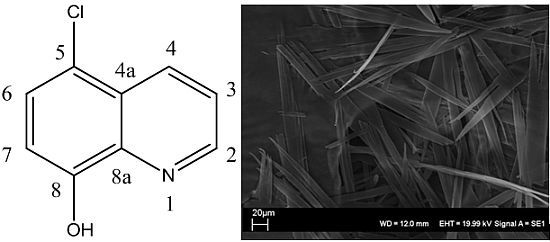
Gelation Behavior of 5-Chloro-8-hydroxyquinoline, an Antituberculosis Agent in Aqueous Alcohol Solutions
Abstract
:1. Introduction
2. Results and Discussion
| Entry | Solvent system | [cloxiquine] gel stability |
|---|---|---|
| 1 | 1:1 EtOH/H2O | 1.0% very stable |
| 2 | 1:1 EtOH/H2O | 0.75% stable |
| 3 | 1:1 EtOH/H2O | 0.5% stable |
| 4 | 1:2 EtOH/H2O | 1.0% stable |
| 5 | 1:2 EtOH/H2O | 0.5% stable |
| 6 | 1:2 EtOH/H2O | 0.2% poor |
| 7 | 1:4 EtOH/H2O | 1.0% stable |
| 8 | 2:1 MeOH/H2O | 1.0% very stable |
| 9 | 2:1 MeOH/H2O | 0.75% stable |
| 10 | 2:1 MeOH/H2O | 0.5% precipitate |
| 11 | 1:2 MeOH/H2O | 1.0% precipitate |
| 12 | 1:4 MeOH/H2O | 1.0% precipitate |
| 13 | 2:1 1-PrOH/H2O | 1.0% precipitate |
| 14 | 1:1 1-PrOH/H2O | 2.0% very stable |
| 15 | 1:1 1-PrOH/H2O | 1.0% stable |
| 16 | 1:1 1-PrOH/H2O | 0.75% poor |
| 17 | 1:1 1-PrOH/H2O | 0.5% precipitate |
| 18 | 1:2 1-PrOH/H2O | 1.0% stable |
| 19 | 1:4 1-PrOH/H2O | 1.0% stable |
| 20 | 1:4 1-PrOH/H2O | 0,5% stable |
| 21 | 1:4 1-PrOH/H2O | 0.2% stable |
| 22 | 1:4 1-PrOH/H2O | 0.1% stable |
| 23 | 1:8 1-PrOH/H2O | 1.0% precipitate |
| 24 | 1:10 1-PrOH/H2O | 1.0% precipitate |
| 25 | 1:1 2-PrOH/H2O | 2.0% stable |
| 26 | 1:1 2-PrOH/H2O | 1.0% stable |
| 27 | 1:1 2-PrOH/H2O | 0.5% stable |
| 28 | 1:1 2-PrOH/H2O | 0.2% poor |
| 29 | 1:2 2-PrOH/H2O | 1% (g) stable |
| 30 | 1:4 2-PrOH/H2O | 1% precipitate |




3. Experimental

4. Conclusions
Acknowledgments
Conflict of Interest
References
- Dye, C. Global epidemiology of tuberculosis. Lancet 2006, 367, 938–940. [Google Scholar] [CrossRef]
- Ahmad, Z.; Makaya, N.H.; Grosset, J. History of drug discovery: Early evaluation studies and lessons learnt from them. Prog. Respir. Res. 2011, 40, 2–9. [Google Scholar] [CrossRef]
- Hongmanee, P.; Rukseree, K.; Buabut, B.; Somsri, B.; Prasit, P. In vitro activities of cloxyquin (5-chloroquinolin-8-ol) against Mycobacterium tuberculosis. Antimicrob. Agents Chemother. 2007, 51, 1105–1106. [Google Scholar] [CrossRef]
- Darby, C.M.; Nathan, C.F. Killing of non-replicating Mycobacterium tuberculosis by 8-hydroxy-quinoline. J. Antimicrob. Chemother. 2010, 65, 1424–1427. [Google Scholar] [CrossRef]
- Enquist, P.-A.; Gylfe, Å.; Hägglund, U.; Lindström, P.; Norberg-Scherman, H.; Sundin, C.; Elofsson, M. Derivatives of 8-hydroxyquinoline-Antibacterial agents that target intra- and extracellular Gram-negative pathogens. Bioorg. Med. Chem. Lett. 2012, 22, 3550–3553. [Google Scholar] [CrossRef]
- Ma, Z.; Moulton, B. A novel polymorph of 5-chloro-8-hydroxyquinoline with improved water solubility and faster dissolution rate. J. Chem. Crystallogr. 2009, 39, 913–918. [Google Scholar] [CrossRef]
- Fernández-Bachiller, M.I.; Pérez, C.; González-Munoz, G.C.; Conde, S.; López, M.G.; Villarroya, M.; García, A.G.; Rodríguez-Franco, M.I. Novel tacrine-8-hydroxyquinoline hybrids as multifunctional agents for the treatment of Alzheimer’s disease, with neuroprotective, cholinergic, antioxidant, and copper-complexing properties. J. Med. Chem. 2010, 53, 4927–4937. [Google Scholar]
- Kharadia, G.J.; Patel, J.R.; Dholakiy, B.Z. Antituberculosis, antifungal and thermal activity of mixed ligand transition metal complexes. Appl. Organomet. Chem. 2010, 24, 821–827. [Google Scholar] [CrossRef]
- Ma, Z.; Moulton, B. Mixed-Ligand Coordination Species: A Promising Approach for “Second-Generation” Drug Development. Cryst. Growth Des. 2007, 7, 196–198. [Google Scholar] [CrossRef]
- Ma, Z.; Moulton, B. Supramolecular medicinal chemistry: Mixed-ligand coordination complexes. Mol. Pharm. 2007, 4, 373–385. [Google Scholar] [CrossRef]
- Pasipanodya, J.G.; Gumbo, T. A new evolutionary and pharmacokinetic-pharmacodynamic scenario for rapid emergence of resistance to single and multiple anti-tuberculosis drugs. Curr. Opin. Pharmacol. 2011, 11, 457–463. [Google Scholar] [CrossRef]
- Sangeetha, N.M.; Maitra, U. Supramolecular gels: Functions and uses. Chem. Soc. Rev. 2005, 34, 821–836. [Google Scholar] [CrossRef]
- Svobodová, H.; Noponen, V.; Kolehmainen, E.; Sievänen, E. Recent advances in steroidal supramolecular gels. RSC Adv. 2012, 2, 7840–7847. [Google Scholar]
- Latosińska, J.N.; Latosińska, M.; Tomczak, M.A.; Seliger, J.; Žagar, V. Supramolecular synthon pattern in solid clioquinol and cloxiquine (APIs of antibacterial, antifungal, antiaging and antituberculosis drugs) studied by 35Cl NQR, 1H-17O and 1H-14N NQDR and DFT/QTAIM. J. Mol. Model. 2011, 17, 1781–1800. [Google Scholar] [CrossRef]
- Svobodová, H.; Nonappa; Lahtinen, M.; Wimmer, Z.; Kolehmainen, E. A steroid-based gelator of A(LS)2 type: Tuning gel properties by metal coordination. Soft Matter 2012, 8, 3840–3847. [Google Scholar]
- Iqbal, S.; Rodríguez-LLansola, F.; Escuder, B.; Miravet, J.F.; Verbruggen, I.; Willemis, R. HRMAS 1H-NMR as a tool for the study of supramolecular gels. Soft Matter 2010, 6, 1875–1878. [Google Scholar] [CrossRef]
- Spectral Database for Organic Compounds. Available online: http://riodb01.ibase.aist.go.jp/sdbs/cgi-bin/cre_index.cgi?lang=eng (accessed on 18 September 2012).
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kolehmainen, E.; Salo, H.; Korpela, J. Gelation Behavior of 5-Chloro-8-hydroxyquinoline, an Antituberculosis Agent in Aqueous Alcohol Solutions. Antibiotics 2012, 1, 17-24. https://doi.org/10.3390/antibiotics1010017
Kolehmainen E, Salo H, Korpela J. Gelation Behavior of 5-Chloro-8-hydroxyquinoline, an Antituberculosis Agent in Aqueous Alcohol Solutions. Antibiotics. 2012; 1(1):17-24. https://doi.org/10.3390/antibiotics1010017
Chicago/Turabian StyleKolehmainen, Erkki, Hannu Salo, and Jukka Korpela. 2012. "Gelation Behavior of 5-Chloro-8-hydroxyquinoline, an Antituberculosis Agent in Aqueous Alcohol Solutions" Antibiotics 1, no. 1: 17-24. https://doi.org/10.3390/antibiotics1010017