Rapid Identification of High-Temperature Responsive Genes Using Large-Scale Yeast Functional Screening System in Potato
Abstract
:1. Introduction
2. Results
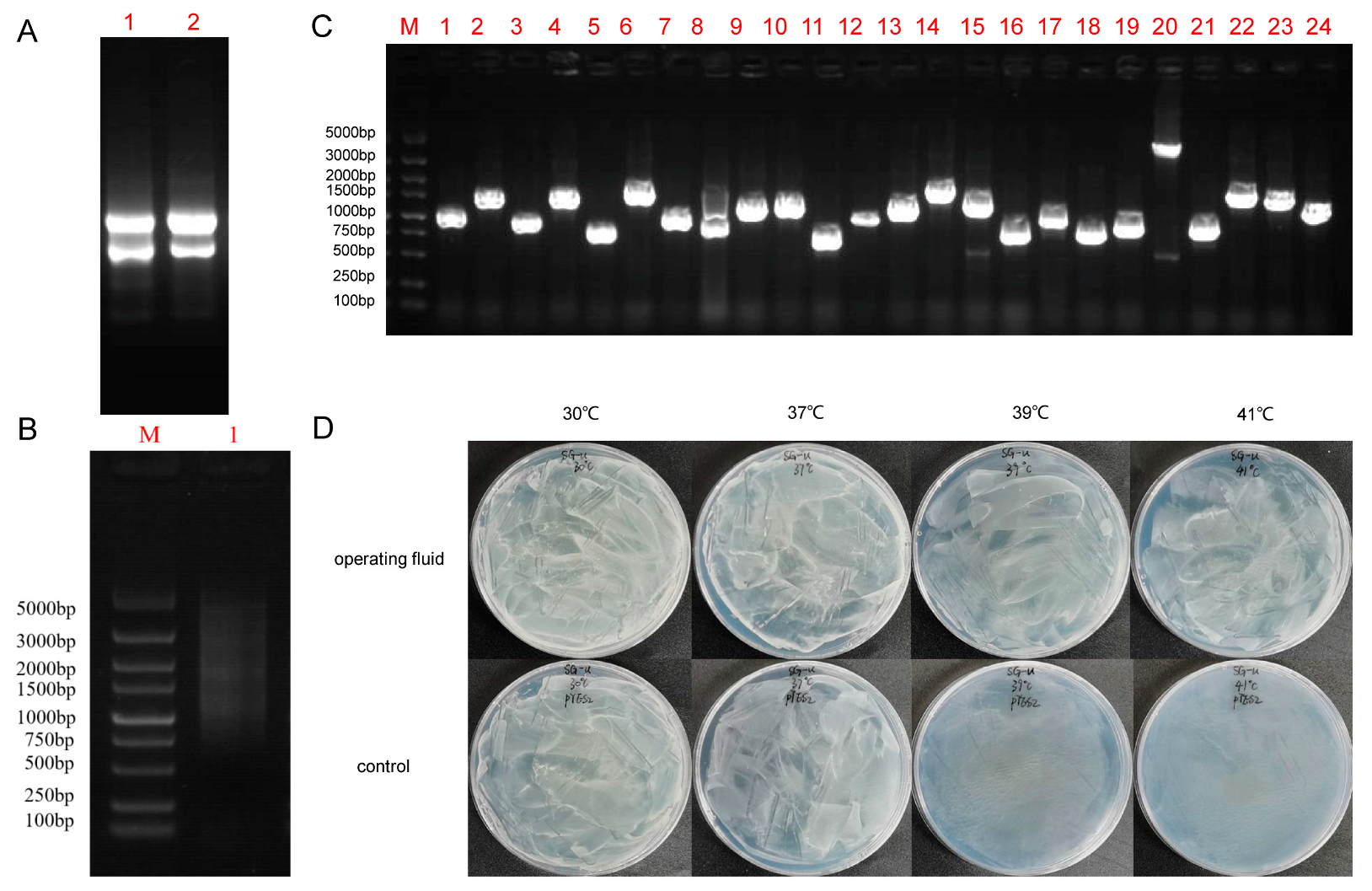
2.1. Determining S. tuberosum Heat-Responsive Genes Using a Yeast Functional Screening System
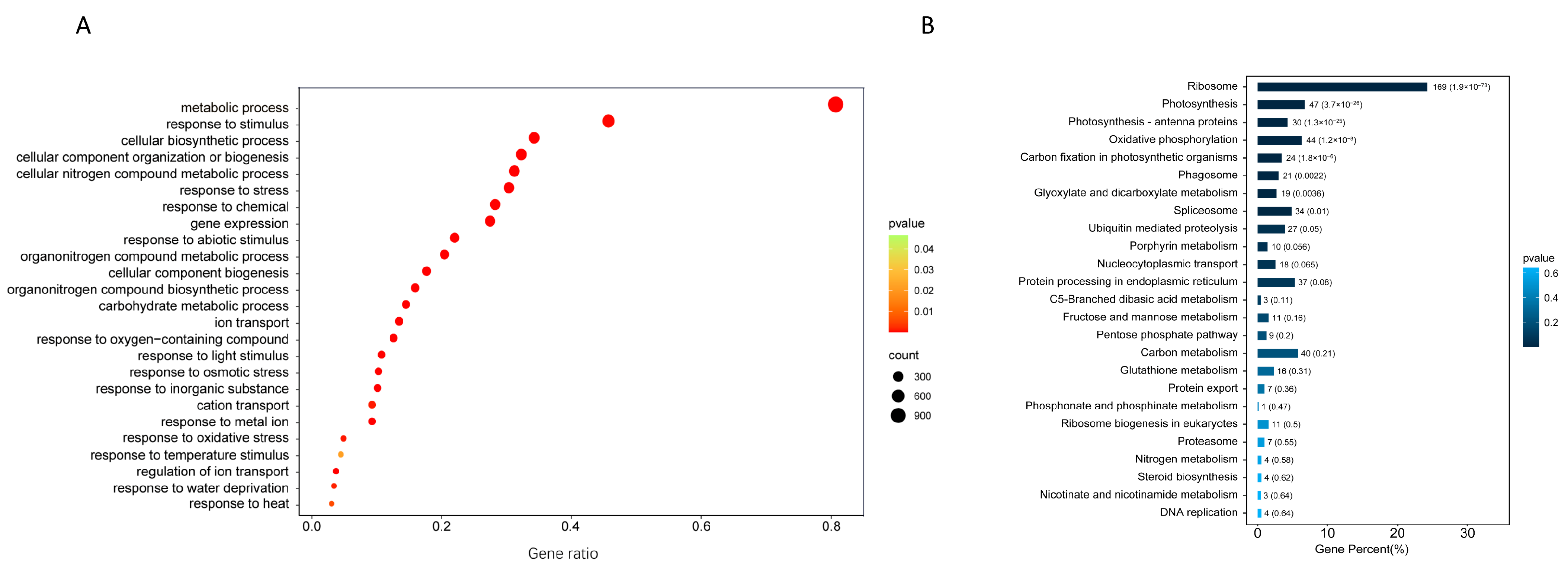
2.2. Gene Functional Annotation and Classification
2.3. Gene Expression Analysis Based on PGSC Expression Data
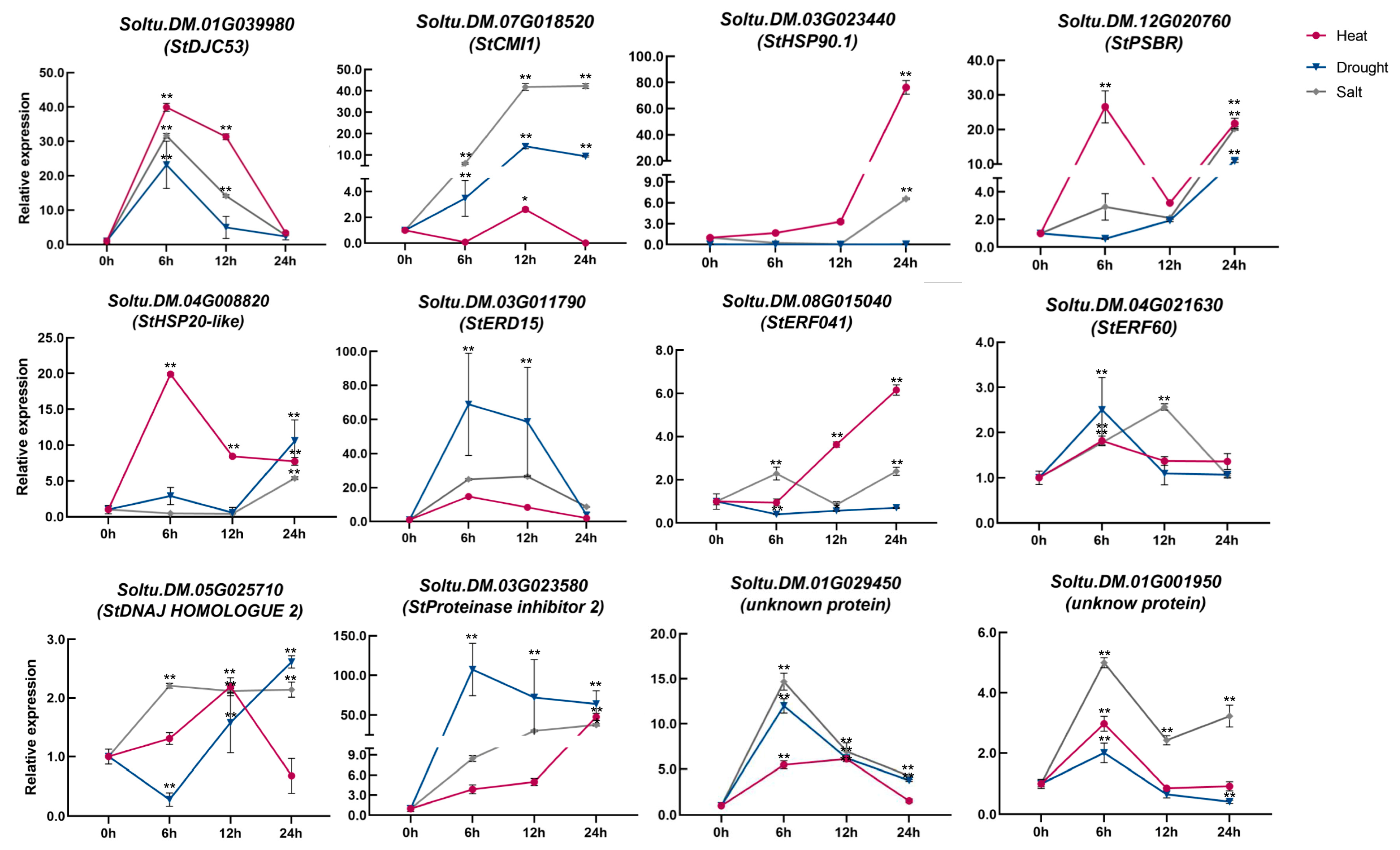
2.4. Expression Pattern of Selected Potato Genes under Heat, Salt, and Drought Stresses
2.5. High-Temperature Candidate Genes’ Transfer Enhances Heat Tolerance in Saccharomyces cerevisiae
3. Discussion
4. Materials and Methods
4.1. Materials, Stress Treatments, and RNA Isolation
4.2. Yeast cDNA Expression Library Construction
4.3. DNA Sequencing and Gene Annotation
4.4. qRT-PCR Analysis
4.5. High-Temperature Sensitivity Assays in Yeast Cells
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Francini, A.; Sebastiani, L. Abiotic Stress Effects on Performance of Horticultural Crops. Horticulturae 2019, 5, 67. [Google Scholar] [CrossRef]
- Hijmans, R.J. Global distribution of the potato crop. Am. J. Potato Res. 2001, 78, 403–412. [Google Scholar] [CrossRef]
- McGill, C.R.; Kurilich, A.C.; Davignon, J. The role of potatoes and potato components in cardiometabolic health: A review. Ann. Med. 2013, 45, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Camire, M.E.; Kubow, S.; Donnelly, D.J. Potatoes and human health. Crit. Rev. Food Sci. Nutr. 2009, 49, 823–840. [Google Scholar] [CrossRef] [PubMed]
- Hancock, R.D.; Morris, W.L.; Ducreux, L.J.; Morris, J.A.; Usman, M.; Verrall, S.R.; Fuller, J.; Simpson, C.G.; Zhang, R.; Hedley, P.E.; et al. Physiological, biochemical and molecular responses of the potato (Solanum tuberosum L.) plant to moderately elevated temperature. Plant Cell Environ. 2014, 37, 439–450. [Google Scholar] [CrossRef] [PubMed]
- Steyn, J.M.; Du Plessis, H.F.; Fourie, P.; Hammes, P.S. Yield response of potato genotypes to different soil water regimes in contrasting seasons of a subtropical climate. Potato Res. 1998, 41, 239–254. [Google Scholar] [CrossRef]
- Chen, X.; Xue, H.; Zhu, L.; Wang, H.; Long, H.; Zhao, J.; Meng, F.; Liu, Y.; Ye, Y.; Luo, X.; et al. ERF49 mediates brassinosteroid regulation of heat stress tolerance in Arabidopsis thaliana. BMC Biol. 2022, 20, 254. [Google Scholar] [CrossRef]
- Ren, Y.; Huang, Z.; Jiang, H.; Wang, Z.; Wu, F.; Xiong, Y.; Yao, J. A heat stress responsive NAC transcription factor heterodimer plays key roles in rice grain filling. J. Exp. Bot. 2021, 72, 2947–2964. [Google Scholar] [CrossRef]
- Zhao, J.; Lu, Z.; Wang, L.; Jin, B. Plant Responses to Heat Stress: Physiology, Transcription, Noncoding RNAs, and Epigenetics. Int. J. Mol. Sci. 2020, 22, 117. [Google Scholar] [CrossRef] [PubMed]
- Nakano, T.; Suzuki, K.; Fujimura, T.; Shinshi, H. Genome-wide analysis of the ERF gene family in Arabidopsis and rice. Plant Physiol. 2006, 140, 411–432. [Google Scholar] [CrossRef] [PubMed]
- Bashir, K.; Matsui, A.; Rasheed, S.; Seki, M. Recent advances in the characterization of plant transcriptomes in response to drought, salinity, heat, and cold stress. F1000Research 2019, 8, 658. [Google Scholar] [CrossRef]
- Ashfaq, W.; Brodie, G.; Fuentes, S.; Gupta, D. Infrared Thermal Imaging and Morpho-Physiological Indices Used for Wheat Genotypes Screening under Drought and Heat Stress. Plants 2022, 11, 3269. [Google Scholar] [CrossRef]
- Wang, J.; Mao, X.; Wang, R.; Li, A.; Zhao, G.; Zhao, J.; Jing, R. Identification of wheat stress-responding genes and TaPR-1-1 function by screening a cDNA yeast library prepared following abiotic stress. Sci. Rep. 2019, 9, 141. [Google Scholar] [CrossRef] [PubMed]
- Eswaran, N.; Parameswaran, S.; Sathram, B.; Anantharaman, B.; Kumar, G.R.K.; Tangirala, S.J. Yeast functional screen to identify genetic determinants capable of conferring abiotic stress tolerance in Jatropha curcas. BMC Biotechnol. 2010, 10, 23. [Google Scholar] [CrossRef] [PubMed]
- Gangadhar, B.H.; Yu, J.W.; Sajeesh, K.; Park, S.W. A systematic exploration of high-temperature stress-responsive genes in potato using large-scale yeast functional screening. Mol. Genet. Genom. 2013, 289, 185–201. [Google Scholar] [CrossRef]
- Wang, Z.; He, Z.; Xu, X.; Shi, X.; Ji, X.; Wang, Y. Revealing the salt tolerance mechanism of Tamarix hispida by large-scale identification of genes conferring salt tolerance. Tree Physiol. 2021, 41, 2153–2170. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.K. Abiotic Stress Signaling and Responses in Plants. Cell 2016, 167, 313–324. [Google Scholar] [CrossRef]
- Yin, Y.; Qin, K.; Song, X.; Zhang, Q.; Zhou, Y.; Xia, X.; Yu, J. BZR1 transcription factor regulates heat stress tolerance through FERONIA receptor-like kinase-mediated reactive oxygen species signaling in tomato. Plant Cell Physiol. 2018, 59, 2239–2254. [Google Scholar] [CrossRef]
- Li, P.; Ma, Q.; Qu, C.; Zhu, S.; Zhao, K.; Ma, X.; Li, Z.; Zhang, X.; Gong, F.; Yin, D. Genome-wide identification and expression analysis of auxin response factors in peanut (Arachis hypogaea L.). PeerJ 2021, 9, e12319. [Google Scholar] [CrossRef]
- Cappetta, E.; Andolfo, G.; Guadagno, A.; Di Matteo, A.; Barone, A.; Frusciante, L.; Ercolano, M.R. Tomato genomic prediction for good performance under high-temperature and identification of loci involved in thermotolerance response. Hortic. Res. 2021, 8, 212. [Google Scholar] [CrossRef]
- Wen, J.; Jiang, F.; Weng, Y.; Sun, M.; Shi, X.; Zhou, Y.; Yu, L.; Wu, Z. Identification of heat-tolerance QTLs and high-temperature stress-responsive genes through conventional QTL mapping, QTL-seq and RNA-seq in tomato. BMC Plant Biol. 2019, 19, 398. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Lim, S.D.; Jang, C.S. Oryza sativa drought-, heat-, and salt-induced RING finger protein 1 (OsDHSRP1) negatively regulates abiotic stress-responsive gene expression. Plant Mol. Biol. 2020, 103, 235–252. [Google Scholar] [CrossRef]
- Iqbal, M.Z.; Jia, T.; Tang, T.; Anwar, M.; Ali, A.; Hassan, M.J.; Zhang, Y.; Tang, Q.; Peng, Y. A Heat Shock Transcription Factor TrHSFB2a of White Clover Negatively Regulates Drought, Heat and Salt Stress Tolerance in Transgenic Arabidopsis. Int. J. Mol. Sci. 2022, 23, 12769. [Google Scholar] [CrossRef] [PubMed]
- Djemal, R.; Khoudi, H. The barley SHN1-type transcription factor HvSHN1 imparts heat, drought and salt tolerances in transgenic tobacco. Plant Physiol. Biochem. 2021, 164, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Meiri, D.; Breiman, A. Arabidopsis ROF1 (FKBP62) modulates thermotolerance by interacting with HSP90.1 and affecting the accumulation of HsfA2-regulated sHSPs. Plant J. 2009, 59, 387–399. [Google Scholar] [CrossRef]
- Imai, J.; Yahara, I. Role of HSP90 in Salt Stress Tolerance via Stabilization and Regulation of Calcineurin. Mol. Cell. Biol. 2000, 20, 9262–9270. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Duan, M.; Zhang, L.; Li, J.; Shan, L.; Zheng, L.; Liu, J. HOP1 and HOP2 are involved in salt tolerance by facilitating the brassinosteroid-related nucleo-cytoplasmic partitioning of the HSP90-BIN2 complex. Plant Cell Environ. 2022, 45, 3551–3565. [Google Scholar] [CrossRef] [PubMed]
- Aalto, M.K.; Helenius, E.; Kariola, T.; Pennanen, V.; Heino, P.; Horak, H.; Puzorjova, I.; Kollist, H.; Palva, E.T. ERD15--an attenuator of plant ABA responses and stomatal aperture. Plant Sci. 2012, 182, 19–28. [Google Scholar] [CrossRef]
- Kariola, T.; Brader, G.; Helenius, E.; Li, J.; Heino, P.; Palva, E.T. EARLY RESPONSIVE TO DEHYDRATION 15, a negative regulator of abscisic acid responses in Arabidopsis. Plant Physiol. 2006, 142, 1559–1573. [Google Scholar] [CrossRef]
- Jin, T.; Sun, Y.; Shan, Z.; He, J.; Wang, N.; Gai, J.; Li, Y. Natural variation in the promoter of GsERD15B affects salt tolerance in soybean. Plant Biotechnol. J. 2021, 19, 1155–1169. [Google Scholar] [CrossRef]
- Sharma, S.; Chahal, A.; Prasad, H.; Walia, A.; Kumar, R.; Dobhal, S. Biotechnology, Identification, phylogeny and transcript profiling of ERF family genes during temperature stress treatment in Pea (Pisum sativum L.). J. Plant Biochem. Biotechnol. 2022, 31, 561–572. [Google Scholar] [CrossRef]
- Jeena, G.S.; Phukan, U.J.; Singh, N.; Joshi, A.; Pandey, A.; Sharma, Y.; Tripathi, V.; Shukla, R.K. AtERF60 negatively regulates ABR1 to modulate salt, drought, and basal resistance in Arabidopsis. bioRxiv 2021. [Google Scholar] [CrossRef]
- So, H.-A.; Chung, E.; Lee, J.-H. Arabidopsis atDjC53 encoding a type III J-protein plays a negative role in heat shock tolerance. Genes Genom. 2014, 36, 733–744. [Google Scholar] [CrossRef]
- Suzuki, N.; Rizhsky, L.; Liang, H.; Shuman, J.; Shulaev, V.; Mittler, R. Enhanced tolerance to environmental stress in transgenic plants expressing the transcriptional coactivator multiprotein bridging factor 1c. Plant Physiol. 2005, 139, 1313–1322. [Google Scholar] [CrossRef]
- Cominelli, E.; Galbiati, M.; Vavasseur, A.; Conti, L.; Sala, T.; Vuylsteke, M.; Leonhardt, N.; Dellaporta, S.L.; Tonelli, C. A guard-cell-specific MYB transcription factor regulates stomatal movements and plant drought tolerance. Curr. Biol. 2005, 15, 1196–1200. [Google Scholar] [CrossRef]
- Davletova, S.; Schlauch, K.; Coutu, J.; Mittler, R. The zinc-finger protein Zat12 plays a central role in reactive oxygen and abiotic stress signaling in Arabidopsis. Plant Physiol. 2005, 139, 847–856. [Google Scholar] [CrossRef] [PubMed]
- Allahverdiyeva, Y.; Suorsa, M.; Rossi, F.; Pavesi, A.; Kater, M.M.; Antonacci, A.; Tadini, L.; Pribil, M.; Schneider, A.; Wanner, G.; et al. Arabidopsis plants lacking PsbQ and PsbR subunits of the oxygen-evolving complex show altered PSII super-complex organization and short-term adaptive mechanisms. Plant J. 2013, 75, 671–684. [Google Scholar] [CrossRef]
- Sasi, S.; Venkatesh, J.; Daneshi, R.F.; Gururani, M.A. Photosystem II Extrinsic Proteins and Their Putative Role in Abiotic Stress Tolerance in Higher Plants. Plants 2018, 7, 100. [Google Scholar] [CrossRef]
- Faiza, T.; Iqbal, M.; Aqib, I. Over-expressed HSP17. 6B, encoding HSP20-like chaperones superfamily protein, confers heat stress tolerance in Arabidopsis thaliana. Pak. J. Bot. 2019, 51, 855–864. [Google Scholar]
- Guo, L.M.; Li, J.; He, J.; Liu, H.; Zhang, H.M. A class I cytosolic HSP20 of rice enhances heat and salt tolerance in different organisms. Sci. Rep. 2020, 10, 1383. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Kong, L.; Zhang, Y.; Liao, Y. Gene and Metabolite Integration Analysis through Transcriptome and Metabolome Brings New Insight into Heat Stress Tolerance in Potato (Solanum tuberosum L.). Plants 2021, 10, 103. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Ma, Z.; Chen, H.; Liu, M. MYB Gene Family in Potato (Solanum tuberosum L.): Genome-Wide Identification of Hormone-Responsive Reveals Their Potential Functions in Growth and Development. Int. J. Mol. Sci. 2019, 20, 4847. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]


| Transcript ID | FPKM | Log2 (Fold Change) | Homologous of Arabidopisis | FunctionalAnnotation | Type | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| CK ① | Salt | Mannitol | CK ② | Heat | Salt | Osmotic | Heat | ||||
| Soltu.DM.01G032700 | 0.01 | 10.42 | 6.82 | 7.39 | 0.01 | 10.03 | 9.41 | −9.53 | AT2G45660.1 | AGAMOUS-like 20 | TF |
| Soltu.DM.02G005400 | 4.24 | 9.25 | 11.38 | 24.45 | 0.28 | 1.13 | 1.42 | −6.47 | AT2G17040.1 | NAC domain containing protein 36 | TF |
| Soltu.DM.03G019380 | 7.30 | 16.39 | 43.31 | 17.69 | 0.21 | 1.17 | 2.57 | −6.36 | AT1G14440.2 | homeobox protein 31 | TF |
| Soltu.DM.10G011180 | 0.01 | 6.84 | 11.78 | 6.53 | 0.47 | 9.42 | 10.20 | −3.78 | AT1G58110.2 | Basic-leucine zipper (bZIP) transcription factor family protein | TF |
| Soltu.DM.03G033840 | 12.54 | 12.52 | 15.35 | 27.29 | 3.13 | 0.00 | 0.29 | −3.12 | AT1G73830.1 | BR enhanced expression 3 | TF |
| Soltu.DM.02G004750 | 4.18 | 4.70 | 7.29 | 11.18 | 2.69 | 0.17 | 0.80 | −2.06 | AT5G37020.1 | auxin response factor 8 | TF |
| Soltu.DM.02G006820 | 74.04 | 54.67 | 102.37 | 224.76 | 75.59 | −0.44 | 0.47 | −1.57 | AT1G75080.2 | Brassinosteroid signaling positive regulator (BZR1) family protein | TF |
| Soltu.DM.06G013280 | 9.46 | 13.22 | 21.68 | 51.89 | 17.67 | 0.48 | 1.20 | −1.55 | AT5G52510.1 | SCARECROW-like 8 | TF |
| Soltu.DM.03G013350 | 18.07 | 29.59 | 60.32 | 13.38 | 4.80 | 0.71 | 1.74 | −1.48 | AT3G56400.1 | WRKY DNA-binding protein 70 | TF |
| Soltu.DM.06G029100 | 98.07 | 90.66 | 93.74 | 123.78 | 45.82 | −0.11 | −0.07 | −1.43 | AT3G20770.1 | Ethylene insensitive 3 family protein | TF |
| Soltu.DM.04G027170 | 6.33 | 6.59 | 13.10 | 19.29 | 7.55 | 0.06 | 1.05 | −1.35 | AT5G44080.1 | Basic-leucine zipper (bZIP) transcription factor family protein | TF |
| Soltu.DM.02G004720 | 9.66 | 11.29 | 12.69 | 11.94 | 5.07 | 0.23 | 0.39 | −1.24 | AT5G37020.1 | auxin response factor 8 | TF |
| Soltu.DM.08G011740 | 17.52 | 46.81 | 72.52 | 126.37 | 54.59 | 1.42 | 2.05 | −1.21 | AT5G11270.1 | overexpressor of cationic peroxidase 3 | TF |
| Soltu.DM.01G003280 | 9.34 | 15.34 | 20.53 | 11.14 | 5.00 | 0.72 | 1.14 | −1.16 | AT2G02080.1 | indeterminate (ID)-domain 4 | TF |
| Soltu.DM.04G018230 | 12.95 | 23.50 | 24.89 | 60.41 | 27.20 | 0.86 | 0.94 | −1.15 | AT3G52250.1 | Duplicated homeodomain-like superfamily protein | TF |
| Soltu.DM.10G001700 | 13.12 | 31.28 | 35.32 | 34.66 | 17.76 | 1.25 | 1.43 | −0.96 | AT5G54680.1 | basic helix-loop-helix (bHLH) DNA-binding superfamily protein | TF |
| Soltu.DM.12G012870 | 7.11 | 12.35 | 20.92 | 29.44 | 15.99 | 0.80 | 1.56 | −0.88 | AT5G23090.4 | nuclear factor Y, subunit B13 | TF |
| Soltu.DM.01G027650 | 23.27 | 55.46 | 79.25 | 207.26 | 121.61 | 1.25 | 1.77 | −0.77 | AT3G08505.1 | zinc finger (CCCH-type/C3HC4-type RING finger) family protein | TF |
| Soltu.DM.03G023990 | 2.14 | 6.24 | 10.47 | 14.83 | 9.96 | 1.55 | 2.29 | −0.57 | AT4G32730.2 | Homeodomain-like protein | TF |
| Soltu.DM.08G023170 | 7.11 | 17.31 | 25.29 | 19.21 | 15.91 | 1.28 | 1.83 | −0.27 | AT5G45420.1 | Duplicated homeodomain-like superfamily protein | TF |
| Soltu.DM.10G029240 | 25.55 | 40.36 | 51.67 | 27.52 | 22.88 | 0.66 | 1.02 | −0.27 | AT5G06770.1 | KH domain-containing protein/zinc finger (CCCH type) family protein | TF |
| Soltu.DM.04G015330 | 5.38 | 19.64 | 32.32 | 59.65 | 50.36 | 1.87 | 2.59 | −0.24 | AT2G33550.1 | Homeodomain-like superfamily protein | TF |
| Soltu.DM.01G007500 | 35.41 | 88.80 | 131.85 | 183.06 | 154.98 | 1.33 | 1.90 | −0.24 | AT5G13180.1 | NAC domain containing protein 83 | TF |
| Soltu.DM.12G006760 | 5.54 | 16.15 | 13.16 | 20.90 | 30.15 | 1.54 | 1.25 | 0.53 | AT5G65070.1 | K-box region and MADS-box transcription factor family protein | TF |
| Soltu.DM.06G030750 | 7.58 | 18.00 | 12.37 | 6.11 | 11.06 | 1.25 | 0.71 | 0.86 | AT2G28510.1 | Dof-type zinc finger DNA-binding family protein | TF |
| Soltu.DM.02G010940 | 8.55 | 16.60 | 34.00 | 19.18 | 39.98 | 0.96 | 1.99 | 1.06 | AT3G27010.1 | TEOSINTE BRANCHED 1, cycloidea, PCF (TCP)-domain family protein 20 | TF |
| Soltu.DM.04G021630 | 96.79 | 304.78 | 316.91 | 477.46 | 1196.88 | 1.65 | 1.71 | 1.33 | AT4G39780.1 | Integrase-type DNA-binding superfamily protein | TF |
| Soltu.DM.04G002000 | 6.21 | 15.14 | 15.82 | 7.72 | 23.04 | 1.29 | 1.35 | 1.58 | AT1G25440.1 | B-box type zinc finger protein with CCT domain | TF |
| Soltu.DM.08G015040 | 5.29 | 10.68 | 9.98 | 3.50 | 15.45 | 1.01 | 0.92 | 2.14 | AT5G11590.1 | Integrase-type DNA-binding superfamily protein | TF |
| Soltu.DM.04G031370 | 0.79 | 2.68 | 1.07 | 4.67 | 40.22 | 1.76 | 0.42 | 3.11 | AT2G14210.1 | AGAMOUS-like 44 | TF |
| Soltu.DM.09G009470 | 1.05 | 3.60 | 23.70 | 46.41 | 2.74 | 1.78 | 4.50 | −4.08 | AT1G53540.1 | HSP20-like chaperones superfamily protein | HSP |
| Soltu.DM.01G039980 | 30.11 | 73.87 | 116.80 | 80.48 | 12.47 | 1.29 | 1.96 | −2.69 | AT1G56300.1 | Chaperone DnaJ-domain superfamily protein | HSP |
| Soltu.DM.04G008820 | 31.69 | 36.28 | 42.81 | 195.65 | 36.36 | 0.20 | 0.43 | −2.43 | AT5G37670.1 | HSP20-like chaperones superfamily protein | HSP |
| Soltu.DM.01G041960 | 0.56 | 5.19 | 10.14 | 13.27 | 3.63 | 3.22 | 4.19 | −1.87 | AT4G10250.1 | HSP20-like chaperones superfamily protein | HSP |
| Soltu.DM.03G023440 | 11.95 | 21.31 | 39.52 | 117.25 | 33.65 | 0.83 | 1.73 | −1.80 | AT5G52640.1 | heat shock protein 90.1 | HSP |
| Soltu.DM.08G025000 | 12.16 | 14.04 | 16.68 | 18.35 | 5.47 | 0.21 | 0.46 | −1.75 | AT5G47590.1 | Heat shock protein HSP20/alpha crystallin family | HSP |
| Soltu.DM.06G031880 | 1.28 | 4.90 | 14.30 | 14.85 | 4.87 | 1.93 | 3.48 | −1.61 | AT1G07400.1 | HSP20-like chaperones superfamily protein | HSP |
| Soltu.DM.07G004660 | 145.00 | 98.96 | 129.47 | 114.99 | 52.36 | −0.55 | −0.16 | −1.13 | AT1G14980.1 | chaperonin 10 | HSP |
| Soltu.DM.08G025550 | 65.78 | 89.42 | 99.43 | 154.44 | 76.63 | 0.44 | 0.60 | −1.01 | AT4G22670.1 | HSP70-interacting protein 1 | HSP |
| Soltu.DM.04G001130 | 5.38 | 16.50 | 10.40 | 19.10 | 14.06 | 1.62 | 0.95 | −0.44 | AT3G08910.1 | DNAJ heat shock family protein | HSP |
| Soltu.DM.06G032250 | 20.15 | 51.01 | 66.49 | 45.28 | 48.49 | 1.34 | 1.72 | 0.10 | AT5G58740.1 | HSP20-like chaperones superfamily protein | HSP |
| Soltu.DM.03G022580 | 23.21 | 53.31 | 34.51 | 51.15 | 57.33 | 1.20 | 0.57 | 0.16 | AT5G53400.1 | HSP20-like chaperones superfamily protein | HSP |
| Soltu.DM.07G027620 | 914.03 | 2243.58 | 2560.85 | 4803.50 | 5901.53 | 1.30 | 1.49 | 0.30 | AT5G56000.1 | HEAT SHOCK PROTEIN 81.4 | HSP |
| Soltu.DM.05G025710 | 1122.25 | 1514.19 | 2365.67 | 3477.91 | 4490.32 | 0.43 | 1.08 | 0.37 | AT5G22060.1 | DNAJ homologue 2 | HSP |
| Soltu.DM.12G026320 | 5.16 | 31.78 | 32.64 | 31.73 | 53.42 | 2.62 | 2.66 | 0.75 | AT1G53540.1 | HSP20-like chaperones superfamily protein | HSP |
| Soltu.DM.06G031840 | 25.66 | 61.71 | 80.93 | 122.40 | 212.18 | 1.27 | 1.66 | 0.79 | AT1G07400.1 | HSP20-like chaperones superfamily protein | HSP |
| Soltu.DM.01G049730 | 7.02 | 17.61 | 29.55 | 12.56 | 22.20 | 1.33 | 2.07 | 0.82 | AT4G39150.2 | DNAJ heat shock N-terminal domain-containing protein | HSP |
| Soltu.DM.04G032140 | 4.12 | 13.02 | 13.34 | 10.61 | 20.67 | 1.66 | 1.69 | 0.96 | AT4G07990.1 | Chaperone DnaJ-domain superfamily protein | HSP |
| Soltu.DM.11G019990 | 22.76 | 45.21 | 43.24 | 9.09 | 24.46 | 0.99 | 0.93 | 1.43 | AT5G19855.1 | Chaperonin-like RbcX protein | HSP |
| Soltu.DM.01G001660 | 58.79 | 130.16 | 142.28 | 49.45 | 137.02 | 1.15 | 1.28 | 1.47 | AT2G34860.2 | DnaJ/Hsp40 cysteine-rich domain superfamily protein | HSP |
| Soltu.DM.04G036440 | 64.68 | 122.78 | 83.19 | 11.34 | 77.58 | 0.92 | 0.36 | 2.77 | AT1G75690.1 | DnaJ/Hsp40 cysteine-rich domain superfamily protein | HSP |
| Soltu.DM.07G023770 | 21.39 | 13.16 | 24.89 | 25.42 | 12.33 | −0.70 | 0.22 | −1.04 | AT1G16670.1 | Protein kinase superfamily protein | Kinase |
| Soltu.DM.11G007950 | 13.36 | 19.69 | 20.75 | 39.74 | 4.01 | 0.56 | 0.63 | −3.31 | AT1G66150.1 | transmembrane kinase 1 | Kinase |
| Soltu.DM.01G037020 | 21.50 | 35.83 | 72.27 | 25.57 | 15.55 | 0.74 | 1.75 | −0.72 | AT5G58140.1 | phototropin 2 | Kinase |
| Soltu.DM.01G026590 | 14.58 | 24.37 | 26.87 | 15.18 | 5.15 | 0.74 | 0.88 | −1.56 | AT3G51990.1 | Protein kinase superfamily protein | Kinase |
| Soltu.DM.12G027440 | 59.20 | 101.21 | 125.91 | 23.31 | 31.84 | 0.77 | 1.09 | 0.45 | AT4G30960.1 | SOS3-interacting protein 3 | Kinase |
| Soltu.DM.08G023690 | 11.93 | 35.87 | 45.58 | 47.01 | 24.58 | 1.59 | 1.93 | −0.94 | AT4G33950.1 | Protein kinase superfamily protein | Kinase |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, K.; Wen, S.; Shang, L.; Li, Y.; Li, Z.; Chen, W.; Li, Y.; Jian, H.; Lyu, D. Rapid Identification of High-Temperature Responsive Genes Using Large-Scale Yeast Functional Screening System in Potato. Plants 2023, 12, 3712. https://doi.org/10.3390/plants12213712
Wang K, Wen S, Shang L, Li Y, Li Z, Chen W, Li Y, Jian H, Lyu D. Rapid Identification of High-Temperature Responsive Genes Using Large-Scale Yeast Functional Screening System in Potato. Plants. 2023; 12(21):3712. https://doi.org/10.3390/plants12213712
Chicago/Turabian StyleWang, Ke, Shiqi Wen, Lina Shang, Yang Li, Ziyan Li, Weixi Chen, Yong Li, Hongju Jian, and Dianqiu Lyu. 2023. "Rapid Identification of High-Temperature Responsive Genes Using Large-Scale Yeast Functional Screening System in Potato" Plants 12, no. 21: 3712. https://doi.org/10.3390/plants12213712





