Lifestyle Risk Factors for Weight Gain in Children with and without Asthma
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. Clinical Assessment
2.3. Anthropometry
2.4. Polysomnography
2.5. Dietary and Physical Activity Assessment
2.6. Metabolic Outcomes
2.7. Statistical Analysis
3. Results
3.1. Sleep Outcomes
3.2. Metabolic Outcomes
3.3. Dietary Intake and Physical Activity
3.4. Correlations
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Han, J.C.; Lawlor, D.A.; Kimm, S.Y. Childhood obesity. Lancet 2010, 375, 1737–1748. [Google Scholar] [CrossRef]
- Peters, J.I.; McKinney, J.M.; Smith, B.; Wood, P.; Forkner, E.; Galbreath, A.D. Impact of obesity in asthma: Evidence from a large prospective disease management study. Ann. Allergy Asthma Immunol. 2011, 106, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Australian Centre for Asthma Monitoring. Asthma in Australian Children: Findings from Growing up in Australia, the Longitudinal Study of Australian Children; Australian Institute of Health and Welfare: Canberra, Australian, 2009. [Google Scholar]
- Magee, C.A.; Caputi, P.; Iverson, D.C. Patterns of health behaviours predict obesity in Australian children. J. Paediatr. Child Health 2013, 49, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Te Velde, S.J.; van Nassau, F.; Uijtdewilligen, L.; van Stralen, M.M.; Cardon, G.; De Craemer, M.; Manios, Y.; Brug, J.; Chinapaw, M.J. Energy balance-related behaviours associated with overweight and obesity in preschool children: A systematic review of prospective studies. Obes. Rev. 2012, 13 (Suppl. 1), 56–74. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Beydoun, M.A.; Wang, Y. Is sleep duration associated with childhood obesity? A systematic review and meta-analysis. Obesity 2008, 16, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Spiegel, K.; Tasali, E.; Penev, P.; Van Cauter, E. Sleep curtailment in healthy young men is associated with decreased leptin levels, elevated ghrelin levels, and increased hunger and appetite. Ann. Intern. Med. 2004, 141, 846–850. [Google Scholar] [CrossRef] [PubMed]
- Schmid, S.M.; Hallschmid, M.; Jauch-Chara, K.; Wilms, B.; Benedict, C.; Lehnert, H.; Born, J.; Schultes, B. Short-term sleep loss decreases physical activity under free-living conditions but does not increase food intake under time-deprived laboratory conditions in healthy men. Am. J. Clin. Nutr. 2009, 90, 1476–1482. [Google Scholar] [CrossRef] [PubMed]
- Garrison, M.; Lozano, P.; Christakis, D. Controller medication use and sleep problems in pediatric asthma: A longitudinal case-crossover analysis. Arch. Pediatr. Adolesc. Med. 2011, 165, 826–830. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.E.; Wood, L.G.; Williams, R.B.; Collins, C.E. Associations between sleep, dietary intake and physical activity in children: A systematic review. JBI Database Syst. Rev. Implement. Rep. 2013, 11, 227–262. [Google Scholar] [CrossRef]
- Division of Nutrition Physical Activity & Obesity; National Center for Chronic Disease Prevention & Health Promotion. A SAS Program for the CDC Growth Charts. Available online: http://www.cdc.gov/nccdphp/dnpao/growthcharts/resources/sas.htm (accessed on 8 March 2012).
- Juniper, E.; Gruffydd-Jones, K.; Svensson, K. Asthma control questionnaire in children: Validation, measurement properties, interpretation. Eur. Respir. J. 2010, 36, 1410–1416. [Google Scholar] [CrossRef] [PubMed]
- Global Initiative for Asthma. Pocket Guide for Asthma Management and Prevention in Children. 2005. Available online: http://www.seicap.es/documentos/archivos/GINA2006guiapediatricaingles.pdf (accessed on 24 February 2017).
- Lewandowski, A.S.; Toliver-Sokol, M.; Palermo, T.M. Evidence-based review of subjective pediatric sleep measures. J. Pediatr. Psychol. 2011, 36, 780–793. [Google Scholar] [CrossRef] [PubMed]
- Hankinson, J.; Odencrantz, J.; Fedan, K. Spirometric reference values from a sample of the general U.S. Population. Am. J. Respir. Crit. Care Med. 1999, 159, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.E.; Gibson, P.G.; Collins, C.E.; Hilton, J.M.; Latham-Smith, F.; Wood, L.G. Increased sleep latency and reduced sleep duration in children with asthma. Sleep Breath 2013, 17, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Iber, C.; Ancoli-Israel, S.; Chesson, A.J.; Quan, S. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology, and Technical Specifications; The American Academy of Sleep Medicine: Westchester, IL, USA, 2007. [Google Scholar]
- American thoracic society. Standards and indications for cardiopulmonary sleep studies in children. Am. J. Respir. Crit. Care Med. 1996, 153, 866–878. [Google Scholar]
- White, M.A.; Whisenhunt, B.L.; Williamson, D.A.; Greenway, F.L.; Netemeyer, R.G. Development and validation of the food-craving inventory. Obesity 2002, 10, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Burrows, T.; Warren, J.; Colyvas, K.; Garg, M.; Collins, C. Validation of overweight children’s fruit and vegetable intake using plasma carotenoids. Obesity 2008, 17, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Marshall, S.; Watson, J.F.; Burrows, T.; Guest, M.; Collin, C. The development and evaluation of the australian child and adolescent recommended food score: A cross-sectional study. Nutr. J. 2012, 11, 96. [Google Scholar] [CrossRef] [PubMed]
- Booth, M.L.; Okely, A.D.; Chey, T.; Bauman, A. The reliability and validity of the adolescent physical activity recall questionnaire. Med. Sci. Sports Exerc. 2002, 34, 1986–1995. [Google Scholar] [CrossRef] [PubMed]
- Hardy, L.L.; Booth, M.L.; Okely, A.D. The reliability of the adolescent sedentary activity questionnaire (ASAQ). Prev. Med. 2007, 45, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, M.; Engleman, H.; Whyte, K.; Deary, I.; Shapiro, C.; Douglas, N. Morbidity in nocturnal asthma: Sleep quality and daytime cognitive performance. Thorax 1991, 46, 569–573. [Google Scholar] [CrossRef] [PubMed]
- Ramagopal, M.; Scharf, S.M.; Roberts, D.W.; Blaisdell, C.J. Obstructive sleep apnea and history of asthma in snoring children. Sleep Breath 2008, 12, 381–392. [Google Scholar] [CrossRef] [PubMed]
- Strunk, R.C.; Sternberg, A.L.; Bacharier, L.B.; Szefler, S.J. Nocturnal awakening caused by asthma in children with mild-to-moderate asthma in the childhood asthma management program. J. Allergy Clin. Immunol. 2002, 110, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Van Maanen, A.; Wijga, A.H.; Gehring, U.; Postma, D.S.; Smit, H.A.; Oort, F.J.; Rodenburg, R.; Meijer, A.M. Sleep in children with asthma: Results of the PIAMA study. Eur. Respir. J. 2013, 41, 832–837. [Google Scholar] [CrossRef] [PubMed]
- Cottrell, L.; Neal, W.A.; Ice, C.; Perez, M.K.; Piedimonte, G. Metabolic abnormalities in children with asthma. Am. J. Respir. Crit. Care Med. 2011, 183, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Vahlkvist, S.; Inman, M.D.; Pedersen, S. Effect of asthma treatment on fitness, daily activity and body composition in children with asthma. Allergy 2011, 65, 1464–1471. [Google Scholar] [CrossRef] [PubMed]
- Landis, A.M.; Parker, K.P.; Dunbar, S.B. Sleep, hunger, satiety, food cravings, and caloric intake in adolescents. J. Nurs. Scholarsh. 2009, 41, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Hitze, B.; Bosy-Westphal, A.; Bielfeldt, F.; Settler, U.; Plachta-Danielzik, S.; Pfeuffer, M.; Schrezenmeir, J.; Monig, H.; Muller, M.J. Determinants and impact of sleep duration in children and adolescents: Data of the Kiel Obesity Prevention Study. Eur. J. Clin. Nutr. 2009, 63, 739–746. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Ortega, F.; Ruiz, J.R.; Rey-Lopez, J.; Beghin, L.; Manios, Y.; Cuenca-Garcia, M.; Plada, M.; Diethelm, K.; Kafatos, A.; et al. Short sleep duration is associated with increased obesity markers in european adolescents: Effect of physical activity and dietary habits. The HELENA study. Int. J. Obes. 2011, 35, 1308–1317. [Google Scholar] [CrossRef] [PubMed]
- Tatone-Tokuda, F.; Dubois, L.; Ramsay, T.; Girard, M.; Touchette, E.; Petit, D.; Montplaisir, J.Y. Sex differences in the association between sleep duration, diet and body mass index: A birth cohort study. J. Sleep Res. 2012, 21, 448–460. [Google Scholar] [CrossRef] [PubMed]
| Subject Characteristics | Non-Asthma (n = 17) | Asthma (n = 17) | p Value |
|---|---|---|---|
| Age (years); mean (SD) | 10.8 (2.3) | 10.7 (2.4) | 0.928 |
| Gender (female); n (%) | 12 (70.6) | 5 (29.4) | 0.016 1 |
| Height (cm); median (IQR) | 146.0 (136.7, 153.0) | 139.8 (133.0, 147.7) | 0.380 |
| Weight (kg); median (IQR) | 37.0 (30.5, 46.1) | 36.0 (28.1, 41.6) | 0.502 |
| BMI z-score (SD); median (IQR) | 0.4 (−1.2, 1.0) | 0.0 (−0.4, 0.6) | 0.986 |
| Waist-to-height (cm/cm); mean (SD) | 0.44 (0.06) | 0.46 (0.05) | 0.228 |
| Evening lung function | |||
| FEV1 %predicted (%); mean (SD) | 94.0 (12.7) | 93.8 (17.3) | 0.974 |
| FVC %predicted (%); mean (SD) | 90.8 (11.7) | 93.3 (11.9) | 0.552 |
| FEV1/FVC (%); mean (SD) | 87.2 (86.4, 91.5) | 86.0 (77.8, 91.1) | 0.207 |
| Morning lung function | |||
| FEV1 %predicted (%); mean (SD) | 93.8 (12.3) | 88.1 (14.0) | 0.258 |
| FVC %predicted (%); mean (SD) | 90.7 (7.9) | 90.3 (10.0) | 0.914 |
| FEV1/FVC (%); mean (SD) | 88.0 (84.5, 89.8) | 79.2 (75.7, 88.7) | 0.125 |
| Sleep Indices | Non-Asthma (n = 17) | Asthma (n = 17) | p Value |
|---|---|---|---|
| Paediatric Daytime Sleepiness Scale (score); mean (SD) | 15.3 (5.2) | 16.2 (5.7) | 0.620 |
| Total sleep time (min); mean (SD) | 428.5 (38.8) | 415.3 (53.5) | 0.416 |
| Total awake time (min); median (IQR) | 44.0 (35.0, 54.5) | 54.5 (32.0, 66.5) | 0.642 |
| Sleep efficiency (%); mean (SD) | 81.6 (6.9) | 78.2 (7.5) | 0.181 |
| Sleep latency (min); mean (SD) | 40.9 (16.9) | 56.6 (25.5) | 0.042 1 |
| Non-REM Sleep Stages: | |||
| Stage 1 (%); median (IQR) | 2.0 (0.7, 2.6) | 0.6 (0.3, 1.6)) | 0.094 |
| Stage 2 (%); median (IQR) | 48.9 (43.3, 52.6) | 49.0 (43.5, 55.8) | 0.931 |
| Stage 3 (%); median (IQR) | 31.4 (29.0, 37.1) | 32.7 (26.1, 38.1) | 0.904 |
| REM latency (min); median (IQR) | 166.5 (139.0, 237.5) | 149.0 (100.5, 192.0) | 0.294 |
| REM sleep (%); mean (SD) | 17.9 (8.5) | 16.9 (6.4) | 0.717 |
| Arousal Index (n/h); median (IQR) | 3.2 (1.9, 10.1) | 3.7 (2.7, 6.4) | 0.836 |
| Respiratory Disturbance Index (n/h); median (IQR) | 0.1 (0.0, 1.3) | 0.2 (0.0, 0.4) | 0.986 |
| Systemic Biomarkers | Non-Asthma (n = 14) | Asthma (n = 13) | p Value |
|---|---|---|---|
| Cortisol (nmol/L); median (IQR) | 363 (310, 424) | 326 (310, 403) | 0.865 |
| IGF-1 (U/ml); median (IQR) | 1.04 (0.74, 1.94) | 0.73 (0.66, 1.08) | 0.190 |
| hsCRP (mg/L); median (IQR) | 1.1 (0.5, 1.5) | 0.7 (0.2, 0.9) | 0.113 |
| HOMA-IR; median (IQR) | 1.4 (0.9, 2.4) | 1.4 (1.0, 1.7) | 0.961 |
| Triglycerides (mmol/L); median (IQR) | 0.7 (0.7, 0.8) | 1.0 (0.8, 1.2) | 0.013 1 |
| Cholesterol (mmol/L); mean (SD) | 4.4 (0.4) | 4.8 (0.2) | 0.380 |
| TC/HDL (ratio); mean (SD) | 3.2 (1.0) | 3.3 (0.9) | 0.786 |
| Dietary Intake Measures | Non-Asthma (n = 17) | Asthma (n = 17) | p Value |
|---|---|---|---|
| Energy (kj); mean (SD) | 8945.5 (2314.5) | 9365.4 (2722.7) | 0.631 |
| Protein (%energy); mean (SD) | 16.4 (2.5) | 17.2 (2.5) | 0.380 |
| Total fat (%energy); mean (SD) | 33.4 (4.1) | 31.5 (3.7) | 0.183 |
| Saturated fat (%fat); mean (SD) | 51.0 (3.5) | 49.1 (4.4) | 0.177 |
| PUFA (%fat); mean (SD) | 11.7 (2.1) | 12.7 (3.1) | 0.276 |
| MUFA (%fat); mean (SD) | 37.2 (2.3) | 38.2 (2.2) | 0.197 |
| Carbohydrate (%energy); mean (SD) | 51.6 (5.3) | 52.0 (4.5) | 0.809 |
| Sugars (g); mean (SD) | 146.4 (47.7) | 159.4 (72.6) | 0.542 |
| Fibre (g); mean (SD) | 23.5 (6.4) | 27.2 (8.4) | 0.151 |
| Australian Recommended Food Score; mean (SD) | 26.6 (8.1) | 27.9 (10.1) | 0.683 |
| Correlations | Total Sleep Time (min) | Paediatric Daytime Sleepiness Scale (Score) |
|---|---|---|
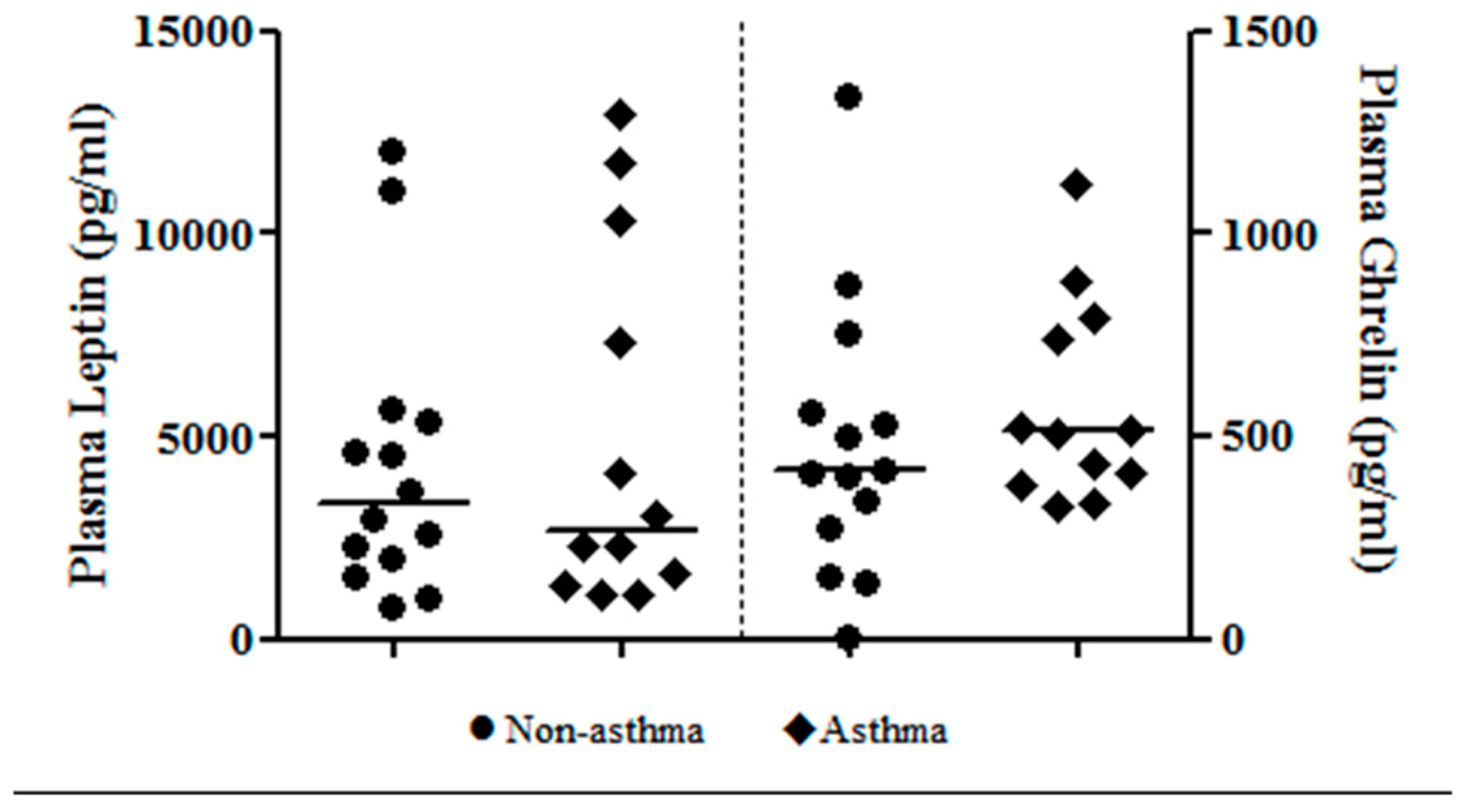
| Ghrelin (pg/mL) | 0.215 | −0.0003 |
| Leptin (pg/mL) | −0.377 | 0.143 |
| TC/HDL (ratio) | −0.090 | 0.614 1 |
| Triglycerides (mmol/L) | −0.159 | 0.307 |
| Food Cravings Index-II (score) | 0.106 | −0.042 |
| Australian Recommended Food Score | 0.357 1 | −0.016 |
| Physical activity (min/week) | 0.315 | −0.143 |
| Physical activity intensity (METS) | 0.107 | 0.077 |
| Sedentary activity (min/week) | −0.270 | 0.386 1 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jensen, M.E.; Gibson, P.G.; Collins, C.E.; Hilton, J.M.; Wood, L.G. Lifestyle Risk Factors for Weight Gain in Children with and without Asthma. Children 2017, 4, 15. https://doi.org/10.3390/children4030015
Jensen ME, Gibson PG, Collins CE, Hilton JM, Wood LG. Lifestyle Risk Factors for Weight Gain in Children with and without Asthma. Children. 2017; 4(3):15. https://doi.org/10.3390/children4030015
Chicago/Turabian StyleJensen, Megan E., Peter G. Gibson, Clare E. Collins, Jodi M. Hilton, and Lisa G. Wood. 2017. "Lifestyle Risk Factors for Weight Gain in Children with and without Asthma" Children 4, no. 3: 15. https://doi.org/10.3390/children4030015
APA StyleJensen, M. E., Gibson, P. G., Collins, C. E., Hilton, J. M., & Wood, L. G. (2017). Lifestyle Risk Factors for Weight Gain in Children with and without Asthma. Children, 4(3), 15. https://doi.org/10.3390/children4030015







