan bionanocomposites by chemical reduction method and their antibacterial activity. Int. J. Nanomed. 2011, 6, 271–284. [Google Scholar] [CrossRef] [Green Version]
- Oves, M.; Rauf, M.A.; Hussain, A.; Qari, A.H.; Parwaz Khan, A.A.; Muhammad, P.; Rehman, M.T.; Alajmi, M.F.; Ismail, I.I.M. Antibacterial Silver Nanomaterial Synthesis from Mesoflavibacter zeaxanthinifaciens and Targeting Biofilm Formation. Front. Pharmacol. 2019, 10, 801. [Google Scholar] [CrossRef] [Green Version]
- Dong, Y.; Zhu, H.; Shen, Y.; Zhang, W.; Zhang, L. Antibacterial activity of silver nanoparticles of different particle size against Vibrio Natriegens. PLoS ONE 2019, 14, e0222322. [Google Scholar] [CrossRef] [Green Version]
- Deshmukh, A.R.; Gupta, A.; Kim, B.S. Ultrasound Assisted Green Synthesis of Silver and Iron Oxide Nanoparticles Using Fenugreek Seed Extract and Their Enhanced Antibacterial and Antioxidant Activities. Biomed. Res. Int. 2019, 2019, 1714358. [Google Scholar] [CrossRef]
- Feroze, N.; Arshad, B.; Younas, M.; Afridi, M.I.; Saqib, S.; Ayaz, A. Fungal mediated synthesis of silver nanoparticles and evaluation of antibacterial activity. Microsc. Res. Tech. 2019. [Google Scholar] [CrossRef]
- Gondil, V.S.; Kalaiyarasan, T.; Bharti, V.K.; Chhibber, S. Antibiofilm potential of Seabuckthorn silver nanoparticles (SBT@AgNPs) against Pseudomonas aeruginosa. 3 Biotech. 2019, 9, 402. [Google Scholar] [CrossRef] [PubMed]
- Mickymaray, S. One-step synthesis of silver nanoparticles using saudi arabian desert seasonal plant Sisymbrium irio and antibacterial activity against multidrug-resistant bacterial strains. Biomolecules 2019, 9, 662. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jayaprakash, N.; Suganya, R. Green synthesis of Ag nanoparticles using Tamarind fruit extract for the antibacterial studies. J. Photochem. Photobiol. B Biol. 2017, 169, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Nakkala, J.R.; Mata, R.; Sadras, S.R. Green synthesized nano silver: Synthesis, physicochemical profiling, antibacterial, anticancer activities and biological in vivo toxicity. J. Colloid Interface Sci. 2017, 499, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Burdușel, A.C.; Gherasim, O.; Grumezescu, A.M.; Mogoantă, L.; Ficai, A.; Andronescu, E. Biomedical applications of silver nanoparticles: An up-to-date overview. Nanomaterials 2018, 8, 681. [Google Scholar] [CrossRef] [Green Version]
- Seiffert, J.; Hussain, F.; Wiegman, C.; Li, F.; Bey, L.; Baker, W.; Porter, A.; Ryan, M.P.; Chang, Y.; Gow, A.; et al. Pulmonary toxicity of instilled silver nanoparticles: Influence of size, coating and rat strain. PLoS ONE 2015, 10, e0119726. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.H.; Jun, B.H. Silver nanoparticles: Synthesis and application for nanomedicine. Int. J. Mol. Sci. 2019, 20, 865. [Google Scholar] [CrossRef] [Green Version]
- Harmon, A.R.; Kennedy, A.J.; Poda, A.R.; Bednar, A.J.; Chappell, M.A.; Steevens, J.A. Determination of nanosilver dissolution kinetics and toxicity in an environmentally relevant aqueous medium. Environ. Toxicol. Chem. 2014, 33, 1783–1791. [Google Scholar] [CrossRef]
- Oukarroum, A.; Samadani, M.; Dewez, D. Influence of pH on the toxicity of silver nanoparticles in the green alga Chlamydomonas acidophila. Water. Air. Soil Pollut. 2014, 225, 1–8. [Google Scholar] [CrossRef]
- Asharani, P.V.; Lianwu, Y.; Gong, Z.; Valiyaveettil, S. Comparison of the toxicity of silver, gold and platinum nanoparticles in developing zebrafish embryos. Nanotoxicology 2012, 5, 43–54. [Google Scholar] [CrossRef]
- Cardoso, P. Nanopartículas de plata: Obtención, utilización como antimicrobiano e impacto en el área de la salud. Rev. Hosp. Niños 2016, 58, 19–28. [Google Scholar]
- Sagee, O.; Dror, I.; Berkowitz, B. Transport of silver nanoparticles (AgNPs) in soil. Chemosphere 2012, 88, 670–675. [Google Scholar] [CrossRef] [PubMed]
- Mirhosseini, M. Synergistic antibacterial effect of metal oxide nanoparticles and ultrasound stimulation. J. Biol. Today’s World 2015, 4, 138–144. [Google Scholar]
- Yoon, K.Y.; Hoon Byeon, J.; Park, J.H.; Hwang, J. Susceptibility constants of Escherichia coli and Bacillus subtilis to silver and copper nanoparticles. Sci. Total Environ. 2007, 373, 572–575. [Google Scholar] [CrossRef]
- Kumar, S.V.; Bafana, A.P.; Pawar, P.; Faltane, M.; Rahman, A.; Dahoumane, S.A.; Kucknoor, A.; Jeffryes, C.S. Optimized production of antibacterial copper oxide nanoparticles in a microwave-assisted synthesis reaction using response surface methodology. Colloids Surf. A 2019, 573, 170–178. [Google Scholar] [CrossRef]
- Moniri Javadhesari, S.; Alipour, S.; Mohammadnejad, S.; Akbarpour, M.R. Antibacterial activity of ultra-small copper oxide (II) nanoparticles synthesized by mechanochemical processing against S. aureus and E. coli. Mater. Sci. Eng. C 2019, 105, 110011. [Google Scholar] [CrossRef]
- Nabila, M.I.; Kannabiran, K. Biosynthesis, characterization and antibacterial activity of copper oxide nanoparticles (CuO NPs) from actinomycetes. Biocatal. Agric. Biotechnol. 2018, 15, 56–62. [Google Scholar] [CrossRef]
- Zhang, Y.J.; Ding, Z.; Zhao, G.; Zhang, T.; Xu, Q.; Cui, B.; Liu, J.X. Transcriptional responses and mechanisms of copper nanoparticle toxicology on zebrafish embryos. J. Hazard. Mater. 2018, 344, 1057–1068. [Google Scholar] [CrossRef]
- Yen, H.J.; Horng, J.L.; Yu, C.H.; Fang, C.Y.; Yeh, Y.H.; Lin, L.Y. Toxic effects of silver and copper nanoparticles on lateral-line hair cells of zebrafish embryos. Aquat. Toxicol. 2019, 215, 105273. [Google Scholar] [CrossRef]
- Jing, X.; Park, J.H.; Peters, T.M.; Thorne, P.S. Toxicity of copper oxide nanoparticles in lung epithelial cells exposed at the air-liquid interface compared with in vivo assessment. Toxicol. In Vitro 2015, 29, 502–511. [Google Scholar] [CrossRef] [Green Version]
- Gerber, A.; Bundschuh, M.; Klingelhofer, D.; Groneberg, D.A. Gold nanoparticles: Recent aspects for human toxicology. J. Occup. Med. Toxicol. 2013, 8, 32. [Google Scholar] [CrossRef] [Green Version]
- Her, S.; Jaffray, D.A.; Allen, C. Gold nanoparticles for applications in cancer radiotherapy: Mechanisms and recent advancements. Adv. Drug Deliv. Rev. 2017, 109, 84–101. [Google Scholar] [CrossRef] [PubMed]
- Newman, J.D.S.; Blanchard, G.J. Formation of gold nanoparticles using amine reducing agents. Langmuir 2006, 22, 5882–5887. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, P.; Vig, K.; Dennis, V.; Singh, S. Functionalized gold nanoparticles and their biomedical applications. Nanomaterials 2011, 1, 31–63. [Google Scholar] [CrossRef]
- Balasubramanian, S.K.; Jittiwat, J.; Manikandan, J.; Ong, C.N.; Yu, L.E.; Ong, W.Y. Biodistribution of gold nanoparticles and gene expression changes in the liver and spleen after intravenous administration in rats. Biomaterials 2010, 31, 2034–2042. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Shareena Dasari, T.P.; Deng, H.; Yu, H. Antimicrobial Activity of Gold Nanoparticles and Ionic Gold. J. Environ. Sci. Heal. Part C Environ. Carcinog. Ecotoxicol. Rev. 2015, 33, 286–327. [Google Scholar] [CrossRef] [PubMed]
- Patil, M.; Kim, G.-D. Eco-friendly approach for nanoparticles synthesis and mechanism behind antibacterial activity of silver and anticancer activity of gold nanoparticles. Appl. Microbiol. Biotechnol. 2017, 101, 79–92. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.P.; Ma, B.Y.; Wei, X.W.; Qian, Z.Y. The in vitro and in vivo toxicity of gold nanoparticles. Chin. Chem. Lett. 2017, 28, 691–702. [Google Scholar] [CrossRef]
- Alkilany, A.M.; Murphy, C.J. Toxicity and cellular uptake of gold nanoparticles: What we have learned so far? J. Nanopart. Res. 2010, 12, 2313–2333. [Google Scholar] [CrossRef] [Green Version]
- Ostroumov, S.A.; Poklonov, V.A.; Kotelevtsev, S.V.; Orlov, S.N. Toxicity of gold nanoparticles for plants in experimental aquatic system. Moscow Univ. Biol. Sci. Bull. 2014, 69, 108–112. [Google Scholar] [CrossRef] [Green Version]
- Botha, T.L.; James, T.E.; Wepener, V. Comparative aquatic toxicity of gold nanoparticles and ionic gold using a species sensitivity distribution approach. J. Nanomater. 2015, 986902. [Google Scholar] [CrossRef]
- Shamaila, S.; Zafar, N.; Riaz, S.; Sharif, R.; Nazir, J.; Naseem, S. Gold nanoparticles: An efficient antimicrobial agent against enteric bacterial human pathogen. Nanomaterials 2016, 6, 71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- MubarakAli, D.; Thajuddin, N.; Jeganathan, K.; Gunasekaran, M. Plant extract mediated synthesis of silver and gold nanoparticles and its antibacterial activity against clinically isolated pathogens. Colloids Surf. B 2011, 85, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Ayaz Ahmed, K.B.; Subramanian, S.; Sivasubramanian, A.; Veerappan, G.; Veerappan, A. Preparation of gold nanoparticles using Salicornia brachiata plant extract and evaluation of catalytic and antibacterial activity. Spectrochim. Acta 2014, 130, 54–58. [Google Scholar] [CrossRef]
- Król, A.; Pomastowski, P.; Rafińska, K.; Railean-Plugaru, V.; Buszewski, B. Zinc oxide nanoparticles: Synthesis, antiseptic activity and toxicity mechanism. Adv. Colloid Interface Sci. 2017, 249, 37–52. [Google Scholar] [CrossRef]
- Darvishi, E.; Kahrizi, D.; Arkan, E. Comparison of different properties of zinc oxide nanoparticles synthesized by the green (using Juglans regia L. leaf extract) and chemical methods. J. Mol. Liq. 2019, 286, 110831. [Google Scholar] [CrossRef]
- Kaliamurthi, S.; Selvaraj, G.; Elibol, Z.; Demir, A. The relationship between Chlorella sp. and zinc oxide nanoparticles: Changes in biochemical, oxygen evolution, and lipid production ability. Process. Biochem. 2019, 85, 43–50. [Google Scholar] [CrossRef]
- Mirzaei, H. Zinc oxide nanoparticles: Biological synthesis and biomedical applications. Ceram. Int. J. 2017, 43, 907–914. [Google Scholar] [CrossRef]
- Santhoshkumar, J.; Kumar, S.V.; Rajeshkumar, S. Synthesis of zinc oxide nanoparticles using plant leaf extract against urinary tract infection pathogen. Resour. Technol. 2017, 3, 459–465. [Google Scholar] [CrossRef]
- Jiang, J.; Pi, J.; Cai, J. The advancing of zinc oxide nanoparticles for biomedical applications. Bioinorg. Chem. Appl. 2018, 2018, 1062562. [Google Scholar] [CrossRef]
- Happy, A.; Soumyaa, M.; Kumara, V.; Rajeshkumar, S.; Sheba, D.R.; Lakshmi, T.; Nallaswamyd, V.D. Phyto-assisted synthesis of zinc oxide nanoparticles using Cassia alata and its antibacterial activity against Escherichia coli. Biochem. Biophys. Rep. 2019, 17, 208–211. [Google Scholar] [CrossRef] [PubMed]
- Mishra, P.K.; Mishra, H.; Ekielski, A.; Talegaonkar, S.; Vaidya, B. Zinc oxide nanoparticles: A promising nanomaterial for biomedical applications. Drug Discov. Today 2017, 22, 1825–1834. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Nanda, A. Antimicrobial and antifungal potential of zinc oxide nanoparticles in comparison to conventional zinc oxide particles. J. Chem. Pharm. Res. 2013, 5, 457–463. [Google Scholar]
- Yu, J.; Zhang, W.; Li, Y.; Wang, G.; Yang, L.; Jin, J.; Chen, Q.; Huang, M. Synthesis, characterization, antimicrobial activity and mechanism of a novel hydroxyapatite whisker/nano zinc oxide biomaterial. Biomed. Mater. 2015, 10, 15001. [Google Scholar] [CrossRef]
- Sirelkhatim, A.; Mahmud, S.; Seeni, A. Review on Zinc Oxide Nanoparticles: Antibacterial Activity and Toxicity Mechanism. Nano-Micro Lett. 2015, 7, 219–242. [Google Scholar] [CrossRef] [Green Version]
- Nur, S.; Shameli, K.; Mei-Theng, W.M.; Teow, S.-Y.; Chew, J.; Ismail, N.A. Cytotoxicity and antibacterial activities of plant-mediated synthesized zinc oxide (ZnO) nanoparticles using Punica granatum (pomegranate) fruit peels extract. J. Mol. Struct. J. 2019, 1189, 57–65. [Google Scholar]
- Jayabalan, J.; Mani, G.; Krishnan, N.; Pernabas, J.; Milton, J.; Tae, H. Green biogenic synthesis of zinc oxide nanoparticles using Pseudomonas putida culture and its In vitro antibacterial and anti-biofilm activity. Biocatal. Agric. Biotechnol. 2019, 21, 1–9. [Google Scholar] [CrossRef]
- Bai, D.-P.; Zhang, X.-F.; Zhang, G.-L.; Huang, Y.-F.; Gurunathan, S. Zinc oxide nanoparticles induce apoptosis and autophagy in human ovarian cancer cells. Int. J. Nanomed. 2017, 12, 6521–6535. [Google Scholar] [CrossRef] [Green Version]
- Bala, N.; Saha, S.; Chakraborty, M.; Maiti, M.; Das, S.; Basu, R.; Nandy, P. Green synthesis of zinc oxide nanoparticles using Hibiscus subdariffa leaf extract: Effect of temperature on synthesis, anti-bacterial activity and anti-diabetic activity. RSC Adv. 2014, 5, 4993–5003. [Google Scholar] [CrossRef]
- El-gharbawy, R.M.; Mahmoud, A.; Abu-risha, S.E. ScienceDirect Zinc oxide nanoparticles and a standard antidiabetic drug restore the function and structure of beta cells in Type-2 diabetes. Biomed. Pharmacother. 2016, 84, 810–820. [Google Scholar] [CrossRef]
- Smijs, T.G.; Pavel, S. Titanium dioxide and zinc oxide nanoparticles in sunscreens: Focus on their safety and effectiveness. Nanotechnol. Sci. Appl. 2011, 4, 95–112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kołodziejczak-radzimska, A.; Jesionowski, T. Zinc Oxide—From Synthesis to Application: A Review. Materials 2014, 7, 2833–2881. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agarwal, H.; Nakara, A.; Menon, S.; Shanmugam, V. Eco-friendly synthesis of zinc oxide nanoparticles using Cinnamomum Tamala leaf extract and its promising effect towards the antibacterial activity. J. Drug Deliv. Sci. Technol. 2019, 53, 1773–2247. [Google Scholar] [CrossRef]
- Shankar, S.; Rhim, J. Effect of types of zinc oxide nanoparticles on structural, mechanical and antibacterial properties of poly (lactide)/poly (butylene adipate-co- terephthalate) composite films. Food Packag. Shelf Life J. 2019, 21, 2214–2894. [Google Scholar] [CrossRef]
- Elumalai, K.; Velmurugan, S. Green synthesis, characterization and antimicrobial activities of zinc oxide nanoparticles from the leaf extract of Azadirachta indica (L.). Appl. Surf. Sci. 2015, 345, 329–336. [Google Scholar] [CrossRef]
- Chandra, H.; Patel, D.; Kumari, P.; Jangwan, J.S.; Yadav, S. Phyto-mediated synthesis of zinc oxide nanoparticles of Berberis aristata: Characterization, antioxidant activity and antibacterial activity with special reference to urinary tract pathogens. Mater. Sci. Eng. C 2019, 102, 212–220. [Google Scholar] [CrossRef]
- Raja, A.; Ashokkumar, S.; Marthandam, R.P.; Jayachandiran, J. Eco-friendly preparation of zinc oxide nanoparticles using Tabernaemontana divaricata and its photocatalytic and antimicrobial activity. J. Photochem. Photobiol. 2018, 181, 53–58. [Google Scholar] [CrossRef]
- Hobman, J.L.; Crossman, L.C. Bacterial antimicrobial metal ion resistance. J. Med. Microbiol. 2014, 64, 471–497. [Google Scholar] [CrossRef]
- Microbes, P. Nanoparticles: Alternatives Against Drug-Resistant. Molecules 2016, 21, 836. [Google Scholar]
- Alessandrini, F.; Vennemann, A.; Gschwendtner, S.; Neumann, A.U.; Rothballer, M.; Seher, T.; Wimmer, M.; Kublik, S.; Traidl-Hoffmann, C.; Schloter, M.; et al. Pro-inflammatory versus Immunomodulatory Effects of silver nanoparticles in the lung: The critical role of dose, size and surface modification. Nanomaterials 2017, 7, 300. [Google Scholar] [CrossRef] [Green Version]
- Drake, P.L.; Hazelwood, K.J. Exposure-related health effects of silver and silver compounds: A review. Ann. Occup. Hyg. 2005, 49, 575–585. [Google Scholar] [PubMed] [Green Version]
- Chung, K.F.; Seiffert, J.; Chen, S.; Theodorou, I.G.; Goode, A.E.; Leo, B.F.; McGilvery, C.M.; Hussain, F.; Wiegman, C.; Rossios, C.; et al. Inactivation, clearance, and functional effects of lung-instilled short and long silver nanowires in rats. ACS Nano 2017, 11, 2652–2664. [Google Scholar] [CrossRef] [PubMed]
- Gosens, I.; Cassee, F.R.; Zanella, M.; Manodori, L.; Brunelli, A.; Costa, A.L.; Bokkers, B.G.; de Jong, W.H.; Brown, D.; Hristozov, D.; et al. Organ burden and pulmonary toxicity of nano-sized copper (II) oxide particles after short-term inhalation exposure. Nanotoxicology 2016, 5390, 1084–1095. [Google Scholar] [CrossRef] [PubMed]
- Raun, N.; Stoeger, T.; van den Brule, S.; Saber, A.T.; Beyerle, A.; Vietti, G.; Mortensen, A.; Szarek, J.; Budtz, H.C.; Kermanizadeh, A.; et al. Acute and subacute pulmonary toxicity and mortality in mice after intratracheal instillation of ZnO nanoparticles in three laboratories. Food Chem. Toxicol. J. 2015, 85, 84–95. [Google Scholar]
- Korani, M.; Ghazizadeh, E.; Korani, S.; Hami, Z.; Mohammadi, A. Effects of silver nanoparticles on human health. Eur. J. Nanomed. 2015, 7, 51–62. [Google Scholar] [CrossRef]
- Almansour, M.I.; Alferah, M.A.; Shraideh, Z.A.; Jarrar, B.M. Zinc oxide nanoparticles hepatotoxicity: Histological and histochemical study. Environ. Toxicol. Pharmacol. 2017, 51, 124–130. [Google Scholar] [CrossRef]
- Sarkar, A.; Das, J.; Manna, P.; Sil, P.C. Nano-copper induces oxidative stress and apoptosis in kidney via both extrinsic and intrinsic pathways. Toxicology 2011, 290, 208–217. [Google Scholar] [CrossRef]
- Ibrahim, K.E.; Al-mutary, M.G.; Khan, H.A. Mice Exposed to Gold Nanoparticles. Molecules 2018, 23, 1848. [Google Scholar] [CrossRef] [Green Version]
- Bulcke, F.; Dringen, R.; Scheiber, I.F. Neurotoxicity of Copper. Adv. Neurobiol. 2017, 18, 313–343. [Google Scholar]
- Attia, H.; Nounou, H.; Shalaby, M. Zinc oxide nanoparticles Induced oxidative DNA damage, inflammation and apoptosis in rat’s brain. Toxics 2018, 6, 29. [Google Scholar] [CrossRef] [Green Version]
- Yan, Y.; Huang, G.; Bu, Y.; Lv, Q.; Deng, L.; Zhou, P.; Wang, J.; Yang, Y.; Liu, Y.; Cen, Q.; et al. Zinc oxide nanoparticles cause nephrotoxicity and kidney metabolism alterations in rat’s. J. Environ. Sci. Health A Tox. Hazard. Subst Environ. Eng. 2012, 47, 577–588. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.N.; Zhang, M.; Xia, L.; Zhang, J.; Xing, G. The toxic effects and mechanisms of CuO and ZnO nanoparticles. Materials 2012, 5, 2850–2871. [Google Scholar] [CrossRef] [Green Version]
- Setyawati, M.I.; Tay, C.Y.; Leong, D.T. Mechanistic Investigation of the Biological Effects of SiO2, TiO2, and ZnO Nanoparticles on Intestinal Cells. Small 2015, 11, 3458–3468. [Google Scholar] [CrossRef] [PubMed]
- Hwan, J.; Kim, S.J.; Kim, Y.H.; Noh, J.R.; Gang, G.T.; Chung, B.H.; Song, N.W.; Lee, C.H. Susceptibility to gold nanoparticle-induced hepatotoxicity is enhanced in a mouse model of nonalcoholic steatohepatitis. Toxicology 2012, 294, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Markets, M. Metal Nanoparticles Market by metal (Platinum, Gold, Silver, Iron, Titanium, Copper, Nickel), End-use industry (Pharmaceutical & healthcare, Electrical & electronics, Catalyst, Personal care & cosmetics), and Region - Global Forecast to 2022. Mark. Res. Rep. 2018, 4489142. [Google Scholar]
- Bondarenko, O.; Juganson, K.; Ivask, A.; Kasemets, K.; Mortimer, M.; Kahru, A. Toxicity of Ag, CuO and ZnO nanoparticles to selected environmentally relevant test organisms and mammalian cells in vitro: A critical review. Arch. Toxicol. 2013, 87, 1181–1200. [Google Scholar] [CrossRef] [Green Version]
- Bergin, I.L.; Wilding, L.A.; Morishita, M.; Walacavage, K.; Ault, A.P.; Axson, J.L.; Stark, D.I.; Hashway, S.A.; Capracotta, S.S.; Leroueil, P.R.; et al. Effects of particle size and coating on toxicologic parameters, fecal elimination kinetics and tissue distribution of acutely ingested silver nanoparticles in a mouse model. J. Autism Dev. Disord. 2017, 47, 549–562. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.D.; Wu, H.Y.; Wu, D.; Wang, Y.Y.; Chang, J.H.; Zhai, Z.B.; Meng, A.M.; Liu, P.X.; Zhang, L.A.; Fan, F.Y.; et al. Toxicologic effects of gold nanoparticles in vivo by different administration routes. Int. J. Nanomed. 2010, 5, 771–781. [Google Scholar] [CrossRef] [Green Version]
- Kermanshahi, R.K.; Hojati, V.; Shiravi, A. Zinc Oxide Nanoparticles Absorption Rate in the Heart Tissue of Female Mice. J. Chem. Heal. Risks 2015, 5, 193–198. [Google Scholar]
- Hoseini, S.M.; Hedayati, A.; Taheri Mirghaed, A.; Ghelichpour, M. Toxic effects of copper sulfate and copper nanoparticles on minerals, enzymes, thyroid hormones and protein fractions of plasma and histopathology in common carp Cyprinus carpio. Exp. Toxicol. Pathol. 2016, 68, 493–503. [Google Scholar] [CrossRef]




| Specie | Microorganism | Morphology | Synthesis | Average Size (nm) | Activity | MIB and MIC Values | References |
|---|---|---|---|---|---|---|---|
| AuNPs | |||||||
| Trichoderma hamatum | fungus | spherical, pentagonal and hexagonal | extracellular | 5–30 | P. aeruginosa; Serratia sp.; B. subtilis; S.aureus | Data not shown | [40] |
| Alternanthera bettzickiana | plant extract | spherical | extracellular | 80–120 | S. typhi; P. aeruginosa; E. Aerogenes; S. aureus; B. subtilis; M. luteus | MIC values (expressed in µL of AuNPs): 10 µL B. subtilis 20 µL S. aureus 30 µL M. luteus 40 µL E. aerogenes, S. typhi and P. aeruginosa | [41] |
| Deinococcus radiodurans | bacteria | spherical, triangular and irregular | intra- and extracellular | ~43.75 | E. coli; S. aureus | Data not shown | [42] |
| Pseudomonas veronii AS41G | bacteria | irregular | extracellular | 5–25 | E. coli; S. aureus (+) | Data not shown | [43] |
| Bacillus licheniformis | bacteria | spherical | extracellular | 20–75 (~38) | E. coli; P. aeroginosa; B. subtilis | Values not shown | [44] |
| Fusarium oxysporum f. sp. cubense JT1 | fungus | n.a.0F | extracellular | ~22 | Pseudomonas sp. | Data not shown | [45] |
| Stoechospermum marginatum | algae | spherical to irregular | extracellular | 18.7–93.7 | P. aeruginosa; V. cholerae; V. parahaemoluticus; S. paratyphi; P. vulgaris; S. typhi; K. pneumoniae; K. oxytoca; E. faecalis(+); | AuNPs more effective against E. faecalis > K. pneumoniae. Non-effective against E. coli | [46] |
| Streptomyces viridogens (HM10) | bacteria | spherical and rod | intracellular | 18–20 | E. coli; S. aureus | Data not shown | [47] |
| CuNPs | |||||||
| Shewanella loihica PV-4 | bacteria | spherical | extracellular | 10–16 | E. coli | 100 µg/mL Cu-NPs inhibits 86% of the bacteria | [48] |
| SeNPs | |||||||
| Enterococcus faecalis | bacteria | spherical | extracellular | 29–195 (~99) | S. aureus (no observed activity against P. aeruginosa, B. subtilis and E. coli) | Data not shown | [49] |
| ZnONPs | |||||||
| Glycosmis pentaphylla | plant extract | spherical | extracellular | 32–36 | S. dysenteriae; S. paratyphi; S. aureus; B. cereus | At 100 µg/mL maximum inhibition is observed | [50] |
| Suaeda aegyptiaca | plant extract | spherical | extracellular | ~60 | P. aeruginosa; E. coli; S. aureus; B. subtilis | P. aeruginosa MIC and MBC: 0.19–0.78 mg/mL E. coli MIC: 1.56–12.50 mg/mL MBC: 6.25–12.50 mg/mL S. aureus MIC and MBC: 0.39–1.56 mg/mL B. subtilis MIC: 0.19–0.39 mg/mL MBC: 0.78–12.50 mg/mL | [51] |
| Pichia kudriavzevii | fungus | hexagonal | extracellular | 10–61 | E. coli(+); S. marcescens; B. subtilis(+); S. aureus (+); S. epidermis (++) | Data not shown | [52] |
| Jacaranda mimosifolia | plant extract | spherical | extracellular | 2–4 | E. coli; E. faecium | Data not shown | [53] |
| CuONPs | |||||||
| Cystoseira trinodis | algae | spherical | intracellular | 6–7.8 | E. coli; S. typhi; E. faecalis; S. aureus; B. subtilis; S. faecalis | E. coli and S. aureus MIC: 2.5 μg/mL E. faecalis MIC: 5 μg/mL S. typhimurium MIC: 10 μg/mL | [54] |
| Nanoparticles Efficacy | Physicochemical Characteristics of the Nanoparticles | Production Method | Therapeutic Efficacy | MIB and MIC Values | Reference |
|---|---|---|---|---|---|
| Coliforms bacteria in water and fecal media | Monodispersed spherical AgNPs Average size 20–60 nm ζ-potential (−30 to −15) mV | (Chemical reduction) Green method from extracts of Olea Europaea leaves (Leccino and Carolea), pH 7 or 8 | Antibacterial activity evaluated with total bacteria detection by plate count techniques. Conducted trials of toxicology and cytotoxicity (WST-8 assay, lactate dehydrogenase (LDH) assay, comet assay) | Data not shown | [105] |
| Human pathogenic Gram-positive and Gram-negative bacteria: Staphylococcus aureus, Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa and methicillin-resistant Staphylococcus aureus (MRSA)) | Spherical or rarely polygonal AgNPs Average size 44 nm | (Chemical reduction) Green method AgNPs were synthesized using Picea abies L. stem bark extract, and sing different surfactants | Effective antioxidant activity | Staphylococcus Aureus: (MIC 0.05 mg/mL, MBC 1.57 mg/mL) MRSA: MIC 0.09 mg/mL, MCB 0.25 mg/mL) E. coli MIC: 0.23 mg/mL, MCB 0.31 mg/mL Klebsiella pneumoniae MIC 0.63 mg/mL, MCB: 1.18 mg/mL Pseudomonas aeruginosa MIC 0.16 mg/mL, MCB 0.31 mg/mL | [106] |
| Staphylococcus aureus, Escherichia coli, and Pseudomonas aeruginosa | Spherical shape Average size 430 nm ζ-potential −15.2 mV | (Chemical reduction) Green method. synthesized using terpenes rich extract of Lantana camara L. leaves | Antibacterial aactivity assessed using agar-well diffusion method Conducted trials of Brine shrimp cytotoxicity and antioxidant potential | Data not shown | [107] |
| Staphylococcus aureus, Escherichia coli | Spherical shape Average size between 10–26 nm | (Chemical reduction) Green method. AgNPs were synthesized using Acalypha wilkesiana extract | Agar-well diffusion method was used to evaluate antibacterial activity | Data not shown | [108] |
| Staphylococcus aureus, Escherichia coli (Extended-Spectrum Beta-lactamase (ESBL), and MRSA | Average size 77.68 ± 33.95 nm ζ- potential −34.6 ± 12.7 mV UV–Vis wavelength: 420 nm | (Fungus-mediated Synthesis) Green method. AgNPs were synthesized using Fusarium oxysporum | MIC, antibacterial combination assay Antimicrobial disk susceptibility test and time-kill curve assay used to evaluate antibacterial activity. Also conducted trials of cytotoxicity assay in human red blood cells | MRSA MIC 0.212 mg/mL ESBL MIC 0.106 mg/mL | [109] |
| Escherichia coli, Salmonella typhi, Staphylococcus aureus, Vibrio cholerae, Enterococcus faecalis, Hafnia alvei, Acinetobacter baumannii | Average size: first method: 428.2 ± 197.0 second method: 190.1 ± 102nm Polydispersity index: 0.4 ζ-Potential first method −22.1 ± 0.9 and second method −26.1 ± 1.4 mV, UV–Vis wavelength 412 and 418 nm. | (Chemical reduction) Green method. AgNPs were synthesized using Andrographis paniculate, aqueous, and ethanolic extracts | The zone of inhibition (ZOI), MIC, trypan blue dye exclusion assay, also conducted trials of CellToxTm green assay, LPO assay, hemocompatibility assay and in vivo intravenous delivery of AgNPs and Investigation of liver and kidney function biomarkers | S. typhi MIC 0.125 and 0.250 μg/mL H. alvei MIC 0.125 and 0.125 μg/mL E. faecalis MIC 0.250 and 0.250 μg/mL A. baumannii MIC 0.250 and 0.125 μg/mL E. coli MIC 0.125 and 0.250 μg/mL V. cholera MIC 0.125 and 0.125 μg/mL | [110] |
| Staphylococcus aureus, Bacillus subtilis, and Escherichia coli | Spherical shape Average size 13.2 ± 2.9 nm ζ-potential −16.6 mV UV–Vis wavelength 420 nm | (bacterial-mediated Synthesis) Green method. AgNPs were synthesized using acidophilic actinobacterial SH11 | Disc diffusion, MIC and LIVE/DEAD analyses to evaluate antibacterial activity | S. aureus MIC 40 μg/mL E. coli MIC 70 μg/mL B. subtilis MIC 40 μg/ml | [111] |
| Staphylococcus aureus, MRSA, Escherichia coli, and Pseudomonas aeruginosa | Average size between 6.28–9.84 nm, UV–Vis wavelength range of 391– 403 nm | (Chemical reduction) Method into the lamellar space layer of montmorillonite/chitosan (MMT/Cts) on using NaBH4 | Disc diffusion method to evaluate antibacterial activity | Data not shown | [112] |
| Bacillus subtilis and MRSA | Average size between 10 and 35 nm Polydispersity index 0.2, ζ-potential of −30 mV UV–Vis wavelength of 421 nm | (bacterial-mediated Synthesis) synthesized AgNps from the exopolysaccharide of recently recovered bacterial strain CEES51 | Zone Inhibition Assay, MIC, MBC, Antibiofilm activity determination, colony-forming unit determination to estimate the bacterial susceptibility against AgNPs, intracellular reactive oxygen species production by AgNPs inside bacterial cells | B. subtilis MIC 10 μg/mL, MBC 50 μg/mL MRSA MIC 10 μg/mL, MBC 12.5 μg/ml | [113] |
| Vibrio natriegens | Average size 10 ± 5 nm, 30 ± 5 nm, 60 ± 5 nm, 90 ± 5 nm UV–Vis wavelength ranged from 400–420 nm | (Chemical reduction) Green method. AgNPs of different size were synthesized using casein hydroly- sate as a reducing reagent and sodium hydroxide (NaOH) as a catalyst | MIC, MCB, reactive oxygen species production by AgNps inside bacterial cells | MIC 1.0–11.5 μg/mL MBC 1.1–11.7 μg/ml | [114] |
| Staphylococcus aureus and Escherichia coli | Average size 20 nm UV–Vis wavelength of 390 nm | (Chemical reduction) Green method AgNPs were synthesized using Ultrasound assisted fabrication and fenugreek seed extract as a reducing and capping agent | The agar diffusion method was used for the antimicrobial assay. And the antioxidant activity | Data not shown | [115] |
| Staphylococcus aureus, Shigella dysenteriae, and Salmonella typhi | Average size from 60 to 80 nm | (fungus-mediated Synthesis) Green method. AgNPs were synthesized using Penicillium oxalicum | Antimicrobial potential in liquid broth by optical density measurements, and disc diffusion method | Data not shown | [116] |
| Pseudomonas aeruginosa, Klebsiella pneumoniae, and Escherichia coli MRSA | Average size 10 to 40 nm ζ-potential −29 ± 0.11 mV | (Chemical reduction) Green method. AgNPs were synthesized using lyophilized Seabuckthorn | MIC, MCB, evaluation of P. aeruginosa biofilm, anti-quorum sensing inhibition assay. Also conducted trials of cytotoxicity assay with human dermal fibroblast | P. aeruginosa MIC 2 μg/mL, MBC 4 μg/mL E. coli MIC 4 μg/mL, MBC 8 μg/mL S. aureus MIC 4 μg/mL, MBC 8 μg/mL K. pneumoniae MIC 8 μg/mL, MBC 16 μg/mL | [117] |
| Escherichia coli- 25922 and multidrug-resistant pathogens of Pseudomonas aeruginosa and Acinetobacter baumannii | Spherical shape Average size from 35 to 50 nm, UV–Vis wavelength of 326 nm | (Chemical reduction) Green method. AgNPs were synthesized using Sisymbrium irio extract | The agar diffusion method was used for the antimicrobial assay | Data not shown | [118] |
| Bacillus cereus, Staphylococcus aureus, Micrococcus Luteus, Bacillus Subtilis, Enerococcus Sp. Pseudomonas aeruginosa, Salmonella typhi, Escherichia coli, and Klebsiella pneumonia | Spherical shape Average size 10 nm UV–Vis wavelength of 432 nm | (Chemical reduction) Green method. AgNPs were synthesized using Tamarindus indica natural fruit extract | The agar diffusion method was used for the antimicrobial assay | Data not shown | [119] |
| Escherichia coli, Bacillus subtilis, Pseudomonous fluorescence and Salmonella typhi | Average size 21 nm ζ-potential −32 mV UV–Vis wavelength of 421nm | (Chemical reduction) Green method. AgNPs were synthesized using Ficus religiosa leaf extract | Kirby–Bauer Disk diffusion method and the growth inhibition curve of E. coli was examined after the exposure of AgNPs. Also conducted trials of anti-cancer activity and in vivo toxicity | Data not shown | [120] |
| Nanoparticles Efficacy | Physicochemical Characteristics of the Nanoparticles | Production Method | Therapeutic Efficacy | MIB and MIC Values | Reference |
|---|---|---|---|---|---|
| Staphylococcus aureus Pseudomonas aeruginosa | Data not shown | Data not shown | Ultrasound increased the antibacterial effect of CuO nanoparticles against S. aureus and P. aeruginosa | Data not shown | [129] |
| E. coli S. epiderdimis methicillin 655 resistant S.s aureus (superbug MRSA) isolate Spore-forming Bacillus megatarium | Nanoparticles ranged from 30 to 60 nm | Data not shown | Reaction of copper nanoparticles of 100 nm with B. subtilis showed the highest susceptibility (Z = 0.0734 mL/μg) whereas the reaction of silver nanoparticles of 40 nm with E. coli showed the lowest one (Z = 0.0236 mL/μg) | Data not shown | [130] |
| B. megatarium, S. epidermidis, E. coli MRSA | Average size of 1.36 ± 0.6 nm | CuCl2 as the precursor, D (+) glucose as the reducing agent, soluble starch as the NP stabilizing agent | Cu1X and Cu10X kill B. megatarium, S. epidermidis, E. coli and MRSA | Data not shown | [131] |
| E. coli S. aureus | Spherical morphology and a narrow size distribution with 7 and 14 nm | Mechanochemical method using two different Cu-containing precursors (i.e., CuSO4·5H2O and CuCl2·2H2O) | CuCl2·2H2O derived nanoparticles showed more antibacterial activity than CuSO4.5H2O derived nanoparticles | E. coli MIC:3.75 mg/mL S. aureus MIC: 2.50 mg/mL | [132] |
| Nanoparticles Efficacy | Physicochemical Characteristics of the Nanoparticles | Production Method | Therapeutic Efficacy (Tests Employed) | MIB and MIC Values | Reference |
|---|---|---|---|---|---|
| P. aeruginosa | Average size 18.32 nm | Biological method (extract of A. comosus) | Disc diffusion method | MIC, MIB: 4 μg/mL | [148] |
| S. aureus | MIC: 3.92 μg/mL | ||||
| E. coli | Average size 150 nm | Biological method (extract of M. piperita) | Disc diffusion method | MIB: 12–16 μg/mL MIC: 4 μg/mL | [149] |
| K. pneumoniae | Average size 77.13 and 38.86 (due to extraction method) | Biological method (extract of G. elongate) | Standard agar well diffusion method | MIC: 3.3 μg/mL | [148] |
| S. typhimurium | Average size 25 to 35 nm | Biological method (extract of S. brachiate) | Disc diffusion method | MIC, MIB: 8 μg/mL | [150] |
| K. oxytoca | Average size 18.7 to 93.7 nm | Biological method (extract of S. marginatum) | Agar well diffusion method | Data not shown | [46] |
| E. faecalis | Data not shown | ||||
| V. cholerae | Data not shown | ||||
| S. paratyphii | Data not shown | ||||
| V. parahaemolyticus | Data not shown | ||||
| P. vulgaris | Data not shown | ||||
| B. subtilis | Average size 6 to 40 nm | Chemical method [sodium borohydride (NaBH4) as a reducing agent+ | Enzyme-linked immunosorbent assay (ELISA) | MIC 7.56 μg/mL | [148] |
| Organism and Specie against the Nanoparticles are Effective | Physicochemical Characteristics of the Nanoparticles | Production Method | Therapeutic Efficacy Assessment | MIB and MIC Values | Reference |
|---|---|---|---|---|---|
| Escherichia coli Enterococcus faecalis | Spherical and hexagonal-shaped UV–Vis absorption 32.98 nm (600 °C) UV–Vis absorption 81.84 nm (700 °C) | Green method Biosynthesis of ZnO-NPs using Punica granatum fruit peels extract | Antimicrobial susceptibility test shows effective antibacterial activities against two strains of bacteria Cell proliferation assay shows selective toxicity towards colon cancer cells (HCT116) and proved non-toxic to normal cell (CCD112) | MIC E. coli – 64.53 µg/mL MIC E. faecalis – 22.09 µg/mL | [162] |
| Pseudomonas otitidis Pseudomonas oleovorans Acinetobacter baumannii Bacillus cereus Enterococcus faecalis | Spherical shape Average size 25–45 nm | Green method Biogenic synthesis of ZnO NPs using Pseudomonas putida broth culture | Antibacterial microsomal triglyceride transfer protein assay shows effective antibacterial activities against all strains of bacteria | MIC 10 µg/mL in all bacteria | [163] |
| Staphylococcus aureus | Hexagonal shape UV–Vis absorption 25.57 nm ζ-potential −20.9 mV | Green method Biosynthesis of ZnO-NPs using Cinnamomum Tamala leaf extract | Broth dilution assay, protein leakage analysis, membrane stability analysis, and growth curve analysis show a time and concentration dependent reduction in bacterial growth | MIC 40 µg/mL | [169] |
| Escherichia coli Listeria monocytogenes | Uniform rod-shape Average size 20-30 nm diameter, 100–150 nm length | Green method Synthesis using KOH as a hydrolysing agent | The viable colony count method shows effective antibacterial activities against both strains of bacteria | Data not shown | [170] |
| Escherichia coli | Spherical shape Average size 60–80 nm | Green method Phyto-assisted synthesis of ZnO-NPs using Cassia alata fresh leaves | Growth kinetic assay demonstrated bacteriostatic effect | MIC 20 µg/mL | [157,171] |
| Bacillus cereus Bacillus subtilis Escherichia coli Klebsiella pneumoniae Staphylococcus aureus Serratia marcescens | Needle like shape Average size 90–110 nm | Green method Phyto-assisted synthesis of ZnO-NPs using Berberis aristata leaf extract | Antibacterial activity assay shows effective antibacterial activities against all strains of bacteria and MIC was determinate. The maximum activity was found against Bacillus subtilis | MIC B. cereus – 128 µg/mL B. subtilis – 64 µg/mL E. coli - 256 µg/mL K. pneumoniae – 256 µg/mL S. aureus - 128 µg/mL S. marcescens 64 µg/mL | [172] |
| Staphylococcus aureus Escherichia coli Salmonella paratyphi | Spherical shape Average size 20–50 nm | Green method Biosynthesis of ZnO-NPs using aqueous Tabermaemontana divaricata leaf extract | Antibacterial activity assay shows effective antibacterial activities against all strains of bacteria | Data not shown | [173] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez-López, E.; Gomes, D.; Esteruelas, G.; Bonilla, L.; Lopez-Machado, A.L.; Galindo, R.; Cano, A.; Espina, M.; Ettcheto, M.; Camins, A.; et al. Metal-Based Nanoparticles as Antimicrobial Agents: An Overview. Nanomaterials 2020, 10, 292. https://doi.org/10.3390/nano10020292
Sánchez-López E, Gomes D, Esteruelas G, Bonilla L, Lopez-Machado AL, Galindo R, Cano A, Espina M, Ettcheto M, Camins A, et al. Metal-Based Nanoparticles as Antimicrobial Agents: An Overview. Nanomaterials. 2020; 10(2):292. https://doi.org/10.3390/nano10020292
Chicago/Turabian StyleSánchez-López, Elena, Daniela Gomes, Gerard Esteruelas, Lorena Bonilla, Ana Laura Lopez-Machado, Ruth Galindo, Amanda Cano, Marta Espina, Miren Ettcheto, Antoni Camins, and et al. 2020. "Metal-Based Nanoparticles as Antimicrobial Agents: An Overview" Nanomaterials 10, no. 2: 292. https://doi.org/10.3390/nano10020292










