Recent Advances in Microneedling-Assisted Cosmetic Applications
Abstract
:1. Introduction
2. Microneedling Devices
2.1. Microneedling in Cosmetic Applications
2.1.1. Treatment of Acne Scars
2.1.2. Treatment of Vitiligo
2.1.3. Treatment of Hair Loss
2.1.4. Treatment of Melasma
2.1.5. Skin Rejuvenation
2.1.6. Treatment of Skin Cancer
3. Fabricated Microneedle Patches
4. Cosmetic-Agent-Loaded Dissolvable Microneedles
4.1. Ascorbic Acid (AA)
4.2. Hyaluronic Acid (HA)
4.3. Retinoids
4.4. Glutathione
4.5. Acetyl-Hexapeptide-3 (AHP-3)
4.6. Niacinamide
4.7. Collagen
4.8. Combinations of Cosmetic Agents
4.9. Other Cosmetic Agents
5. Safety of Microneedling
6. Regulations Related to Microneedling Products
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Huang, Y.; Yu, H.; Wang, L.; Shen, D.; Ni, Z.; Ren, S.; Lu, Y.; Chen, X.; Yang, J.; Hong, Y. Research progress on cosmetic microneedle systems: Preparation, property and application. Eur. Polym. J. 2021, 163, 110942. [Google Scholar] [CrossRef]
- Krasuska, M.; Lavda, A.; Thompson, A.; Millings, A. The role of adult attachment orientation and coping in psychological adjustment to living with skin conditions. Br. J. Dermatol. 2018, 178, 1396–1403. [Google Scholar] [CrossRef] [PubMed]
- Ganceviciene, R.; Liakou, A.I.; Theodoridis, A.; Makrantonaki, E.; Zouboulis, C.C. Skin anti-aging strategies. Derm. Endocrinol. 2012, 4, 308–319. [Google Scholar] [CrossRef] [PubMed]
- Lawson, C.N.; Hollinger, J.; Sethi, S.; Rodney, I.; Sarkar, R.; Dlova, N.; Callender, V.D. Updates in the understanding and treatments of skin & hair disorders in women of color. Int. J. Womens Dermatol. 2017, 3, S21–S37. [Google Scholar] [PubMed]
- Ganesan, P.; Choi, D.-K. Current application of phytocompound-based nanocosmeceuticals for beauty and skin therapy. Int. J. Nanomed. 2016, 11, 1987. [Google Scholar] [CrossRef] [PubMed]
- Abu-Huwaij, R.; Al-Assaf, S.F.; Hamed, R. Recent exploration of nanoemulsions for drugs and cosmeceuticals delivery. J. Cosmet. Dermatol. 2022, 21, 3729–3740. [Google Scholar] [CrossRef]
- Bouwstra, J.A.; Ponec, M. The skin barrier in healthy and diseased state. Biochim. Biophys. Acta 2006, 1758, 2080–2095. [Google Scholar] [CrossRef]
- Hamed, R.; Al Baraghthi, T.; Alkilani, A.Z.; Abu-Huwaij, R. Correlation between rheological properties and in vitro drug release from penetration enhancer-loaded Carbopol® gels. J. Pharm. Innov. 2016, 11, 339–351. [Google Scholar] [CrossRef]
- Alkilani, A.Z.; Nasereddin, J.; Hamed, R.; Nimrawi, S.; Hussein, G.; Abo-Zour, H.; Donnelly, R.F. Beneath the Skin: A Review of Current Trends and Future Prospects of Transdermal Drug Delivery Systems. Pharmaceutics 2022, 14, 1152. [Google Scholar] [CrossRef]
- Bos, J.D.; Meinardi, M.M. The 500 Dalton rule for the skin penetration of chemical compounds and drugs. Exp. Dermatol. 2000, 9, 165–169. [Google Scholar] [CrossRef]
- Andrews, S.N.; Jeong, E.; Prausnitz, M.R. Transdermal delivery of molecules is limited by full epidermis, not just stratum corneum. Pharm. Res. 2013, 30, 1099–1109. [Google Scholar] [CrossRef] [PubMed]
- Hamed, R.; Mahmoud, N.N.; Alnadi, S.H.; Alkilani, A.Z.; Hussein, G. Diclofenac diethylamine nanosystems-loaded bigels for topical delivery: Development, rheological characterization, and release studies. Drug Dev. Ind. Pharm. 2020, 46, 1705–1715. [Google Scholar] [CrossRef] [PubMed]
- McCrudden, M.T.; McAlister, E.; Courtenay, A.J.; González-Vázquez, P.; Raj Singh, T.R.; Donnelly, R.F. Microneedle applications in improving skin appearance. Exp. Dermatol. 2015, 24, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Carrier, A.; Chen, Y.; Lin, S.; Wang, J.; Cui, S.; Zhang, X. Polymeric microneedles for controlled transdermal drug delivery. J. Control. Release 2019, 315, 97–113. [Google Scholar] [CrossRef] [PubMed]
- Bonati, L.M.; Epstein, G.K.; Strugar, T.L. Microneedling in all skin types: A review. J. Drugs Dermatol. 2017, 16, 308–313. [Google Scholar] [PubMed]
- Henry, S.; McAllister, D.V.; Allen, M.G.; Prausnitz, M.R. Microfabricated microneedles: A novel approach to transdermal drug delivery. J. Pharm. Sci. 1998, 87, 922–925. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.Y.; Zhang, J.N.; Chen, B.Z.; Wang, Q.L.; Guo, X.D. A solid polymer microneedle patch pretreatment enhances the permeation of drug molecules into the skin. RSC Adv. 2017, 7, 15408–15415. [Google Scholar] [CrossRef]
- Park, J.-H.; Allen, M.G.; Prausnitz, M.R. Polymer microneedles for controlled-release drug delivery. Pharm. Res. 2006, 23, 1008–1019. [Google Scholar] [CrossRef]
- Ripolin, A.; Quinn, J.; Larrañeta, E.; Vicente-Perez, E.M.; Barry, J.; Donnelly, R.F. Successful application of large microneedle patches by human volunteers. Int. J. Pharm. 2017, 521, 92–101. [Google Scholar] [CrossRef]
- Nguyen, H.X. Safety of microneedles for transdermal drug delivery. J. Pharmacovigil. 2018, 6, e172. [Google Scholar] [CrossRef]
- Gill, H.S.; Denson, D.D.; Burris, B.A.; Prausnitz, M.R. Effect of microneedle design on pain in human volunteers. Clin. J. Pain 2008, 24, 585–594. [Google Scholar] [CrossRef]
- Prausnitz, M.R.; Mikszta, J.A.; Cormier, M.; Andrianov, A.K. Microneedle-based vaccines. Curr. Top. Microbiol. Immunol. 2009, 333, 369–393. [Google Scholar]
- Dharadhar, S.; Majumdar, A.; Dhoble, S.; Patravale, V. Microneedles for transdermal drug delivery: A systematic review. Drug Dev. Ind. Pharm. 2019, 45, 188–201. [Google Scholar] [CrossRef]
- Cheung, K.; Das, D.B. Microneedles for drug delivery: Trends and progress. Drug Deliv. 2016, 23, 2338–2354. [Google Scholar] [CrossRef]
- Iriarte, C.; Awosika, O.; Rengifo-Pardo, M.; Ehrlich, A. Review of applications of microneedling in dermatology. Clin. Cosmet. Investig. Dermatol. 2017, 10, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Larraneta, E.; Lutton, R.E.; Woolfson, A.D.; Donnelly, R.F. Microneedle arrays as transdermal and intradermal drug delivery systems: Materials science, manufacture and commercial development. Mater. Sci. Eng. R Rep. 2016, 104, 1–32. [Google Scholar] [CrossRef]
- Harris, A.G.; Naidoo, C.; Murrell, D.F. Skin needling as a treatment for acne scarring: An up-to-date review of the literature. Int. J. Womens Dermatol. 2015, 1, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Dsouza, L.; Ghate, V.M.; Lewis, S.A. Derma rollers in therapy: The transition from cosmetics to transdermal drug delivery. Biomed. Microdevices 2020, 22, 77. [Google Scholar] [CrossRef] [PubMed]
- El-Domyati, M.; Barakat, M.; Awad, S.; Medhat, W.; El-Fakahany, H.; Farag, H. Microneedling therapy for atrophic acne scars: An objective evaluation. J. Clin. Aesthet. Dermatol. 2015, 8, 36. [Google Scholar]
- Hou, A.; Cohen, B.; Haimovic, A.; Elbuluk, N. Microneedling: A comprehensive review. Dermatol. Surg. 2017, 43, 321–339. [Google Scholar] [CrossRef]
- Alster, T.S.; Graham, P.M. Microneedling: A review and practical guide. Dermatol. Surg. 2018, 44, 397–404. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Yang, H.; Kim, H.; Jung, H.; Jung, H. Novel cosmetic patches for wrinkle improvement: Retinyl retinoate-and ascorbic acid-loaded dissolving microneedles. Int. J. Cosmet. Sci. 2014, 36, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, H.; Kolli, C.S.; Banga, A.K. Characterization of microchannels created by metal microneedles: Formation and closure. AAPS J. 2011, 13, 473–481. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Zhao, D.; Wang, X.; Li, C.; Yang, T.; Du, L.; Wei, Z.; Cheng, Q.; Cao, H.; Liang, Z. Efficient delivery of nucleic acid molecules into skin by combined use of microneedle roller and flexible interdigitated electroporation array. Theranostics 2018, 8, 2361–2376. [Google Scholar] [CrossRef] [PubMed]
- Matilda, I.; Drejøe, J.B. Microneedling in Mature Burn Scars. J. Med. Case Rep. Rev. 2019, 2, 277–281. [Google Scholar]
- Fabbrocini, G.; De Vita, V.; Fardella, N.; Pastore, F.; Annunziata, M.; Mauriello, M.; Monfrecola, A.; Cameli, N. Skin needling to enhance depigmenting serum penetration in the treatment of melasma. Plast. Surg. Int. 2011, 2011, 158241. [Google Scholar] [CrossRef] [PubMed]
- Sabri, A.H.; Cater, Z.; Ogilvie, J.; Scurr, D.J.; Marlow, M.; Segal, J. Characterisation of mechanical insertion of commercial microneedles. J. Drug Deliv. Sci. Technol. 2020, 58, 101766. [Google Scholar] [CrossRef]
- Scott, J.A.; Banga, A.K. Cosmetic devices based on active transdermal technologies. Ther. Deliv. 2015, 6, 1089–1099. [Google Scholar] [CrossRef]
- Majid, I.; Sheikh, G.; September, P. Microneedling and its applications in dermatology. Prime Int. J. Aesthetic Anti-Ageing Med. Healthc. 2014, 4, 44–49. [Google Scholar]
- Doddaballapur, S. Microneedling with dermaroller. J. Cutan. Aesthet. Surg. 2009, 2, 110–111. [Google Scholar] [CrossRef]
- Fabbrocini, G.; Fardella, N.; Monfrecola, A.; Proietti, I.; Innocenzi, D. Acne scarring treatment using skin needling. Clin. Exp. Dermatol. 2009, 34, 874–879. [Google Scholar] [CrossRef] [PubMed]
- Kochhar, J.S.; Tan, J.J.; Kwang, Y.C.; Kang, L. Recent trends in microneedle development & applications in medicine and cosmetics (2013–2018). In Microneedles for Transdermal Drug Delivery; Springer: Cham, Switzerland, 2019; pp. 95–144. [Google Scholar]
- Amer, M.; Farag, F.; Amer, A.; ElKot, R.; Mahmoud, R. Dermapen in the treatment of wrinkles in cigarette smokers and skin aging effectively. J. Cosmet. Dermatol. 2018, 17, 1200–1204. [Google Scholar] [CrossRef] [PubMed]
- McAlister, E.; McCrudden, M.T.; Donnelly, R.F. Microneedles in improving skin appearance and enhanced delivery of cosmeceuticals. In Microneedles for Drug and Vaccine Delivery and Patient Monitoring; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2018. [Google Scholar]
- Zduńska, K.; Kołodziejczak, A.; Rotsztejn, H. Is skin microneedling a good alternative method of various skin defects removal. Dermatol. Ther. 2018, 31, e12714. [Google Scholar] [CrossRef] [PubMed]
- Ebrahim, H.M.; Albalate, W. Efficacy of microneedling combined with tacrolimus versus either one alone for vitiligo treatment. J. Cosmet. Dermatol. 2020, 19, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Soltani-Arabshahi, R.; Wong, J.W.; Duffy, K.L.; Powell, D.L. Facial allergic granulomatous reaction and systemic hypersensitivity associated with microneedle therapy for skin rejuvenation. JAMA Dermatol. 2014, 150, 68–72. [Google Scholar] [CrossRef] [PubMed]
- Lewis, W. Is Microneedling Really the Next Big Thing? Practice 2014, 7, 24–28. [Google Scholar]
- Fucci-da-Costa, A.P.C.; Camasmie, H.R. Drug delivery after microneedling: Report of an adverse reaction. Dermatol. Surg. 2018, 44, 593–594. [Google Scholar] [CrossRef] [PubMed]
- Bahuguna, A. Micro needling-facts and fictions. Asian J. Med. Sci. 2013, 4, 1–4. [Google Scholar] [CrossRef]
- Agarwal, M. Dermaroller: The Transepidermal Delivery System. In Aesthetic Medicine; Springer: Berlin/Heidelberg, Germany, 2012; pp. 273–275. [Google Scholar]
- Bin Sabri, A.H. Application of Microneedles for the Treatment of Nodular Basal Cell Carcinoma. Ph.D. Thesis, University of Nottingham, Nottingham, UK, 2021. [Google Scholar]
- Singh, A.; Yadav, S. Microneedling: Advances and widening horizons. Indian Dermatol. Online J. 2016, 7, 244–254. [Google Scholar]
- Koo, H. Effectiveness of Dyes as Skin Biopsy Markers; North Carolina State University: Raleigh, NC, USA, 2018; p. 165. [Google Scholar]
- Sharad, J. Combination of microneedling and glycolic acid peels for the treatment of acne scars in dark skin. J. Cosmet. Dermatol. 2011, 10, 317–323. [Google Scholar] [CrossRef]
- Saadawi, A.N.; Esawy, A.M.; Kandeel, A.H.; El-Sayed, W. Microneedling by dermapen and glycolic acid peel for the treatment of acne scars: Comparative study. J. Cosmet. Dermatol. 2019, 18, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Garg, S.; Baveja, S. Combination therapy in the management of atrophic acne scars. J. Cutan. Aesthet. Surg. 2014, 7, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Costa, I.M.; Costa, M.C. Microneedling for varicella scars in a dark-skinned teenager. Dermatol. Surg. 2014, 40, 333–334. [Google Scholar] [CrossRef] [PubMed]
- Ali, B.; ElMahdy, N.; Elfar, N.N. Microneedling (Dermapen) and Jessner’s solution peeling in treatment of atrophic acne scars: A comparative randomized clinical study. J. Cosmet. Laser Ther. 2019, 21, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-K.; Jang, Y.-H.; Son, Y.-H.; Lee, C.-S.; Bae, J.-Y.; Park, J.-M. Management of Hypertrophic Scar after Burn Wound Using Microneedling Procedure (Dermastamp (R)). J. Korean. Burn. Soc. 2009, 121–124. [Google Scholar] [CrossRef]
- Claytor, R.B.; Sheck, C.G.; Chopra, V. Microneedling outcomes in early postsurgical scars. Plast. Reconst. Surg. 2022, 150, 557e–561e. [Google Scholar] [CrossRef] [PubMed]
- Ramaut, L.; Hoeksema, H.; Pirayesh, A.; Stillaert, F.; Monstrey, S. Microneedling: Where do we stand now? A systematic review of the literature. J. Plast. Reconstr. Aesthet. Surg. 2018, 71, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Juhasz, M.L.; Cohen, J.L. Microneedling for the treatment of scars: An update for clinicians. Clin. Cosmet. Investig. Dermatol. 2020, 13, 997–1003. [Google Scholar] [CrossRef]
- Lima, E.; Lima, M.; Lima, E.; Lima, M. Correcting Post-surgical Scar Using PCI. In Percutaneous Collagen Induction with Microneedling: A Step-by-Step Clinical Guide; Springer: Cham, Switzerland, 2021; pp. 133–139. [Google Scholar]
- Salloum, A.; Bazzi, N.; Maalouf, D.; Habre, M. Microneedling in vitiligo: A systematic review. Dermatol. Ther. 2020, 33, e14297. [Google Scholar] [CrossRef]
- Dillon, A.B.; Sideris, A.; Hadi, A.; Elbuluk, N. Advances in vitiligo: An update on medical and surgical treatments. J. Clin. Aesthet. Dermatol. 2017, 10, 15–28. [Google Scholar]
- Lima, E.V.A.; Lima, M.M.D.A.; Miot, H.A. Induction of pigmentation through microneedling in stable localized vitiligo patients. Dermatol. Surg. 2020, 46, 434–435. [Google Scholar] [CrossRef] [PubMed]
- Jha, A.K.; Sonthalia, S. 5-Fluorouracil as an adjuvant therapy along with microneedling in vitiligo. J. Am. Acad. Dermatol. 2019, 80, e75–e76. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Bharti, R.; Agarwal, S. Microneedling with Dermaroller 192 needles along with 5-fluorouracil solution in the treatment of stable vitiligo. J. Am. Acad. Dermatol. 2019, 81, e67–e69. [Google Scholar] [CrossRef] [PubMed]
- Lei, T.-C.; Hearing, V.J. Deciphering skin re-pigmentation patterns in vitiligo: An update on the cellular and molecular events involved. Chin. Med. J. 2020, 133, 1231–1238. [Google Scholar] [CrossRef] [PubMed]
- Bailey, A.J.-M.; Li, H.O.-Y.; Zheng, D.; Glassman, S.J.; Tan, M.G. Microneedling as an Adjuvant to Local Therapies for Vitiligo: A Systematic Review and Meta-Analysis. Dermatol. Surg. 2021, 47, 1314–1316. [Google Scholar] [CrossRef] [PubMed]
- Attwa, E.M.; Khashaba, S.A.; Ezzat, N.A. Evaluation of the additional effect of topical 5-fluorouracil to needling in the treatment of localized vitiligo. J. Cosmet. Dermatol. 2020, 19, 1473–1478. [Google Scholar] [CrossRef] [PubMed]
- Mina, M.; Elgarhy, L.; Al-saeid, H.; Ibrahim, Z. Comparison between the efficacy of microneedling combined with 5-fluorouracil vs microneedling with tacrolimus in the treatment of vitiligo. J. Cosmet. Dermatol. 2018, 17, 744–751. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, Z.A.; Hassan, G.F.; Elgendy, H.Y.; Al-shenawy, H.A. Evaluation of the efficacy of transdermal drug delivery of calcipotriol plus betamethasone versus tacrolimus in the treatment of vitiligo. J. Cosmet. Dermatol. 2019, 18, 581–588. [Google Scholar] [CrossRef]
- Martinez-Lopez, A.; Montero-Vilchez, T.; Sierra-Sánchez, Á.; Molina-Leyva, A.; Arias-Santiago, S. Advanced medical therapies in the management of non-scarring alopecia: Areata and androgenic alopecia. Int. J. Mol. Sci. 2020, 21, 8390. [Google Scholar] [CrossRef]
- Starace, M.; Alessandrini, A.; Brandi, N.; Piraccini, B.M. Preliminary results of the use of scalp microneedling in different types of alopecia. J. Cosmet. Dermatol. 2020, 19, 646–650. [Google Scholar] [CrossRef]
- Ocampo-Garza, S.S.; Fabbrocini, G.; Ocampo-Candiani, J.; Cinelli, E.; Villani, A. Micro needling: A novel therapeutic approach for androgenetic alopecia, A Review of Literature. Dermatol. Ther. 2020, 33, e14267. [Google Scholar] [CrossRef] [PubMed]
- Faghihi, G.; Nabavinejad, S.; Mokhtari, F.; Fatemi Naeini, F.; Iraji, F. Microneedling in androgenetic alopecia; comparing two different depths of microneedles. J. Cosmet. Dermatol. 2021, 20, 1241–1247. [Google Scholar] [CrossRef] [PubMed]
- Fertig, R.M.; Gamret, A.C.; Cervantes, J.; Tosti, A. Microneedling for the treatment of hair loss? J. Eur. Acad. Dermatol. Venereol. 2018, 32, 564–569. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Chen, Q.; Wen, D.; Chen, Z.; Wang, J.; Chen, G.; Wang, Z.; Zhang, X.; Zhang, Y.; Hu, Q. A therapeutic microneedle patch made from hair-derived keratin for promoting hair regrowth. ACS Nano 2019, 13, 4354–4360. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Zhao, J.; Li, H.; Yu, M.; Zhang, W.; Qin, D.; Qiu, K.; Chen, X.; Kong, M. A Drug-Free, Hair Follicle Cycling Regulatable, Separable, Antibacterial Microneedle Patch for Hair Regeneration Therapy. Adv. Healthc. Mater. 2022, 11, 2200908. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.Y. Targeting Wnt/β-catenin pathway for developing therapies for hair loss. Int. J. Mol. Sci. 2020, 21, 4915. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.; Zhang, G.; Zhang, W.; Liu, J.; Zhang, J.; Chen, Y.; Peng, H.; Cheng, Y.; Ding, X.; Xin, H. Hair grows hair: Dual-effective hair regrowth through a hair enhanced dissolvable microneedle patch cooperated with the pure yellow light irradiation. Appl. Mater. Today 2021, 25, 101188. [Google Scholar] [CrossRef]
- Ogbechie-Godec, O.A.; Elbuluk, N. Melasma: An up-to-date comprehensive review. Dermatol. Ther. 2017, 7, 305–318. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.Z.; Muddasani, S.; Alam, M. A Systematic Review of the Efficacy and Safety of Microneedling in the Treatment of Melasma. Dermatol. Surg. 2020, 46, 1636–1641. [Google Scholar] [CrossRef] [PubMed]
- Arenas-Soto, C. Microneedles: A therapeutic alternative in melasma. J. Dermat. Cosmetol. 2018, 2, 207–210. [Google Scholar] [CrossRef]
- Lima, E.d.A. Microneedling in facial recalcitrant melasma: Report of a series of 22 cases. An. Bras. Dermatol. 2015, 90, 919–921. [Google Scholar] [CrossRef] [PubMed]
- Farshi, S.; Mansouri, P. Study of efficacy of microneedling and mesoneedling in the treatment of epidermal melasma: A pilot trial. J. Cosmet. Dermatol. 2020, 19, 1093–1098. [Google Scholar] [CrossRef] [PubMed]
- Shamsi Meymandi, S.; Mozayyeni, A.; Shamsi Meymandi, M.; Aflatoonian, M. Efficacy of microneedling plus topical 4% tranexamic acid solution vs 4% hydroquinone in the treatment of melasma: A single-blind randomized clinical trial. J. Cosmet. Dermatol. 2020, 19, 2906–2911. [Google Scholar] [CrossRef] [PubMed]
- Speeckaert, R.; Bulat, V.; Speeckaert, M.M.; van Geel, N. The Impact of Antioxidants on Vitiligo and Melasma: A Scoping Review and Meta-Analysis. Antioxidants 2023, 12, 2082. [Google Scholar] [CrossRef] [PubMed]
- Aust, M.C.; Fernandes, D.; Kolokythas, P.; Kaplan, H.M.; Vogt, P.M. Percutaneous collagen induction therapy: An alternative treatment for scars, wrinkles, and skin laxity. Plast. Reconstr. Surg. 2008, 121, 1421–1429. [Google Scholar] [CrossRef] [PubMed]
- Roohaninasab, M.; Gandomkar, K.; Goodarzi, A. Microneedling in vitiligo: A systematic review. Surg. Cosmet. Dermatol. 2022, 14, e20220123. [Google Scholar] [CrossRef]
- Gao, F.L.; Jin, R.; Zhang, L.; Zhang, Y.G. The contribution of melanocytes to pathological scar formation during wound healing. Int. J. Clin. Exp. Med. 2013, 6, 609–613. [Google Scholar] [PubMed]
- El-Domyati, M.; Abdel-Wahab, H.; Hossam, A. Combining microneedling with other minimally invasive procedures for facial rejuvenation: A split-face comparative study. Int. J. Dermatol. 2018, 57, 1324–1334. [Google Scholar] [CrossRef] [PubMed]
- Alessa, D.; Bloom, J.D. Microneedling Options for Skin Rejuvenation, Including Non-temperature-controlled Fractional Microneedle Radiofrequency Treatments. Facial Plast. Surg. Clin. N. Am. 2020, 28, 1–7. [Google Scholar] [CrossRef]
- Dayan, E.; Chia, C.; Burns, A.J.; Theodorou, S. Adjustable depth fractional radiofrequency combined with bipolar radiofrequency: A minimally invasive combination treatment for skin laxity. Aesthet. Surg. J. 2019, 39 (Suppl. 3), S112. [Google Scholar] [CrossRef]
- Hidajat, D.; Murlistyarini, S. Successful treatment of rare adverse event after radiofrequency microneedle on Fitzpatrick skin type IV: A case report. J. Cosmet. Laser. Ther. 2023, 25, 102–106. [Google Scholar] [CrossRef] [PubMed]
- Chandrashekar, B.S.; Sriram, R.; Mysore, R.; Bhaskar, S.; Shetty, A. Evaluation of microneedling fractional radiofrequency device for treatment of acne scars. J. Cutan. Aesthet. Surg. 2014, 7, 93–97. [Google Scholar] [CrossRef]
- Baek, G.; Kim, M.H.; Jue, M.S. Efficacy of microneedle radiofrequency therapy in the treatment of senile purpura: A prospective study. Skin Res. Technol. 2022, 28, 856–864. [Google Scholar] [CrossRef]
- Eubanks, S.W.; Solomon, J.A. Safety and efficacy of fractional radiofrequency for the treatment and reduction of acne scarring: A prospective study. Lasers. Surg. Med. 2022, 54, 74–81. [Google Scholar] [CrossRef]
- Li, J.; Duan, F.; Kuang, J. Meta-analysis of fractional radiofrequency treatment for acne and/or acne scars. J. Cosmet. Dermatol. 2022, 21, 6754–6766. [Google Scholar] [CrossRef]
- Kim, J.; Kim, S.M.; Jung, B.K.; Oh, S.H.; Kim, Y.-K.; Lee, J.H. Laser-assisted Delivery of a Combined Antioxidant Formulation Enhances the Clinical Efficacy of Fractional Microneedle Radiofrequency Treatment: A Pilot Study. Med. Lasers Eng. Basic Res. Clin. Appl. 2021, 10, 161–169. [Google Scholar] [CrossRef]
- Sabri, A.; Ogilvie, J.; McKenna, J.; Segal, J.; Scurr, D.; Marlow, M. Intradermal Delivery of an Immunomodulator for Basal Cell Carcinoma; Expanding the Mechanistic Insight into Solid Microneedle-Enhanced Delivery of Hydrophobic Molecules. Mol. Pharm. 2020, 17, 2925–2937. [Google Scholar] [CrossRef]
- Naguib, Y.W.; Kumar, A.; Cui, Z. The effect of microneedles on the skin permeability and antitumor activity of topical 5-fluorouracil. Acta. Pharm. Sin. B 2014, 4, 94–99. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, K.S.; Shan, X.; Mao, J.; Qiu, L.; Chen, J. Derma roller® microneedles-mediated transdermal delivery of doxorubicin and celecoxib co-loaded liposomes for enhancing the anticancer effect. Mater. Sci. Eng. C 2019, 99, 1448–1458. [Google Scholar] [CrossRef]
- Al-Mayahy, M.H.; Sabri, A.H.; Rutland, C.S.; Holmes, A.; McKenna, J.; Marlow, M.; Scurr, D.J. Insight into imiquimod skin permeation and increased delivery using microneedle pre-treatment. Eur. J. Pharm. Biopharm. 2019, 139, 33–43. [Google Scholar] [CrossRef]
- Zhi, D.; Yang, T.; Zhang, T.; Yang, M.; Zhang, S.; Donnelly, R.F. Microneedles for gene and drug delivery in skin cancer therapy. J. Control. Release 2021, 335, 158–177. [Google Scholar] [CrossRef] [PubMed]
- Seetharam, A.A.; Choudhry, H.; Bakhrebah, M.A.; Abdulaal, W.H.; Gupta, M.S.; Rizvi, S.M.D.; Alam, Q.; Siddaramaiah; Gowda, D.V.; Moin, A. Microneedles Drug Delivery Systems for Treatment of Cancer: A Recent Update. Pharmaceutics 2020, 12, 1101. [Google Scholar] [CrossRef] [PubMed]
- Waghule, T.; Singhvi, G.; Dubey, S.K.; Pandey, M.M.; Gupta, G.; Singh, M.; Dua, K. Microneedles: A smart approach and increasing potential for transdermal drug delivery system. Biomed. Pharmacother. 2019, 109, 1249–1258. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Alba, M.; Tieu, T.; Tong, Z.; Minhas, R.S.; Rudd, D.; Voelcker, N.H.; Cifuentes-Rius, A.; Elnathan, R. Engineering Micro–Nanomaterials for Biomedical Translation. Adv. Biomed. Res. 2021, 1, 2100002. [Google Scholar] [CrossRef]
- Dugam, S.; Tade, R.; Dhole, R.; Nangare, S. Emerging era of microneedle array for pharmaceutical and biomedical applications: Recent advances and toxicological perspectives. Future J. Pharm. Sci. 2021, 7, 19. [Google Scholar] [CrossRef]
- Dalvi, M.; Kharat, P.; Thakor, P.; Bhavana, V.; Singh, S.B.; Mehra, N.K. Panorama of dissolving microneedles for transdermal drug delivery. Life Sci. 2021, 284, 119877. [Google Scholar] [CrossRef] [PubMed]
- Alkilani, A.Z.; McCrudden, M.T.C.; Donnelly, R.F. Transdermal drug delivery: Innovative pharmaceutical developments based on disruption of the barrier properties of the stratum corneum. Pharmaceutics 2015, 7, 438–470. [Google Scholar] [CrossRef] [PubMed]
- Aldawood, F.K.; Andar, A.; Desai, S. A Comprehensive Review of Microneedles: Types, Materials, Processes, Characterizations and Applications. Polymers 2021, 13, 2815. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Yang, S.; Yuan, W.; Jin, T. Challenges and strategies in developing microneedle patches for transdermal delivery of protein and peptide therapeutics. Curr. Pharm. Biotechnol. 2012, 13, 1292–1298. [Google Scholar] [CrossRef]
- Avcil, M.; Çelik, A. Microneedles in Drug Delivery: Progress and Challenges. Micromachines 2021, 12, 1321. [Google Scholar] [CrossRef]
- Luo, X.; Yang, L.; Cui, Y. Microneedles: Materials, fabrication, and biomedical applications. Biomed. Microdevices 2023, 25, 20. [Google Scholar] [CrossRef] [PubMed]
- Sargioti, N.; Levingstone, T.J.; O’Cearbhaill, E.D.; McCarthy, H.O.; Dunne, N.J. Metallic microneedles for transdermal drug delivery: Applications, fabrication techniques and the effect of geometrical characteristics. Bioengineering 2022, 10, 24. [Google Scholar] [CrossRef]
- Bao, L.; Park, J.; Bonfante, G.; Kim, B. Recent advances in porous microneedles: Materials, fabrication, and transdermal applications. Drug Deliv. Transl. Res. 2022, 12, 395–414. [Google Scholar] [CrossRef] [PubMed]
- Ebrahiminejad, V.; Rad, Z.F.; Prewett, P.D.; Davies, G.J. Fabrication and testing of polymer microneedles for transdermal drug delivery. Beilstein J. Nanotechnol. 2022, 13, 629–640. [Google Scholar] [CrossRef] [PubMed]
- Al-Japairai, K.A.S.; Mahmood, S.; Almurisi, S.H.; Venugopal, J.R.; Hilles, A.R.; Azmana, M.; Raman, S. Current trends in polymer microneedle for transdermal drug delivery. Int. J. Pharm. 2020, 587, 119673. [Google Scholar] [CrossRef] [PubMed]
- Tucak, A.; Sirbubalo, M.; Hindija, L.; Rahić, O.; Hadžiabdić, J.; Muhamedagić, K.; Čekić, A.; Vranić, E. Microneedles: Characteristics, materials, production methods and commercial development. Micromachines 2020, 11, 961. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Wang, W.; Li, Z. An improved manufacturing approach for discrete silicon microneedle arrays with tunable height-pitch ratio. Sensors 2016, 16, 1628. [Google Scholar] [CrossRef]
- Rajabi, M.; Roxhed, N.; Shafagh, R.Z.; Haraldson, T.; Fischer, A.C.; Wijngaart, W.v.d.; Stemme, G.; Niklaus, F. Flexible and stretchable microneedle patches with integrated rigid stainless steel microneedles for transdermal biointerfacing. PLoS ONE 2016, 11, e0166330. [Google Scholar] [CrossRef] [PubMed]
- Salwa; Chevala, N.T.; Jitta, S.R.; Marques, S.M.; Vaz, V.M.; Kumar, L. Polymeric microneedles for transdermal delivery of nanoparticles: Frontiers of formulation, sterility and stability aspects. J. Drug. Deliv. Sci. Technol. 2021, 65, 102711. [Google Scholar] [CrossRef]
- Parhi, R. Review of microneedle based transdermal drug delivery systems. Int. J. Pharm. Sci. Nanotechnol. (IJPSN) 2019, 12, 4511–4523. [Google Scholar] [CrossRef]
- Kim, Y.C.; Park, J.H.; Prausnitz, M.R. Microneedles for drug and vaccine delivery. Adv. Drug Deliv. Rev. 2012, 64, 1547–1568. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, R.F.; Singh, T.R.R.; Morrow, D.I.; Woolfson, A.D. Microneedle-Mediated Transdermal and Intradermal Drug Delivery; Wiley-Blackwell: Hoboken, NJ, USA, 2012. [Google Scholar]
- Liu, T.T.; Chen, K.; Wang, Q. Skin drug permeability and safety through a vibrating solid micro-needle system. Drug Deliv. Transl. Res. 2018, 8, 1025–1033. [Google Scholar] [CrossRef]
- Lee, C.A.; Baek, J.S.; Kwag, D.G.; Lee, H.J.; Park, J.; Cho, C.W. Enhancement of skin permeation of vitamin C using vibrating microneedles. Transl. Clin. Pharmacol. 2017, 25, 15–20. [Google Scholar] [CrossRef]
- Lhernould, M.S.; Deleers, M.; Delchambre, A. Hollow polymer microneedles array resistance and insertion tests. Int. J. Pharm. 2015, 480, 152–157. [Google Scholar] [CrossRef] [PubMed]
- Kumar Singla, S.; Muthuraman, A.; Sahai, D.; Mangal, N.; Dhamodharan, J. Therapeutic applications of transdermal microneedles. Front. Biosci. (Elite Ed.) 2021, 13, 158–184. [Google Scholar] [PubMed]
- Yang, D.; Chen, M.; Sun, Y.; Jin, Y.; Lu, C.; Pan, X.; Quan, G.; Wu, C. Microneedle-mediated transdermal drug delivery for treating diverse skin diseases. Acta Biomater. 2021, 121, 119–133. [Google Scholar] [CrossRef] [PubMed]
- Menon, I.; Bagwe, P.; Gomes, K.B.; Bajaj, L.; Gala, R.; Uddin, M.N.; D’souza, M.J.; Zughaier, S.M. Microneedles: A new generation vaccine delivery system. Micromachines 2021, 12, 435. [Google Scholar] [CrossRef] [PubMed]
- Vinayakumar, K.; Kulkarni, P.G.; Nayak, M.; Dinesh, N.; Hegde, G.M.; Ramachandra, S.; Rajanna, K. A hollow stainless steel microneedle array to deliver insulin to a diabetic rat. J. Micromech. Microeng. 2016, 26, 065013. [Google Scholar] [CrossRef]
- Lee, J.W.; Prausnitz, M.R. Drug Delivery Using Microneedle Patches: Not Just for Skin; Taylor & Francis: Abingdon, UK, 2018; pp. 541–543. [Google Scholar]
- Kim, J.; Jeong, D. Dissolvable Microneedles: Applications and Opportunities. ONdrugDelivery Mag. 2018, 84, 24–29. [Google Scholar]
- Obaidat, R.; BaniAmer, F.; Assaf, S.M.; Yassin, A. Fabrication and Evaluation of Transdermal Delivery of Carbamazepine Dissolving Microneedles. AAPS PharmSciTech 2021, 22, 253. [Google Scholar] [CrossRef]
- Lahiji, S.F.; Dangol, M.; Jung, H. A patchless dissolving microneedle delivery system enabling rapid and efficient transdermal drug delivery. Sci. Rep. 2015, 5, 7914. [Google Scholar] [CrossRef] [PubMed]
- McCrudden, M.T.; Alkilani, A.Z.; McCrudden, C.M.; McAlister, E.; McCarthy, H.O.; Woolfson, A.D.; Donnelly, R.F. Design and physicochemical characterisation of novel dissolving polymeric microneedle arrays for transdermal delivery of high dose, low molecular weight drugs. J. Control. Release 2014, 180, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.H.; Jin, S.G. Microneedle for transdermal drug delivery: Current trends and fabrication. J. Pharm. Investig. 2021, 51, 503–517. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y. Development and Characterization of Polymeric Formulations and Microneedles for Dermal Drug Delivery. Ph.D. Thesis, Mercer University, Macon, GA, USA, 2020. [Google Scholar]
- Donnelly, R.F.; McCrudden, M.T.C.; Zaid Alkilani, A.; Larrañeta, E.; McAlister, E.; Courtenay, A.J.; Kearney, M.-C.; Singh, T.R.R.; McCarthy, H.O.; Kett, V.L. Hydrogel-forming microneedles prepared from “super swelling” polymers combined with lyophilised wafers for transdermal drug delivery. PLoS ONE 2014, 9, e111547. [Google Scholar] [CrossRef]
- Raj Singh, T.R.; McCarron, P.A.; Woolfson, A.D.; Donnelly, R.F. Investigation of swelling and network parameters of poly(ethylene glycol)-crosslinked poly(methyl vinyl ether-co-maleic acid) hydrogels. Eur. Polym. J. 2009, 45, 1239–1249. [Google Scholar] [CrossRef]
- Aung, N.N.; Ngawhirunpat, T.; Rojanarata, T.; Patrojanasophon, P.; Pamornpathomkul, B.; Opanasopit, P. Fabrication, characterization and comparison of α-arbutin loaded dissolving and hydrogel forming microneedles. Int. J. Pharm. 2020, 586, 119508. [Google Scholar] [CrossRef] [PubMed]
- Kwon, K.M.; Lim, S.-M.; Choi, S.; Kim, D.-H.; Jin, H.-E.; Jee, G.; Hong, K.-J.; Kim, J.Y. Microneedles: Quick and easy delivery methods of vaccines. Clin. Exp. Vaccine Res. 2017, 6, 156–159. [Google Scholar] [CrossRef] [PubMed]
- Guillot, A.J.; Cordeiro, A.S.; Donnelly, R.F.; Montesinos, M.C.; Garrigues, T.M.; Melero, A. Microneedle-based delivery: An overview of current applications and trends. Pharmaceutics 2020, 12, 569. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.M.; Cornwell, M.; Hill, J.; Prausnitz, M.R. Precise microinjection into skin using hollow microneedles. J. Investig. Dermatol. 2006, 126, 1080–1087. [Google Scholar] [CrossRef]
- Tarbox, T.N.; Watts, A.B.; Cui, Z.; Williams, R.O. An update on coating/manufacturing techniques of microneedles. Drug Deliv. Transl. Res. 2018, 8, 1828–1843. [Google Scholar] [CrossRef]
- Mizuno, Y.; Takasawa, K.; Hanada, T.; Nakamura, K.; Yamada, K.; Tsubaki, H.; Hara, M.; Tashiro, Y.; Matsuo, M.; Ito, T. Fabrication of novel-shaped microneedles to overcome the disadvantages of solid microneedles for the transdermal delivery of insulin. Biomed. Microdevices 2021, 23, 38. [Google Scholar] [CrossRef] [PubMed]
- Turner, J.G.; White, L.R.; Estrela, P.; Leese, H.S. Hydrogel-Forming Microneedles: Current Advancements and Future Trends. Macromol. Biosci. 2021, 21, 2000307. [Google Scholar] [CrossRef]
- Alimardani, V.; Abolmaali, S.S.; Yousefi, G.; Rahiminezhad, Z.; Abedi, M.; Tamaddon, A.; Ahadian, S. Microneedle arrays combined with nanomedicine approaches for transdermal delivery of therapeutics. J. Clin. Med. 2021, 10, 181. [Google Scholar] [CrossRef] [PubMed]
- Ravetti, S.; Clemente, C.; Brignone, S.; Hergert, L.; Allemandi, D.; Palma, S. Ascorbic acid in skin health. Cosmetics 2019, 6, 58. [Google Scholar] [CrossRef]
- Juncan, A.M.; Moisă, D.G.; Santini, A.; Morgovan, C.; Rus, L.-L.; Vonica-Țincu, A.L.; Loghin, F. Advantages of hyaluronic acid and its combination with other bioactive ingredients in cosmeceuticals. Molecules 2021, 26, 4429. [Google Scholar] [CrossRef]
- Zasada, M.; Budzisz, E. Retinoids: Active molecules influencing skin structure formation in cosmetic and dermatological treatments. Postep. Dermatol. Alergol. 2019, 36, 392–397. [Google Scholar] [CrossRef]
- Burger, P.; Landreau, A.; Azoulay, S.; Michel, T.; Fernandez, X. Skin whitening cosmetics: Feedback and challenges in the development of natural skin lighteners. Cosmetics 2016, 3, 36. [Google Scholar] [CrossRef]
- Raikou, V.; Varvaresou, A.; Panderi, I.; Papageorgiou, E. The efficacy study of the combination of tripeptide-10-citrulline and acetyl hexapeptide-3. A prospective, randomized controlled study. J. Cosmet. Dermatol. 2017, 16, 271–278. [Google Scholar] [CrossRef]
- Tadini, K.A.; Mercurio, D.G.; Campos, P.M.B.G.M. Acetyl hexapeptide-3 in a cosmetic formulation acts on skin mechanical properties-clinical study. Braz. J. Pharm. Sci. 2015, 51, 901–909. [Google Scholar] [CrossRef]
- Lee, C.; Yang, H.; Kim, S.; Kim, M.; Kang, H.; Kim, N.; An, S.; Koh, J.; Jung, H. Evaluation of the anti-wrinkle effect of an ascorbic acid-loaded dissolving microneedle patch via a double-blind, placebo-controlled clinical study. Int. J. Cosmet. Sci. 2016, 38, 375–381. [Google Scholar] [CrossRef]
- Zaid Alkilani, A.; Hamed, R.; Hussein, G.; Alnadi, S. Nanoemulsion-based patch for the dermal delivery of ascorbic acid. J. Dispers. Sci. Technol. 2021, 43, 1801–1811. [Google Scholar] [CrossRef]
- Kim, Y.; Bhattaccharjee, S.A.; Beck-Broichsitter, M.; Banga, A.K. Fabrication and characterization of hyaluronic acid microneedles to enhance delivery of magnesium ascorbyl phosphate into skin. Biomed. Microdevices 2019, 21, 104. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.-H.; Ha, S.K.; Choi, I.; Kim, K.S.; Park, J.; Choi, N.; Kim, B.; Sung, J.H. Fabrication of degradable carboxymethyl cellulose (CMC) microneedle with laser writing and replica molding process for enhancement of transdermal drug delivery. Biotechnol. Bioprocess. Eng. 2016, 21, 110–118. [Google Scholar] [CrossRef]
- Park, Y.; Kim, K.S.; Chung, M.; Sung, J.H.; Kim, B. Fabrication and characterization of dissolving microneedle arrays for improving skin permeability of cosmetic ingredients. J. Ind. Eng. Chem. 2016, 39, 121–126. [Google Scholar] [CrossRef]
- Sawutdeechaikul, P.; Kanokrungsee, S.; Sahaspot, T.; Thadvibun, K.; Banlunara, W.; Limcharoen, B.; Sansureerungsikul, T.; Rutwaree, T.; Oungeun, M.; Wanichwecharungruang, S. Detachable dissolvable microneedles: Intra-epidermal and intradermal diffusion, effect on skin surface, and application in hyperpigmentation treatment. Sci. Rep. 2021, 11, 24114. [Google Scholar] [CrossRef]
- Avcil, M.; Akman, G.; Klokkers, J.; Jeong, D.; Çelik, A. Efficacy of bioactive peptides loaded on hyaluronic acid microneedle patches: A monocentric clinical study. J. Cosmet. Dermatol. 2020, 19, 328–337. [Google Scholar] [CrossRef] [PubMed]
- Huh, I.; Kim, S.; Yang, H.; Jang, M.; Kang, G.; Jung, H. Effects of two droplet-based dissolving microneedle manufacturing methods on the activity of encapsulated epidermal growth factor and ascorbic acid. Eur. J. Pharm. Sci. 2018, 114, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.Y.; Kwon, H.J.; Ahn, G.R.; Ko, E.J.; Yoo, K.H.; Kim, B.J.; Lee, C.; Kim, D. Hyaluronic acid microneedle patch for the improvement of crow’s feet wrinkles. Dermatol. Ther. 2017, 30, e12546. [Google Scholar] [CrossRef] [PubMed]
- Han, S.-K.; Lee, S.-J.; Ha, H.-Y. Skin Moisturizing Effects of a Microneedle Patch Containing Hyaluronic Acid and Lonicerae flos. Processes 2021, 9, 321. [Google Scholar] [CrossRef]
- Fonseca, D.F.; Vilela, C.; Pinto, R.J.; Bastos, V.; Oliveira, H.; Catarino, J.; Faísca, P.; Rosado, C.; Silvestre, A.J.; Freire, C.S. Bacterial nanocellulose-hyaluronic acid microneedle patches for skin applications: In vitro and in vivo evaluation. Mater. Sci. Eng. C 2021, 118, 111350. [Google Scholar] [CrossRef]
- Kim, H.; Kim, B.; Kim, H.; Um, S.; Lee, J.; Ryoo, H.; Jung, H. Synthesis and in vitro biological activity of retinyl retinoate, a novel hybrid retinoid derivative. Bioorg. Med. Chem. 2008, 16, 6387–6393. [Google Scholar] [CrossRef]
- Kim, H.; Kim, N.; Jung, S.; Mun, J.; Kim, J.; Kim, B.; Lee, J.; Ryoo, H.; Jung, H. Improvement in skin wrinkles from the use of photostable retinyl retinoate: A randomized controlled trial. Br. J. Dermatol. 2010, 162, 497–502. [Google Scholar] [CrossRef]
- Toprangkobsin, P.; Banlunara, W.; Limcharoen, B.; Leelahavanichkul, A.; Asawanonda, P.; Kumtornrut, C.; Sansureerungsikul, T.; Rutwaree, T.; Wanichwecharungruang, S. Delivery and diffusion of retinal in dermis and epidermis through the combination of prodrug nanoparticles and detachable dissolvable microneedles. Drug Deliv. Transl. Res. 2022, 12, 2751–2761. [Google Scholar] [CrossRef]
- Limcharoen, B.; Toprangkobsin, P.; Kröger, M.; Darvin, M.E.; Sansureerungsikul, T.; Rutwaree, T.; Wanichwecharungruang, S.; Banlunara, W.; Lademann, J.; Patzelt, A. Microneedle-facilitated intradermal proretinal nanoparticle delivery. Nanomaterials 2020, 10, 368. [Google Scholar] [CrossRef]
- Hirobe, S.; Otsuka, R.; Iioka, H.; Quan, Y.-S.; Kamiyama, F.; Asada, H.; Okada, N.; Nakagawa, S. Clinical study of a retinoic acid-loaded microneedle patch for seborrheic keratosis or senile lentigo. Life Sci. 2017, 168, 24–27. [Google Scholar] [CrossRef]
- Hiraishi, Y.; Hirobe, S.; Iioka, H.; Quan, Y.-S.; Kamiyama, F.; Asada, H.; Okada, N.; Nakagawa, S. Development of a novel therapeutic approach using a retinoic acid-loaded microneedle patch for seborrheic keratosis treatment and safety study in humans. J. Control. Release 2013, 171, 93–103. [Google Scholar] [CrossRef]
- Lee, Y.; Kumar, S.; Kim, S.H.; Seong, K.-Y.; Lee, H.; Kim, C.; Jung, Y.-S.; Yang, S.Y. Odorless glutathione microneedle patches for skin whitening. Pharmaceutics 2020, 12, 100. [Google Scholar] [CrossRef]
- Gorouhi, F.; Maibach, H.I. Role of topical peptides in preventing or treating aged skin. Int. J. Cosmet. Sci. 2009, 31, 327–345. [Google Scholar] [CrossRef]
- Schagen, S.K. Topical peptide treatments with effective anti-aging results. Cosmetics 2017, 4, 16. [Google Scholar] [CrossRef]
- Lim, S.H.; Tiew, W.J.; Zhang, J.; Ho, P.C.-L.; Kachouie, N.N.; Kang, L. Geometrical optimisation of a personalised microneedle eye patch for transdermal delivery of anti-wrinkle small peptide. Biofabrication 2020, 12, 035003. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.H.; Kathuria, H.; Amir, M.H.B.; Zhang, X.; Duong, H.T.T.; Ho, P.C.; Kang, L. High resolution photopolymer for 3D printing of personalised microneedle for transdermal delivery of anti-wrinkle small peptide. J. Control. Release 2021, 329, 907–918. [Google Scholar] [CrossRef] [PubMed]
- An, J.H.; Lee, H.J.; Yoon, M.S.; Kim, D.H. Anti-wrinkle efficacy of cross-linked hyaluronic acid-based microneedle patch with acetyl hexapeptide-8 and epidermal growth factor on Korean skin. Ann. Dermatol. 2019, 31, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Bissett, D.L.; Oblong, J.E.; Berge, C.A. Niacinamide: AB vitamin that improves aging facial skin appearance. Dermatol. Surg. 2005, 31, 860–866. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Kim, B. Skin permeability of compounds loaded within dissolving microneedles dependent on composition of sodium hyaluronate and carboxymethyl cellulose. Korean J. Chem. Eng. 2017, 34, 133–138. [Google Scholar] [CrossRef]
- Shin, C.I.; Kim, M.; Kim, Y.-C. Delivery of niacinamide to the skin using microneedle-like particles. Pharmaceutics 2019, 11, 326. [Google Scholar] [CrossRef] [PubMed]
- Reilly, D.M.; Lozano, J. Skin collagen through the lifestages: Importance for skin health and beauty. Aesthet. Surg. J. 2021, 8, 10–20517. [Google Scholar] [CrossRef]
- Zhang, S.; Duan, E. Fighting against skin aging: The way from bench to bedside. Cell Transplant. 2018, 27, 729–738. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Inayathullah, M.; Manoukian, M.A.; Malkovskiy, A.V.; Manickam, S.; Marinkovich, M.P.; Lane, A.T.; Tayebi, L.; Seifalian, A.M.; Rajadas, J. Transdermal delivery of functional collagen via polyvinylpyrrolidone microneedles. Ann. Biomed. Eng. 2015, 43, 2978–2990. [Google Scholar] [CrossRef] [PubMed]
- Aditya, A.; Kim, B.; Koyani, R.D.; Oropeza, B.; Furth, M.; Kim, J.; Kim, N.P. Kinetics of collagen microneedle drug delivery system. J. Drug Deliv. Sci. Technol. 2019, 52, 618–623. [Google Scholar] [CrossRef]
- Kochhar, J.S.; Anbalagan, P.; Shelar, S.B.; Neo, J.K.; Iliescu, C.; Kang, L. Direct microneedle array fabrication off a photomask to deliver collagen through skin. Pharm. Res. 2014, 31, 1724–1734. [Google Scholar] [CrossRef]
- Al-Atif, H. Collagen supplements for aging and wrinkles: A paradigm shift in the fields of dermatology and cosmetics. Dermatol. Pract. Concept. 2022, 12, e2022018. [Google Scholar] [CrossRef] [PubMed]
- Lo, S.; Fauzi, M.B. Current update of collagen nanomaterials—Fabrication, characterisation and its applications: A review. Pharmaceutics 2021, 13, 316. [Google Scholar] [CrossRef] [PubMed]
- Jang, M.; Baek, S.; Kang, G.; Yang, H.; Kim, S.; Jung, H. Dissolving microneedle with high molecular weight hyaluronic acid to improve skin wrinkles, dermal density and elasticity. Int. J. Cosmet. Sci. 2020, 42, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Kim, S.; Jang, M.; Kim, H.; Lee, S.; Kim, Y.; Eom, Y.A.; Kang, G.; Chiang, L.; Baek, J.H.; et al. Two-phase delivery using a horse oil and adenosine-loaded dissolving microneedle patch for skin barrier restoration, moisturization, and wrinkle improvement. J. Cosmet. Dermatol. 2019, 18, 936–943. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Eom, Y.A.; Yang, H.; Jang, M.; Jung, S.U.; Park, Y.O.; Lee, S.E.; Jung, H. Skin Barrier Restoration and Moisturization Using Horse Oil-Loaded Dissolving Microneedle Patches. Skin Pharmacol. Physiol. 2018, 31, 163–171. [Google Scholar] [CrossRef]
- Kang, G.; Tu, T.; Kim, S.; Yang, H.; Jang, M.; Jo, D.; Ryu, J.; Baek, J.; Jung, H. Adenosine-loaded dissolving microneedle patches to improve skin wrinkles, dermal density, elasticity and hydration. Int. J. Cosmet. Sci. 2018, 40, 199–206. [Google Scholar] [CrossRef]
- Hong, J.Y.; Ko, E.J.; Choi, S.Y.; Li, K.; Kim, A.R.; Park, J.O.; Kim, B.J. Efficacy and safety of a novel, soluble microneedle patch for the improvement of facial wrinkle. J. Cosmet. Dermatol. 2018, 17, 235–241. [Google Scholar] [CrossRef] [PubMed]
- El-Leithy, E.S.; Makky, A.M.; Khattab, A.M.; Hussein, D.G. Optimization of nutraceutical coenzyme Q10 nanoemulsion with improved skin permeability and anti-wrinkle efficiency. Drug Dev. Ind. Pharm. 2018, 44, 316–328. [Google Scholar] [CrossRef]
- Bradley, E.J.; Griffiths, C.E.; Sherratt, M.J.; Bell, M.; Watson, R.E. Over-the-counter anti-ageing topical agents and their ability to protect and repair photoaged skin. Maturitas 2015, 80, 265–272. [Google Scholar] [CrossRef]
- Hunt, K.J.; Hung, S.K.; Ernst, E. Botanical extracts as anti-aging preparations for the skin. Drugs Aging 2010, 27, 973–985. [Google Scholar] [CrossRef]
- Rafique, M.; Hussain Shah, S.N. Anti-Ageing Potential of a Cream (W/O Emulsion) Containing Grape Seed Extract (GSE): Formulation and in vivo Evaluation of Effectiveness Using Non-Invasive Biophysical Technique. J. Clin. Exp. Dermatol. Res. 2019, 10, 1000500. [Google Scholar] [CrossRef]
- Rattanawiwatpong, P.; Wanitphakdeedecha, R.; Bumrungpert, A.; Maiprasert, M. Anti-aging and brightening effects of a topical treatment containing vitamin C, vitamin E, and raspberry leaf cell culture extract: A split-face, randomized controlled trial. J. Cosmet. Dermatol. 2020, 19, 671–676. [Google Scholar] [CrossRef]
- Chondrogianni, N.; Kapeta, S.; Chinou, I.; Vassilatou, K.; Papassideri, I.; Gonos, E.S. Anti-ageing and rejuvenating effects of quercetin. Exp. Gerontol. 2010, 45, 763–771. [Google Scholar] [CrossRef]
- Sunoqrot, S.; Al-Shalabi, E.; Hasan Ibrahim, L.; Zalloum, H. Nature-Inspired Polymerization of Quercetin to Produce Antioxidant Nanoparticles with Controlled Size and Skin Tone-Matching Colors. Molecules 2019, 24, 3815. [Google Scholar] [CrossRef]
- Bal, S.M.; Caussin, J.; Pavel, S.; Bouwstra, J.A. In vivo assessment of safety of microneedle arrays in human skin. Eur. J. Pharm. Sci. 2008, 35, 193–202. [Google Scholar] [CrossRef]
- Jeong, H.-R.; Lee, H.-S.; Choi, I.-J.; Park, J.-H. Considerations in the use of microneedles: Pain, convenience, anxiety and safety. J. Drug Target. 2017, 25, 29–40. [Google Scholar] [CrossRef]
- U.S. Food & Drug Administration. Microneedling Devices: Getting to the Point on Benefits, Risks and Safety; U.S. Food & Drug Administration: Silver Spring, DA, USA, 2021.
- Donnelly, R.F.; Singh, T.R.R.; Larrañeta, E.; McCrudden, M.T. Microneedles for Drug and Vaccine Delivery and Patient Monitoring; John Wiley & Sons: Hoboken, NJ, USA, 2018. [Google Scholar]
- Cary, J.H.; Li, B.S.; Maibach, H.I. Dermatotoxicology of microneedles (MNs) in man. Biomed. Microdevices 2019, 21, 66. [Google Scholar] [CrossRef]
- Park, J.H.; Prausnitz, M.R. Analysis of Mechanical Failure of Polymer Microneedles by Axial Force. J. Korean Phys. Soc. 2010, 56, 1223–1227. [Google Scholar] [CrossRef]
- Hoesly, F.J.; Borovicka, J.; Gordon, J.; Nardone, B.; Holbrook, J.S.; Pace, N.; Ibrahim, O.; Bolotin, D.; Warycha, M.; Kwasny, M.; et al. Safety of a novel microneedle device applied to facial skin: A subject- and rater-blinded, sham-controlled, randomized trial. Arch. Dermatol. 2012, 148, 711–717. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, R.F.; Singh, T.R.; Alkilani, A.Z.; McCrudden, M.T.; O’Neill, S.; O’Mahony, C.; Armstrong, K.; McLoone, N.; Kole, P.; Woolfson, A.D. Hydrogel-forming microneedle arrays exhibit antimicrobial properties: Potential for enhanced patient safety. Int. J. Pharm. 2013, 451, 76–91. [Google Scholar] [CrossRef] [PubMed]
- Davis, S.P.; Landis, B.J.; Adams, Z.H.; Allen, M.G.; Prausnitz, M.R. Insertion of microneedles into skin: Measurement and prediction of insertion force and needle fracture force. J. Biomech. 2004, 37, 1155–1163. [Google Scholar] [CrossRef]
- Huang, H.; Fu, C. Different fabrication methods of out-of-plane polymer hollow needle arrays and their variations. J. Micromech. Microeng. 2007, 17, 393–402. [Google Scholar] [CrossRef]
- FDA. Regulatory Considerations for Microneedling Products. Guidance for Industry and Food and Drug Administration Staff. 2020. Available online: https://www.fda.gov/media/107708/download (accessed on 10 November 2020).


| Specifications | Dermaroller | Dermapen |
|---|---|---|
| Needle length | 200–3000 µm [28] | Adjustable in length from 250 to 2500 µm [38] |
| Needles depth | Adjustable needle penetration depth in automated Dermarollers only [28] | Adjustable needle penetration depth during use [28,37] |
| Disposable needles | Disposable head in automated rollers only [28] | Disposable needles [28] |
| Applications | In cosmetics and transdermal applications, where the skin is pretreated with the Dermaroller, followed by the use of the transdermal formulation [28] | Primarily in cosmetics [13] |
| Uniform pressure on the skin | Depending on the user, no way to control how much pressure is on the skin, except for automated Dermarollers [28] | Uniform pressure [28] |
| Advantages | Easy-to-use, home-usable Dermaroller, applied across the skin vertically, horizontally, and diagonally [43,44] | An inexpensive office maneuver, risk-free procedure as the needles are hidden in a guide, less painful, used to treat facial wrinkles due to aging and smoking, penetrates the skin at a perpendicular angle, is suitable for delicate and specific areas, and does not require pressure on the skin as in the case of Dermaroller [43,45,46] |
| Disadvantages | Controlling the pressure has to come with practice and experience (except for automated devices); difficult to treat small areas or localized scars [43] | The disadvantages of Dermaroller are overcome by Dermapen [44] |
| Side effects | Bleeding, swelling, bruising, redness, temporary erythema, pain, burning sensation, edema, itching, and peeling (these typically go after a few days or weeks), along with a risk of tips breaking in the skin [27,39] | Redness and swelling that disappears within 2–3 days [43] |
| Microneedling setup | Accessible for home use at low cost or by skin professionals in clinics [25,47,48] | |
| Recovery of the skin barrier function | Several hours to 72 h of device usage, based on age, skin elasticity, skin application site, needle length, number of applications, and application pressure [44,49] | |
| Microneedling Product | Company Name | Description of the Device | Uses |
|---|---|---|---|
| Dermaroller® [109] | White Lotus, Germany | A cylindrical roller with solid microneedles of 200–2500 µm in length | Improve skin texture and treat scars and hyperpigmentation |
| Dermaroller® [110] | DermaSpark, Vancouver, Canada | Solid or metal microneedles | Induce the production of collagen and elastin and enhance the penetration of cosmetic agents Promote skin repair and reduce the appearance of wrinkles, scars, and stretch marks |
| Dermaroller® Genosys [110] | Hansderma, Downey, CA, USA | Different needle lengths of 250–2000 µm | Induce the production of collagen and elastin Treat wrinkles and acne scars |
| Dermaroller® C-8 [111] | Dermaroller Series by Anastassakis K. | The needles’ length is 130 μm, with 24 circular arrays of 8 needles each (total 192 needles) | Enhance the penetration of topical agents |
| Dermaroller® CIT-8 [111] | Dermaroller Series by Anastassakis K. | The needle’s length of 500 μm | Induce the production of collagen and remodel the skin |
| Dermaroller® MF-8 [111] | Dermaroller Series by Anastassakis K. | The needle’s length of 1500 μm | Treat scars |
| Dermaroller® MS-4 [111] | Dermaroller Series by Anastassakis K. | A small cylinder of 1 cm in length and 2 cm in diameter with four circular arrays of 1500 μm needle length | Treat scars |
| Dermaroller® C-8HE [53] | Dermaroller Series by Anastassakis K. | Has a 200 µm needle length | Used in hair-bearing surfaces like the scalp |
| Dermapen® [13] | MDerma FDS (USA) | Made up of 12 microneedles | Induce the production of collagen and elastin |
| Dermapen® [112] | Dermapenworld, Sydney, Austria | A 33-gauge gamma-sterilized stainless steel needles | Treat various skin conditions such as acne, stretch marks, and hair loss Enhance drug absorption |
| Exceed Microneedling® device [112] | MT. Derm GmbH, Berlin, Germany | Needle length can be adjusted between 0.0 and 1500 µm. | Improve the appearance of facial acne scars in Fitzpatrick skin types I, II, III, and IV in adults |
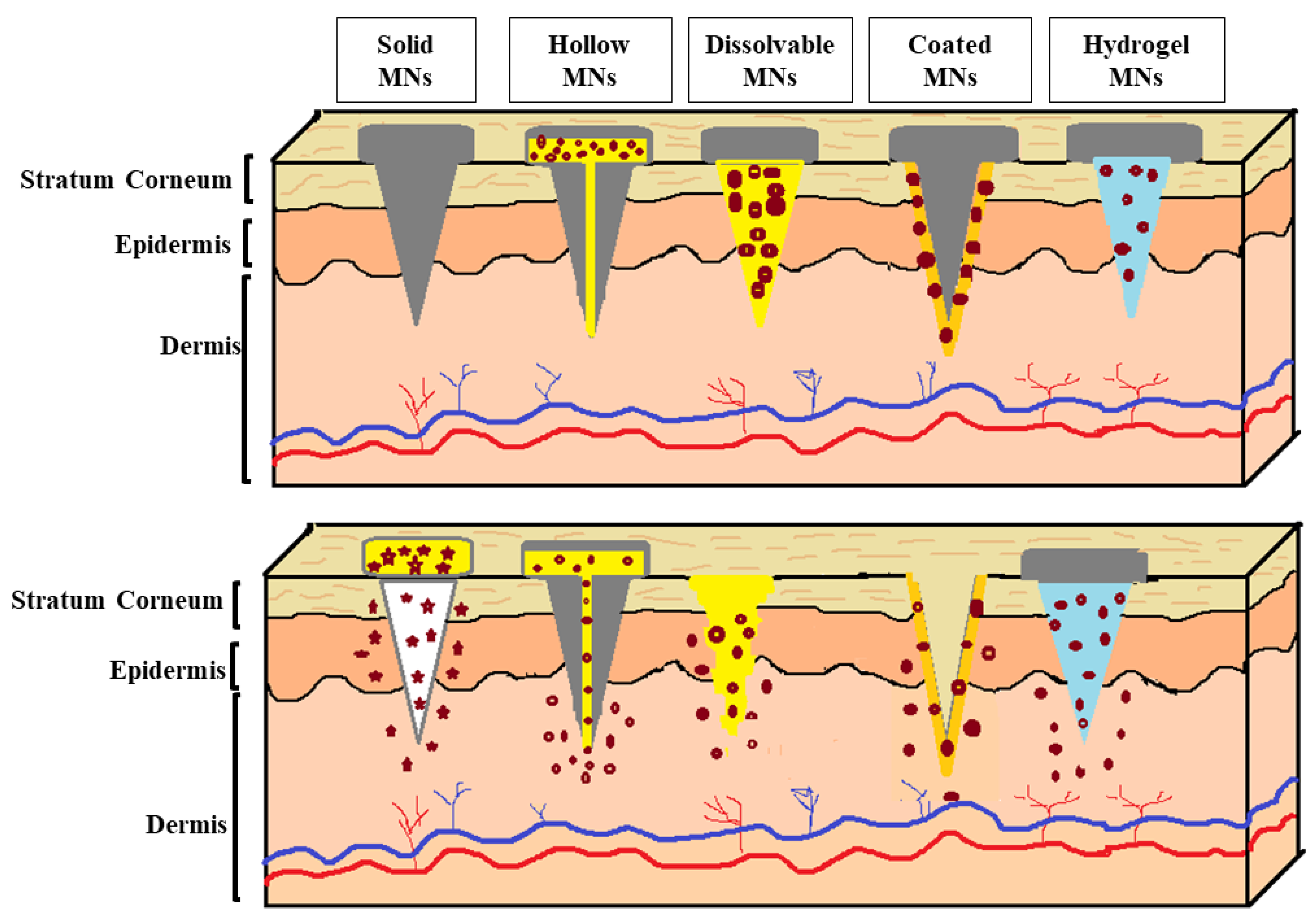
| Types of MNs | Benefits | Risks | Limitations |
|---|---|---|---|
| Solid | Suitable for the delivery of cosmetic agents into the lower skin layers, as microneedles create channels due to their sharp needles [114] | It might cause damage to the skin [114] | Allow slow diffusion through the skin [146] Micropores remain open for a limited time, stopping the delivery of cosmetic agents [147] Two-step application [128] |
| Hollow | Have an empty shape that is filled with a large amount of cosmetic agents [114] Ability to control the release over time [114] | Might cause leakage and clogging [114] | Deposit the cosmetic agent directly into the epidermis or the upper dermis layer [109] Increasing the microneedle bore in an attempt to increase the flow rate may decrease the microneedles’ strength and sharpness, making insertion into the skin more difficult [109,148] Weaker than solid MNs [148] |
| Dissolvable | Biocompatible [114] One-step application, as microneedles can pierce the skin and are kept inserted until complete dissolution [147] Tuning the delivery rate by controlling the dissolution rate of the polymer used in the microneedles’ formulation [147,149] Avoiding the generation of sharps waste, minimizing the cost of waste management, and reducing needle-stick injuries [147,149] | Potential accumulation of polymer within the skin upon repeated application of microneedles [149] | Limited loading capacity and limited ability to perforate the SC [147] Long-term safety for repeated use has not been established in humans [149] Requires technical expertise to manufacture [114] Takes time to dissolve [114] |
| Coated | Fast delivery to the skin [114] Tuning the delivery rate by altering the polymer(s) architecture and the thickness of the film [149] | Potential accumulation of polymer within the skin upon repeated application of microneedles [149] | Relatively low loading capacity on the surface of the microneedles [147,150] The coating’s thickness can decrease the sharpness of the microneedles, impacting their ability to perforate the skin [147] The shape of the microneedles must be designed to ensure delivery and overcome the insertion forces into the skin [150] Biohazardous sharps waste after use [149] Lack of safety data [149] |
| Hydrogel | Tuning the delivery rate by controlling the the density of crosslinking [149,151] Delivering cosmetic agents in a molecular weight range of 0.17–67 kDa through the hydrogel matrix [149] Removal of swollen microneedles after use reduces the risk of intradermal material accumulation [149] The release profile is characterized by an initial burst release followed by a steady rate of release [151] | Cytotoxicity due to the accumulation of unreacted polymers during crosslinking [151] | Restricted to agents that are stable to crosslinking conditions such as heat and UV exposure [149] Restricted to polymeric materials capable of crosslinking under mild conditions such as freeze/thaw [149] Biohazardous waste after use [149] |
| Fabricated MNs | Company Name | Type | Uses |
|---|---|---|---|
| MicroHyala® [109,110,111] | CosMED Pharmaceutical, Kyoto city, Japan | Dissolvable MNs | Intradermal delivery of HA for skin aging |
| LiteClear® [109,152] | Nanomed Skincare, Delaware, USA | Solid MNs made of silicon | Treat acne and skin blemishes Used to pretreat the skin before topical application |
| 3M® Hollow Microstructured Transdermal System [111] | 3M, Minnesota, USA | Hollow MNs (1 cm2 array with a needle length of 1500 μm) | Intradermal delivery |
| 3M® Solid Microstructured Transdermal System [111] | 3M, Minnesota, USA | Solid MNs (1 cm2 array with needle lengths of 250, 500, and 700 μm) | Intradermal delivery |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamed, R.; Abu Nahia, B.J.; Alkilani, A.Z.; Al-Adhami, Y.; Obaidat, R. Recent Advances in Microneedling-Assisted Cosmetic Applications. Cosmetics 2024, 11, 51. https://doi.org/10.3390/cosmetics11020051
Hamed R, Abu Nahia BJ, Alkilani AZ, Al-Adhami Y, Obaidat R. Recent Advances in Microneedling-Assisted Cosmetic Applications. Cosmetics. 2024; 11(2):51. https://doi.org/10.3390/cosmetics11020051
Chicago/Turabian StyleHamed, Rania, Baraah Jehad Abu Nahia, Ahlam Zaid Alkilani, Yasmeen Al-Adhami, and Rana Obaidat. 2024. "Recent Advances in Microneedling-Assisted Cosmetic Applications" Cosmetics 11, no. 2: 51. https://doi.org/10.3390/cosmetics11020051






