Fabrication of Magnetic Molecularly Imprinted Polymers for Selective Extraction of Dibutyl Phthalates in Food Matrices
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis
2.2.1. Preparation of Fe3O4@MOF
2.2.2. Preparation of Fe3O4@MOF@MIP
2.3. Characterization of Fe3O4@MOF@MIP160
2.4. Sample Preparation
2.5. GC-MS Analysis Parameters for the Samples
2.6. Dynamic Binding Experiments
2.7. Stability Experiment
2.8. Selective Research
2.9. Regeneration of Adsorption of Fe3O4@MOF@MIP160
3. Results
3.1. Optimization of the Synthesis Conditions
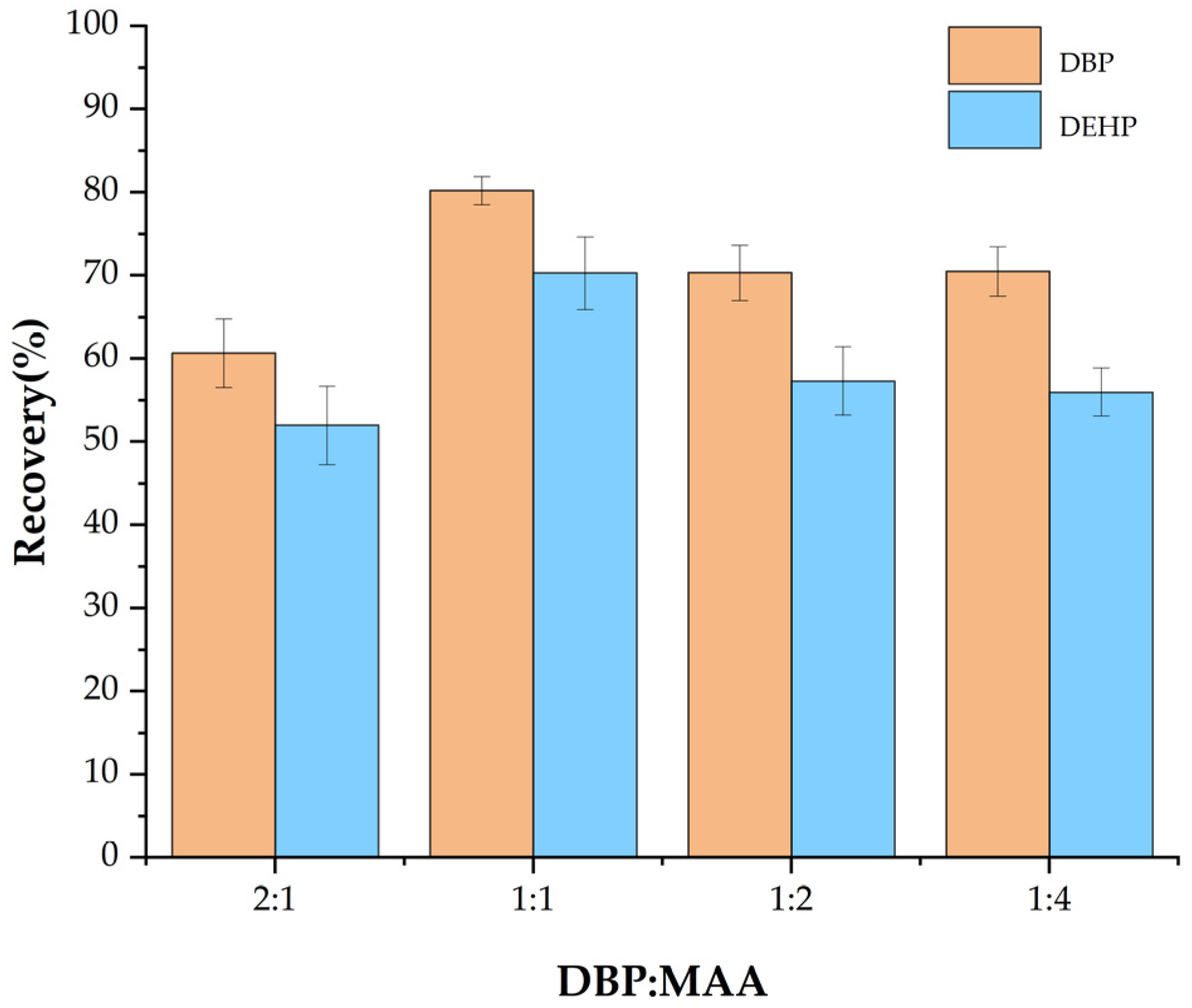
3.1.1. Effect of the Ratio of Template Molecules to Functional Monomer
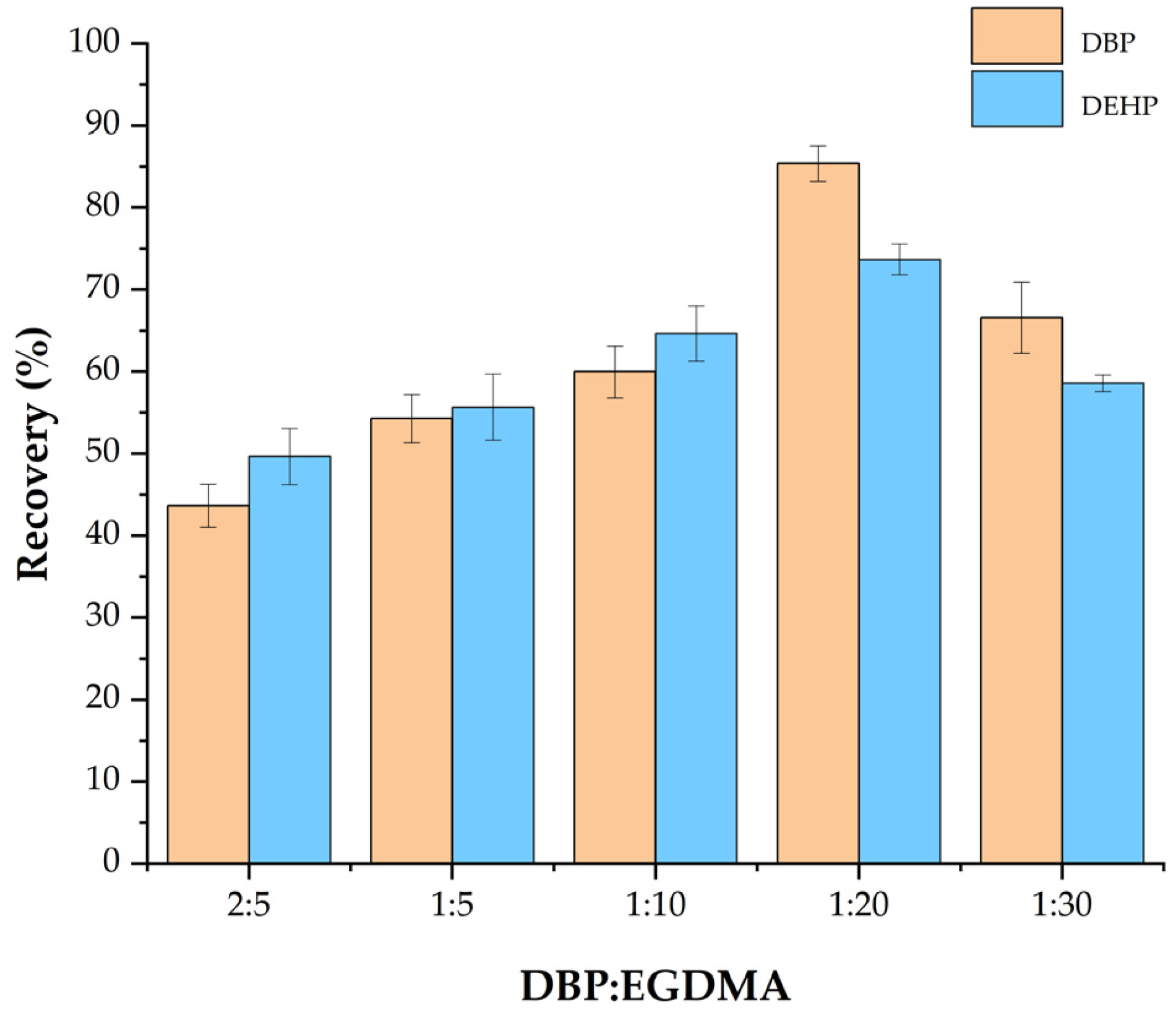
3.1.2. Effect of the Ratio of Template Molecules to EGDMA
3.1.3. Effect of Reaction Time
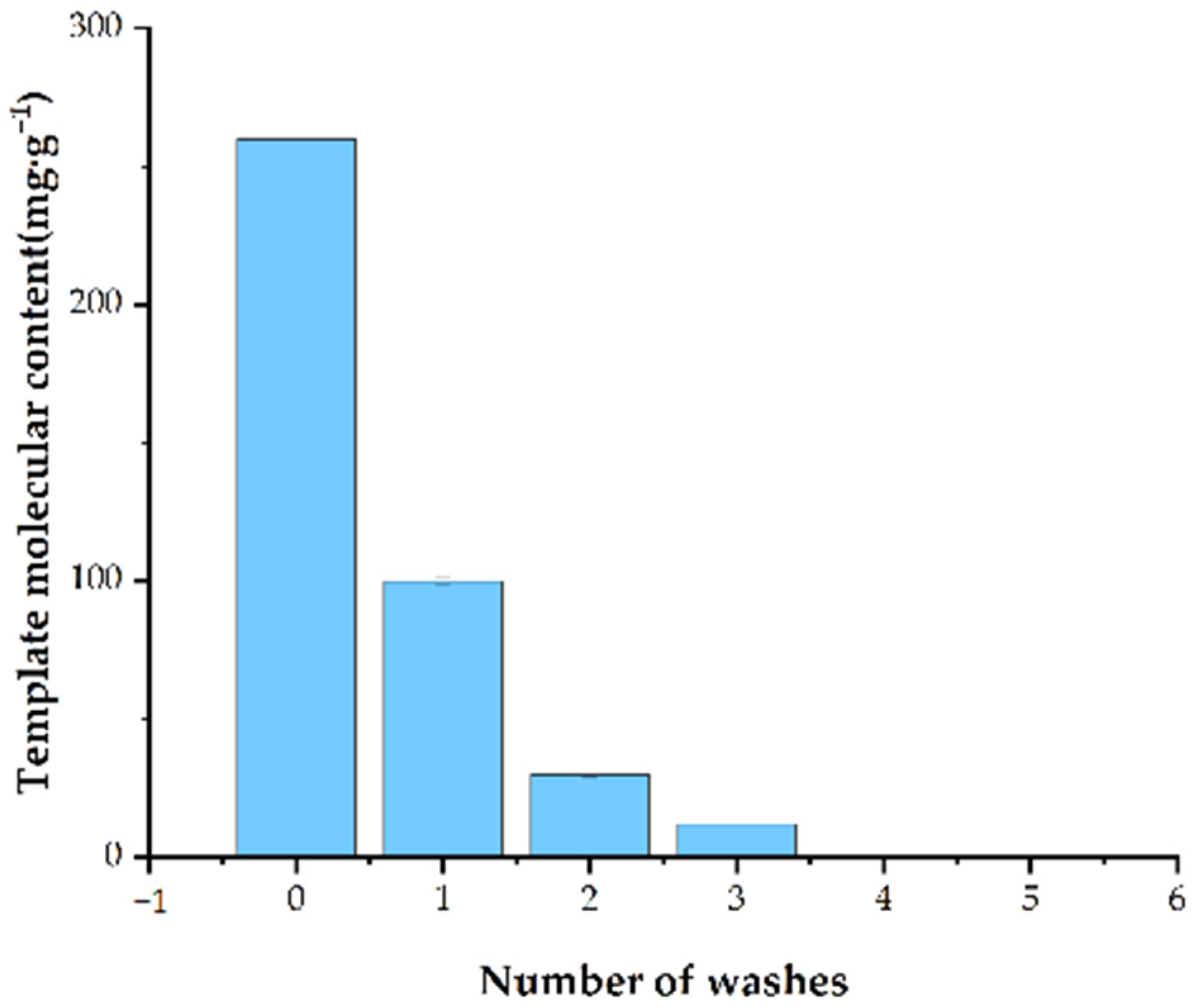
3.1.4. Number of Washes Required to Completely Remove Stencil Molecules
3.2. Characterization of Adsorbent Materials
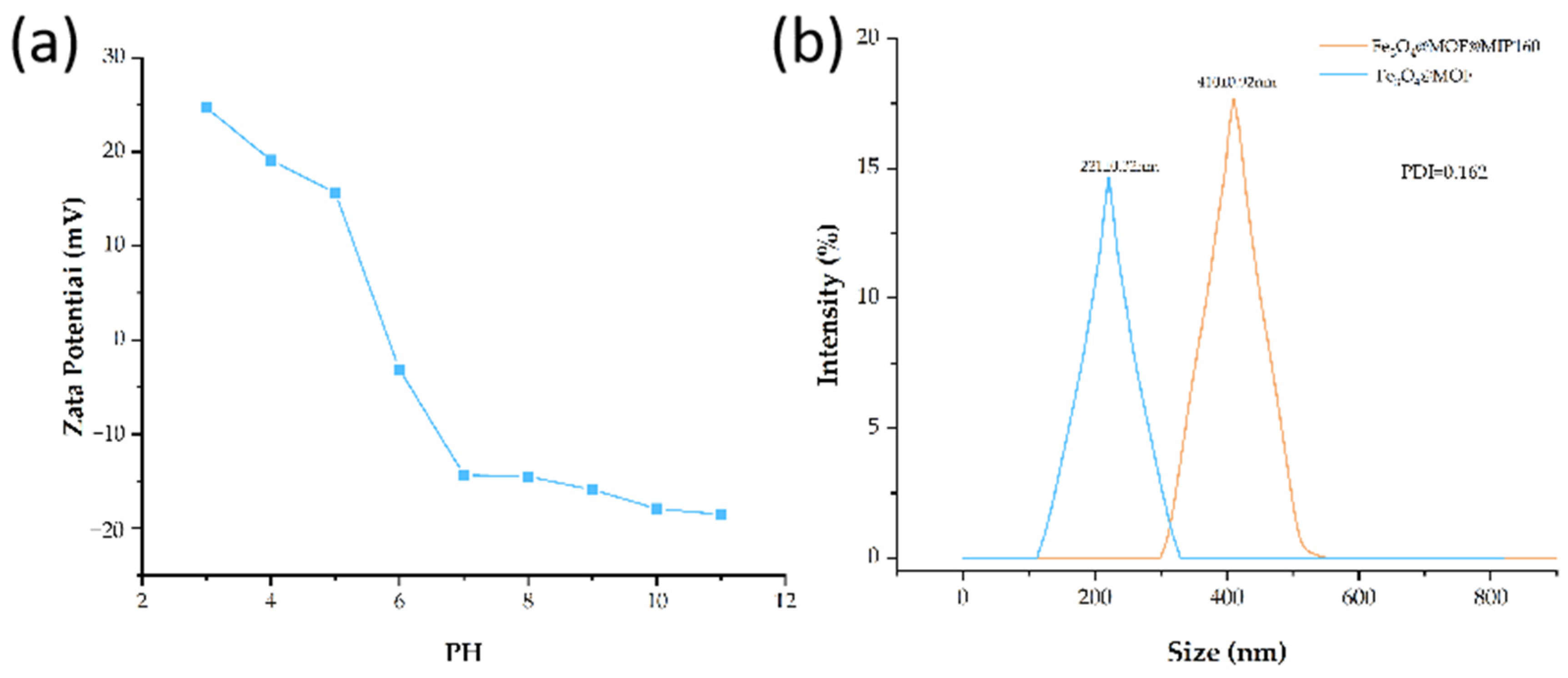
3.2.1. TEM, DLS, and Zeta Analysis
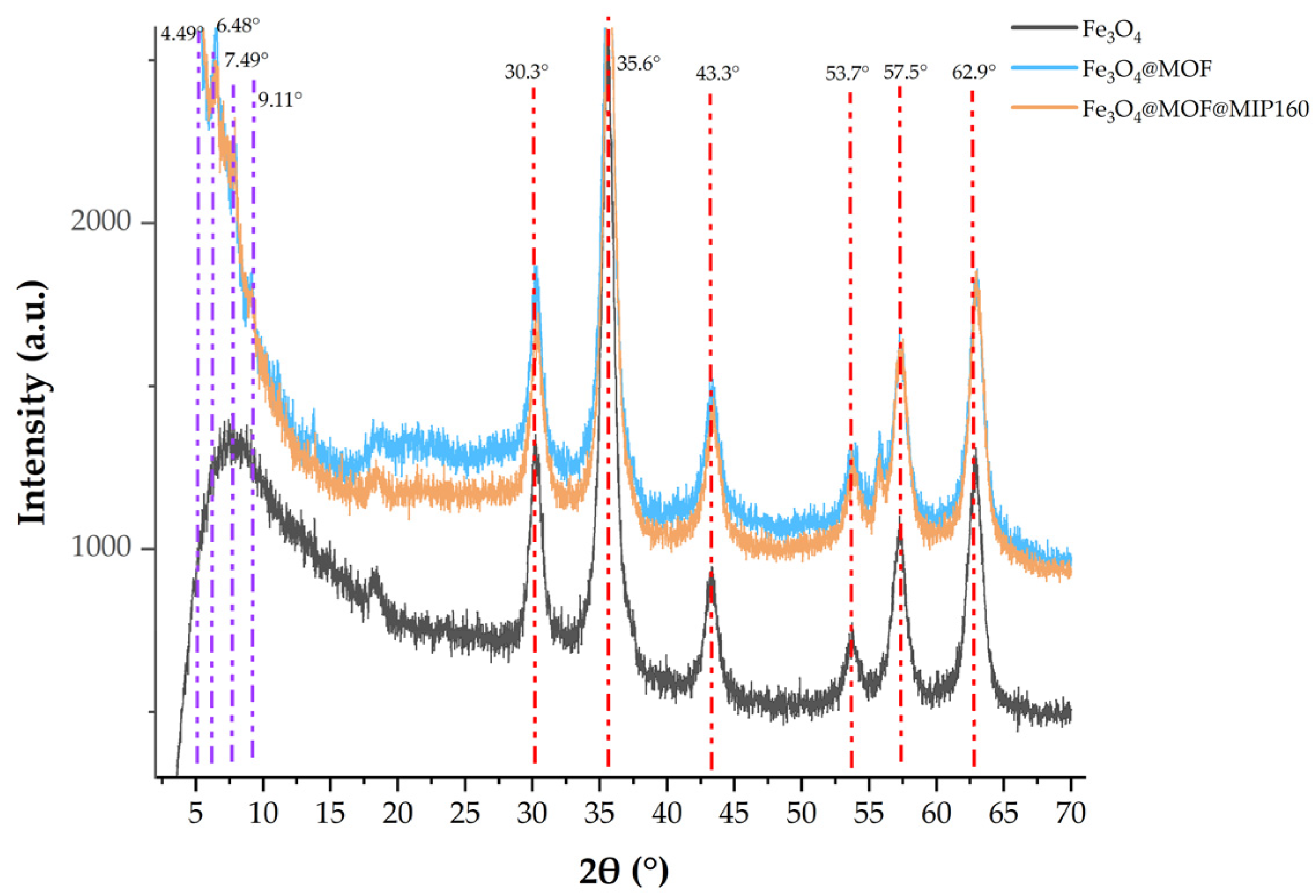
3.2.2. XRD Analysis
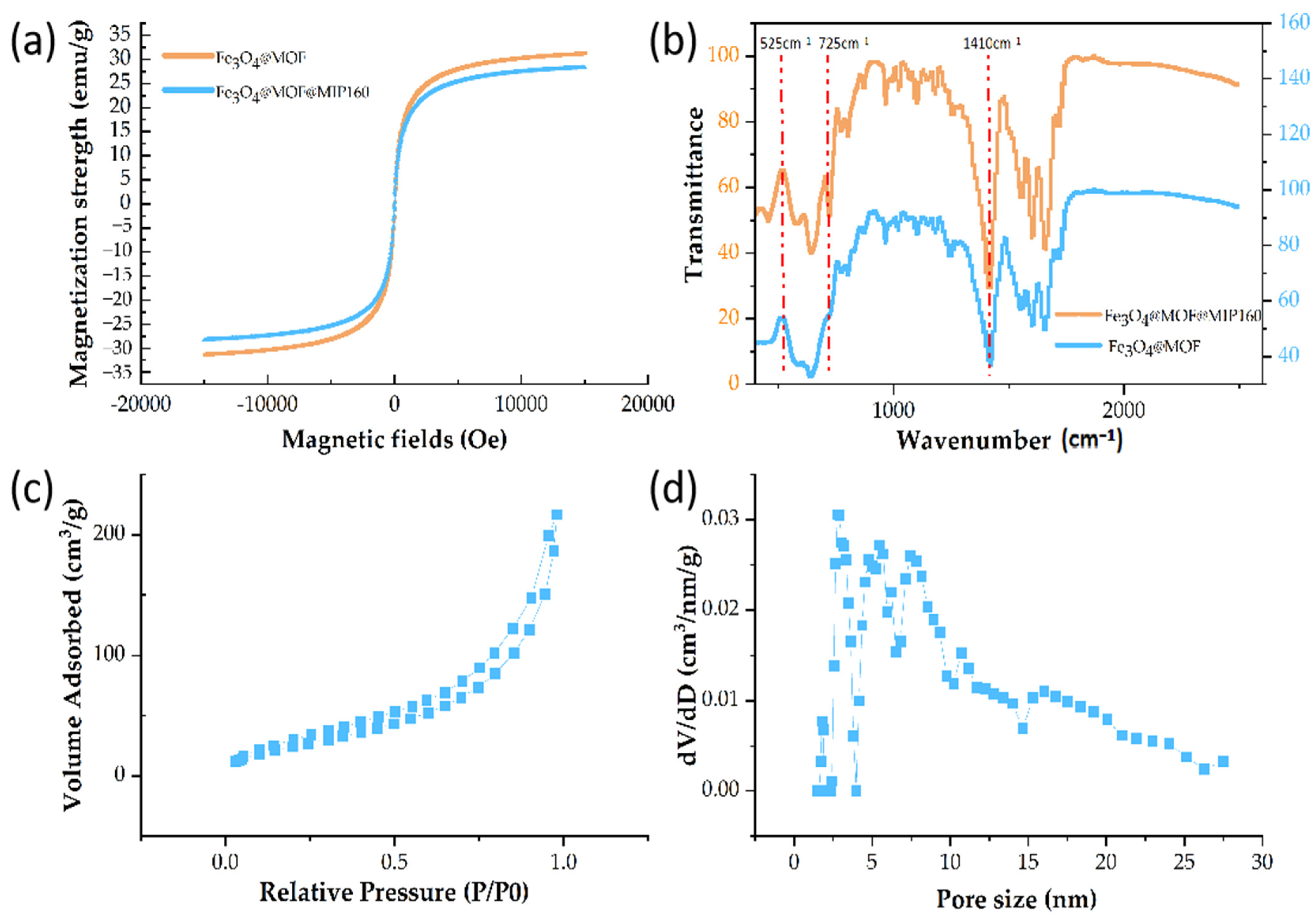
3.2.3. VSM Analysis
3.2.4. FT-IR Analysis
3.2.5. Nitrogen Adsorption–Desorption
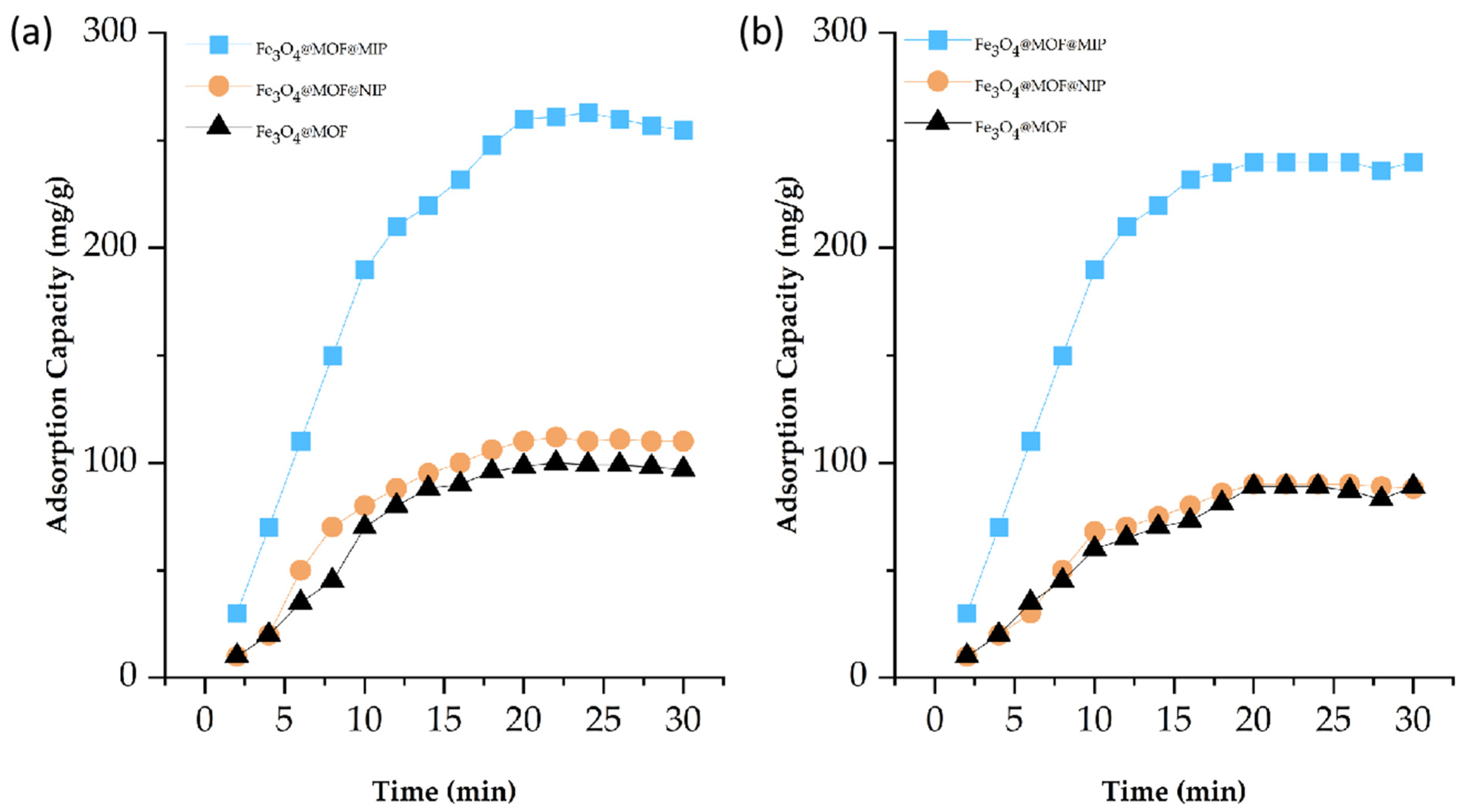
3.3. Analysis of Adsorption Performance of Fe3O4@MOF@MIP160
3.4. Stability of Fe3O4@MOF@MIP160
3.5. Selectivity of Fe3O4@MOF@MIP160
3.6. Regeneration of Fe3O4@MOF@MIP160
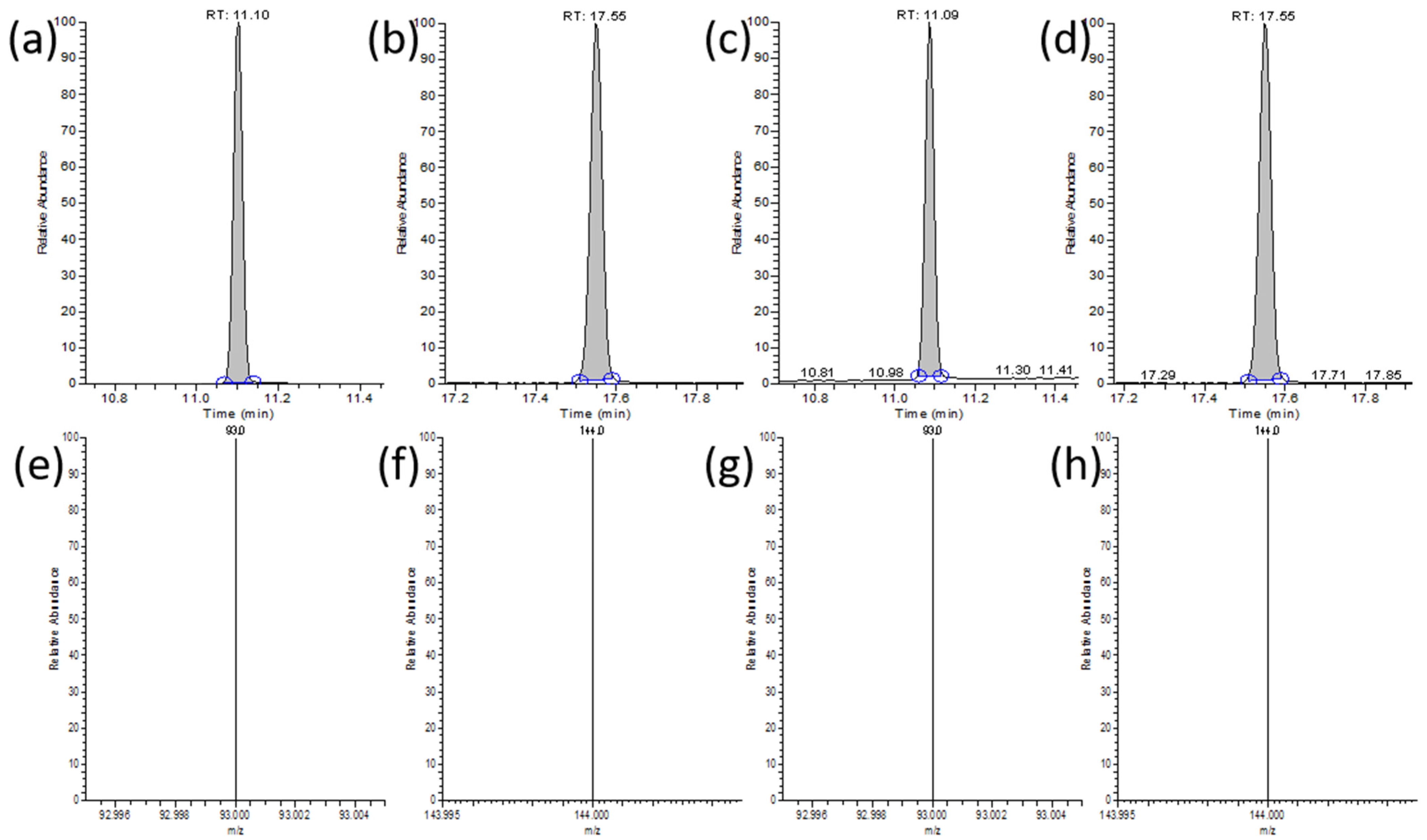
3.7. GC-MS Methodology Evaluation
3.8. Application to Real Sample Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chen, M.; Niu, Z.; Zhang, X.; Zhang, Y. Pollution Characteristics and Health Risk of Sixty-Five Organics in One Drinking Water System: PAEs Should Be Prioritized for Control. Chemosphere 2024, 350, 141171. [Google Scholar] [CrossRef] [PubMed]
- Kato, K.; Silva, M.J.; Reidy, J.A.; Hurtz, D.; Malek, N.A.; Needham, L.L.; Nakazawa, H.; Barr, D.B.; Calafat, A.M. Mono(2-Ethyl-5-Hydroxyhexyl) Phthalate and Mono-(2-Ethyl-5-Oxohexyl) Phthalate as Biomarkers for Human Exposure Assessment to Di-(2-Ethylhexyl) Phthalate. Environ. Health Perspect. 2004, 112, 327–330. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wang, Q.; Jiang, N.; Lv, H.; Liang, C.; Yang, H.; Yao, X.; Wang, J. Occurrence, Source, Ecological Risk, and Mitigation of Phthalates (PAEs) in Agricultural Soils and the Environment: A Review. Environ. Res. 2023, 220, 115196. [Google Scholar] [CrossRef]
- Fasano, E.; Bono-Blay, F.; Cirillo, T.; Montuori, P.; Lacorte, S. Migration of Phthalates, Alkylphenols, Bisphenol A and Di(2-Ethylhexyl)Adipate from Food Packaging. Food Control. 2012, 27, 132–138. [Google Scholar] [CrossRef]
- Sun, Q.; Zhang, X.; Liu, C.; Nier, A.; Ying, S.; Zhang, J.; Zhao, Y.; Zhang, Y.; Wang, Z.; Shi, M. The Content of PAEs in Field Soils Caused by the Residual Film Has a Periodical Peak. Sci. Total Environ. 2023, 864, 161078. [Google Scholar] [CrossRef]
- Gong, J.; Yi, X.; Liang, J.; Liu, Z.; Du, Z. Inhibitory Effects of Phthalate Esters (PAEs) and Phthalate Monoesters towards Human Carboxylesterases (CESs). Toxicol. Appl. Pharmacol. 2024, 482, 116785. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yang, Y.; Tao, Y.; Guo, X.; Cui, Y.; Li, Z. Phthalates (PAEs) and Reproductive Toxicity: Hypothalamic-Pituitary-Gonadal (HPG) Axis Aspects. J. Hazard. Mater. 2023, 459, 132182. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Liu, Y.; Chuo, Y.; Wang, X.; Jiao, Y.; Shi, W.; Bao, Y. Cuscuta chinensis flavonoids alleviate ovarian damage in offspring female mice induced by BPA exposure during pregnancy by regulating the central carbon metabolism pathway. Ecotoxicol. Environ. Saf. 2024, 275, 116253. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Jiao, Y.; Li, Z.; Tao, Y.; Yang, Y. Hazards of Phthalates (PAEs) Exposure: A Review of Aquatic Animal Toxicology Studies. Sci. Total Environ. 2021, 771, 145418. [Google Scholar] [CrossRef]
- Isci, G. Assessment of Phthalate Esters in Packaged Fruit Juices Sold in the Turkish Market and Their Implications on Human Health Risk. Food Chem. 2024, 435, 137658. [Google Scholar] [CrossRef]
- Mikula, P.; Svobodová, Z.; Smutná, M. Phthalates: Toxicology and Food Safety—A Review. Czech J. Food Sci. 2005, 23, 217–223. [Google Scholar] [CrossRef]
- Arfaeinia, L.; Dobaradaran, S.; Nasrzadeh, F.; Shamsi, S.; Poureshgh, Y.; Arfaeinia, H. Phthalate Acid Esters (PAEs) in Highly Acidic Juice Packaged in Polyethylene Terephthalate (PET) Container: Occurrence, Migration and Estrogenic Activity-Associated Risk Assessment. Microchem. J. 2020, 155, 104719. [Google Scholar] [CrossRef]
- Wang, R.; Ma, X.; Zhang, X.; Li, X.; Li, D.; Dang, Y. C8-Modified Magnetic Graphene Oxide Based Solid-Phase Extraction Coupled with Dispersive Liquid-Liquid Microextraction for Detection of Trace Phthalate Acid Esters in Water Samples. Ecotoxicol. Environ. Saf. 2019, 170, 789–795. [Google Scholar] [CrossRef]
- Jose Varghese, R.; Zikalala, N.; Sakho, E.H.M.; Oluwafemi, O.S. Green Synthesis Protocol on Metal Oxide Nanoparticles Using Plant Extracts, in Colloidal Metal Oxide Nanoparticles; Elsevier: Amsterdam, The Netherlands, 2020; pp. 67–82. [Google Scholar] [CrossRef]
- Keshavarzi, M.; Ghorbani, M.; Mohammadi, P.; Pakseresht, M.; Ziroohi, A.; Rastegar, A. Development of a Magnetic Sorbent Based on Synthesis of MOF-on-MOF Composite for Dispersive Solid-Phase Microextraction of Five Phthalate Esters in Bottled Water and Fruit Juice Samples. Microchem. J. 2022, 182, 107934. [Google Scholar] [CrossRef]
- Prokůpková, G.; Holadová, K.; Poustka, J.; Hajšlová, J. Development of a Solid-Phase Microextraction Method for the Determination of Phthalic Acid Esters in Water. Anal. Chim. Acta 2002, 457, 211–223. [Google Scholar] [CrossRef]
- Ghaedi, M.; Sadeghian, B.; Pebdani, A.A.; Sahraei, R.; Daneshfar, A.; Duran, C. Kinetics, Thermodynamics and Equilibrium Evaluation of Direct Yellow 12 Removal by Adsorption onto Silver Nanoparticles Loaded Activated Carbon. Chem. Eng. J. 2012, 187, 133–141. [Google Scholar] [CrossRef]
- Ahn, C.K.; Park, D.; Woo, S.H.; Park, J.M. Removal of Cationic Heavy Metal from Aqueous Solution by Activated Carbon Impregnated with Anionic Surfactants. J. Hazard. Mater. 2009, 164, 1130–1136. [Google Scholar] [CrossRef]
- Moazzen, M.; Mousavi Khaneghah, A.; Shariatifar, N.; Ahmadloo, M.; Eş, I.; Baghani, A.N.; Yousefinejad, S.; Alimohammadi, M.; Azari, A.; Dobaradaran, S.; et al. Multi-Walled Carbon Nanotubes Modified with Iron Oxide and Silver Nanoparticles (MWCNT-FeO/Ag) as a Novel Adsorbent for Determining PAEs in Carbonated Soft Drinks Using Magnetic SPE-GC/MS Method34. Arab. J. Chem. 2019, 12, 476–488. [Google Scholar] [CrossRef]
- Zhang, S.; Wang, R.; Wu, Y.; Chen, Z.; Tong, P.; He, Y.; Lin, Z.; Cai, Z. One-Pot Synthesis of Magnetic Covalent Organic Frameworks for Highly Efficient Enrichment of Phthalate Esters from Fine Particulate Matter. J. Chromatogr. A 2022, 1667, 462906. [Google Scholar] [CrossRef]
- Liu, Y.; Song, W.; Zhou, D.; Han, F.; Gong, X.; Pan, P. A New Core-Shell Magnetic Mesoporous Surface Molecularly Imprinted Composite and Its Application as an MSPE Sorbent for Determination of Phthalate Esters. RSC Adv. 2022, 12, 7253–7261. [Google Scholar] [CrossRef]
- Javed, R.; Zia, M.; Naz, S.; Aisida, S.O.; Ain, N.; Ao, Q. Role of capping agents in the application of nanoparticles in biomedicine and environmental remediation: Recent trends and future prospects. J. Nanobiotechnology 2020, 18, 172. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.; Gao, H.T.; Zhu, F.; Cao, W.X.; Yan, Y.H.M.; Zhou, X.; Xu, Q.; Ji, W.L. SPE–UPLC–MS/MS for the Determination of Phthalate Monoesters in Rats Urine and Its Application to Study the Effects of Food Emulsifier on the Bioavailability of Priority Controlling PAEs. J. Chromatogr. B 2016, 1012–1013, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Meng, L.; Lan, T.; Xu, J.; Zhao, P.; Lei, J. Adjusting Structure-Activity Relationship to Obtain Hybrid Proton Exchange Membrane with Enhanced Transport Efficiency by Introducing Functionalized Nano-Coated MOFs. J. Membr. Sci. 2024, 694, 122409. [Google Scholar] [CrossRef]
- Cheng, L.; Huang, R.; Cao, Q.; Liu, N.; Li, P.; Sun, M.; Qin, H.; Wu, L. Magnetic Metal-Organic Frameworks as Adsorbents for the Detection of Azo Pigments in Food Matrices. Food Chem. 2023, 402, 134134. [Google Scholar] [CrossRef] [PubMed]
- Assen, A.H.; Yassine, O.; Shekhah, O.; Eddaoudi, M.; Salama, K.N. MOFs for the Sensitive Detection of Ammonia: Deployment of Fcu-MOF Thin Films as Effective Chemical Capacitive Sensors. ACS Sens. 2017, 2, 1294–1301. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Sun, Z.; Liu, H.; Zhou, Y.; Li, J.; Wang, X.; Gong, B. Molecularly Imprinted Polymer Coating on Metal-Organic Frameworks for Solid-Phase Extraction of Fluoroquinolones from Water. J. Sep. Sci. 2019, 42, 3302–3310. [Google Scholar] [CrossRef] [PubMed]
- Ke, F.; Wang, L.; Zhu, J. Multifunctional Au-Fe3O4@MOF Core-Shell Nanocomposite Catalysts with Controllable Reactivity and Magnetic Recyclability. Nanoscale 2014, 7, 1201–1208. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.; Xia, Z.-Z.; Sun, X.; Jiang, H.-L.; Wang, L.-L.; Ai, S.; Zhao, R.-S. Recent Advance and Applications of Covalent Organic Frameworks Based on Magnetic Solid-Phase Extraction Technology for Food Safety Analysis. TrAC Trends Anal. Chem. 2023, 162, 117054. [Google Scholar] [CrossRef]
- Xu, H.; Zhu, J.; Wu, X.; Cheng, Y.; Wang, D.; Cai, D. Recognition and Quantitative Analysis for Six Phthalate Esters (PAEs) through Functionalized ZIF-67@Ag Nanowires as Surface-Enhanced Raman Scattering Substrate. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2023, 284, 121735. [Google Scholar] [CrossRef]
- Yan, Y.; Yang, B.; Ji, G.; Lu, K.; Zhao, Z.; Zhang, H.; Xia, M.; Wang, F. Tunable Zirconium-Based Metal Organic Frameworks Synthesis for Dibutyl Phthalate Efficient Removal: An Investigation of Adsorption Mechanism on Macro and Micro Scale. J. Colloid Interface Sci. 2023, 650, 222–235. [Google Scholar] [CrossRef]
- Zhou, Q.; Guo, M.; Wu, S.; Fornara, D.; Sarkar, B.; Sun, L.; Wang, H. Electrochemical Sensor Based on Corncob Biochar Layer Supported Chitosan-MIPs for Determination of Dibutyl Phthalate (DBP). J. Electroanal. Chem. 2021, 897, 115549. [Google Scholar] [CrossRef]
- Gao, J.; Fan, D.; Chu, Q.; Lyu, H.; Xie, Z. Fabrication of a Novel Surface Molecularly Imprinted Polymer Based on Zeolitic Imidazolate Framework-7 for Selective Extraction of Phthalates. Microchem. J. 2022, 178, 107399. [Google Scholar] [CrossRef]
- Yeganegi, A.; Fardindoost, S.; Tasnim, N.; Hoorfar, M. Molecularly Imprinted Polymers (MIP) Combined with Raman Spectroscopy for Selective Detection of Δ9-Tetrahydrocannabinol (THC). Talanta 2024, 267, 125271. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Ma, X.; Xie, X.; Huang, R.; Zhang, M.; Li, J.; Zeng, G.; Fan, Y. Preparation of Dual-Dummy-Template Molecularly Imprinted Polymers Coated Magnetic Graphene Oxide for Separation and Enrichment of Phthalate Esters in Water. Chem. Eng. J. 2019, 361, 245–255. [Google Scholar] [CrossRef]
- He, J.; Wang, L.; Liu, H.; Sun, B. Recent Advances in Molecularly Imprinted Polymers (MIPs) for Visual Recognition and Inhibition of α-Dicarbonyl Compound-Mediated Maillard Reaction Products. Food Chem. 2024, 446, 138839. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, J.; Zhao, W.; Guo, R.; Cui, S.; Huang, J.; Lu, J.; Liu, H.; Liu, Y. Integrated Treatment of Tetracycline in Complex Environments with MIPs-Based FeO-CuO-Au Nanocomposites: Selective SERS Detection and Targeted Photocatalytic Degradation342. J. Alloys Compd. 2024, 982, 173796. [Google Scholar] [CrossRef]
- Movlaee, K.; Ganjali, M.R.; Norouzi, P.; Neri, G. Iron-Based Nanomaterials/Graphene Composites for Advanced Electrochemical Sensors. Nanomaterials 2017, 7, 406. [Google Scholar] [CrossRef] [PubMed]
- Hou, F.; Chang, Q.; Wan, N.; Li, J.; Zang, X.; Zhang, S.; Wang, C.; Wang, Z. A Novel Porphyrin-Based Conjugated Microporous Nanomaterial for Solid-Phase Microextraction of Phthalate Esters Residues in Children’s Food. Food Chem. 2022, 388, 133015. [Google Scholar] [CrossRef]
- Gao, J.-J.; Lang, X.-X.; Yu, Q.-Q.; Li, H.-Y.; Wang, H.-J.; Wang, M.-Q. Amphiphilic BODIPY-Based Nanoparticles as “Light-up” Fluorescent Probe for PAEs Detection by an Aggregation/Disaggregation Approach. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 252, 119492. [Google Scholar] [CrossRef]
- Wu, Q.; Song, Y.; Wang, Q.; Liu, W.; Hao, L.; Wang, Z.; Wang, C. Combination of magnetic solid-phase extraction and HPLC-UV for simultaneous determination of four phthalate esters in plastic bottled juice. Food Chem. 2021, 339, 127855. [Google Scholar] [CrossRef]
- Liu, X.; Sun, Z.; Chen, G.; Zhang, W.; Cai, Y.; Kong, R.; Wang, X.; Suo, Y.; You, J. Determination of phthalate esters in environmental water by magnetic Zeolitic Imidazolate Framework-8 solid-phase extraction coupled with high-performance liquid chromatography. J. Chromatogr. A 2015, 1409, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Zhou, Q.; Yuan, Y.; Wang, H.; Tong, Y.; Zhan, Y.; Sheng, X.; Sun, Y.; Zhou, X. Enrichment and sensitive determination of phthalate esters in environmental water samples: A novel approach of MSPE-HPLC based on PAMAM dendrimers-functionalized magnetic-nanoparticles. Talanta 2020, 206, 120213. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.-B.; Yu, Q.-W.; Yuan, B.-F.; Feng, Y.-Q. Fast Microextraction of Phthalate Acid Esters from Beverage, Environmental Water and Perfume Samples by Magnetic Multi-Walled Carbon Nanotubes. Talanta 2012, 90, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Niu, J.; Zhang, Y.; Liu, H.; Huang, S.; Yuan, S.; Dong, G.; Bu, L.; Song, D.; Zhou, Q. High Enrichment and Sensitive Measurement of Seventeen Phthalates in Beverages with Metal Organic Framework Functionalized Magnetic MXene Nanocomposite Based on Magnetic Solid Phase Extraction Prior to Gas Chromatography-Triple Quadrupole Mass Spectrometry. Sep. Purif. Technol. 2024, 339, 126595. [Google Scholar] [CrossRef]
- Cao, X.-L. Phthalate Esters in Foods: Sources, Occurrence, and Analytical Methods. Compr. Rev. Food Sci. Food Saf. 2010, 9, 21–43. [Google Scholar] [CrossRef] [PubMed]
- Martín-Gómez, B.; Stephen Elmore, J.; Valverde, S.; Ares, A.M.; Bernal, J. Recent Applications of Chromatography for Determining Microplastics and Related Compounds (Bisphenols and Phthalate Esters) in Food. Microchem. J. 2024, 197, 109903. [Google Scholar] [CrossRef]
- Yang, R.; Liu, Y.; Yan, X.; Liu, S. Simultaneous Extraction and Determination of Phthalate Esters in Aqueous Solution by Yolk-Shell Magnetic Mesoporous Carbon-Molecularly Imprinted Composites Based on Solid-Phase Extraction Coupled with Gas Chromatography-Mass Spectrometry. Talanta 2016, 161, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Liu, M.; Sun, M.; Chen, K.; Cao, X.; Hu, Y. Magnetic Solid-Phase Extraction of Phthalate Esters (PAEs) in Apparel Textile by Core-Shell Structured FeO@silica@triblock-Copolymer Magnetic Microspheres34. Talanta 2016, 150, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Lokhat, D.; Brijlal, S.; Naidoo, D.E.; Premraj, C.; Kadwa, E. Synthesis of Size-and-Shape-Controlled Iron Oxide Nanoparticles via Coprecipitation and In Situ Magnetic Separation. Ind. Eng. Chem. Res. 2022, 61, 16980–16991. [Google Scholar] [CrossRef]
- de Jesús Ruíz-Baltazar, Á.; Reyes-López, S.Y.; de Lourdes Mondragón-Sánchez, M.; Robles-Cortés, A.I.; Pérez, R. Eco-Friendly Synthesis of FeO Nanoparticles: Evaluation of Their Catalytic Activity in Methylene Blue Degradation by Kinetic Adsorption Models34. Results Phys. 2019, 12, 989–995. [Google Scholar] [CrossRef]
- Pathak, G.; Rajkumari, K.; Rokhum, S.L. Wealth from Waste: M. Acuminata Peel Waste-Derived Magnetic Nanoparticles as a Solid Catalyst for the Henry Reaction. Nanoscale Adv. 2019, 1, 1013–1020. [Google Scholar] [CrossRef] [PubMed]





| Range | DBP | DEHP | |||
|---|---|---|---|---|---|
| 10–1000 ppb | 1000–5000 ppb | 10–1000 ppb | 1000–5000 ppb | ||
| Drinking water | standard curve | y = 0.432x + 0.562 | y = 1.22x + 0.322 | y = 0.426x + 0.228 | y = 1.82x + 0.237 |
| R2 | 0.9998 | 0.9999 | 0.9991 | 0.9993 | |
| Fruit juice | standard curve | y = 0.335x + 0.340 | y = 0.763x + 0.036 | y = 0.662x + 0.716 | y = 0.613x + 0.028 |
| R2 | 0.9998 | 0.9997 | 0.9992 | 0.9991 | |
| White spirits | standard curve | y = 0.55216x + 4.046 | y = 0.287x + 0.887 | y = 0.629x + 1.22 | y = 3.27x + 0.008 |
| R2 | 0.9993 | 0.9996 | 0.9991 | 0.9991 | |
| Sample | Analyte | Added (0.1 μg/L) | Added (1 μg/L) | Added (10 μg/L) | |||
|---|---|---|---|---|---|---|---|
| Extraction Rate (%) | RSD (%) | Extraction Rate (%) | RSD (%) | Extraction Rate (%) | RSD (%) | ||
| Drinking water | DBP | 93.5 ± 2.6 | 2.4 | 90.9 ± 1.9 | 2.4 | 100.7 ± 3.4 | 3.37 |
| DEHP | 85.3 ± 2.4 | 1.7 | 87.3 ± 1.4 | 2.2 | 90.3 ± 2.5 | 2.34 | |
| Fruit juice | DBP | 90.8 ± 1.3 | 3.2 | 90.1 ± 1.2 | 1.9 | 97.7 ± 2.1 | 2.18 |
| DEHP | 80.3 ± 2.1 | 1.4 | 81.3 ± 1.6 | 3.7 | 87.3 ± 1.5 | 3.16 | |
| White spirits | DBP | 80.8 ± 2.3 | 1.5 | 86.1 ± 1.2 | 3.28 | 97.3 ± 3.1 | 1.29 |
| DEHP | 70.3 ± 2.1 | 2.7 | 78.3 ± 1.9 | 2.45 | 87.3 ± 1.0 | 1.96 | |
| Sample 1 | Sample 2 | Sample 3 | Sample 4 (White Spirits) | |
|---|---|---|---|---|
| DBP (mg/kg) | 0.01 | 0.19 | 0.09 | 0.71 |
| DEHP (mg/kg) | 0.82 | 0.66 | 1.02 | 3.22 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, L.; Lu, Y.; Wang, C.; Cheng, L. Fabrication of Magnetic Molecularly Imprinted Polymers for Selective Extraction of Dibutyl Phthalates in Food Matrices. Foods 2024, 13, 1397. https://doi.org/10.3390/foods13091397
Li L, Lu Y, Wang C, Cheng L. Fabrication of Magnetic Molecularly Imprinted Polymers for Selective Extraction of Dibutyl Phthalates in Food Matrices. Foods. 2024; 13(9):1397. https://doi.org/10.3390/foods13091397
Chicago/Turabian StyleLi, Lina, Yunzhu Lu, Chengtao Wang, and Lei Cheng. 2024. "Fabrication of Magnetic Molecularly Imprinted Polymers for Selective Extraction of Dibutyl Phthalates in Food Matrices" Foods 13, no. 9: 1397. https://doi.org/10.3390/foods13091397







