Artemisia brevifolia Wall. Ex DC Enhances Cefixime Susceptibility by Reforming Antimicrobial Resistance
Abstract
:1. Introduction
2. Results
2.1. Percentage Yield
2.2. Phytochemical Analysis
2.2.1. Total Flavonoid Content Estimation
2.2.2. Total Phenolic Content Estimation
2.2.3. Secondary Metabolite Estimation
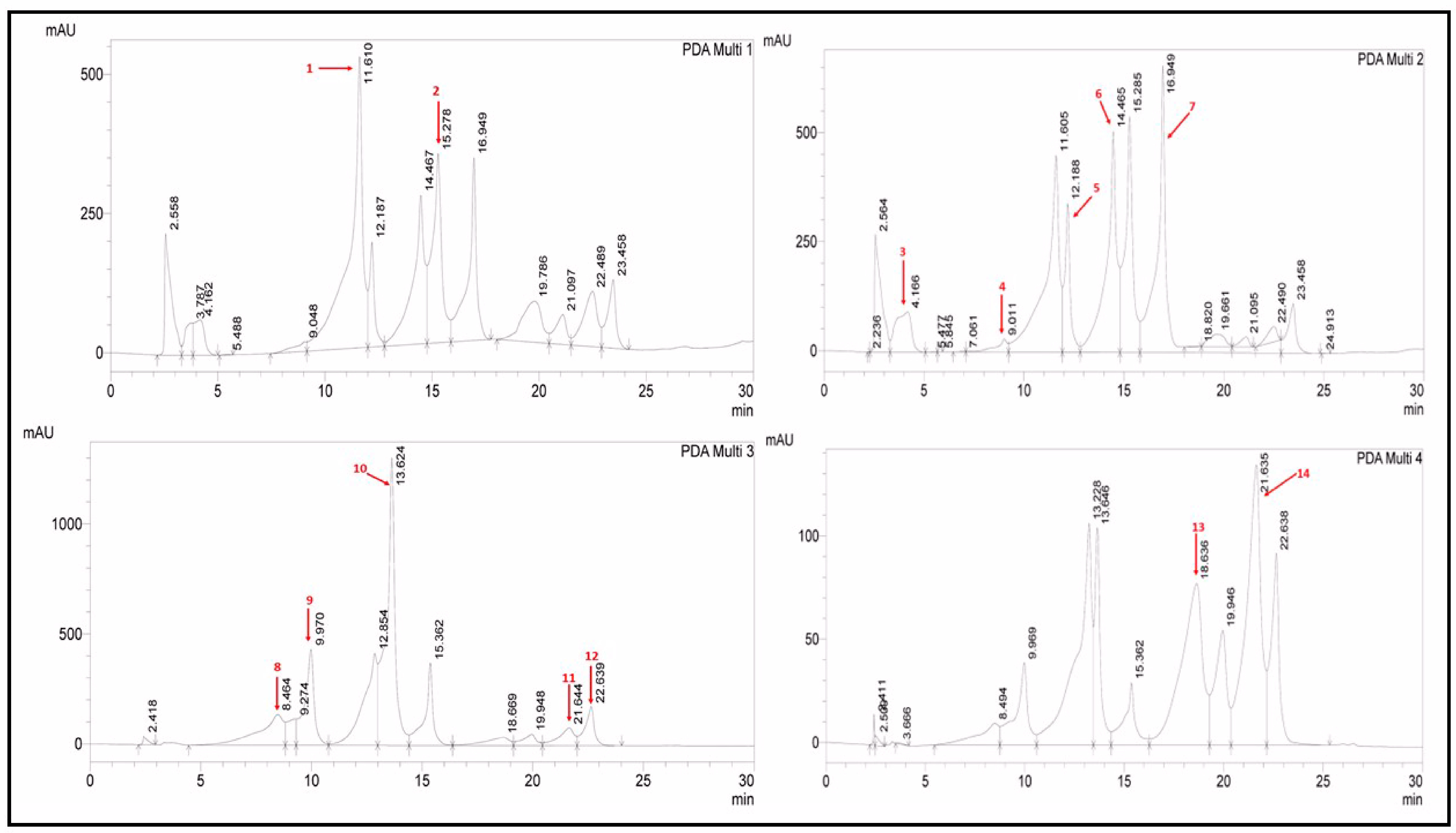
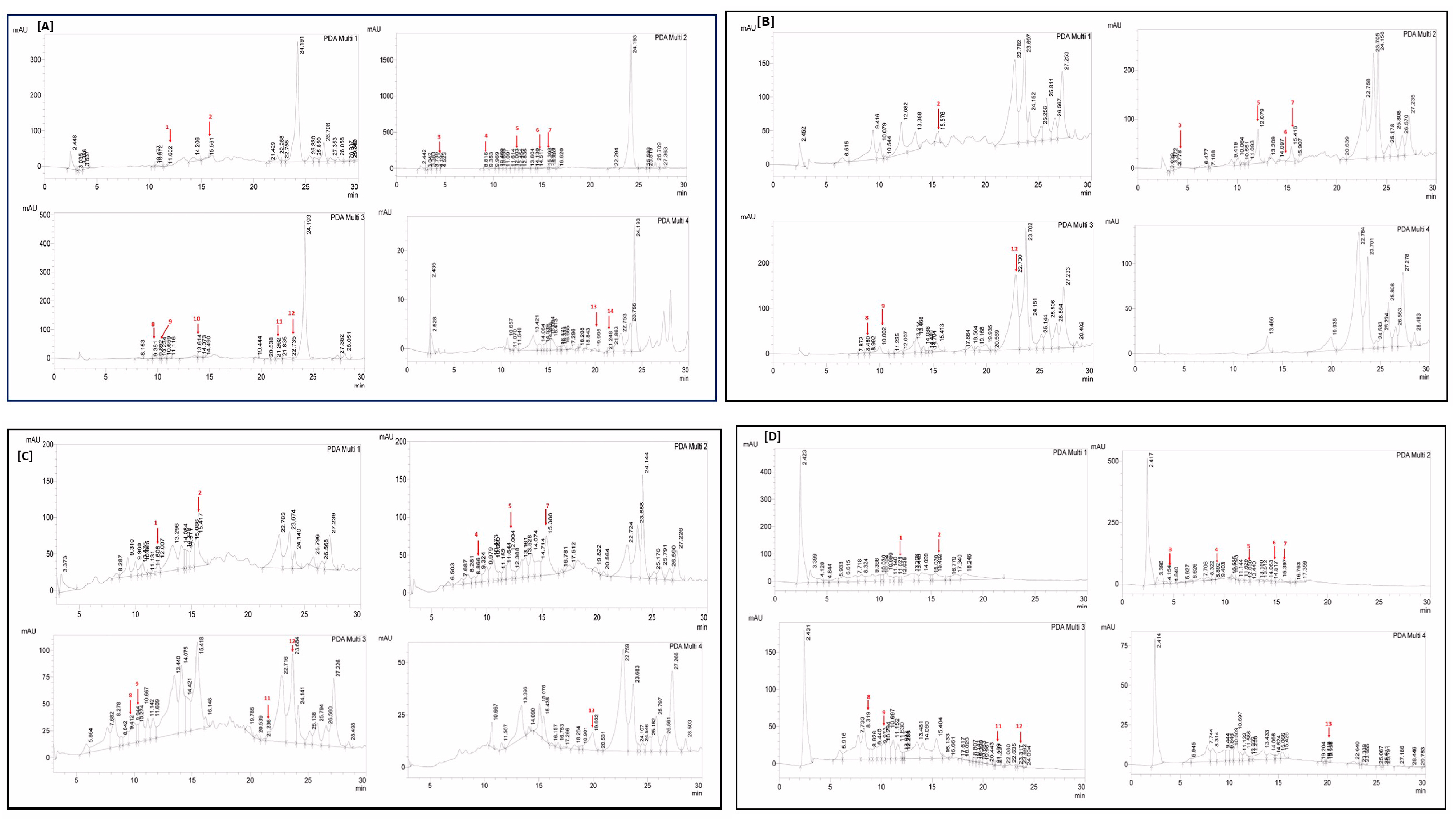
2.2.4. RP-HPLC Analysis: Detection of Polyphenolic Content
2.3. Clinical Bacterial Isolates Were Resistant to Cefixime
| Polyphenol | LOD | LOQ | RT | Λ [20] | Polyphenols (µg/mg of Sample) | |||
|---|---|---|---|---|---|---|---|---|
| NH | EA | M | Aq | |||||
| Vanillic Acid | 0.02 | 0.06 | 11.610 | 257 | - | - | 0.21 ± 0.5 h | 0.13 ± 0.04 f |
| Rutin | 0.18 | 0.54 | 15.278 | 257 | - | 0.55 ± 0.5 c | 2.85 ± 0.025 b | 1.11 ± 0.04 b |
| Gallic Acid | 0.05 | 0.16 | 4.166 | 279 | 0.16 ± 0.3 d | 0.33 ± 0.3 e | 0.00 ± 0.01 | 0.156 ± 0.02 f |
| Catechin | 0.04 | 0.11 | 9.011 | 279 | 0.82 ± 0.09 a | - | 2.43 ± 0.08 c | 1.35 ± 0.08 a |
| Syringic Acid | 0.05 | 0.14 | 12.188 | 279 | 0.24 ± 0.08 c | 2.54 ± 0.04 b | 1.22 ± 0.09 e | 0.28 ± 0.09 d |
| Apocynin | 0.01 | 0.04 | 14.405 | 279 | 0.04 ± 0.04 f | 0.08 ± 0.05 g | - | 0.07 ± 0.05 g |
| Coumaric Acid | 0.08 | 0.23 | 16.949 | 279 | 0.26 ± 0.03 c | 0.42 ± 0.01 d | 1.19 ± 0.09 e | 0.23 ± 0.04 e |
| Gentisic Acid | 0.06 | 0.18 | 8.464 | 325 | - | 0.00 ± 0.01 | 0.43 ± 0.07 g | 0.90 ± 0.01 c |
| Caffeic Acid | 0.03 | 0.09 | 9.970 | 325 | 0.10 ± 0.05 e | 0.21 ± 0.01 f | 0.44 ± 0.02 g | 0.22 ± 0.015 e |
| Ferulic Acid | 0.03 | 0.10 | 13.624 | 325 | 0.14 ± 0.05 de | - | - | - |
| Luteolin | 0.25 | 0.76 | 21.644 | 325 | - | - | 0.97 ± 0.3 f | 0.22 ± 0.01 e |
| Apigenin | 0.12 | 0.35 | 22.639 | 325 | 0.38 ± 0.01 b | 8.65 ± 0.012 a | 3.13 ± 0.015 a | - |
| Quercetin | 0.44 | 1.34 | 18.636 | 368 | 0.01 ± 0.02 g | - | 1.54 ± 0.02 d | - |
| Kaempferol | 0.09 | 0.28 | 21.635 | 368 | - | - | - | - |
| Cumulative | 2.15 ± 0.02 | 12.63 ± 0.01 | 14.41 ± 0.02 | 4.45 ± 0.02 | ||||
A. brevifolia Extracts Possess Mild to Moderate Antibacterial Activity
2.4. A. brevifolia Ethyl Acetate Extract Showed Total Synergism with Cefixime
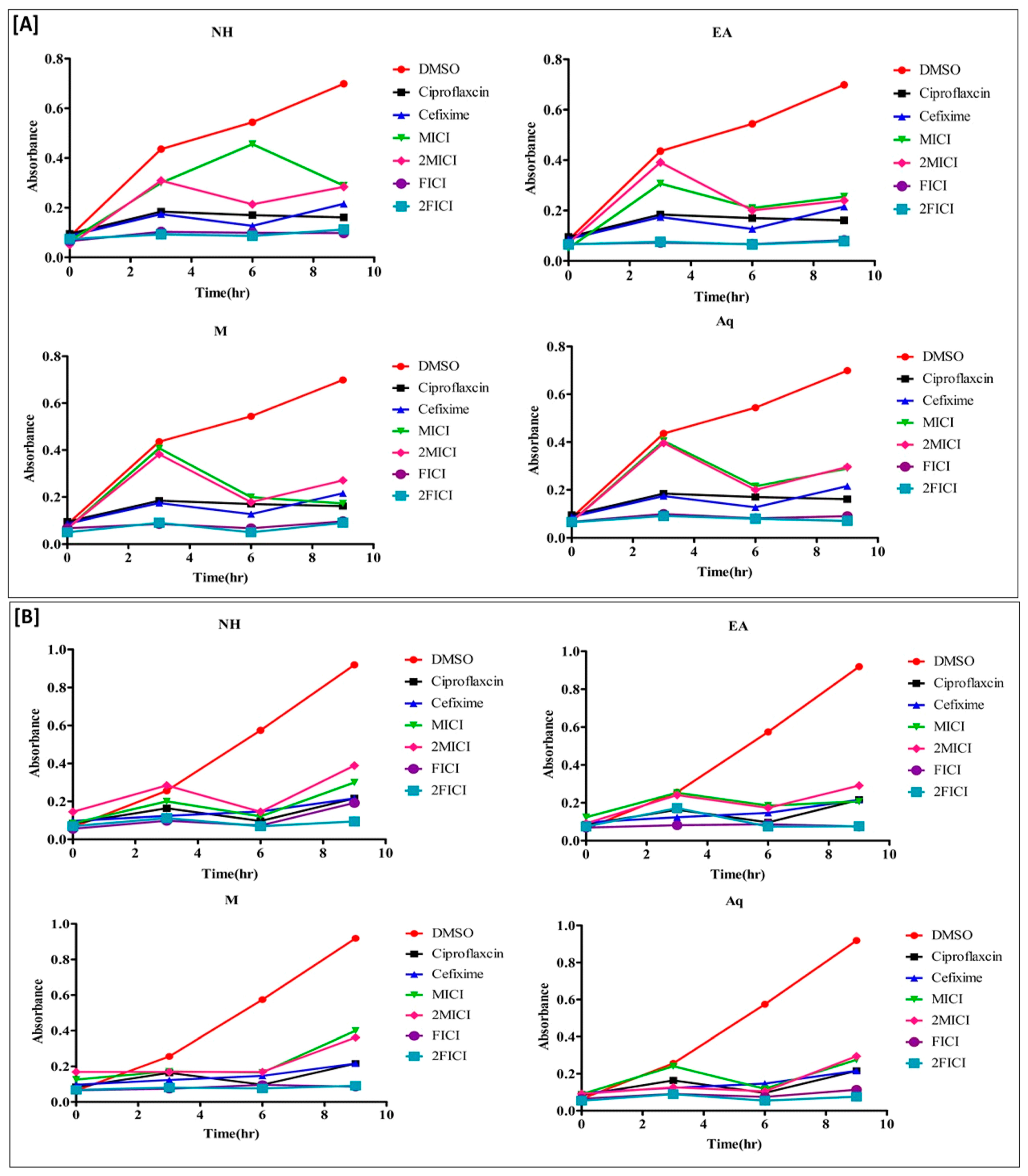
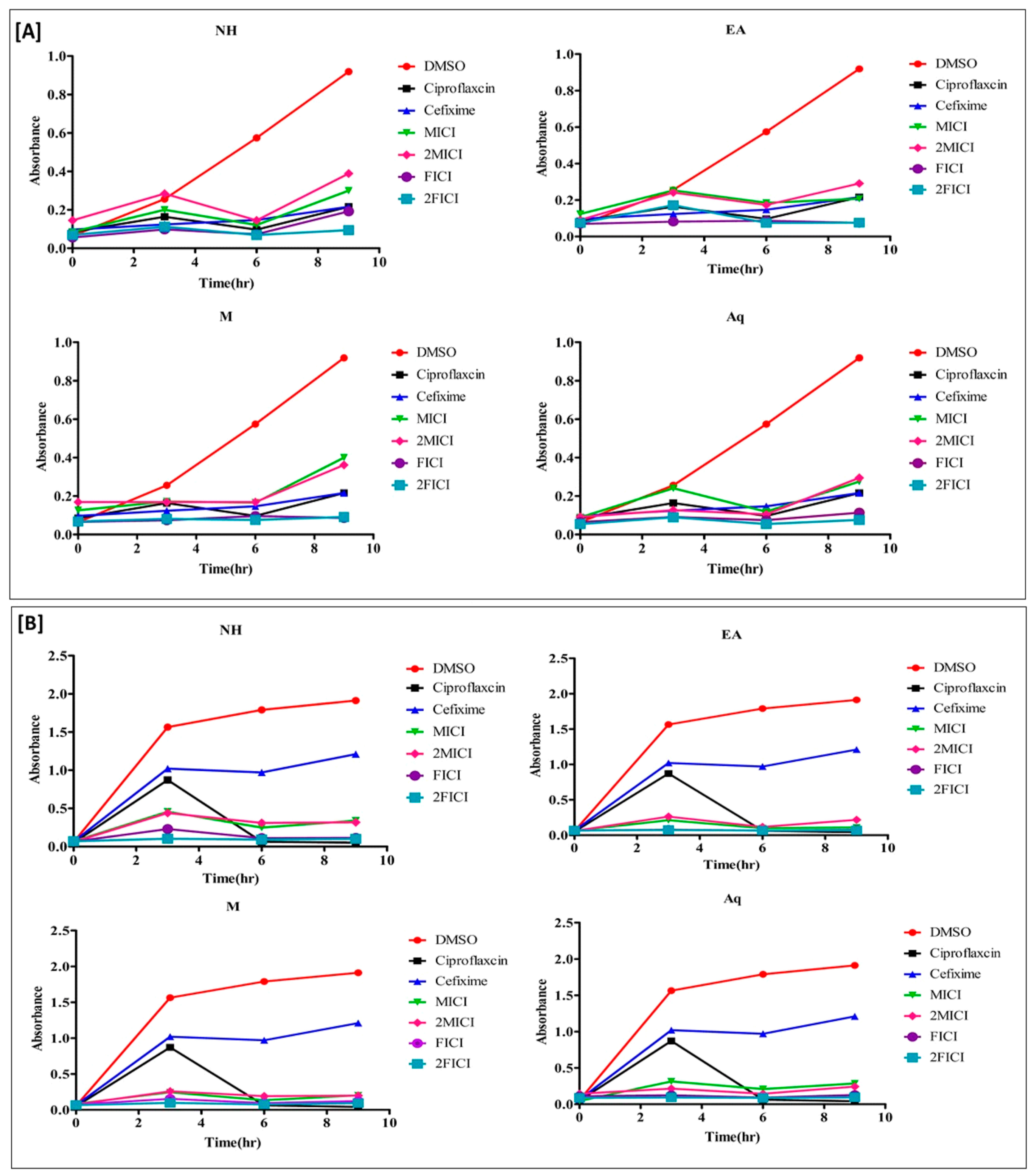
2.5. The Effect of the Extracts Alone and in Combination Is Time Dependent
2.6. A. brevifolia Extracts Reduce Bacterial Protein Content
2.7. Hemolytic Analysis
3. Discussion
- Extraction can be a potentially rate-limiting step when preparing samples for screening bioactive compounds of interest. The efficiency of this step is affected by many factors such as solvent polarity, extraction method, physical characteristics size of sample particles, and period of extraction. These contingency factors were addressed first by selecting four solvents for extraction, n-hexane, methanol, ethyl acetate, and water, depicting variable polarity.
- Second, samples were macerated for 72 h, ensuring sufficient time for solvents to penetrate the fine particles of powdered plant.
- Third, maceration was combined with periodic sonication, aiding the diffusion of solvent and extraction of phytoconstituents from powdered plant material. A significant extraction yield (6.1%) was obtained with Aq extract, which indicated the presence of more polar content.
4. Materials and Methods
4.1. Materials
4.1.1. Chemicals and reagents
4.1.2. Cultures
4.2. Methods
4.2.1. Preparation of Extracts
4.2.2. Total Phenolic Content
4.2.3. Glycoside, Alkaloidal, Tannin, Saponin, and Terpenoid Contents
4.2.4. Antimicrobial Evaluation: Preliminary Resistance Profiling of Antibiotics
4.2.5. RP-HPLC Analysis
LOD and LOQ Determination
Analysis of Polyphenols
4.2.6. Antibacterial Assay
4.2.7. Hemolytic Assay
4.3. Statistical Evaluation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hamad, B. The antibiotics market. Nat. Rev. Drug Discov. 2010, 9, 675–676. [Google Scholar] [CrossRef] [PubMed]
- Bower, C.; Daeschel, M. Resistance responses of microorganisms in food environments. Int. J. Food Microbiol. 1999, 50, 33–44. [Google Scholar] [CrossRef]
- Singh, G.; Katoch, M. Antimicrobial activities and mechanism of action of Cymbopogon khasianus (Munro ex Hackel) Bor essential oil. BMC Complement. Med. Ther. 2020, 20, 331. [Google Scholar] [CrossRef]
- Thomford, N.E.; Senthebane, D.A.; Rowe, A.; Munro, D.; Seele, P.; Maroyi, A.; Dzobo, K. Natural products for drug discovery in the 21st century: Innovations for novel drug discovery. Int. J. Mol. Sci. 2018, 19, 1578. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M.; Snader, K.M. The influence of natural products upon drug discovery. Nat. Prod. Rep. 2000, 17, 215–234. [Google Scholar] [CrossRef] [PubMed]
- Bungãu, S.G.; Popa, V.-C. Between religion and science some aspects concerning illness and healing in antiquity. Transylv. Rev. 2015, 24, 3–18. [Google Scholar]
- Koehn, F.E.; Carter, G.T. The evolving role of natural products in drug discovery. Nat. Rev. Drug Discov. 2005, 4, 206–220. [Google Scholar] [CrossRef]
- Nogueira, J.O.E.; Campolina, G.A.; Batista, L.R.; Alves, E.; Caetano, A.R.S.; Brandão, R.M.; Nelson, D.L.; Cardoso, M.D.G. Mechanism of action of various terpenes and phenylpropanoids against Escherichia coli and Staphylococcus aureus. FEMS Microbiol. Lett. 2021, 368, fnab052. [Google Scholar] [CrossRef]
- Tit, D.M.; Bungau, S.G. Antioxidant activity of essential oils. Antioxidants 2023, 12, 383. [Google Scholar]
- Rufai, M.S. Essential Oils and Their Antimicrobial Activity: A Review. UMYU J. Microbiol. Res. 2017, 2, 87–93. [Google Scholar] [CrossRef]
- Cowan, M.M. Plant products as antimicrobial agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar] [CrossRef] [PubMed]
- Scalbert, A. Antimicrobial properties of tannins. Phytochemistry 1991, 30, 3875–3883. [Google Scholar] [CrossRef]
- Khan, N.; Ahmed, M.; Ahmed, A.; Shaukat, S.; Wahab, M.; Ajaib, M.; Siddiqui, M.F.; Nasir, N. Important medicinal plants of chitral gol National park (cgnp) Pakistan. Pak. J. Bot. 2011, 43, 797–809. [Google Scholar]
- Ashraf, M.; Hayat, M.Q.; Jabeen, S.; Shaheen, N.; Khan, M.A.; Yasmin, G. Artemisia L. species recognized by the local community of northern areas of Pakistan as folk therapeutic plants. J. Med. Plants Res. 2010, 4, 112–119. [Google Scholar]
- Ahsan, H.; Ijaz, M.U.; Ashraf, A.; Ehsan, N.; Noreen, R.; Zafar, S.; Samad, A.; Al-Ghanim, K.A.; Al-Misned, F.; Al-Mulahim, N.; et al. Methanol extract of Artemisia brevifolia as a curative agent against CCl4 induced nephrotoxicity in albino rats. J. King Saud Univ. Sci. 2020, 32, 3072–3078. [Google Scholar] [CrossRef]
- Hayat, M.Q.; Khan, M.A.; Ashraf, M.; Jabeen, S. Ethnobotany of the genus Artemisia L. (Asteraceae) in Pakistan. Ethnobot. Res. Appl. 2009, 7, 147–162. [Google Scholar] [CrossRef]
- Verma, S.; Singh, S. Current and future status of herbal medicines. Vet. World 2008, 1, 347. [Google Scholar] [CrossRef]
- Ashraf, M.; Hayat, M.Q.; Mumtaz, A.S. A study on elemental contents of medicinally important species of Artemisia L. (Asteraceae) found in Pakistan. J. Med. Plants Res. 2010, 4, 2256–2263. [Google Scholar]
- Humphries, R.; Bobenchik, A.M.; Hindler, J.A.; Schuetz, A.N. Overview of changes to the clinical and laboratory standards institute performance standards for antimicrobial susceptibility testing, M100. J. Clin. Microbiol. 2021, 59, e00213–e00221. [Google Scholar] [CrossRef]
- Sarkar, T.; Bharadwaj, K.K.; Salauddin, M.; Pati, S.; Chakraborty, R. Phytochemical characterization, antioxidant, anti-inflammatory, anti-diabetic properties, molecular docking, pharmacokinetic profiling, and network pharmacology analysis of the major phytoconstituents of raw and differently dried mangifera indica (himsagar cultivar): An in vitro and in silico investigations. Appl. Biochem. Biotechnol. 2021, 194, 950–987. [Google Scholar]
- Arabski, M.; Węgierek-Ciuk, A.; Czerwonka, G.; Lankoff, A.; Kaca, W. Effects of Saponins against Clinical E. coli Strains and Eukaryotic Cell Line. J. Biomed. Biotechnol. 2012, 2012, 286216. [Google Scholar] [CrossRef]
- Elahi, M.F.; Guan, G.; Wang, L. Hemocompatibility of surface modified silk fibroin materials: A review. Rev. Adv. Mater. Sci. 2014, 38, 148–159. [Google Scholar]
- Bungau, S.; Tit, D.M.; Behl, T.; Aleya, L.; Zaha, D.C. Aspects of excessive antibiotic consumption and environmental influences correlated with the occurrence of resistance to antimicrobial agents. Curr. Opin. Environ. Sci. Health 2021, 19, 100224. [Google Scholar] [CrossRef]
- Lee, K.-H. Discovery and development of natural product-derived chemotherapeutic agents based on a medicinal chemistry approach. J. Nat. Prod. 2010, 73, 500–516. [Google Scholar] [CrossRef]
- Rout, S.P.; Choudary, K.; Kar, D.; Das, L.; Jain, A. Plants in traditional medicinal system -future source of new drugs. Int. J. Pharm. Pharm. Sci. 2009, 1, 1–23. [Google Scholar]
- Vică, M.L.; Glevitzky, M.; Tit, D.M.; Behl, T.; Heghedűş-Mîndru, R.C.; Zaha, D.C.; Ursu, F.; Popa, M.; Glevitzky, I.; Bungău, S. The antimicrobial activity of honey and propolis extracts from the central region of Romania. Food Biosci. 2021, 41, 101014. [Google Scholar] [CrossRef]
- Bogdan, M.A.; Bungau, S.; Tit, D.M.; Zaha, D.C.; Nechifor, A.C.; Behl, T.; Chambre, D.; Lupitu, A.I.; Copolovici, L.; Copolovici, D.M. Chemical profile, antioxidant capacity, and antimicrobial activity of essential oils extracted from three different varieties (Moldoveanca 4, Vis Magic 10, and Alba 7) of Lavandula angustifolia. Molecules 2021, 26, 4381. [Google Scholar] [CrossRef] [PubMed]
- Tabassum, S.; Ahmed, M.; Mirza, B.; Naeem, M.; Zia, M.; Shanwari, Z.K.; Khan, G.M. Appraisal of phytochemical and in vitro biological attributes of an unexplored folklore: Rhus Punjabensis Stewart. BMC Complement. Altern. Med. 2017, 17, 146. [Google Scholar] [CrossRef]
- Behl, T.; Sharma, A.; Sharma, L.; Sehgal, A.; Zengin, G.; Brata, R.; Fratila, O.; Bungau, S. Exploring the multifaceted therapeutic potential of withaferin A and its derivatives. Biomedicines 2020, 8, 571. [Google Scholar] [CrossRef]
- Souri, E.; Amin, G.; Farsam, H.; Barazandeh, T.M. Screening of antioxidant activity and phenolic content of 24 medicinal plant extracts. DARU J. Pharm. Sci. 2008, 16, 83–87. [Google Scholar]
- Fatima, H.; Khan, K.; Zia, M.; Ur-Rehman, T.; Mirza, B.; Haq, I.U. Extraction optimization of medicinally important metabolites from Datura innoxia Mill.: An in vitro biological and phytochemical investigation. BMC Complement. Altern. Med. 2015, 15, 376. [Google Scholar] [CrossRef] [PubMed]
- Othman, L.; Sleiman, A.; Abdel-Massih, R.M. Antimicrobial activity of polyphenols and alkaloids in middle eastern plants. Front. Microbiol. 2019, 10, 911. [Google Scholar] [CrossRef]
- Soulef, K.; Yahia, A. Anti-Bacterial Effects of Glycosides Extract of Glycyrrhiza Glabra L. from the Region of Djamâa (South-East of Algeria). J. Med. Herbs Ethnomedicine 1970, 3, 22–25. [Google Scholar] [CrossRef]
- Edewor, T.; Owa, S.; Ologan, A.; Akinfemi, F. Quantitative determination of the saponin content and GC-MS study of the medicinal plant Cassytha fiiformis (linn.) leaves. J. Coast. Life Med. 2016, 4, 154–156. [Google Scholar] [CrossRef]
- Foster, T.J. Antibiotic resistance in Staphylococcus aureus. Current status and future prospects. FEMS Microbiol. Rev. 2017, 41, 430–449. [Google Scholar] [CrossRef] [PubMed]
- Kranjec, C.; Kristensen, S.S.; Bartkiewicz, K.T.; Brønner, M.; Cavanagh, J.P.; Srikantam, A.; Mathiesen, G.; Diep, D.B. A bacteriocin-based treatment option for Staphylococcus haemolyticus biofilms. Sci. Rep. 2021, 11, 13909. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Ding, Y.; Yao, K.; Gao, W.; Wang, Y. Antimicrobial resistance analysis of clinical Escherichia coli isolates in neonatal ward. Front. Pediatr. 2021, 9, 670470. [Google Scholar] [CrossRef]
- Hussain, A.; Hayat, M.Q.; Sahreen, S.; ul Ain, Q.; Bokhari, S.A. Pharmacological Promises of Genus Artemisia (Asteraceae): A Review: Pharmacological Promises of Genus Artemisia. Proc. Pak. Acad. Sci. B Life Environ. Sci. 2017, 54, 265–287. [Google Scholar]
- Kazmi, S.T.B.; Naz, I.; Saniya Zahra, S.; Nasar, H.; Fatima, H.; Shuja Farooq, A.; Haq, I.U. Phytochemical analysis and comprehensive evaluation of pharmacological potential of Artemisia brevifolia Wall. ex DC. Saudi Pharm. J. 2022, 30, 793–814. [Google Scholar] [CrossRef]
- Zhou, X.; Seto, S.W.; Chang, D.; Kiat, H.; Razmovski-Naumovski, V.; Chan, K.; Bensoussan, A. Synergistic effects of Chinese herbal medicine: A comprehensive review of methodology and current research. Front. Pharmacol. 2016, 7, 201. [Google Scholar] [CrossRef]
- Aiyegoro, O.A.; Afolayan, A.J.; Okoh, A.I. Synergistic interaction of Helichrysum pedunculatum leaf extracts with antibiotics against wound infection associated bacteria. Biol. Res. 2009, 42, 327–338. [Google Scholar] [CrossRef]
- Ahmad, I.; Aqil, F. In vitro efficacy of bioactive extracts of 15 medicinal plants against ESβL-producing multidrug-resistant enteric bacteria. Microbiol. Res. 2007, 162, 264–275. [Google Scholar] [CrossRef]
- Al-hebshi, N.; Al-haroni, M.; Skaug, N. In vitro antimicrobial and resistance-modifying activities of aqueous crude khat extracts against oral microorganisms. Arch. Oral Biol. 2006, 51, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Haroun, M.F.; Al-Kayali, R.S. Synergistic effect of Thymbra spicata L. extracts with antibiotics against multidrug-resistant Staphylococcus aureus and Klebsiella pneumoniae strains. Iran. J. Basic Med. Sci. 2016, 19, 1193. [Google Scholar] [PubMed]
- Gibbons, S.; Ohlendorf, B.; Johnsen, I. The genus Hypericum—A valuable resource of anti-staphylococcal leads. Fitoterapia 2002, 73, 300–304. [Google Scholar] [CrossRef] [PubMed]
- Sader, H.S.; Streit, J.M.; Fritsche, T.R.; Jones, R.N. Antimicrobial activity of daptomycin against multidrug-resistant Gram-positive strains collected worldwide. Diagn. Microbiol. Infect. Dis. 2004, 50, 201–204. [Google Scholar] [CrossRef]
- Jacqueline, C.; Caillon, J.; Le Mabecque, V.; Miegeville, A.F.; Donnio, P.Y.; Bugnon, D.; Potel, G. In vitro activity of linezolid alone and in combination with gentamicin, vancomycin or rifampicin against methicillin-resistant Staphylococcus aureus by time–kill curve methods. J. Antimicrob. Chemother. 2003, 51, 857–864. [Google Scholar] [CrossRef]
- Sib, T.; Olaniran, A.; Okoh, A. In vitro antibacterial activities of crude extracts of Garcinia kola seeds against wound sepsis associated Staphylococcus strains. J. Med. Plants Res. 2010, 4, 710–716. [Google Scholar]
- Iten, F.; Saller, R.; Abel, G.; Reichling, J. Additive antmicrobial effects of the active components of the essential oil of Thymus vulgaris–chemotype carvacrol. Planta Medica 2009, 75, 1231–1236. [Google Scholar] [CrossRef]
- Mabeku, L.B.K.; Emmanuel, T.; Kouam, J.; Zra, T.; Louis, O.E.J. Synergetic effects of plant extracts and antibiotics on Vibrio cholerae O1 strains isolated from clinical specimens. Int. J. Biol. 2013, 5, 64. [Google Scholar]
- Katzung, B.G.; Masters, S.B.; Trevor, A.J. Basic Clinical Pharmacology; McGraw Hill: New York, NY, USA, 2004. [Google Scholar]
- Qian, W.; Yang, M.; Wang, T.; Sun, Z.; Liu, M.; Zhang, J.; Zeng, Q.; Cai, C.; Li, Y. Antibacterial mechanism of vanillic acid on physiological, morphological, and biofilm properties of carbapenem-resistant enterobacter hormaechei. J. Food Prot. 2020, 83, 576–583. [Google Scholar] [CrossRef]
- Kim, D.-H.; Je, J.-Y. Antimicrobial activity of gallic acid-grafted-chitosan against fish pathogens. J. Carbohydr. Chem. 2015, 34, 163–171. [Google Scholar] [CrossRef]
- Jayaraman, P.; Sakharkar, M.K.; Lim, C.S.; Tang, T.H.; Sakharkar, K.R. Activity and interactions of antibiotic and phytochemical combinations against Pseudomonas aeruginosa in vitro. Int. J. Biol. Sci. 2010, 6, 556. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.-H.; Hu, Z.Q.; Okubo, S.; Hara, Y.; Shimamura, T. Mechanism of synergy between epigallocatechin gallate and β-lactams against methicillin-resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 2001, 45, 1737–1742. [Google Scholar] [CrossRef] [PubMed]
- Zohra, M.; Fawzia, A. Hemolytic activity of different herbal extracts used in Algeria. Int. J. Pharma Sci. Res. 2014, 5, 495–500. [Google Scholar]
- Jasmine, R.; Selvakumar, B.N.; Daisy, P. Investigating the mechanism of action of terpenoids and the effect of interfering substances on an Indian medicinal plant extract demonstrating antibacterial. Int. J. Pharm. Stud. Res. 2011, 5, 1832–1837. [Google Scholar]
- Shaikh, J.; Patil, M. Qualitative tests for preliminary phytochemical screening: An overview. Int. J. Chem. Stud. 2020, 8, 603–608. [Google Scholar] [CrossRef]
- De, S.; Dey, Y.; Ghosh, A. Phytochemical investigation and chromatographic evaluation of the different extracts of tuber of Amorphaphallus paeoniifolius (Araceae). Int. J. Pharm. Biol. Res. 2010, 1, 150–157. [Google Scholar]
- Devmurari, V. Phytochemical screening study and antibacterial evaluation of Symplocos racemosa Roxb. Arch. Appl. Sci. Res. 2010, 2, 354–359. [Google Scholar]
- Indumathi, C.; Durgadevi, G.; Nithyavani, S.; Gayathri, P. Estimation of terpenoid content and its antimicrobial property in Enicostemma litorrale. Int. J. ChemTech Res. 2014, 6, 4264–4267. [Google Scholar]
- Zahra, S.S.; Ahmed, M.; Qasim, M.; Gul, B.; Zia, M.; Mirza, B.; Haq, I.U. Polarity based characterization of biologically active extracts of Ajuga bracteosa Wall. ex Benth. and RP-HPLC analysis. BMC Complement. Altern. Med. 2017, 17, 443. [Google Scholar] [CrossRef] [PubMed]
- Fadli, M.; Saad, A.; Sayadi, S.; Chevalier, J.; Mezrioui, N.E.; Pagès, J.M.; Hassani, L. Antibacterial activity of Thymus maroccanus and Thymus broussonetii essential oils against nosocomial infection–bacteria and their synergistic potential with antibiotics. Phytomedicine 2012, 19, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Das, B.; Dash, S.K.; Mandal, D.; Ghosh, T.; Chattopadhyay, S.; Tripathy, S.; Das, S.; Dey, S.K.; Das, D.; Roy, S. Green synthesized silver nanoparticles destroy multidrug resistant bacteria via reactive oxygen species mediated membrane damage. Arab. J. Chem. 2017, 10, 862–876. [Google Scholar] [CrossRef]
- Nouroozi, R.V.; Noroozi, M.V.; Ahmadizadeh, M. Determination of protein concentration using bradford microplate protein quantification assay. Dis. Diagn. 2015, 4, 11–17. [Google Scholar] [CrossRef]
| Extract | TFC (µgQE/mgE) | TPC (µgGAE/mgE) |
|---|---|---|
| NH | 1.72 ± 0.02 d | 1.52 ± 0.029 d |
| EA | 2.82 ± 0.3 c | 5.2 ± 0.04 c |
| M | 9.93 ± 0.5 a | 15.98 ± 0.1 a |
| Aq | 7.65 ± 0.025 b | 11.25 ± 0.3 b |
| Extract | Glycosides | Alkaloids | Saponins | Tannins | Terpenoids | ||
|---|---|---|---|---|---|---|---|
| Cardiac | Anthraquinone | Coumarin | |||||
| NH | + | − | − | + | − | − | + |
| EA | + | + | − | − | − | − | + |
| M | + | + | + | + | − | + | − |
| Aq | + | + | + | + | + | + | − |
| Antibiotic (20 µg/disc) | Antibacterial Activity (ZOI mm ± SD) | |||
|---|---|---|---|---|
| R. E. coli | R. P. aeruginosa | R. S. haemolyticus | MRSA | |
| Ciprofloxacin | 17 ± 0.6 c | 16 ± 0.1 c | 24 ± 0.4 b | 20 ± 0.12 c |
| Doxycycline | 30 ± 0.1 a | 25 ± 0.76 b | 24 ± 0.01 b | 28 ± 0.11 b |
| Cefixime | - | - | - | - |
| Clarithromycin | 30 ± 0.3 a | 35 ± 0.23 a | 37 ± 0.1 a | 35 ± 0.10 a |
| Lincomycin | 21 ± 0.3 b | 25 ± 0.5 b | 24 ± 0.1 b | 20 ± 0.01 c |
| Pathogen | A. brevifolia Extracts (µg/mL) | Cefixime (µg/mL) | Ciprofloxacin (µg/mL) | |||
|---|---|---|---|---|---|---|
| NH | EA | M | Aq | |||
| R. P. aeruginosa | 80.9 | 66.3 | 66.6 | 100.2 | 40 | 1.11 |
| R. E. coli | 69.56 | 84.53 | 73.5 | 105.7 | 40 | 1.11 |
| R. S. haemolyticus | 79.17 | 79.3 | 79 | 100.4 | 40 | 0.37 |
| MRSA | 86.4 | 67.9 | 82 | 100.2 | 20 | 3.33 |
| Strain | MIC (µg/mL) (Alone) | MIC (µg/mL) Combination | Fold Reduction | Fractional Inhibitory Concentration Index (FICI) | Synergism | |
|---|---|---|---|---|---|---|
| R. P. aeruginosa | NH | 80.9 | 30 | 3 | 0.62 | Partial |
| Cefixime | 40 | 10 | 4 | |||
| EA | 66.3 | 20 | 3 | 0.36 | Total | |
| Cefixime | 40 | 2.5 | 16 | |||
| M | 66.6 | 30 | 2 | 0.58 | Partial | |
| Cefixime | 40 | 5 | 8 | |||
| Aq | 100.2 | 50 | 2 | 0.75 | Partial | |
| Cefixime | 40 | 10 | 4 | |||
| R. E. coli | NH | 69.56 | 40 | 2 | 0.83 | Indifferent |
| Cefixime | 40 | 10 | 4 | |||
| EA | 84.53 | 20 | 4 | 0.36 | Total | |
| Cefixime | 40 | 5 | 8 | |||
| M | 73.5 | 30 | 2 | 0.66 | Partial | |
| Cefixime | 40 | 10 | 4 | |||
| Aq | 105.7 | 25 | 4 | 0.74 | Partial | |
| Cefixime | 40 | 20 | 2 | |||
| R. S. haemolyticus | NH | 79.17 | 20 | 4 | 0.75 | Partial |
| Cefixime | 40 | 20 | 2 | |||
| EA | 79.3 | 15 | 5 | 0.44 | Total | |
| Cefixime | 40 | 10 | 4 | |||
| M | 79 | 20 | 4 | 0.75 | Partial | |
| Cefixime | 40 | 20 | 2 | |||
| Aq | 100.4 | 50 | 2 | 1.00 | Indifferent | |
| Cefixime | 40 | 20 | 2 | |||
| MRSA | NH | 86.4 | 40 | 2 | 0.71 | Partial |
| Cefixime | 20 | 5 | 4 | |||
| EA | 67.9 | 15 | 5 | 0.28 | Total | |
| Cefixime | 20 | 1.25 | 16 | |||
| M | 82 | 40 | 2 | 0.74 | Partial | |
| Cefixime | 20 | 5 | 4 | |||
| Aq | 100.2 | 25 | 4 | 0.75 | Partial | |
| Cefixime | 20 | 10 | 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zafar, A.; Wasti, Y.; Majid, M.; Muntaqua, D.; Bungau, S.G.; Haq, I.u. Artemisia brevifolia Wall. Ex DC Enhances Cefixime Susceptibility by Reforming Antimicrobial Resistance. Antibiotics 2023, 12, 1553. https://doi.org/10.3390/antibiotics12101553
Zafar A, Wasti Y, Majid M, Muntaqua D, Bungau SG, Haq Iu. Artemisia brevifolia Wall. Ex DC Enhances Cefixime Susceptibility by Reforming Antimicrobial Resistance. Antibiotics. 2023; 12(10):1553. https://doi.org/10.3390/antibiotics12101553
Chicago/Turabian StyleZafar, Aroosa, Yusra Wasti, Muhammad Majid, Durdana Muntaqua, Simona Gabriela Bungau, and Ihsan ul Haq. 2023. "Artemisia brevifolia Wall. Ex DC Enhances Cefixime Susceptibility by Reforming Antimicrobial Resistance" Antibiotics 12, no. 10: 1553. https://doi.org/10.3390/antibiotics12101553






