Freeze Substitution Accelerated via Agitation: New Prospects for Ultrastructural Studies of Lichen Symbionts and Their Extracellular Matrix
Abstract
:1. Introduction
2. Results
2.1. FS under Continuous Agitation—A Means to Cryopreserve Lichens for TEM
2.2. Improvement in Section Quality by Using an Ultrasonic Diamond Knife
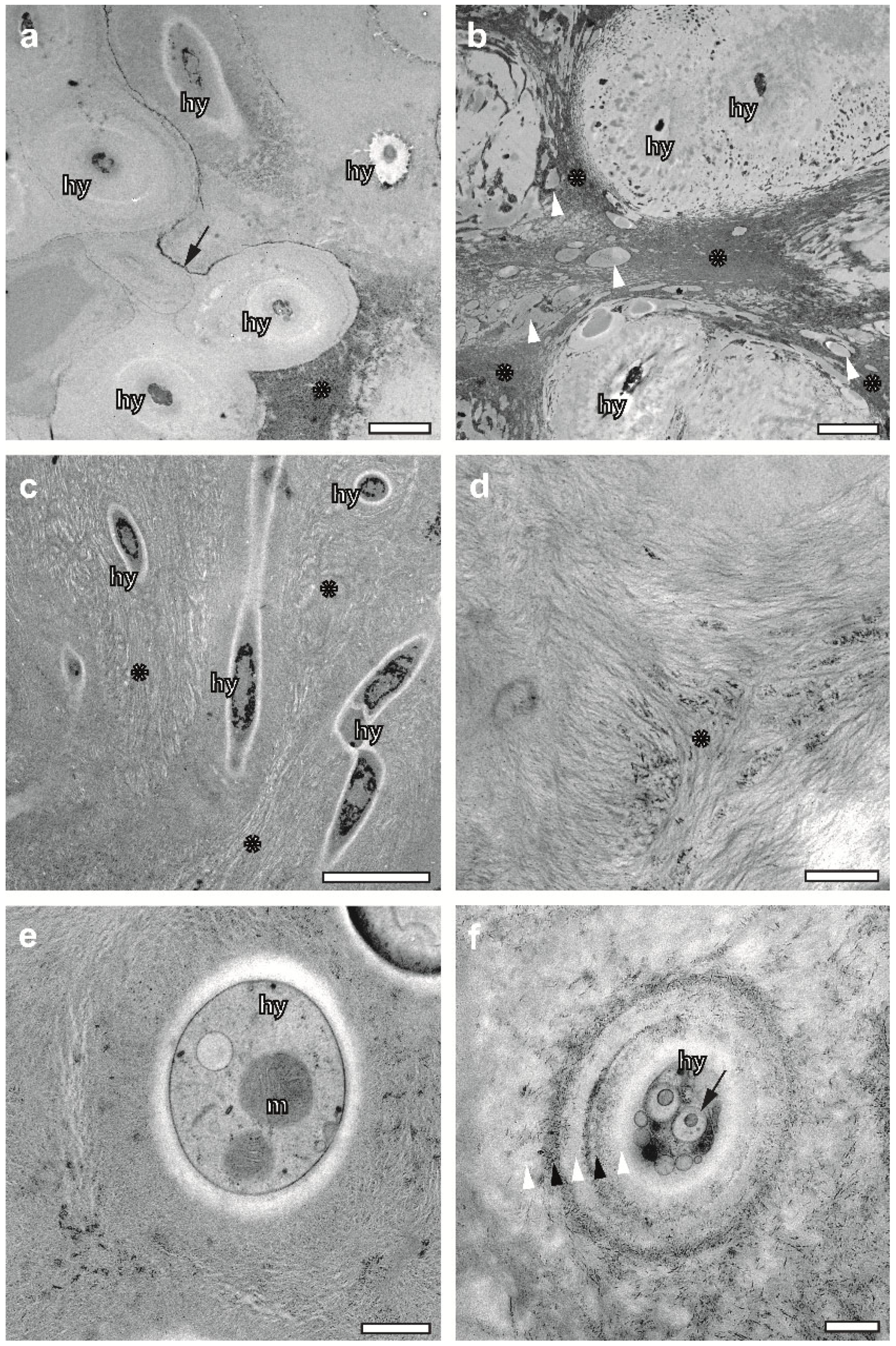
2.3. Cryoimmobilized Hyphal/Algal Junction in X. parietina Displays Patterns of Intraparietal Cell Wall Contact
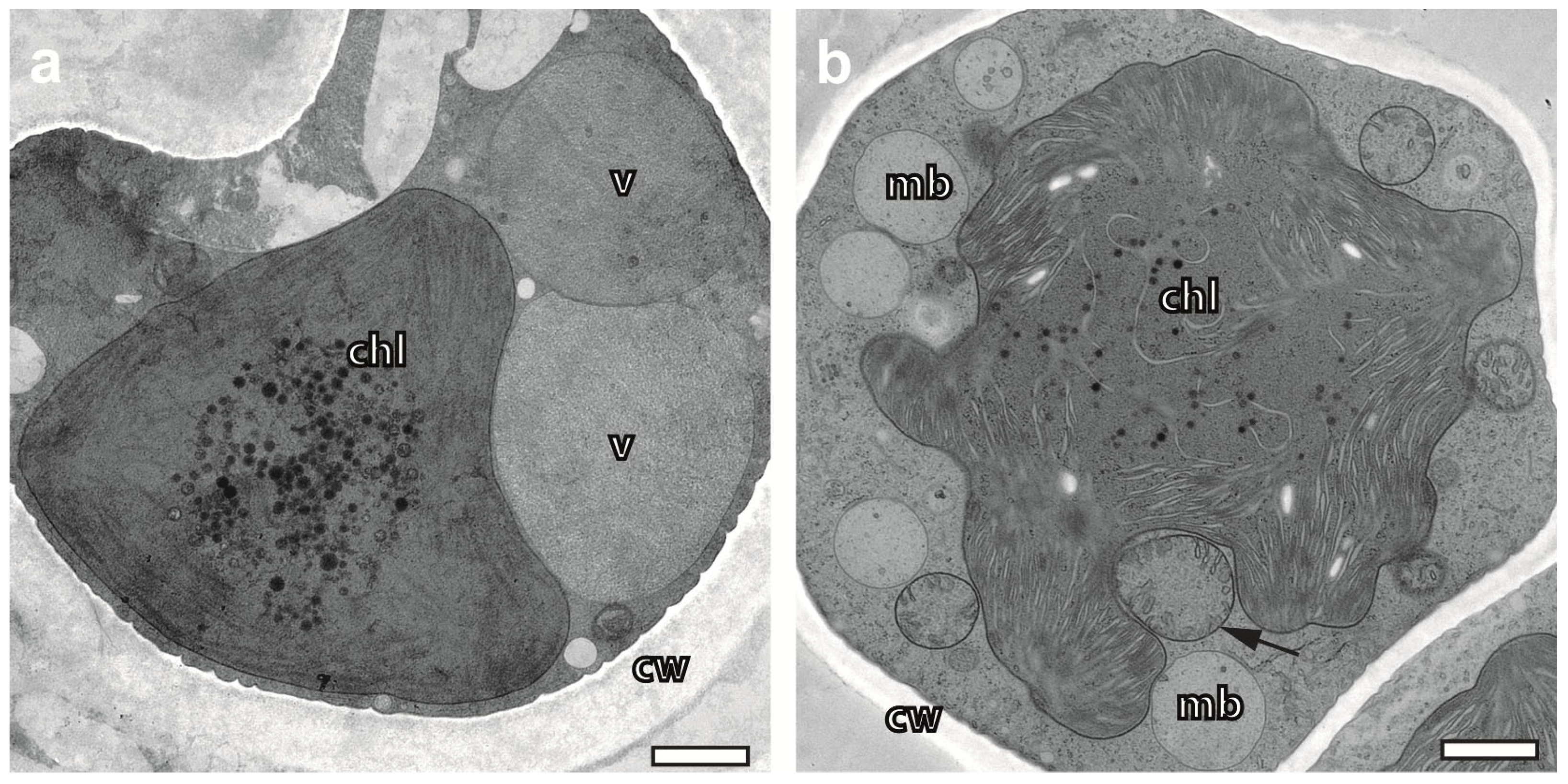
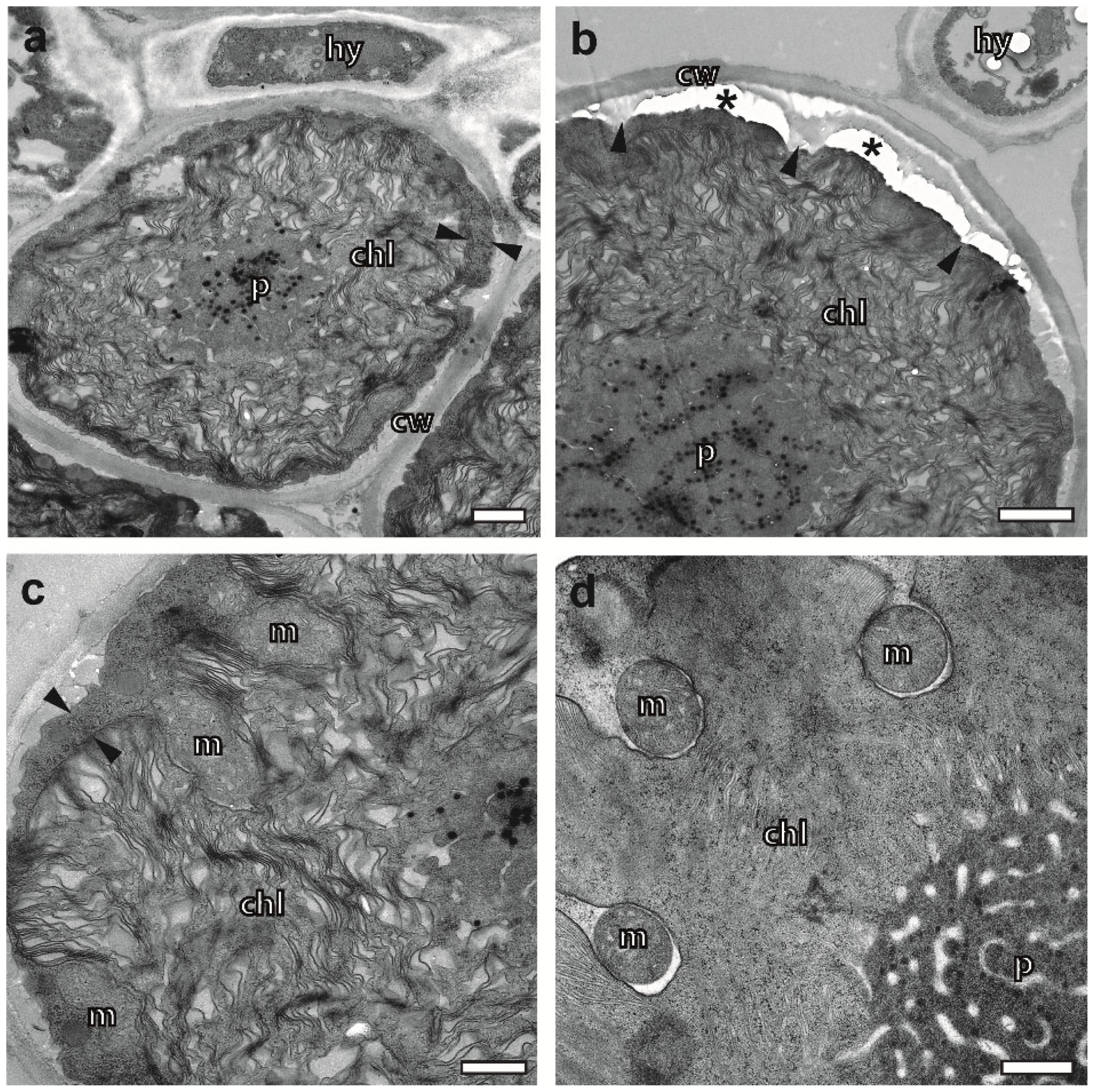
2.4. Cryopreparation Suits the Preservation of Photobionts at Various Degrees of Hydration
2.5. FS Enables Superb Preservation of the ECM in Association with Hyphae
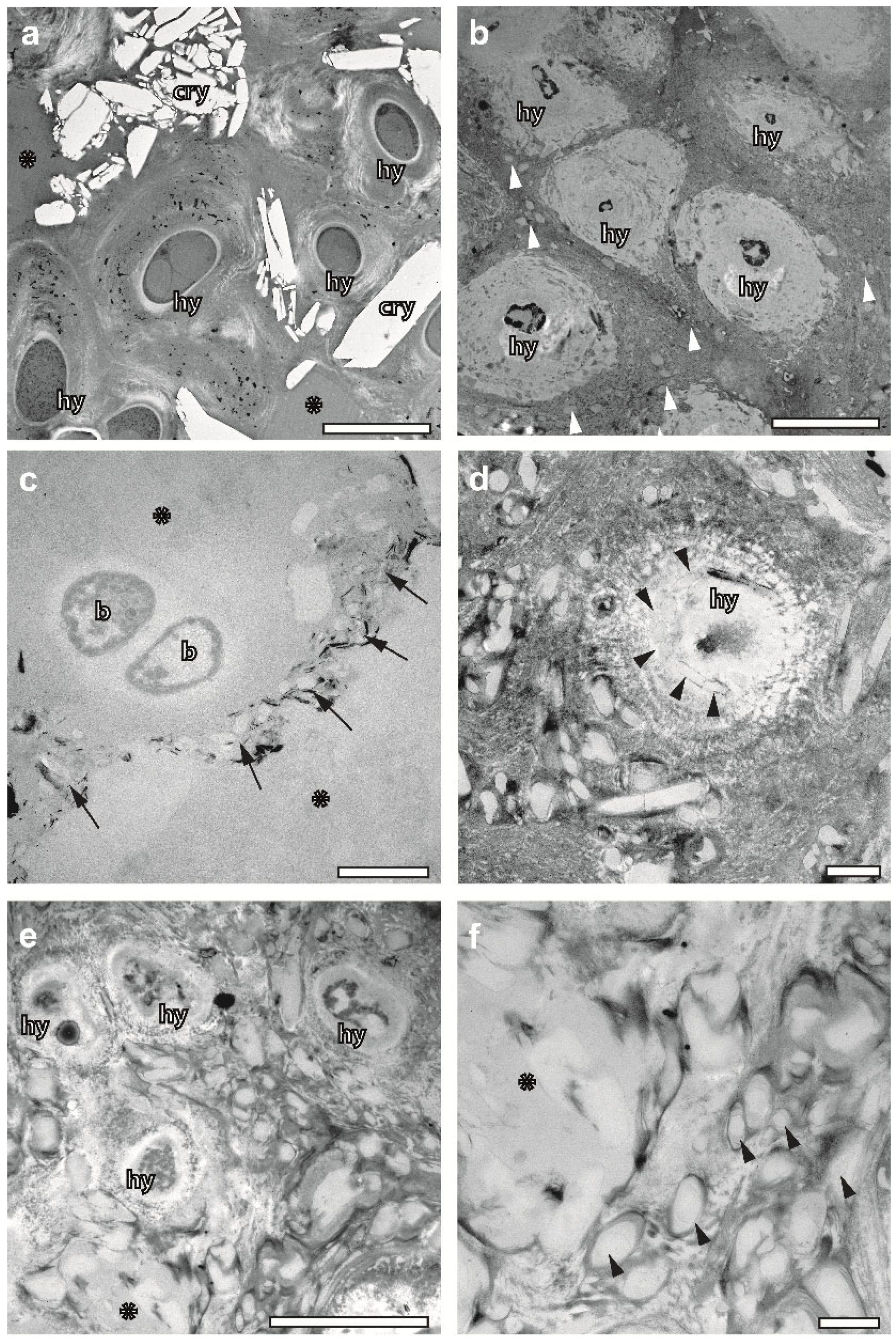
2.6. FS Draws Attention on Structural Diversity of Hyphae
2.7. FS Preserves Crystalline Metabolites inside the Thallus
3. Discussion
4. Materials and Methods
4.1. Sample Collection
4.2. Chemical Fixation and Embedding at Room Temperature
4.3. Freezing
4.4. Freeze Substitution under Agitation
4.5. Light Microscopy
4.6. Sectioning
4.7. Contrasting and TEM
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Molins, A.; Moya, P.; García-Breijo, F.J.; Reig-Armiñana, J.; Barreno, E. A multi-tool approach to assess microalgal diversity in lichens: Isolation, Sanger sequencing, HTS and ultrastructural correlations. Lichenologist 2018, 50, 123–138. [Google Scholar] [CrossRef]
- Català, S.; Campo, E.D.; Barreno, E.; García-Breijo, F.; Reig-Armiñana, J.; Casano, L. Coordinated ultrastructural and phylogenomic analyses shed light on the hidden phycobiont diversity of Trebouxia microalgae in Ramalina Fraxinea. Mol. Phylogenet. Evol. 2016, 94, 765–777. [Google Scholar] [CrossRef]
- Muggia, L.; Perez-Ortega, S.; Kopun, T.; Zellnig, G.; Grube, M. Photobiont selectivity leads to ecological tolerance and evolutionary divergence in a polymorphic complex of lichenized fungi. Ann. Bot. 2014, 114, 463–475. [Google Scholar] [CrossRef]
- De los Ríos, A.; Ascaso, C. Preparative techniques for transmission electron microscopy and confocal laser scanning microscopy of lichens. In Springer Lab Manuals Protocols in Lichenology; Kranner, I.C., Beckett, R.P., Varma, A.K., Eds.; Springer: Berlin/Heidelberg, Germany, 2002; pp. 87–117. [Google Scholar]
- Brown, D.H.; Rapsch, S.; Beckett, A.; Ascaso, C. The effect of desiccation on cell shape in the lichen Parmelia slucata taylor. New Phytol. 1987, 105, 295–299. [Google Scholar] [CrossRef]
- Honegger, R. Ultrastructural studies in lichens I. Haustorial types and their frequencies in a range of lichens with trebouxioid photobionts. New Phytol. 1986, 103, 785–795. [Google Scholar] [CrossRef]
- Honegger, R. The symbiotic phenotype of lichen-forming ascomycetes and their endo- and epibionts. In Fungal Associations; Hock, B., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 287–339. [Google Scholar]
- Casano, L.M.; del Campo, E.M.; García-Breijo, F.J.; Reig-Armiñana, J.; Gasulla, F.; Del Hoyo, A.; Guéra, A.; Barreno, E. Two Trebouxia algae with different physiological performances are ever-present in lichen thalli of Ramalina farinacea. Coexistence versus competition? Environ. Microbiol. 2011, 13, 806–818. [Google Scholar] [CrossRef]
- Spagnuolo, V.; Zampella, M.; Giordano, S.; Adamo, P. Cytological stress and element uptake in moss and lichen exposed in bags in urban area. Ecotoxicol. Environ. Saf. 2011, 74, 1434–1443. [Google Scholar] [CrossRef]
- Sanders, W.B.; de Los Ríos, A. Development of thallus axes in Usnea longissima (Parmeliaceae, Ascomycota), a fruticose lichen showing diffuse growth. Am. J. Bot. 2012, 99, 998–1009. [Google Scholar] [CrossRef]
- Molins, A.; García-Breijo, F.J.; Reig-Armiñana, J.; del Campo, E.M.; Casano, L.; Barreno, E. Coexistence of different intrathalline symbiotic algae and bacterial biofilms in the foliose Canarian lichen Parmotrema Pseudotinctorum. Vieraea. Folia Sci. Biol. Canar. 2013, 41, 349–370. [Google Scholar] [CrossRef]
- Škaloud, P.; Moya, P.; Molins, A.; Peksa, P.; Santos-Guerra, A.; Barreno, E. Untangling the hidden intrathalline microalgal diversity in Parmotrema pseudotinctorum: Trebouxia crespoana sp. nov. Lichenologist 2018, 50, 357–369. [Google Scholar] [CrossRef]
- Moya, P.; Škaloud, P.; Chiva, S.; García-Breijo, F.J.; Reig-Arminana, J.; Vančurová, L.; Barreno, E. Molecular phylogeny and ultrastructure of the lichen microalga Asterochloris mediterranea sp. nov. from Mediterranean and Canary Islands ecosystems. Int. J. Syst. Evol. Microbiol. 2015, 65, 1838–1854. [Google Scholar] [CrossRef] [PubMed]
- Sanders, W.B.; de los Ríos, A. Structure and in situ development of the microlichen Gyalectidium paolae (Gomphillaceae, Ascomycota), an overlooked colonist on palm leaves in southwest Florida. Am. J. Bot. 2015, 102, 1403–1412. [Google Scholar] [CrossRef] [PubMed]
- Vannini, A.; Guarnieri, M.; Paoli, L.; Sorbo, S.; Basile, A.; Loppi, S. Bioaccumulation, physiological and ultrastructural effects of glyphosate in the lichen Xanthoria parietina (L.) Th. Fr. Chemosphere 2016, 164, 233–240. [Google Scholar] [CrossRef]
- Vannini, A.; Paoli, L.; Ceccarelli, S.; Sorbo, S.; Basile, A.; Carginale, V.; Nali, C.; Lorenzini, G.; Pica, M.; Loppi, S. Physiological and ultrastructural effects of acute ozone fumigation in the lichen Xanthoria parietina: The role of parietin and hydration state. Environ. Sci. Pollut. Res. Int. 2018, 25, 8104–8112. [Google Scholar] [CrossRef]
- Lorenz, C.; Bianchi, E.; Poggiali, G.; Alemanno, G.; Benesperi, R.; Brucato, J.R.; Garland, S.; Helbert, J.; Loppi, S.; Lorek, A.; et al. Survivability of the lichen Xanthoria parietina in simulated Martian environmental conditions. Sci. Rep. 2023, 13, 4893. [Google Scholar] [CrossRef] [PubMed]
- Brandt, A.; Posthoff, E.; de Vera, J.-P.; Onofri, S.; Ott, S. Characterisation of growth and ultrastructural effects of the Xanthoria elegans photobiont after 1.5 years of space exposure on the international space station. Orig. Life Evol. Biosph. 2016, 46, 311–321. [Google Scholar] [CrossRef]
- Sanders, W.B.; de Los Ríos, A. Parenchymatous cell division characterizes the fungal cortex of some common foliose lichens. Am. J. Bot. 2017, 104, 207–217. [Google Scholar] [CrossRef] [PubMed]
- De la Torre Noetzel, R.; Ortega Garcia, M.V.; Miller, A.Z.; Bassy, O.; Granja, C.; Cubero, B.; Jordão, L.; Martínez Frías, J.; Rabbow, E.; Backhaus, T.; et al. Lichen vitality after a space flight on board the EXPOSE-R2 facility outside the international space station: Results of the biology and Mars experiment. Astrobiology 2020, 20, 583–600. [Google Scholar] [CrossRef]
- Daminova, A.G.; Rogov, A.M.; Rassabina, A.E.; Beckett, R.P.; Minibayeva, F.V. Effect of melanization on thallus microstructure in the lichen Lobaria pulmonaria. J. Fungi 2022, 8, 791. [Google Scholar] [CrossRef]
- McDonald, K. Cryopreparation methods for electron microscopy of selected model systems. Methods Cell Biol. 2007, 79, 23–56. [Google Scholar]
- Humbel, B.M. Freeze substitution. In Hand Book of Cryo-Preparation Methods for Electron Microscopy; Cavalier, A., Spehner, D., Humbel, B.M., Eds.; CRC Press: Boca Raton, FL, USA, 2009; pp. 319–341. [Google Scholar]
- Hurbain, I.; Sachse, M. The future is cold: Cryo-preparation methods for transmission electron microscopy of cells. Biol. Cell 2011, 103, 405–420. [Google Scholar] [CrossRef] [PubMed]
- Honegger, R.; Peter, M. Routes of solute translocation and the location of water in heteromerous lichens visualized with cryo-techniques in light and electron microscopy. Symbiosis 1994, 16, 167–186. [Google Scholar]
- Honegger, R.; Peter, M.; Scherrer, S. Drought-induced structural alterations at the mycobiont-photobiont interface in a range of foliose macrolichens. Protoplasma 1996, 190, 221–232. [Google Scholar] [CrossRef]
- Honegger, R.; Edwards, D.; Lindsey, A. The earliest records of internally stratified cyanobacterial and algal lichens from the Lower Devonian of the Welsh Borderland. New Phytol. 2013, 197, 264–275. [Google Scholar] [CrossRef]
- Sanders, W.B.; Masumoto, H. Lichen algae: The photosynthetic partners in lichen symbioses. Lichenologist 2021, 53, 347–393. [Google Scholar] [CrossRef]
- Sluiman, H.J.; Lokhorst, G.M. The ultrastructure of cellular division (autosporogenesis) in the coccoid green alga, Trebouxia aggregata, revealed by rapid freeze fixation and freeze substitution. Protoplasma 1988, 144, 149–159. [Google Scholar] [CrossRef]
- Barreno, E.; Muggia, L.; Chiva, S.; Molins, A.; Bordenave, C.; García-Breijo, F.; Moya, P. Trebouxia lynnae sp. nov. (former Trebouxia sp.TR9): Biology and biogeography of an epitome lichen symbiotic microalga. Biology 2022, 11, 1196. [Google Scholar] [CrossRef]
- González-Hourcade, M.; Braga, M.R.; del Campo, E.M.; Ascaso, C.; Patiño, C.; Casano, L.M. Ultrastructural and biochemical analyses reveal cell wall remodelling in lichenforming microalgae submitted to cyclic desiccation–rehydration Ann. Bot. 2020, 125, 459–469. [Google Scholar]
- Feng, J.-J.; He, C.-Y.; Jiang, S.-H.; Zhang, T.; Yu, L.-Y. Saccharomycomorpha psychra n. g., n. sp., a novel member of Glissmonadida (Cercozoa) isolated from Arctic and Antarctica. J. Eukaryot. Microbiol. 2021, 68, e12840. [Google Scholar] [CrossRef]
- McDonald, K.L.; Webb, R.I. Freeze substitution in 3 hours or less. J. Microsc. 2011, 243, 227–233. [Google Scholar] [CrossRef]
- Goldammer, H.; Hollergschwandtner, E.; Elisabeth, N.H.; Frade, P.R.; Reipert, S. Automatized freeze substitution of algae accelerated by a novel agitation module. Protist 2016, 167, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Reipert, S.; Goldammer, H.; Richardson, C.; Goldberg, M.W.; Hawkins, T.J.; Hollergschwandtner, E.; Kaufmann, W.A.; Antreich, S.; Stierhof, Y.D. Agitation modules: Flexible means to accelerate automated freeze substitution. J. Histochem. Cytochem. 2018, 66, 903–921. [Google Scholar] [CrossRef]
- Bordenave, C.D.; García-Breijo, F.; Gazquez, A.; Muggia, L.; Carrasco, P.; Barreno, E. Low Temperature Scanning Electron Microscopy (LTSEM) Findings on the Ultrastructure of Trebouxia lynnae (Trebouxiophyceae, Lichenized Microalgae). Diversity 2023, 15, 170. [Google Scholar] [CrossRef]
- Roth, R.; Wagner, R.; Goodenough, U. Lichen 3. Outer layers. Algal Res. 2021, 56, 102332. [Google Scholar] [CrossRef]
- Spribille, T.; Tagirdzhanova, G.; Goyette, S.; Tuovinen, V.; Case, R.; Zandberg, W.F. 3D biofilms: In search of the polysaccharides holding together lichen symbioses. FEMS Microbiol. Lett. 2020, 367, fnaa023. [Google Scholar] [CrossRef] [PubMed]
- Grimm, M.; Grube, M.; Schiefelbein, U.; Zühlke, D.; Bernhardt, J.; Riedel, K. The Lichens’ Microbiota, still a mystery? Front. Microbiol. Sec. Syst. Microbiol. 2021, 12, 2021. [Google Scholar] [CrossRef]
- Zvěřina, O.; Coufalík, P.; Barták, M.; Petrov, M.; Komárek, J. The contents and distributions of cadmium, mercury, and lead in Usnea antarctica lichens from Solorina Valley, James Ross Island (Antarctica). Environ. Monit. Assess 2018, 190, 13. [Google Scholar] [CrossRef]
- Studer, D.; Gnaegi, H. Minimal compression of ultrathin sections with use of an oscillating diamond knife. J. Microsc. 2000, 197, 94–100. [Google Scholar] [CrossRef]
- Goodenough, U.; Wagner, R.; Roth, R. Lichen 4. The algal layer. Algal Res. 2021, 58, 102355. [Google Scholar] [CrossRef]
- Honegger, R.; Haisch, A. Immunocytochemical location of the (1→3) (1→4)-β-glucan lichenin in the lichen-forming ascomycete Cetraria islandica (Icelandic moss). New Phytol. 2001, 150, 739–746. [Google Scholar] [CrossRef]
- Honegger, R. Cytological aspects of the mycobiont-phycobiont relationship in lichens. Lichenologist 1984, 16, 111–127. [Google Scholar] [CrossRef]
- Goodenough, U.; Roth, R. Lichen 5. Medullary and bacterial biofilm layers. Algal Res. 2021, 58, 102333. [Google Scholar] [CrossRef]
- Bordenave, C.D.; Muggia, L.; Chiva, S.; Leavitt, S.D.; Carrasco, P.; Barreno, E. Chloroplast morphology and pyrenoid ultrastructural analyses reappraise the diversity of the lichen phycobiont genus Trebouxia (Chlorophyta). Algal Res. 2022, 61, 102561. [Google Scholar] [CrossRef]
- Sánchez, F.J.; Meeßen, J.; del Carmen Ruiz, M.; Leopoldo, G.; Ott, S.; Vílchez, C.; Horneck, G.; Sadowsky, A.; de la Torre, R. UV-C tolerance of symbiotic Trebouxia sp. in the space-tested lichen species Rhizocarpon geographicum and Circinaria gyrosa: Role of the hydration state and cortex/screening substances. Int. J. Astrobiol. 2014, 13, 1–18. [Google Scholar] [CrossRef]
- Ball, P. Water is an active matrix of life for cell and molecular biology. Proc. Natl. Acad. Sci. USA 2017, 114, 13327–13335. [Google Scholar] [CrossRef]
- Honegger, R. Chapter 9: Water relations in lichens. In Fungi in the Environment; Gadd, G.M., Watkinson, S.C., Dyer, P., Eds.; Cambridge University Press: Cambridge, UK, 2007. [Google Scholar]
- Gielwanowska, I.; Olech, M. New ultrastructural and physiological features of the thallus in antartic lichens. Acta Biol. Cracoviensia Ser. Bot. 2012, 54, 40–52. [Google Scholar]
- Hosiner, D.; Sponder, G.; Graschopf, A.; Reipert, S.; Schweyen, R.J.; Schüller, C.; Aleschko, M. Pun1p is a metal ion-inducible, calcineurin/Crz1p-regulated plasma membrane protein required for cell wall integrity. Biochim. Biophys. Acta 2011, 1808, 1108–1119. [Google Scholar] [CrossRef]
- Meeßen, J.; Backhaus, T.; Brandt, A.; Raguse, M.; Böttger, U.; de Vera, J.P.; de la Torre, R. The effect of high-dose ionizing radiation on the isolated photobiont of the astrobiological model lichen Circinaria gyrosa. Astrobiology 2017, 17, 15–4162. [Google Scholar] [CrossRef]
- Gielwanowska, I.; Szczuka, E. New ultrastructural features of organelles in leaf cells of Deschampsia antarctica Desv. Polar Biol. 2005, 28, 951–955. [Google Scholar] [CrossRef]
- Yamane, K.; Oi, T.; Enomoto, S.; Nakao, T.; Arai, S.; Miyake, H.; Taniguchi, M. Three-dimensional ultrastructure of chloroplast pockets formed under salinity stress. Plant Cell Environ. 2018, 41, 563–575. [Google Scholar] [CrossRef]
- Beckett, R.P.; Kranner, I.; Minibayeva, F.V. Stress physiology and the symbiosis. In Lichen Biology, 2nd ed.; Thomas, H., Nash, T.H., Eds.; Cambridge University Press: Cambridge, UK, 2008; pp. 134–151. [Google Scholar]
- De Carolis, R.; Cometto, A.; Moya, P.; Barreno, E.; Grube, M.; Tretiach, M.; Leavitt, S.D.; Muggia, L. Photobiont diversity in lichen symbioses from extreme environments. Front. Microbiol. 2022, 13, 809804. [Google Scholar] [CrossRef] [PubMed]
- Hinojosa-Vidal, E.; Marco, F.; Martínez-Alberola, F.; Escaray, F.J.; García-Breijo, F.J.; Reig-Armiñana, J.; Carrasco, P.; Barreno, E. Characterization of the responses to saline stress in the symbiotic green microalga Trebouxia sp. TR9. Planta 2018, 248, 1473–1486. [Google Scholar] [CrossRef] [PubMed]
- De los Ríos, A.; Sancho, L.G.; Grube, M.; Wierzchos, J.; Ascaso, C. Endolithic growth of two Lecidea lichens in granite from continental Antarctica detected by molecular and microscopy techniques. New Phytol. 2005, 165, 181–190. [Google Scholar] [CrossRef]
- Böttger, U.; Meeßen, J.; Martinez-Frias, J.; Hübers, H.W.; Rull, F.; Sánchez, F.J.; De La Torre, R.; De Vera, J.P. Raman spectroscopic analysis of the calcium oxalate producing extremotolerant lichen Circinaria gyrosa. Int. J. Astrobiol. 2013, 13, 19–27. [Google Scholar] [CrossRef]
- Nakata, P.A. Engineering calcium oxalate crystal formation in Arabidopsis. Plant Cell Physiol. 2012, 53, 1275–1282. [Google Scholar] [CrossRef]
- Perez-Leblic, M.I.; Reyes, F.; Martinez, M.J.; Lahoz, R. Cell wall degradation in the autolysis of filamentous fungi. Mycopathologia 1982, 80, 147–155. [Google Scholar] [CrossRef]
- Kalinowska, R.; Bačkor, M.; Pawlik-Skowrońska, B. Parietin in the tolerant lichen Xanthoria parietina (L.) Th. Fr. increases protection of Trebouxia photobionts from cadmium excess. Ecol. Indic. 2015, 58, 132–138. [Google Scholar] [CrossRef]
- Balarinová, K.; Váczi, P.; Barták, M.; Hazdrová, J.; Forbelská, M. Temperature-dependent growth rate and phytosynthetic performance of Antarctic symbotic alga Trebouxia sp. cultivated in a bioreactor. Czech Polar Rep. 2013, 3, 19–27. [Google Scholar] [CrossRef]
- Sohrabi, M. Taxonomy and Phylogeny of the Manna Lichens and Allied Species (Mewgasporaceae). Ph.D. Thesis, University of Helsinki, Helsinki, Finland, 2012. [Google Scholar]
- Crespo, A.; Barreno, E. About the earthling communities of vagrant lichens (Sphaerothalia, Xanthoparmelion vagantis al. Nova). Acta Bot. Malacit. 1978, 4, 55–62. [Google Scholar] [CrossRef]
- Sánchez, F.J.; Mateo-Martí, E.; Raggio, J.; Meeßen, J.; Martínez-Frías, J.; Sancho, L.G.; Ott, S.; de la Torre, R. The resistance of the lichen Circinaria gyrosa (nom. provis.) towards simulated Mars conditions—A model test for the survival capacity of an eukaryotic extremophile. Planet. Space Sci. 2012, 72, 102–110. [Google Scholar] [CrossRef]
- Barták, M.; Hájek, J.; Očenášová, P. Photoinhibition of photosynthesis in Antarctic lichen Usnea antarctica. I. Light intensity- and light duration-dependent changes in functioning of photosystem II. Czech Polar Rep. 2012, 2, 42–51. [Google Scholar] [CrossRef]
- Kuipers, J.; Giepmans, B.N.G. Neodymium as an alternative contrast for uranium in electron microscopy. Histochem. Cell Biol. 2020, 153, 271–277. [Google Scholar] [CrossRef]


| Short FS under agitation | Long FS under agitation |
| HPF sample transfer onto liquid N2-frozen substitution medium (1% OsO4 in acetone) in Sarstedt tubes | |
| Insertion of the filled tubes in the agitation module within the precooled AFS (ca. −104 °C programmed) | |
| FS for 10 h at−85 °C | FS for 60 h at−85 °C |
| Warming up slope (5°−6 °C/h) | |
| −90 °C for 2 h | |
| Warming up slope (15 °C/h) | |
| 60 °C for 2 h | |
| Warming up slope to room temperature (20 °C/h) | |
| 1 h room temperature (to enhance contrast by OsO4) | |
| Removal of the Sarstedt tubes from the AFS for washing and embedding of samples at room temperature | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reipert, S.; Gruber, D.; Cyran, N.; Schmidt, B.; de la Torre Noetzel, R.; Sancho, L.G.; Goga, M.; Bačkor, M.; Schmidt, K. Freeze Substitution Accelerated via Agitation: New Prospects for Ultrastructural Studies of Lichen Symbionts and Their Extracellular Matrix. Plants 2023, 12, 4039. https://doi.org/10.3390/plants12234039
Reipert S, Gruber D, Cyran N, Schmidt B, de la Torre Noetzel R, Sancho LG, Goga M, Bačkor M, Schmidt K. Freeze Substitution Accelerated via Agitation: New Prospects for Ultrastructural Studies of Lichen Symbionts and Their Extracellular Matrix. Plants. 2023; 12(23):4039. https://doi.org/10.3390/plants12234039
Chicago/Turabian StyleReipert, Siegfried, Daniela Gruber, Norbert Cyran, Brigitte Schmidt, Rosa de la Torre Noetzel, Leopoldo G. Sancho, Michal Goga, Martin Bačkor, and Katy Schmidt. 2023. "Freeze Substitution Accelerated via Agitation: New Prospects for Ultrastructural Studies of Lichen Symbionts and Their Extracellular Matrix" Plants 12, no. 23: 4039. https://doi.org/10.3390/plants12234039
APA StyleReipert, S., Gruber, D., Cyran, N., Schmidt, B., de la Torre Noetzel, R., Sancho, L. G., Goga, M., Bačkor, M., & Schmidt, K. (2023). Freeze Substitution Accelerated via Agitation: New Prospects for Ultrastructural Studies of Lichen Symbionts and Their Extracellular Matrix. Plants, 12(23), 4039. https://doi.org/10.3390/plants12234039







