Phenolic Extracts from Wild Olive Leaves and Their Potential as Edible Oils Antioxidants
Abstract
:1. Introduction
2. Experimental Section
2.1. Samples and Reagents
2.2. Extraction of Phenolic Compounds
2.2.1. Solvent Extraction
2.2.2. Supercritical Fluid Extraction
2.3. Phenolic Content Determination
2.4. Antioxidant Activity
2.4.1. DPPH Radical Scavenging Method
2.4.2. Rancimat Method
2.4.3. Peroxide Value Determination
2.5. Statistical Analysis
3. Results and Discussion
3.1. Composition and Phenol Determination
| Parameter | Value ± SD |
|---|---|
| Leaves composition (% w/w) | |
| Moisture | 50.5 ± 1.9 |
| Total solids | 49.5 ± 0.8 |
| Ash | 3.7 ± 0.9 |
| Fat | 1.2 ± 0.2 |
| Total phenols (% w/w) 1 | |
| Methanol extract 2 | 2.06 ± 0.18 a |
| Ethanol extract 2 | 2.73 ± 0.31 b |
| Ethanol:water 1:1 extract 2 | 2.48 ± 0.28 ab |
| n-Propanol extract 2 | 1.37 ± 0.14 c |
| Isopropanol extract 2 | 1.35 ± 0.19 c |
| Ethyl acetate extract 2 | 1.35 ± 0.12 c |
| SFE/CO2 extract | 1.28 ± 0.22 c |
3.2. Solvent Extraction Kinetics
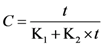
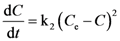
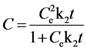
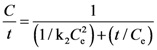
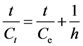
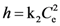 expressing the initial extraction rate h, and Ce equilibrium concentration (extraction capacity).
expressing the initial extraction rate h, and Ce equilibrium concentration (extraction capacity). 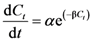
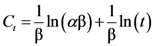

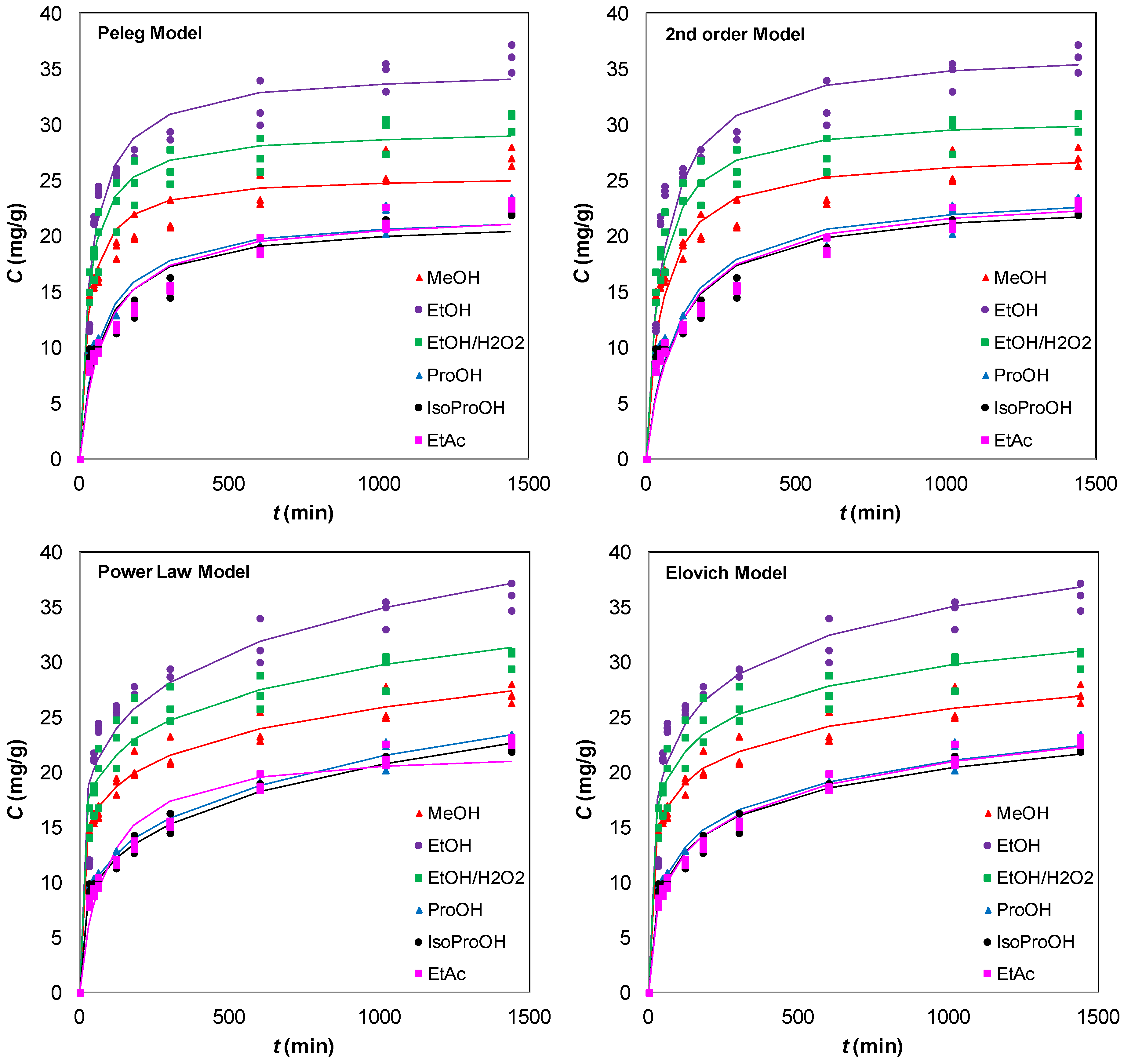
| Methanol | Ethanol | Ethanol:Water 1:1 | n-Propanol | Isopropanol | Ethyl Acetate | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Parameter Values | R2 | Parameter Values | R2 | Parameter Values | R2 | Parameter Values | R2 | Parameter Values | R2 | Parameter Values | R2 | |
| Peleg | ||||||||||||
| K1 | 1.159 ± 0.116 | 0.959 | 1.114 ± 0.102 | 0.967 | 1.034 ± 0.089 | 0.971 | 3.233 ± 0.368 | 0.945 | 3.424 ± 0.376 | 0.949 | 3.734 ± 0.355 | 0.963 |
| K2 | 0.039 ± 0.001 | 0.029 ± 0.001 | 0.034 ± 0.001 | 0.045 ± 0.002 | 0.047 ± 0.002 | 0.045 ± 0.001 | ||||||
| Second-order | ||||||||||||
| k2 | 0.0007 ± 0.0001 | 0.995 | 0.0005 ± 0.0001 | 0.996 | 0.0007 ± 0.0001 | 0.996 | 0.0004 ± 0.00005 | 0.990 | 0.0004 ± 0.00004 | 0.993 | 0.0004 ± 0.00004 | 0.992 |
| h | 0.519 ± 0.089 | 0.644 ± 0.093 | 0.697 ± 0.128 | 0.229 ± 0.027 | 0.225 ± 0.023 | 0.218 ± 0.022 | ||||||
| Ce | 27.535 ± 0.394 | 36.731 ± 0.478 | 30.807 ± 0.393 | 24.215 ± 0.482 | 23.313 ± 0.386 | 23.935 ± 0.421 | ||||||
| Elovich | ||||||||||||
| α | 10.043 ± 2.363 | 0.979 | 5.438 ± 1.880 | 0.935 | 11.501 ± 4.545 | 0.943 | 1.079 ± 0.144 | 0.980 | 1.028 ± 0.134 | 0.980 | 0.850 ± 0.078 | 0.980 |
| β | 0.312 ± 0.013 | 0.200 ± 0.015 | 0.271 ± 0.019 | 0.269 ± 0.011 | 0.277 ± 0.011 | 0.258 ± 0.008 | ||||||
| Power law | ||||||||||||
| B | 8.935 ± 0.356 | 0.988 | 10.269 ± 0.996 | 0.945 | 10.541 ± 0.766 | 0.960 | 3.790 ± 0.158 | 0.994 | 3.649 ± 0.152 | 0.994 | 3.367 ± 0.119 | 0.996 |
| n | 0.154 ± 0.007 | 0.177 ± 0.016 | 0.150 ± 0.012 | 0.251 ± 0.007 | 0.251 ± 0.007 | 0.266 ± 0.006 | ||||||
3.3. Antioxidant Activity of Phenolic Extracts
| Olive Leaf Extract | Antioxidant Activity, as % Inhibition |
|---|---|
| Methanol extract | 46.2 ± 0.75 a |
| Ethanol extract | 55.0 ± 1.09 b |
| Ethanol/water 1:1 extract | 40.9 ± 1.22 c |
| n-Propanol extract | 18.8 ± 0.58 d |
| Isopropanol extract | 32.8 ± 1.41 e |
| Ethyl acetate extract | 22.5 ± 0.63 f |
| SFE/CO2 extract | 33.9 ± 1.81 e |
| Sample | Induction Period 1 (h) | Protection Factor 2 |
|---|---|---|
| Sunflower oil | 7.45 ± 0.07 a | 1.00 |
| Sunflower oil + ascorbyl palmitate 3 | 9.97 ± 0.38 b | 1.34 |
| Sunflower oil + BHT 3 | 10.23 ± 0.13 b | 1.37 |
| Sunflower oil + vitamin E 3 | 9.20 ± 0.41 b | 1.23 |
| Sunflower oil + ethanol extract 4 | 12.97 ± 0.49 d | 1.74 |
| Sunflower oil + ethanol extract 4 + Ascorbyl palmitate 3 | 33.05 ± 0.45 g | 4.44 |
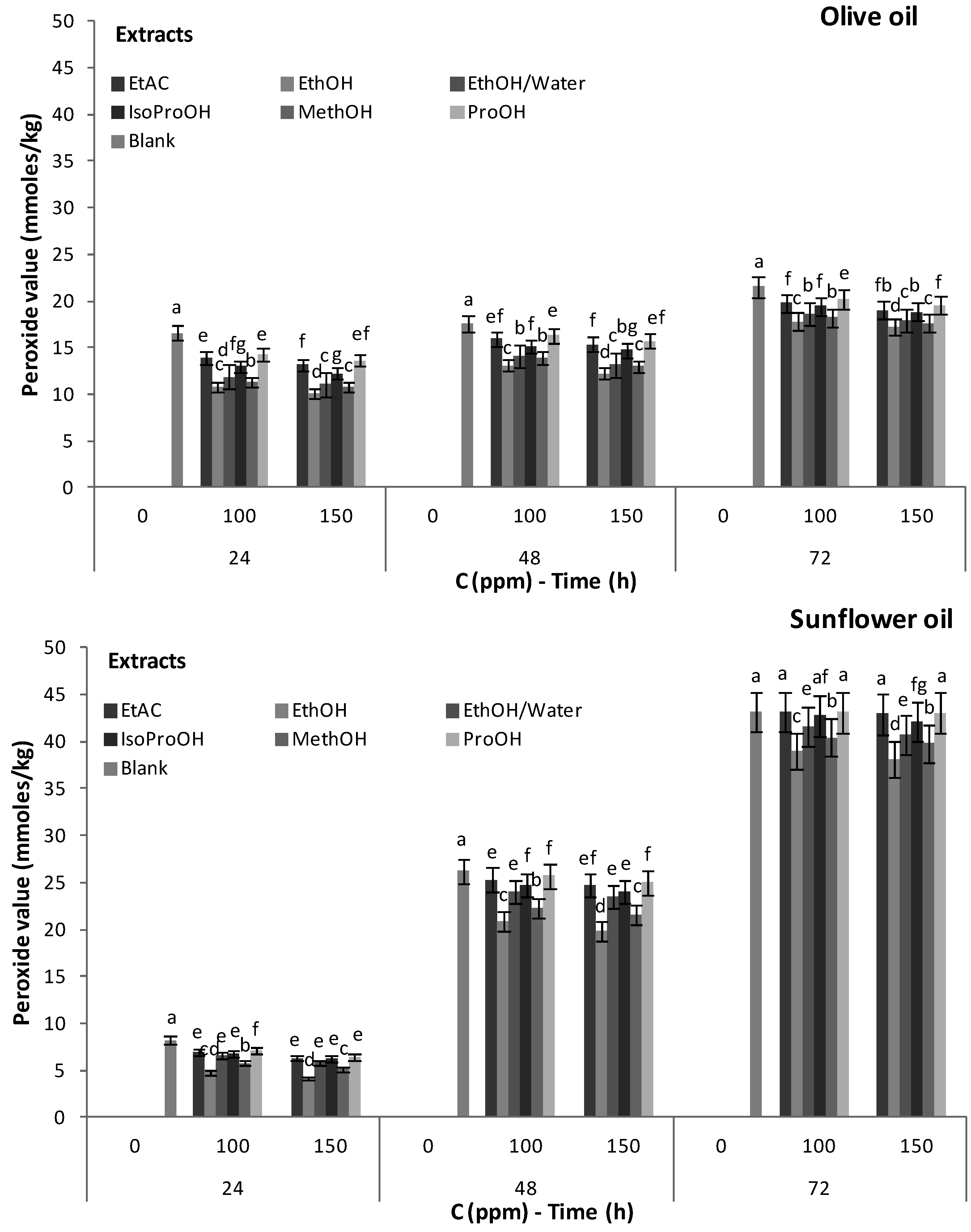
4. Conclusions
Conflicts of Interest
References
- De Leonardis, A.; Aretini, A.; Alfano, G.; Macciola, V.; Ranalli, G. Isolation of a hydroxytyrosol-rich extract from olive leaves (Olea Europaea L.) and evaluation of its antioxidant properties and bioactivity. Eur. Food Res. Technol. 2008, 226, 653–659. [Google Scholar]
- Fki, I.; Allouche, N.; Sayadi, S. The use of polyphenolic extract, purified hydroxytyrosol and 3,4-dihydroxyphenyl acetic acid from olive mill wastewater for the stabilization of refined oils: A potential alternative to synthetic antioxidants. Food Chem. 2005, 93, 197–204. [Google Scholar] [CrossRef]
- Guinda, Á. Use of solid residue from the olive industry. Grasas Y Aceites 2006, 57, 107–115. [Google Scholar]
- Lafka, T.-I.; Lazou, A.E.; Sinanoglou, V.J.; Lazos, E.S. Phenolic and antioxidant potential of olive oil mill wastes. Food Chem. 2011, 125, 92–98. [Google Scholar] [CrossRef]
- Taamalli, A.; Arráez-Román, D.; Zarrouk, M.; Valverde, J.; Segura-Carretero, A.; Fernández-Gutiérrez, A. The occurrence and bioactivity of polyphenols in Tunisian olive products and by-products: A review. J. Food Sci. 2012, 77, R83–R92. [Google Scholar] [CrossRef]
- Fares, R.; Bazzi, S.; Baydoun, S.; Abdel-Massih, R.M. The antioxidant and anti-proliferative activity of the Lebanese Olea europaea extract. Plant Foods Hum. Nutr. 2011, 66, 58–63. [Google Scholar] [CrossRef]
- Massei, G.; Hartley, S.E. Disarmed by domestication? Induced responses to browsing in wild and cultivated olive. Oecologia 2000, 122, 225–231. [Google Scholar] [CrossRef]
- Abaza, L.; Youssef, N.B.; Manai, H.; Haddada, F.M.; Methenni, K.; Zarrouk, M. Chétoui olive leaf extracts: Influence of the solvent type on phenolics and antioxidant activities. Grasas Y Aceites 2011, 62, 96–104. [Google Scholar] [CrossRef]
- Chirinos, R.; Rogez, H.; Campos, D.; Pedreschi, R.; Larondelle, Y. Optimization of extraction conditions of antioxidant phenolic compounds from mashua (Tropaeolum tuberosum Ruíz & Pavón) tubers. Sep. Purif. Technol. 2007, 55, 217–225. [Google Scholar] [CrossRef]
- Lafka, T.-I.; Sinanoglou, V.; Lazos, E.S. On the extraction and antioxidant activity of phenolic compounds from winery wastes. Food Chem. 2007, 104, 1206–1214. [Google Scholar] [CrossRef]
- Le Floch, F.; Tena, M.T.; Ríos, A.; Valcárcel, M. Supercritical fluid extraction of phenol compounds from olive leaves. Talanta 1998, 46, 1123–1130. [Google Scholar] [CrossRef]
- ahin, S.; Bilgin, M. Study on oleuropein extraction from olive tree (Olea europaea) leaves by means of SFE: Comparison of water and ethanol as co-solvent. Sep. Sci. Technol. 2012, 47, 2391–2398. [Google Scholar]
- Association of Official Analytical Chemists (AOAC), Official Methods of Analysis of the Association of Official Analytical Chemists International, 16th ed; AOAC: Arlington, VA, USA, 1995.
- Gutfinger, T. Polyphenols in olive oils. J. Am. Oil Chem. Soc. 1981, 58, 966–968. [Google Scholar] [CrossRef]
- Bandonienė, D.; Murkovic, M.; Pfannhauser, W.; Venskutonis, P.R.; Gruzdienė, D. Detection and activity evaluation of radical scavenging compounds by using DPPH free radical and on-line HPLC-DPPH methods. Eur. Food Res. Technol. 2002, 214, 143–147. [Google Scholar] [CrossRef]
- EEC. Characteristics of olive and olive-pomance oils and their analytical methods. Regulation EEC/2568/1991. Off. J. Eur. Communities 1991, L248, 1–82.
- Peleg, M. An empirical model for the description of moisture sorption curves. J. Food Sci. 1988, 53, 1216–1217. [Google Scholar] [CrossRef]
- Amendola, D., de Faveri; Spigno, G. Grape marc phenolics: Extraction kinetics, quality and stability of extracts. J. Food Eng. 2010, 97, 384–392. [Google Scholar]
- Bucić-Kojić, A.; Planinić, M.; Tomas, S.; Bilić, M.; Velić, D. Study of solid-liquid extraction kinetics of total polyphenols from grape seeds. J. Food Eng. 2007, 81, 236–242. [Google Scholar] [CrossRef]
- Bouaziz, M.; Sayadi, S. Isolation and evaluation of antioxidants from leaves of a Tunisian cultivar olive tree. Eur. J. Lipid Sci. Technol. 2005, 107, 497–504. [Google Scholar] [CrossRef]
- Kiritsakis, K.; Kontominas, M.G.; Kontogiorgis, C.; Hadjipavlou-Litina, D.; Moustakas, A.; Kiritsakis, A. Composition and antioxidant activity of olive leaf extracts from Greek olive cultivars. J. Am. Oil Chem. Soc. 2010, 87, 369–376. [Google Scholar] [CrossRef]
- Kontogianni, V.G.; Gerothanassis, I.P. Phenolic compounds and antioxidant activity of olive leaf extracts. Nat. Prod. Res. 2011, 26, 186–189. [Google Scholar]
- Benavente-García, O.; Castillo, J.; Lorente, J.; Ortuño, A.; Del Rio, J.A. Antioxidant activity of phenolics extracted from Olea europaea L. leaves. Food Chem. 2000, 68, 457–462. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lafka, T.-I.; Lazou, A.E.; Sinanoglou, V.J.; Lazos, E.S. Phenolic Extracts from Wild Olive Leaves and Their Potential as Edible Oils Antioxidants. Foods 2013, 2, 18-31. https://doi.org/10.3390/foods2010018
Lafka T-I, Lazou AE, Sinanoglou VJ, Lazos ES. Phenolic Extracts from Wild Olive Leaves and Their Potential as Edible Oils Antioxidants. Foods. 2013; 2(1):18-31. https://doi.org/10.3390/foods2010018
Chicago/Turabian StyleLafka, Theodora-Ioanna, Andriana E. Lazou, Vassilia J. Sinanoglou, and Evangelos S. Lazos. 2013. "Phenolic Extracts from Wild Olive Leaves and Their Potential as Edible Oils Antioxidants" Foods 2, no. 1: 18-31. https://doi.org/10.3390/foods2010018




