1. Introduction
Lignin, the most abundant natural resource next to cellulose and hemicellulose [
1,
2,
3,
4] contains various functional groups that provide active sites for chemical modification such as polarity adjustment to enhance the compatibility of lignin with other polymeric matrices in lignin/polymer composites [
4,
5] or to improve antioxidant properties [
6,
7,
8,
9,
10,
11]. Furthermore, studies reported lignin-derived encapsulation of various drugs for biomedical and agricultural applications. Richter et al. reported the encapsulation of silver nanoparticles in lignin-coated polymers [
12]. Gregorova et al. studied the encapsulation of lignin nanoparticles in polyethylene films (Björkman lignin from beech wood flour) [
13]. In other studies, the delivery of Resveratrol
® [
14], the controlled release of Avermectin
® [
15], lignin–polyurea microcapsules with anti-photolysis and sustained-release performances [
16], montmorillonite–lignin hybrid hydrogel as super-sorbent for dye removal from wastewater [
17], cellulose–lignin hydrogels and their controlled release of polyphenols [
18], lignin-stimulated protection of polypropylene films and DNA in cells of mice against oxidation damage [
19] have been tested. Gao [
20] and Bshena [
21] studied the antimicrobial activity of various textiles, using lignin incorporated into polyethylene films and applied in the finishing processes. For textiles, there are special requirements such as non-toxicity to the consumer, namely cytotoxicity, allergy or irritation and sensitization. In other recent studies, lignosulfonic acid is reported to exhibit broad-spectrum anti-HIV (human immunodeficiency virus) and anti-HSV (herpes simplex virus) properties [
22,
23]. Thus, Qiu investigated the anti-HIV-1 activity-potential of lignosulfonates as a microbicide to prevent HIV-1 sexual transmission [
23]. Another recently reported study revealed that the antimicrobial capacity of lignin correlates with the phenolic components, specifically the side chain structure and the nature of further functional groups [
24]. Typically, the presence of a double bond in α, β positions of the side chain and a methyl group in the γ position grants the phenolic fragments with the most potency against microorganisms. However, none of the hitherto published studies included the investigation of the antibacterial activity of lignin when included in polymeric matrices.
Unmodified lignin is widely studied as a component for polymer production with a focus on phenol–formaldehyde resins and polyurethanes (PUs) [
25], where lignin is used as polyol substitute due to the high amount of hydroxyl groups resulting in high crosslinking densities and variable mechanical properties [
26,
27,
28]. In previous studies, lignin-derived polyurethane coatings have been prepared using Kraft lignin isolated at room temperature from aqueous media (black liquor) at different pH values [
29]. In addition, their antioxidative activity has been investigated using the Folin–Ciocalteu (FC) assay [
30]. Although lignin contains many functionalities, they are often difficult to access due to rather strong steric hindrance. So far, various procedures have been explored to incorporate more OH groups into the lignin structure including hydroxymethylation, phenolation, demethylation, oxidation and reduction [
31]. These modifications have been studied primarily in conjunction with phenol–formaldehyde (PF) resins or PU research using lignin as a replacement for fossil-based phenols and polyols [
32].
In 2016, Li et al. reported using demethylation to enhance the chemical reactivity at atmospheric pressure to produce fast curing phenolic resins [
33]. Another possibility for lignin demethylation is an enzymatically catalyzed reaction using fungi (i.e., white and brown rot fungi) or bacteria (i.e.,
Pseudomonas,
Sphingomonas). Mainly laccase was investigated, which oxidizes the guaiacyl into catechol units [
34]. Industrially, demethylated lignin is recovered as a byproduct in dimethylsulfoxide (DMSO) production. For this purpose, black liquor is mixed with molten sulfur at about 230 °C. Two methyl groups are transferred from the lignin to the sulfur, forming dimethyl sulfide, which is oxidized to DMSO with nitrogen dioxide. Based on this process, Kraft lignin was demethylated with sulfur at 225 to 235 °C under high pressure and successfully increased its reactivity for the synthesis of phenol–formaldehyde (PF) resins [
34]. Sulfur and halogen compounds are also used as nucleophiles for the chemical demethylation of lignin. For example, Chung and Washburn have demethylated softwood Kraft lignin with hydrobromic acid under the catalytic action of hexadecyltributylphosphonium bromide at 115 °C for 20 h, resulting in an increase in the OH content of 28% [
35]. PU foams synthesized from the modified Kraft lignins showed a higher compressive strength than conventional ones [
36,
37]. Song et al. used the same method for white straw alkali lignin, with results that showed a significant increase in the total hydroxy content of demethylated lignin [
38] when samples were explored for the synthesis of bio-based PF resins by demethylations with sulfur-containing compounds (sulfur,
n-dodecyl mercaptan, sodium hydrogen sulfide and sodium sulfite). Here, soda lignin was heated with the reagent for 1 h at 90 °C. This research aimed to provide a cost-effective and efficient method for the chemical demethylation of lignin. The best results in terms of an increase in OH content and use for PF resins was the sample demethylated with Na
2SO
3. Other authors used Na
2SO
3 for demethylation performed under high-pressure reactors [
39] or under reflux [
15,
40]. Podschun et al. chose a different approach in which organosolv lignin was demethylated under microwave radiation [
41].
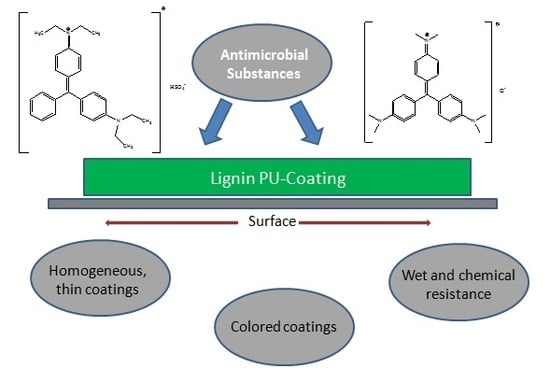
The antimicrobial properties of various dyes, in particular triphenylmethane (TPM) derivatives such as malachite green and crystal violet, have been studied since their first successful application as bioactive additives more than a hundred years ago (
Figure 1,
Table 1).
In 1891, methylene blue, another TPM derivative was discovered by Paul Ehrlich to be efficient in malaria treatment, a few years later followed by the discovery of the antiseptic capacity of brilliant green (BG) [
42,
43,
44]. For many years, malachite green was one of the most frequently used disinfectants in aquaculture due to its fungicidal effects. Due to the discovery of antibiotics and biocide polymers, antimicrobial dyes have not only been displaced in biomedicine but also other applications. Bolous et al. intensively studied the mechanisms of antimicrobial effects [
45,
46]. In detail, it was reported that the evidence to link the antimicrobial properties of TPM dyes, especially brilliant green, to the activity of mechanosensitive ion channel (MIC) of large conductance, which is known to be highly specific and ubiquitous in various bacterial species [
47]. In 2012, Vilela et al. reported a study using methylene blue (MB) and malachite green photosensitizer microbial reduction of
Staphylococcus aureus by synthesizing biofilms with it. The best results showed microbial reduction with 3000 µM of malachite green with a microbial reduction of 1.6–4.0 log
10 [
48]. In oral cavities, biofilm formation is considered to cause resistance to antimicrobial agents. Photodynamic therapies using phenothiazinic photosensitizers first confirmed the antimicrobial effect in biofilms. Malachite green was then compared with the phenothiazinic photosensitizers (methylene blue and toluidine blue) on
Staphylococcus aureus and
Escherichia coli biofilms. Noimark et al. reported the synthesis of modified photobactericidal silicones for medical applications. In detail, crystal violet and/or methylene blue were incorporated into the silicone bulk and gold nanoparticles were coated using a dipping method. The polymers showed good photostability, the photobactericidal activity was determined against
Staphylococcus epidermidis and
Escherichia coli. The results showed that these multi-dye–nanogold-polymers exhibit strong photobactericidal activity both under light and dark conditions [
49]. Bartoszewicz et al. filed a patent claiming lubricious antimicrobial coatings containing silver, pyrrolidone carboxylic acid (PCA) and a TPM dye (malachite green). The coating composition of the invention provides photostability to the silver ions contained therein and is hydrophilic and antimicrobial [
50]. In 2016, Santos et al. comprehensively reviewed various classes of antimicrobial polymers and discussed their bioactivity mechanisms including biocidal activity, antifungal and antibacterial capacity against numerous microorganisms (i.e., gram positive and gram negative bacteria and fungi) [
51].
Table 1 summarizes literature reporting the antimicrobial activity of lignins and triphenylmethane derivatives such as malachite green, brilliant green, methylene blue and crystal violet.
In a recently published study, chitosan/hydroxypropylmethylcellulose (HPMC) composites with varying ratio up to 30% of Kraft lignins (isolated from black liquor and purified via solvent extraction) were prepared and tested against spoilage bacteria that grow at low temperatures. The results revealed the activity against both
B. thermosphacta and
P. fluorescens for samples with 30% lignin. In HPMC/lignin/chitosan films, the 5% addition exhibited activity against both
B. thermosphacta and
P. fluorescens [
52]. Currently, these lignin-derived composites are studied regarding their applications as scaffold component for mesenchymal stem cell differentiation and bone regeneration [
55]. To do so, lignin as feedstock component has to be specified including protocols for quality control using novel chemometric data analysis methods [
56,
57].
In the present study, lignins isolated from black liquor at different pH values were used to explore the potential of these compounds as an antimicrobial component in polyurethane coatings. First, the extraction conditions that favored high lignin yields were optimized. Unmodified and demethylated lignins were used to prepare the lignin–polyurethane (LPU) coatings. The last part of the study aimed to correlate the antimicrobial properties with extraction conditions (i.e., pH value) and molecular structures (unmodified versus demethylated lignins). Furthermore, the influence of additional antimicrobial dyes (brilliant green and crystal violet) on the LPU coating bioactivity, color and morphology was studied.
2. Materials and Methods
2.1. Extraction of Kraft Lignin (KL) and Organosolv Lignin (OL)
The Kraft lignin (KL) was extracted through the acidic precipitation from black liquor according to a procedure reported by Garcia et al. [
58]. First, about 450 mL of black liquor was filtered with a vacuum filter. The filter cake was rejected. Of the filtrate, 400 mL was heated to 50–60 °C. Sulfuric acid (160 mL, 25 vol.%) was added while stirring. The mixture was stirred for another hour at room temperature and then vacuum filtered. The filter cake was washed with distilled water and sulfuric acid (25 vol.%) until the requested pH value was reached (pH 2 to pH 5). Finally, the precipitated lignin was dried in a freeze dryer for 48 h. The organosolv lignin (OL) was isolated according to a procedure recently reported [
10].
2.2. Synthesis of Demethylated Kraft Lignin
For the demethylation, a procedure reported by Li et al. was used and slightly modified [
33]. The sample (1 g), 0.1 g of Na
2SO
3 as the demethylating reagent and 6 g of 2.5 mol NaOH solution were introduced into a 15 mL rolled rim glass on an analytical balance and homogenized. The solution was heated with stirring to 90 or 72 °C and stirred for 1 h at this temperature. After cooling to room temperature (RT), the pH was adjusted to pH 2 by means of 1% HCl. The demethylated lignin precipitated as a brown solid. The suspension was transferred to a 45 mL tube and centrifuged for 10 min at 3000 rpm to separate the demethylated lignin from the aqueous solution. The lignin was washed with distilled water and the pH adjusted to pH 7 with 2.5 molar NaOH solution. It was again centrifuged (for 30 min at 4000 rpm) to separate the aqueous phase from the demethylated lignin. The product was first stored at 40 °C in a drying oven and then freeze-dried at 80 °C and 0.10 mbar. Subsequently, the samples were homogenized and transferred for storage in rolled edge glasses, which were closed with snap lids. Furthermore, the samples were protected against UV radiation.
2.3. Size Exclusion Chromatography
Size exclusion chromatography was used to determine the number-average (
Mn) and weight-average (
Mw) molecular weights of lignins and their polydispersities, analogue to recently reported methods [
29,
30]. A PSS SECurity
2 GPC System was used with tetrahydrofuran as the mobile phase, a run time of 30 min and an injection volume of 100 µL. The system was calibrated using polystyrene standards at different molecular weights.
2.4. Determination of Hydroxyl Groups
The content of hydroxyl groups was determined via two different methods. ISO 14900:2001(E) developed for polyether polyols with steric hindrance was recently reported [
29]. Shortly, each lignin sample was boiled under reflux in 25 mL of acetylation reagent solution with a blank sample simultaneously under the same conditions. After three hours at reflux, the flasks were left to cool down to room temperature. Twenty-five milliliters of sample and blank, respectively, were filled up with water to 100 mL and were titrated with sodium hydroxide (0.5 M). The split up of the acetylated samples and blanks allowed a triple determination via titration. Different amounts of sample and blank were needed. The differences were used to determine the total hydroxyl content.
2.5. Antibacterial Activity of Lignin
The antimicrobial activity of the lignin powders samples was analyzed in a quantitative way by modifying the test for antimicrobial activity and efficacy (JIS Z 2801:2010) of liquid samples [
59]. The JIS is based on a comparison of bacteria counts in saline solution on reference and sample materials after a defined incubation temperature and time.
Staphylococcus aureus (DSM No. 799) was applied as the test organism. The inoculum was prepared in the same way as described above. According to the McFarland-standard the inoculum was adjusted in physiological saline solution with tryptone (Blank, Vörstetten, Germany; VWR International, Darmstadt, Germany) to a concentration of 108 cfu mL
−1. This inoculum suspension was diluted in physiological saline solution with tryptone (Blank, Vörstetten, Germany; VWR International, Darmstadt, Germany) to a final concentration of 105 cfu mL
−1. Lignin powder was added into tubes with 5 mL physiological saline solution with tryptone to a final concentration of 0.1, 0.01 and 0.001 g mL
−1. Each tube was inoculated with 50 µL of the inoculum. The same measurements were done in nutrient broth instead of physiological saline solution. The measurements were carried out in triplicates.
The inoculum (1 mL) was incubated at 37 °C for 24 h in a mixture of 9 mL nutrient broth (Merck KGaA, Darmstadt, Germany) and 1 mL of sample or reference. Afterwards viable counts were determined by counting the colonies on plate-count agar after incubation at 37 °C for 24 h.
The value of antimicrobial activity was calculated by subtracting the logarithmic value of viable counts of the sample from the logarithmic value of reference material after inoculation and incubation:
where as
cgew(reference) = arithmetic mean of bacterial counts of reference 24 h after inoculation, and
cgew(sample) = arithmetic mean of bacterial counts of sample material 24 h after inoculation. According to the JIS Z 2801:2010 a material can be characterized as antimicrobial, if the calculated log
10-reduction is ≥2.0 after 24 h at 37 °C [
59].
2.6. Hemmhoff Test
The antimicrobial activity of the lignin was tested according to the disk diffusion test of the National Committee for Clinical Laboratory Standards (NCCLS) standard method. The disk diffusion test is based on the diffusion of the sampling material in agar. If the bacterium is sensitive to the tested substance, the growth of the bacterium is inhibited and a visible inhibition zone arises. The inhibition zone is the defined area between the punched out area and the beginning of the grown bacterium. If there is no inhibition zone, the bacterium is not sensitive to the tested substance.
Staphylococcus aureus (DSM No. 799) was used as a test organism. The inoculum was prepared by transferring a frozen culture to 10 mL of nutrient broth (Merck KGaA, Darmstadt, Germany). The nutrient broth with the inoculum was incubated at 37 °C for 24 h. According to the McFarland-standard the inoculum was adjusted in physiological saline solution with tryptone (Blank, Vörstetten, Germany; VWR International, Darmstadt, Germany) to a final concentration of 108 cfu mL−1. In each Petri dish (Sarstedt AG, Nümbrecht, Germany) 100 µL of the inoculum was spatulated on Mueller–Hinton agar (VWR International, Darmstadt, Germany) which were impregnated with the different lignins and blank filter papers as references and were put on the inoculated agar plates.
The agar plates were incubated at 37 °C for 24 h. Afterwards, the diameter of the inhibition zone was measured with a digital caliper (Traceable Digital Caliper 6, VWR International, Darmstadt, Germany).
2.7. Synthesis of Lignin-Based Polyurethane Coatings
PEG400 was obtained from Sigma-Aldrich (Steinheim, Germany). 4,4-Diphenylmethane diisocyanate (MDI, for synthesis) was purchased from Merck in Darmstadt and triethylamine (TEA, for synthesis) was received from Carl Roth GmbH in Karlsruhe. All chemicals were used without further purification. PEG400 was mixed with lignin to obtain 1 g of polyol blend. Coatings prepared from lignins isolated at different pH values were produced analogously to the previously described procedure, with the MDI amount adapted to the hydroxyl number of the lignin and the resulting polyol blend. Lignin-based PU coatings were prepared using unmodified and demethylated lignins, respectively, and 4,4-diphenylmethandiisocyanate (MDI). The NCO:OH ratio was 1.7. The calculation was performed according to literature reference [
60,
61]:
where
wMDI,
wL and
wP are the weights (g) of MDI, lignin and polyol, respectively. [NCO]
MDI is the molar content of isocyanate groups in MDI, 8.0 mmol/g for 4,4′-MDI. [OH]
L and [OH]
P are the molar contents of total hydroxyl groups in the lignin and the polyol, respectively. Masses of lignin and polyol were kept constant. Thus, 1 g of lignin was dissolved in 6 mL THF under constant stirring. MDI was added and the mixture was transferred on a polyethylene (PE) transparency and dried for 1 h at room temperature. Finally, the pre-films were cured at 37 °C for 3 h to obtain the final lignin PU films. The synthesis of lignin-modified PU coatings with brilliant green (BG) and crystal violet (CV) followed the same procedure, using 0.8% (
w/
v) of the corresponding triphenylmethane derivative.
Analogously, 1 g of demethylated lignin was dissolved in 6 mL of THF under constant stirring to prepare the LPU coatings. MDI was added and the mixture was transferred onto a PE-transparency and dried for 1 h at room temperature. Finally, the pre-films were cured at 35 °C for 3 h to obtain the final lignin-derived PU films.
2.8. Antimicrobial Activity of the LPU Coatings
The antimicrobial activity of the coatings was analyzed based on the Japanese Industrial Standard (JIS) Z 2801:2010 [
59]. The JIS is based on the comparison of bacteria counts on sample coating/surface and reference material after a defined storage temperature and time (35 °C, 24 h). The reduction of bacteria counts were calculated and represented as log
10-reduction. The log
10-reduction is a measure for the antimicrobial activity and effectiveness of the coatings. According to the JIS a material is called antimicrobial when the log
10-reduction is ≥2 log
10.
Staphylococcus aureus (DSM No. 799) and Listeria monocytogenes were used as test organisms. The inoculum was prepared by transferring a frozen culture to 10 mL of nutrient broth (Merck KGaA, Darmstadt, Germany). The nutrient broth with the inoculum was incubated at 37 °C for 24 h. According to the McFarland-standard the inoculum was adjusted in physiological saline solution with tryptone (Blank, Vörstetten, Germany; VWR International, Darmstadt, Germany) to a final concentration of 108 cfu mL−1. This inoculum suspension was diluted in physiological saline solution with tryptone to a final concentration of 105 cfu mL−1.
The coatings and references were inoculated with 400 µL of the inoculum suspension. To enlarge the contact area of the coatings with the inoculum, the inoculum was covered with a sterile foil (Interscience, Saint-Nom-la-Bretèche, France). The plates were incubated at 37 °C for 24 h. After incubation the inoculated suspension was washed out with 10 mL soybean casein lecithin polysorbate 80 broth (SCDLP) solution (Merck KGaA, Darmstadt, Germany). This served as the first solution stage and was used for further decimal solution series. The bacteria counts were determined by using the drop-plate-technique and counting the colonies on plate-count agar (Merck KGaA, Darmstadt, Germany) after incubation at 37 °C for 24 h.
The value of antimicrobial activity was calculated by subtracting the logarithmic value of viable counts of the sample from the logarithmic value of reference material after inoculation and incubation:
where
cgew (reference) = arithmetic mean of bacterial counts of reference 24 h after inoculation, and
cgew(sample) = arithmetic mean of bacterial counts of sample material 24 h after inoculation. According to the JIS Z 2801:2010 a material can be characterized as antimicrobial, if the calculated log
10-reduction is ≥2.0 after 24 h at 37 °C.
2.9. Thermogravimetric Analysis
TGA measurements were performed with about 10 mg of lignin using a Netzsch (Selb, Germany) TGA 209 F1 with a heating rate of 10 °C min−1 under a nitrogen atmosphere. The temperature ranged from ambient to 800 °C.
2.10. Optical Contact Angle
Static optical contact angle (OCA) measurements were performed on the PU films at room temperature using an OCA device equipped with a charge-coupled device (CCD) photocamera (DataPhysics Instruments, Filderstadt, Germany). A 40 μL volume of distilled water was used to dispense liquid droplets.
2.11. Scanning Electron Microscopy
Scanning electron microscopy (SEM) from ThermoFischer was combined with X-ray analysis (SEM-EDX). Characterization of the texture, phases and the thin LPU layer were determined by SEM-EDX microscopy using an ESEM Quanta FEG 250 FEI with Apollo XL30 EDX (Thermo Fisher Scientific Inc., Huntsville, AL, USA).