Development of the Zn-ZnO(Nw)@CuMnO2 Heterojunction by Low Temperature Zn Foil Oxidation for Gas Sensor Fabrication
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Fabrication of Zn-ZnO(Nw)@CuMnO2 Heterostructures
2.3. Sensor Testing
2.4. Morpho-Structural, Electrical and Optical Characterization
3. Results and Discussion
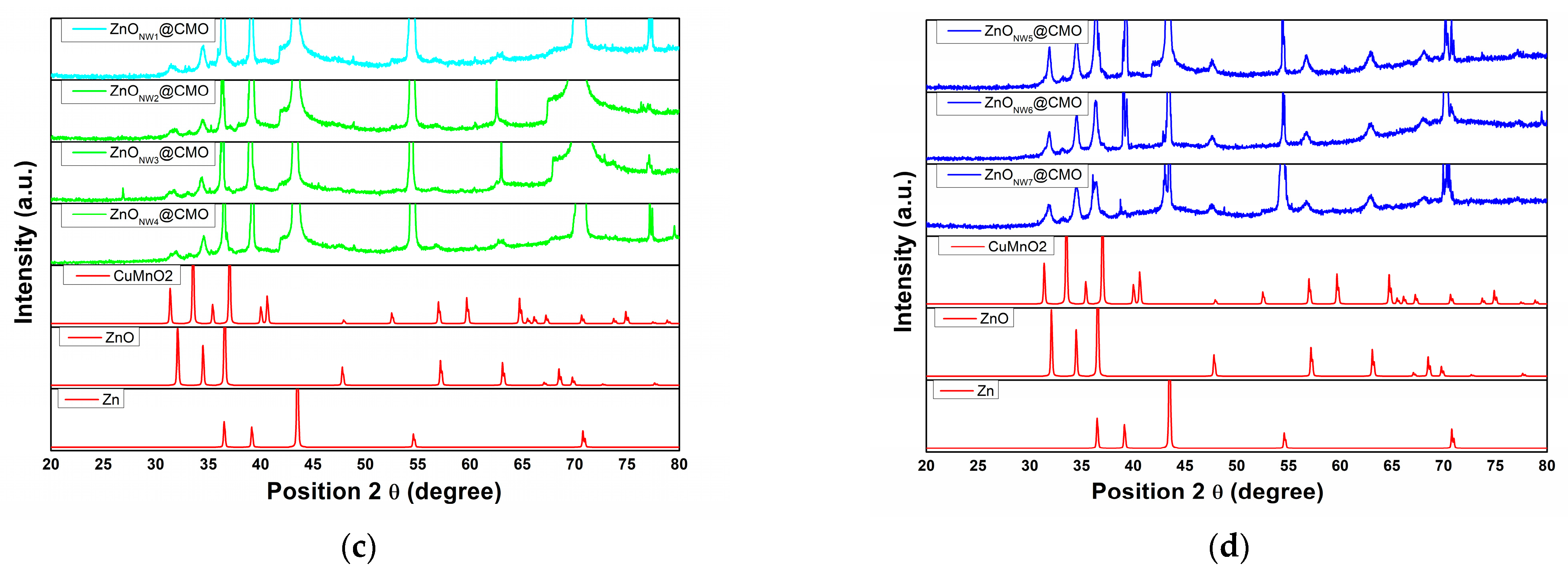
3.1. Structural and Morphological Characteristics
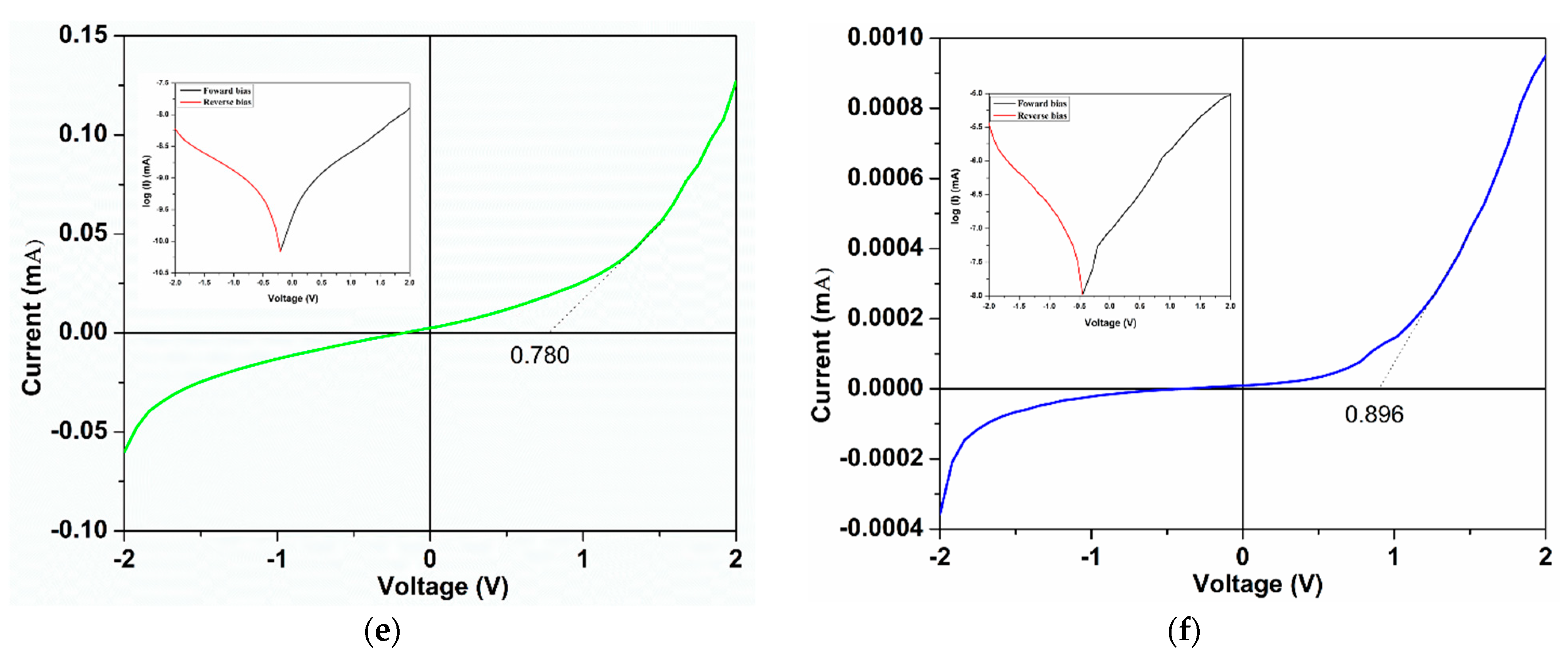
3.2. Optical and Electrical Properties
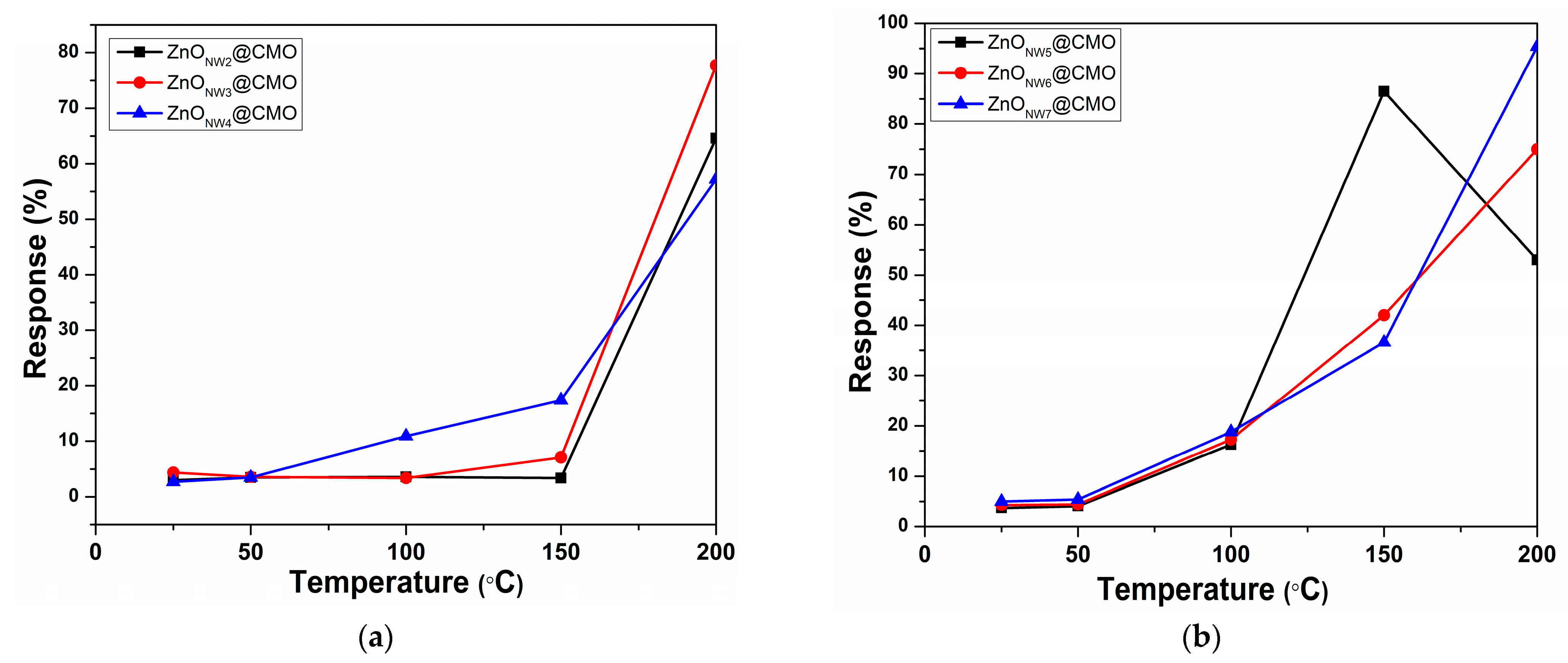
3.3. Sensing Properties of the ZnONw@CMO Sensors
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Srinivasulu, K.; Shiv, G.S. Chemiresistive sensor based on zinc oxide nanoflakes for CO2 detection. ACS Appl. Nano Mater. 2019, 2, 700–706. [Google Scholar] [CrossRef]
- Zhang, C.; Xu, K.; Liu, K.; Xu, J.; Zheng, Z. Metal oxide resistive sensors for carbon dioxide detection. Coord. Chem. Rev. 2022, 472, 214758. [Google Scholar] [CrossRef]
- Malcolm, A.; Wright, S.; Syms, R.R.; Dash, N.; Schwab, M.A.; Finlay, A. Miniature mass spectrometer systems based on a microengineered quadrupole filter. Anal. Chem. 2010, 82, 1751–1758. [Google Scholar] [CrossRef] [PubMed]
- Severinghaus, J.W.; Bradley, A.F. Electrodes for blood pO2 and pCO2 determination. J. Appl. Physiol. 1958, 13, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Bültzingslowen, C.; McEvoy, A.K.; McDonagh, C.; MacCraith, B.D.; Klimant, I.; Krause, C.; Wolfbeis, O.S. Sol–gel based optical carbon dioxide sensor employing dual luminophore referencing for application in food packaging technology. Analyst 2002, 127, 1478–1483. [Google Scholar] [CrossRef] [PubMed]
- Morio, M.; Hyodo, T.; Shimizu, Y.; Egashira, M. Effect of macrostructural control of an auxiliary layer on the CO2 sensing properties of NASICON-based gas sensors. Sens. Actuators B Chem. 2009, 139, 563–569. [Google Scholar] [CrossRef] [Green Version]
- Xu, S.; Li, C.; Li, H.; Li, M.; Qu, C.; Yang, B. Carbon dioxide sensors based on a surface acoustic wave device with a graphene-nickel-l-alanine multilayer film. J. Mater. Chem. 2015, C3, 3882–3890. [Google Scholar] [CrossRef]
- Tanvir, N.B.; Yurchenko, O.; Urban, G. Optimization study for work function-based CO2 sensing using CuO-nanoparticles in respect to humidity and temperature. Procedia Eng. 2015, 120, 667–670. [Google Scholar] [CrossRef] [Green Version]
- Herran, J.; Mandayo, G.G.; Castano, E. Physical behaviour of BaTiO3–CuO thin film under carbon dioxide atmospheres. Sens. Actuators B Chem. 2007, 127, 370–375. [Google Scholar] [CrossRef]
- Yueqiang, L.; Zhuangjun, F. Compositing strategies to enhance the performance of chemiresistive CO2 gas sensors. Mater. Sci. Semicond. Process. 2020, 107, 104820–104841. [Google Scholar] [CrossRef]
- Saruhan, B.; Fomekong, R.L.; Nahirniak, S. Review: Influences of Semiconductor Metal Oxide Properties on Gas Sensing Characteristics. Front. Sens. 2021, 2, 657931. [Google Scholar] [CrossRef]
- Ren, H.; Weng, H.; Zhao, P.; Zuo, R.; Lu, X.; Huang, J. Preparation of porous sea-urchin-like CuO/ZnO composite nanostructure consisting of numerous nanowires with improved gas-sensing performance. Front. Mater. Sci. 2022, 16, 220583. [Google Scholar] [CrossRef]
- Zhang, Y.; Ram, M.K.; Stefanakos, E.K.; Goswami, D.Y. Synthesis, characterization and applications of ZnO nanowires. J. Nanomater. 2012, 2012, 22. [Google Scholar] [CrossRef] [Green Version]
- Wan, H.; Ruda, H.E. A study of the growth mechanism of CVD-grown ZnO nanowires. J. Mater. Sci. Mater. Electron. 2010, 21, 1014–1019. [Google Scholar] [CrossRef]
- Wang, X.D.; Song, J.H.; Wang, Z.L. Nanowire and nanobelt arrays of zinc oxide from synthesis to properties and to novel devices. J. Mater. Chem. 2007, 17, 711–720. [Google Scholar] [CrossRef]
- Chu, D.; Masuda, Y.; Ohji, T.; Kato, K. Formation and photocatalytic application of ZnO nanotubes using aqueous solution. Langmuir 2009, 26, 2811–2815. [Google Scholar] [CrossRef]
- Yu, H.; Zhang, Z.; Han, M.; Hao, X.; Zhu, F. A general low-temperature route for large-scale fabrication of highly oriented ZnO nanorod/nanotube arrays. J. Am. Chem. Soc. 2005, 127, 2378–2379. [Google Scholar] [CrossRef]
- Ding, Y.; Kong, X.Y.; Wang, Z.L. Doping and planar defects in the formation of single-crystal ZnO nanorings. Phys. Rev. B 2004, 70, 235408. [Google Scholar] [CrossRef] [Green Version]
- Tneh, S.S.; Abu Hassan, H.; Saw, K.G.; Yam, F.K.; Hassan, Z. Structural and optical properties of large-scale ZnO nanowires and nanosheets prepared by dry thermal oxidation. Surf. Rev. Lett. 2009, 16, 901–904. [Google Scholar] [CrossRef]
- Li, J.; Peng, H.; Liu, J.; Everitt, H.O. Facile gram-scale growth of single-crystalline nanotetrapod-assembled ZnO through a rapid process. Eur. J. Inorg. Chem. 2008, 20, 3172–3176. [Google Scholar] [CrossRef]
- Park, W.I.; Yi, G.C.; Kim, M.; Pennycook, S.J. ZnO nanoneedles grown vertically on Si substrates by non-catalytic vapor-phase Epitaxy. Adv. Mater. 2002, 14, 1841–1843. [Google Scholar] [CrossRef]
- Dai, Z.R.; Pan, Z.W.; Wang, Z.L. Novel nanostructures of functional oxides synthesized by thermal evaporation. Adv. Funct. Mater. 2003, 13, 9–24. [Google Scholar] [CrossRef]
- Duan, X.F.; Lieber, C.M. Laser-assisted catalytic growth of single crystal GaN nanowires. J. Am. Chem. Soc. 2000, 112, 188–189. [Google Scholar] [CrossRef]
- Li, Y.; Meng, G.W.; Zhang, L.D. Ordered semiconductor ZnO nanowire arrays and their photoluminescence properties. Appl. Phys. Lett. 2000, 76, 126238. [Google Scholar] [CrossRef]
- Ren, S.; Bai, Y.F.; Chen, J.; Deng, S.Z.; Xu, N.S.; Wu, Q.B.; Yang, S.; Phillip, F. Catalyst-free synthesis of ZnO nanowire arrays on zinc substrate by low temperature thermal oxidation. Mater. Lett. 2007, 61, 666–670. [Google Scholar] [CrossRef]
- Yuan, L.; Wang, C.; Cai, R.; Wang, Y.; Zhou, G. Temperature-dependent growth mechanism and microstructure of ZnO nanostructures grown from the thermal oxidation of zinc. J. Cryst. Growth 2014, 390, 101–108. [Google Scholar] [CrossRef]
- Florica, C.; Preda, N.; Costas, A.; Zgura, I.; Enculescu, I. ZnO nanowires grown directly on zinc foils by thermal oxidation in air: Wetting and water adhesion properties. Mat. Lett. 2016, 170, 156–159. [Google Scholar] [CrossRef]
- Galstyan, V.; Moumen, A.; Kumarage, G.W.C.; Comini, E. Progress towards chemical gas sensors: Nanowires and 2D semiconductors. Sens. Actuators B Chem. 2022, 357, 131466. [Google Scholar] [CrossRef]
- Huang, B.; Zhang, Z.; Zhao, C.; Cairang, L.; Bai, J.; Zhang, Y.; Mu, X.; Du, J.; Wang, H.; Pan, X.; et al. Enhanced gas-sensing performance of ZnO@In2O3 core@ shell nanofibers prepared by coaxial electrospinning. Sens. Actuators 2018, 255, 2248–2257. [Google Scholar] [CrossRef]
- Chang, C.-J.; Hung, S.-T.; Lin, C.-K.; Chen, C.-Y.; Kuo, E.-H. Selective growth of ZnO nanorods for gas sensors using ink-jet printing and hydrothermal processes. Thin Solid Films 2010, 519, 1693–1698. [Google Scholar] [CrossRef]
- Chang, C.-J.; Lin, C.-Y.; Chen, J.-K.; Hsu, M.-H. Ce-doped ZnO nanorods based low operation temperature NO2 gas sensors. Ceram. Int. 2014, 40, 10867–10875. [Google Scholar] [CrossRef]
- Han, C.; Li, X.; Shao, C.; Li, X.; Ma, J.; Zhang, X.; Liu, Y. Composition-controllable p-CuO/n-ZnO hollow nanofibers for high-performance H2S detection. Sens. Actuators 2019, 285, 495–503. [Google Scholar] [CrossRef]
- Kang, Y.; Yu, F.; Zhang, L.; Wang, W.; Chen, L.; Li, Y. Review of ZnO-based nanomaterials in gas sensors. Solid State Ion. 2021, 360, 115544–115566. [Google Scholar] [CrossRef]
- Keerthana, S.; Rathnakannan, K. Hierarchical ZnO/CuO nanostructures for room temperature detection of carbon dioxide. J. Alloys Compd. 2022, 897, 162988. [Google Scholar] [CrossRef]
- Hsu, C.L.; Chang, E.C.; Hsueh, H.T.; Liu, Y.H. Solution-synthesized p-type CuMnO2 and n-type ZnO to form the core-shell nanowires for photo and gas sensing. J. Alloys Compd. 2022, 899, 163380. [Google Scholar] [CrossRef]
- Lazau, C.; Poienar, M.; Orha, C.; Ursu, D.; Nicolaescu, M.; Vajda, M.; Bandas, C. Development of a new “n-p” heterojunction based on TiO2 and CuMnO2 synergy materials. Mater. Chem. Phys. 2021, 272, 124999. [Google Scholar] [CrossRef]
- Nicolaescu, M.; Bandas, C.; Orha, C.; Şerban, V.; Lazău, C.; Căprărescu, S. Fabrication of a UV photodetector based on n-TiO2/p-CuMnO2 heterostructures. Coatings 2021, 11, 1380. [Google Scholar] [CrossRef]
- Lazau, C.; Nicolaescu, M.; Orha, C.; Şerban, V.; Bandas, C. Self-Powered Photodetector Based on FTO/n-TiO2/p-CuMnO2 Transparent Thin Films. Materials 2022, 15, 5229. [Google Scholar] [CrossRef]
- Poienar, M.; Banica, R.; Sfirloaga, P.; Ianasi, C.; Mihali, C.V.; Vlazan, P. Microwave-assisted hydrothermal synthesis and catalytic activity study of crednerite-type CuMnO2 materials. Ceram. Int. 2018, 44, 6157–6161. [Google Scholar] [CrossRef]
- Lin, C.-F.; Chao, L.-C. ZnO nanowires prepared by thermal oxidation of metallic zinc films. In Proceedings of the 3rd International Nanoelectronics Confference (INEC), Hong Kong, China, 3–8 January 2010; pp. 1074–1075. [Google Scholar] [CrossRef]
- Khanlary, M.R.; Vahedi, V.; Reyhani, A. Synthesis and characterization of ZnO nanowires by thermal oxidation of Zn thin films at various temperatures. Molecules 2012, 17, 5021–5029. [Google Scholar] [CrossRef] [Green Version]
- Zhang, B.P.; Binh, N.T.; Segawa, Y.; Wakatsuki, K.; Usami, N. Optical properties of ZnO rods formed by metalorganic chemical vapor deposition. Appl. Phys. Lett. 2003, 83, 1635–1637. [Google Scholar] [CrossRef]
- Yang, S.; Lei, G.; Xu, H.; Lan, Z.; Wang, Z.; Gu, H. Metal Oxide Based Heterojunctions for Gas Sensors: A Review. Nanomaterials 2021, 11, 1026. [Google Scholar] [CrossRef] [PubMed]
- Brillson, L.J.; Lu, Y. ZnO Schottky barriers and Ohmic contacts. J. Appl. Phys. 2011, 109, 121301. [Google Scholar] [CrossRef]
- Hossein-Babaei, F.; Lajvardi, M.M.; Alaei-Sheini, N. The energy barrier at noble metal/TiO2 junctions. Appl. Phys. Lett. 2015, 106, 083503. [Google Scholar] [CrossRef]
- Brötzmann, M.; Vetter, U.; Hofsäss, H. BN/ZnO heterojunction diodes with apparently giant ideality factors. J. Appl. Phys. 2009, 106, 063704. [Google Scholar] [CrossRef]
- Harnack, O.; Pacholski, C.; Weller, H.; Yasuda, A.; Wessels, J.M. Rectifying Behavior of Electrically Aligned ZnO Nanorods. Nano Lett. 2003, 3, 1097–1101. [Google Scholar] [CrossRef]
- Lao, C.S.; Liu., J.; Gao, P.; Zhang, L.; Davidovic, D.; Tummala, R.; Wang, Z.L. ZnO Nanobelt/Nanowire Schottky Diodes Formed by Dielectrophoresis Alignment across Au Electrodes. Nano Lett. 2006, 6, 263–266. [Google Scholar] [CrossRef] [Green Version]
- Kumar, M.; Bhat, T.N.; Rajpalke, M.K.; Roul, B.; Kalghatgi, A.T.; Krupanidhi, S.B. Transport and infrared photoresponse properties of InN nanorods/Si heterojunction. Nanoscale Res. Lett. 2011, 6, 609. [Google Scholar] [CrossRef] [Green Version]
- Um, H.-D.; Moiz, S.A.; Park, K.-T.; Jung, J.-Y.; Jee, S.-W.; Ahn, C.H.; Kim, D.C.; Cho, H.K.; Kim, D.-W.; Lee, J.-H. Highly selective spectral response with enhanced responsivity of n-ZnO/p-Si radial heterojunction nanowire photodiodes. Appl. Phys. Lett. 2011, 98, 033102. [Google Scholar] [CrossRef]
- Breitenstein, O.; Altermatt, P.; Ramspeck, K.; Schenk, A. The Origin of ideality factors n > 2 of shunts and surfaces in the dark I-V curves of Si solar cells. In Proceedings of the 21st European Photovoltaic Solar Energy Conference, Dresden, Germany, 4–8 September 2006. [Google Scholar]
- Hsu, K.-C.; Fang, T.-H.; Hsiao, Y.-J.; Wu, P.-C. Response and characteristics of TiO2/perovskite heterojunctions for CO gas sensors. J. Alloys Compd. 2019, 794, 576–584. [Google Scholar] [CrossRef]
- Amarnath, M.; Gurunathan, K. Highly selective CO2 gas sensor using stabilized NiO-In2O3 nanospheres coated reduced graphene oxide sensing electrodes at room temperature. J. Alloys Compd. 2021, 857, 157584. [Google Scholar] [CrossRef]
- González-Garnica, M.; Galdámez-Martínez, A.; Malagón, F.; Ramos, C.D.; Santana, G.; Abolhassani, R.; Kumar Panda, P.; Kaushik, A.; Mishra, Y.K.; Karthik, T.V.K.; et al. One dimensional Au-ZnO hybrid nanostructures based CO2 detection: Growth mechanism and role of the seed layer on sensing performance. Sens. Actuators B Chem. 2021, 337, 129765. [Google Scholar] [CrossRef]
- Jeong, Y.J.; Balamurugan, C.; Lee, D.W. Enhanced CO2 gas-sensing performance of ZnO nanopowder by La loaded during simple hydrothermal method. Sens. Actuators B Chem. 2016, 229, 288–296. [Google Scholar] [CrossRef]
- Kumar, A.; Sanger, A.; Kumar, A.; Chandra, R. Highly sensitive and selective CO gas sensor based on a hydrophobic SnO2/CuO bilayer. RSC Adv. 2016, 6, 47178–47184. [Google Scholar] [CrossRef]
- Hung, S.-T.; Chang, C.-J.; Hsu, C.-H.; Chu, B.H.; Lo, C.F.; Hsu, C.-C.; Pearton, S.J.; Holzworth, M.R.; Whiting, P.G.; Rudawski, N.G.; et al. SnO2 functionalized AlGaN/GaN high electron mobility transistor for hydrogen sensing applications. Int. J. Hydrog. Energy 2012, 37, 13783–13788. [Google Scholar] [CrossRef]
- Chen, Y.; Zhu, C.L.; Xiao, G. Reduced-temperature ethanol sensing characteristics of flower-like ZnO nanorods synthesized by a sonochemical method. Nanotechnology 2006, 17, 4537–4541. [Google Scholar] [CrossRef]








| Sample Support | ZnO Crystallite Size (nm) | ZnO (NWs) Length (nm) | Temperature (°C) | Time (h) | Sensor Code |
|---|---|---|---|---|---|
| Zn-ZnONw1 | 15.40 | - | 300 | 6 | ZnONw1@CMO |
| Zn-ZnONw2 | 15.40 | 54.49 | 350 | 2 | ZnONw2@CMO |
| Zn-ZnONw3 | 16.20 | 46.18 | 4 | ZnONw3@CMO | |
| Zn-ZnONw4 | 17.70 | 62.17 | 6 | ZnONw4@CMO | |
| Zn-ZnONw5 | 22.06 | 91.03 | 400 | 2 | ZnONw5@CMO |
| Zn-ZnONw6 | 23.28 | 134.33 | 4 | ZnONw6@CMO | |
| Zn-ZnONw7 | 22.34 | 85.29 | 6 | ZnONw7@CMO |
| Sensors | VT (V) | IF (mA) | IR (mA) | n | I0 (A) |
|---|---|---|---|---|---|
| ZnONW2@CMO | 0.801 | 8.2 × 10−4 | 8.9 × 10−5 | 4.17 | 8.41 × 10−7 |
| ZnONw3@CMO | 0.290 | 6.7 × 10−2 | 5.6 × 10−2 | 23.03 | 130 × 10−7 |
| ZnONw4@CMO | 0.780 | 12 × 10−2 | 6.0 × 10−2 | 27.81 | 134 × 10−7 |
| ZnONw5@CMO | 0.856 | 4.3 × 10−2 | 1.5 × 10−2 | 11.45 | 0.52 × 10−7 |
| ZnONw6@CMO | 0.359 | 6.6 × 10−3 | 3.1 × 10−3 | 21.16 | 3.72 × 10−7 |
| ZnONw7@CMO | 0.896 | 9.5 × 10−4 | 3.5 × 10−4 | 32.71 | 11.69 × 10−7 |
| Material | Morphologies | CO2 (ppm) | Operating Temperature (℃) | Response (%) | Reference |
|---|---|---|---|---|---|
| Ti/TiO2/LSCNO | Nanotubes | 400 | 200 | 38.41 | [52] |
| rGO/NiO-In2O3 | Nanospheres | 50 | 25 | 40 | [53] |
| Au-ZnO | Nanowires | 200 | 250 | 80 | [54] |
| La-ZnO | Nanorods | 5000 | 400 | 65 | [55] |
| SnO2/CuO | nanofilm | 500 | 180 | 12 | [56] |
| Zn/ZnO/CuMnO2 | Nanowires | 400 | 150 | 85.5 | This work |
| Zn/ZnO/CuMnO2 | Nanowires | 400 | 200 | 95.4 | This work |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nicolaescu, M.; Bandas, C.; Orha, C.; Purcar, V.; Lazau, C. Development of the Zn-ZnO(Nw)@CuMnO2 Heterojunction by Low Temperature Zn Foil Oxidation for Gas Sensor Fabrication. Coatings 2022, 12, 1630. https://doi.org/10.3390/coatings12111630
Nicolaescu M, Bandas C, Orha C, Purcar V, Lazau C. Development of the Zn-ZnO(Nw)@CuMnO2 Heterojunction by Low Temperature Zn Foil Oxidation for Gas Sensor Fabrication. Coatings. 2022; 12(11):1630. https://doi.org/10.3390/coatings12111630
Chicago/Turabian StyleNicolaescu, Mircea, Cornelia Bandas, Corina Orha, Violeta Purcar, and Carmen Lazau. 2022. "Development of the Zn-ZnO(Nw)@CuMnO2 Heterojunction by Low Temperature Zn Foil Oxidation for Gas Sensor Fabrication" Coatings 12, no. 11: 1630. https://doi.org/10.3390/coatings12111630
APA StyleNicolaescu, M., Bandas, C., Orha, C., Purcar, V., & Lazau, C. (2022). Development of the Zn-ZnO(Nw)@CuMnO2 Heterojunction by Low Temperature Zn Foil Oxidation for Gas Sensor Fabrication. Coatings, 12(11), 1630. https://doi.org/10.3390/coatings12111630









