Prebiotics: Definition, Types, Sources, Mechanisms, and Clinical Applications
Abstract
:1. Introduction
2. Definition
3. Types of Prebiotics
3.1. Fructans
3.2. Galacto-Oligosaccharides
3.3. Starch and Glucose-Derived Oligosaccharides
3.4. Other Oligosaccharides
3.5. Non-Carbohydrate Oligosaccharides
4. Production of Prebiotics
4.1. FOS
4.2. GOS
5. Prebiotics Mechanisms for Alteration of Gut Microbiota
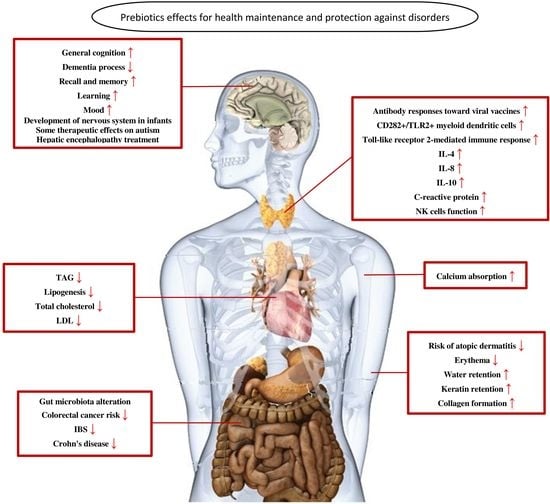
6. Prebiotics Mechanisms for Health Maintenance and Protection against Disorders
6.1. Prebiotics and Gastrointestinal Disorders
6.1.1. Irritable Bowel Syndrome and Crohn’s Disease
6.1.2. Colorectal Cancer
6.1.3. Necrotizing Enterocolitis
6.2. Prebiotics and the Immune System
- I-
- Oligofructose and inulin mixture: The mixture of oligofructans and inulin can improve antibody responses toward viral vaccines, such as influenza and measles [129].
- II-
- FOS: Studies have shown the improvement of antibody response to influenza vaccine following FOS consumption. Moreover, the side effects of the influenza vaccine are reduced [130,131]. Diarrhea-associated fever in infants is also reduced by this category of prebiotics. Apart from these, it can decrease the use of antibiotics, duration of disease, and the incidence of febrile seizures in infants [132,133].β(2→1) fructans can up-regulate the level of interleukin 4 (IL-4) in serum, CD282+/TLR2+ myeloid dendritic cells, and a toll-like receptor 2-mediated immune response in healthy volunteers [134]. In contrast, another study demonstrated that the salivary immunoglobulin A (IgA), immune cells in serum, and activation and proliferation of T cells and natural killer (NK) cells were not changed after consuming β(2→1) fructans [135]. It has been noted that FOS reduces the risk of some immune diseases in infants, such as atopic dermatitis [136,137]. This type of prebiotic decreases the expression of IL-6 and phagocytosis in monocytes and granulocytes [138].
- III-
- GOS: Studies showed that GOS increased the blood level of interleukin 8 (IL-8), interleukin 10 (IL-10), and C-reactive protein in adults, but decreased IL-1β. It has been found that the function of NK cells improves by consuming GOS [139,140]. In infants, GOS reduces the risk of atopic dermatitis and eczema [136,137,141].
- IV-
- AOS (acidic oligosaccharides): The possibility of atopic dermatitis is reduced by AOS in low-risk infants [136].
6.3. Prebiotics and the Nervous System
- I-
- Neural Pathway: The products of prebiotics fermentation can affect the brain by the vagus nerve [146]. Some prebiotics, such as FOS and GOS, have regulatory effects on brain-derived neurotrophic factors, neurotransmitters (e.g., d-serine), and synaptic proteins (e.g., synaptophysin and N-methyl-D-aspartate or NMDA receptor subunits) [147,148].
- II-
- III-
- Immune Pathway: As discussed before, prebiotics can affect different aspects of the immune system. Beside neurological functions, prebiotics are also capable of influencing mood, memory, learning, and some psychiatry disorders by changing the activity and/or composition of gut microbiota [145] (Table 3).
- IV-
- Mood: Stress hormones are able to affect anxiety-related behaviors [150,151]. It was demonstrated that the level of stress hormones (adrenocorticotropic hormone (ACTH) and corticosterone) increased in germ-free mice following exposure to controlled stress. After administrating Bifidobacterium infantis, corticosterone and ACTH reached normal levels [149].
- V-
- Memory, concentration, and learning: Recently, a number of studies have shown the relation between memory and administration of fermentable compounds in both animals and humans [152]. Investigations on a different kind of prebiotics have implicated memory improvement in middle-aged adults [153,154]. Some prebiotics, such as arabinoxylan and arabinose, can enhance general cognition and attenuate the accumulation process of dementia-related glial fibrillary acidic protein in mice [155]. Prebiotics may be more efficient in preserving recall and learning rather than the development process.In 2015, a randomized, double-blind, and placebo-controlled study was performed to examine the effects of FOS and GOS daily consumption for three weeks on the level of salivary cortisol and emotional alteration regarding this hormone. FOS had no significant effect, but 5.5 g GOS intake increased the level of cortisol in saliva and enhanced the concentration in adults [156]. A randomized, double-blind, placebo-controlled trial demonstrated that administration of non-starch polysaccharides (3.6 g per day) for twelve weeks enhanced recall and memory processes in the middle-aged adult [153,154]. In contrast, the mixture of FOS, GOS, and AOS could not enhance the development of the nervous system in preterm infants after 24 months [157]. In two other clinical investigations, Smith et al. observed that administration of inulin-enriched oligofructose might enhance mood, recognition, immediate memory, and recall (after 4 hours). However, this prebiotic failed to recover long-term memory (after 43 days) [158,159].
- VI-
- Autism: 70% of people with autism are suffering from concomitant gastrointestinal disorders compared to 9% of healthy individuals. Chronic constipation (and other diseases as a result of constipation), abdominal pain with or without diarrhea, gastroesophageal reflux disease, abdominal bloating, disaccharide deficiencies, gastrointestinal tract inflammation, and enteric nervous system abnormalities are examples of gastrointestinal symptoms and signs that are reported for patients with autism spectrum disorders [162]. The severity of autism is shown to be correlated to higher gastrointestinal disorders [163]. Interestingly, a review article published in 2016 confirmed these statements [164].The composition of gut microbiota is changed in patients with autism disorders. Some studies have shown high levels of Clostridium and depleted Bifidobacterium in feces. In children with autism, gut metabolites are different from healthy individuals. For example, the amount of SCFAs in children with autism is lower than healthy ones [163,165]. Various prebiotics, such as wheat fiber, may have therapeutic effects on patients with autism by decreasing the population of Clostridium perfringens and increasing the rate of Bifidobacteria [166].Catecholamines, which are a category of neurotransmitters, are increased in individuals with autism. These neurotransmitters are produced by tyrosine hydroxylase. An in vitro study in a rat adrenal medulla cell line demonstrated that SCFAs, the products of prebiotic fermentation, could induce the expression of tyrosine hydroxylase [167]. However, further investigations are required to understand which prebiotics have therapeutic effects on human autism.
- VII-
- Hepatic encephalopathy: Hepatic encephalopathy happens when the liver does not function properly. The main reason for hepatic encephalopathy is the increases in the level of blood ammonia. This condition causes numerous psychiatric and neurologic complications, including personality, speech, and movement disorders, as well as cognition impairment, and may eventually result in coma and death.In 1966, it was demonstrated that lactulose could effectively treat hepatic encephalopathy by decreasing the level of ammonia in the gut. Lactulose can improve the life quality of people suffering from hepatic encephalopathy. This prebiotic also has preventive effects on hepatic encephalopathy [143,168,169,170]. Lactulose exerts its beneficial effects on hepatic encephalopathy through different pathways. First, the product of lactulose fermentation is lactic acid, which is able to reduce the colonic lumen pH by releasing H+. The ammonia in the gut reacts with proton and produces ammonium. This conversion develops a concentration gradient that increases the amount of ammonia reuptake from the blood into the gastrointestinal tract [171]. Second, in the presence of lactulose in the gastrointestinal tract, the bacteria utilize the energy of lactulose fermentation instead of the conversion of amino acids to ammonia energy. Third, lactulose can inhibit glutaminase and prevent the production of ammonia from glutamine [143]. Finally, lactulose shortens the colonic transit time. Thus, it can reduce the level of ammonia in the gastrointestinal tract. Other compounds, such as lactitol, may also be as effective as lactulose in the treatment of hepatic encephalopathy. Interestingly, the side effects of lactitol are much fewer than lactulose (e.g., flatulence and nausea) [172,173,174].
6.4. Prebiotics and Skin
6.5. Prebiotics and Cardiovascular System
6.6. Prebiotics and Calcium Absorption
7. Prebiotics Safety
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Collins, S.; Reid, G. Distant site effects of ingested prebiotics. Nutrients 2016, 8, 523. [Google Scholar] [CrossRef]
- Louis, P.; Flint, H.J.; Michel, C. How to manipulate the microbiota: Prebiotics. In Microbiota of the Human Body; Springer: Basel, Switzerland, 2016; pp. 119–142. [Google Scholar]
- Walker, A.W.; Ince, J.; Duncan, S.H.; Webster, L.M.; Holtrop, G.; Ze, X.; Brown, D.; Stares, M.D.; Scott, P.; Bergerat, A. Dominant and diet-responsive groups of bacteria within the human colonic microbiota. ISME J. 2011, 5, 220–230. [Google Scholar] [CrossRef]
- Glenn, G.; Roberfroid, M. Dietary modulation of the human colonic microbiota: Introducing the concept of prebiotics. J. Nutr. 1995, 125, 1401–1412. [Google Scholar]
- Gibson, G.R.; Probert, H.M.; Van Loo, J.; Rastall, R.A.; Roberfroid, M.B. Dietary modulation of the human colonic microbiota: Updating the concept of prebiotics. Nutr. Res. Rev. 2004, 17, 259–275. [Google Scholar] [CrossRef] [PubMed]
- Bouhnik, Y.; Raskine, L.; Simoneau, G.; Vicaut, E.; Neut, C.; Flourié, B.; Brouns, F.; Bornet, F.R. The capacity of nondigestible carbohydrates to stimulate fecal bifidobacteria in healthy humans: A double-blind, randomized, placebo-controlled, parallel-group, dose-response relation study. Am. J. Clin. Nutr. 2004, 80, 1658–1664. [Google Scholar] [CrossRef] [PubMed]
- Flint, H.J.; Scott, K.P.; Louis, P.; Duncan, S.H. The role of the gut microbiota in nutrition and health. Nat. Rev. Gastroenterol. Hepatol. 2012, 9, 577–589. [Google Scholar] [CrossRef] [PubMed]
- Turroni, F.; Ventura, M.; Buttó, L.F.; Duranti, S.; O’Toole, P.W.; Motherway, M.O.C.; van Sinderen, D. Molecular dialogue between the human gut microbiota and the host: A lactobacillus and bifidobacterium perspective. Cell. Mol. Life Sci. 2014, 71, 183–203. [Google Scholar] [CrossRef]
- Roberfroid, M. Health benefits of non-digestible oligosaccharides. In Dietary Fiber in Health and Disease; Springer: New York, NY, USA, 1997; pp. 211–219. [Google Scholar]
- Morowvat, M.H.; Nezafat, N.; Ghasemi, Y.; Zare, M.H.; Mohkam, M. Probiotic potential of five lactobacillus strains isolated from traditional persian yoghurt in fars province, iran: Viewing through the window of phylogenetics. Biosci. Biotechnol. Res. Asia 2015, 12, 1265–1272. [Google Scholar]
- Shokri, D.; Khorasgani, M.R.; Mohkam, M.; Fatemi, S.M.; Ghasemi, Y.; Taheri-Kafrani, A. The inhibition effect of lactobacilli against growth and biofilm formation of pseudomonas aeruginosa. Probiot. Antimicrob. Proteins 2018, 10, 34–42. [Google Scholar] [CrossRef]
- Stinson, L.F.; Payne, M.S.; Keelan, J.A. Planting the seed: Origins, composition, and postnatal health significance of the fetal gastrointestinal microbiota. Crit. Rev. Microbiol. 2017, 43, 352–369. [Google Scholar] [CrossRef]
- Trompette, A.; Gollwitzer, E.S.; Yadava, K.; Sichelstiel, A.K.; Sprenger, N.; Ngom-Bru, C.; Blanchard, C.; Junt, T.; Nicod, L.P.; Harris, N.L.; et al. Gut microbiota metabolism of dietary fiber influences allergic airway disease and hematopoiesis. Nat. Med. 2014, 20, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Hernot, D.C.; Boileau, T.W.; Bauer, L.L.; Middelbos, I.S.; Murphy, M.R.; Swanson, K.S.; Fahey Jr, G.C. In vitro fermentation profiles, gas production rates, and microbiota modulation as affected by certain fructans, galactooligosaccharides, and polydextrose. J. Agric. Food Chem. 2009, 57, 1354–1361. [Google Scholar] [CrossRef]
- Zhou, Z.; Zhang, Y.; Zheng, P.; Chen, X.; Yang, Y. Starch structure modulates metabolic activity and gut microbiota profile. Anaerobe 2013, 24, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Clarke, T.B.; Davis, K.M.; Lysenko, E.S.; Zhou, A.Y.; Yu, Y.; Weiser, J.N. Recognition of peptidoglycan from the microbiota by nod1 enhances systemic innate immunity. Nat. Med. 2010, 16, 228–231. [Google Scholar] [CrossRef] [PubMed]
- Hamer, H.M.; Jonkers, D.; Venema, K.; Vanhoutvin, S.; Troost, F.; Brummer, R.J. Review article: The role of butyrate on colonic function. Aliment. Pharmacol. Ther. 2008, 27, 104–119. [Google Scholar] [CrossRef] [PubMed]
- den Besten, G.; van Eunen, K.; Groen, A.K.; Venema, K.; Reijngoud, D.-J.; Bakker, B.M. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J. Lipid Res. 2013, 54, 2325–2340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gibson, G.R.; Scott, K.P.; Rastall, R.A.; Tuohy, K.M.; Hotchkiss, A.; Dubert-Ferrandon, A.; Gareau, M.; Murphy, E.F.; Saulnier, D.; Loh, G.; et al. Dietary prebiotics: Current status and new definition. Food Sci. Technol. Bull. Funct. Foods 2010, 7, 1–19. [Google Scholar] [CrossRef]
- Howlett, J.F.; Betteridge, V.A.; Champ, M.; Craig, S.A.; Meheust, A.; Jones, J.M. The definition of dietary fiber-discussions at the ninth vahouny fiber symposium: Building scientific agreement. Food Nutr. Res. 2010, 54, 5750. [Google Scholar] [CrossRef]
- Slavin, J. Fiber and prebiotics: Mechanisms and health benefits. Nutrients 2013, 5, 1417–1435. [Google Scholar] [CrossRef]
- Roberfroid, M.B. Prebiotics: Concept, definition, criteria, methodologies, and products. In Handbook of Prebiotics; CRC Press: Boca Raton, FL, USA, 2008; pp. 49–78. [Google Scholar]
- Bindels, L.B.; Delzenne, N.M.; Cani, P.D.; Walter, J. Towards a more comprehensive concept for prebiotics. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 303–310. [Google Scholar] [CrossRef]
- Scott, K.P.; Gratz, S.W.; Sheridan, P.O.; Flint, H.J.; Duncan, S.H. The influence of diet on the gut microbiota. Pharmacol. Res. 2013, 69, 52–60. [Google Scholar] [CrossRef]
- Hutkins, R.W.; Krumbeck, J.A.; Bindels, L.B.; Cani, P.D.; Fahey, G.; Goh, Y.J.; Hamaker, B.; Martens, E.C.; Mills, D.A.; Rastal, R.A.; et al. Prebiotics: Why definitions matter. Curr. Opin. Biotechnol. 2016, 37, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Scott, K.P.; Martin, J.C.; Duncan, S.H.; Flint, H.J. Prebiotic stimulation of human colonic butyrate-producing bacteria and bifidobacteria, in vitro. FEMS Microbiol. Ecol. 2014, 87, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Macfarlane, G.; Steed, H.; Macfarlane, S. Bacterial metabolism and health-related effects of galacto-oligosaccharides and other prebiotics. J. Appl. Microbiol. 2008, 104, 305–344. [Google Scholar] [CrossRef] [PubMed]
- Johnson, C.R.; Combs, G.F.; Thavarajah, P. Lentil (lens culinaris l.): A prebiotic-rich whole food legume. Food Res. Int. 2013, 51, 107–113. [Google Scholar] [CrossRef]
- Whelan, K. Mechanisms and effectiveness of prebiotics in modifying the gastrointestinal microbiota for the management of digestive disorders. Proc. Nutr. Soc. 2013, 72, 288–298. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fuentes-Zaragoza, E.; Sánchez-Zapata, E.; Sendra, E.; Sayas, E.; Navarro, C.; Fernández-López, J.; Pérez-Alvarez, J.A. Resistant starch as prebiotic: A review. Starch-Stärke 2011, 63, 406–415. [Google Scholar] [CrossRef] [Green Version]
- Ze, X.; Duncan, S.H.; Louis, P.; Flint, H.J. Ruminococcus bromii is a keystone species for the degradation of resistant starch in the human colon. ISME J. 2012, 6, 1535–1543. [Google Scholar] [CrossRef] [Green Version]
- Costabile, A.; Fava, F.; Röytiö, H.; Forssten, S.D.; Olli, K.; Klievink, J.; Rowland, I.R.; Ouwehand, A.C.; Rastall, R.A.; Gibson, G.R.; et al. Impact of polydextrose on the faecal microbiota: A double-blind, crossover, placebo-controlled feeding study in healthy human subjects. Br. J. Nutr. 2012, 108, 471–481. [Google Scholar] [CrossRef]
- Yoo, H.-D.; Kim, D.-J.; Paek, S.-H.; Oh, S.-E. Plant cell wall polysaccharides as potential resources for the development of novel prebiotics. Biomol. Ther. 2012, 20, 371–379. [Google Scholar] [CrossRef]
- Gullón, B.; Gómez, B.; Martínez-Sabajanes, M.; Yáñez, R.; Parajó, J.; Alonso, J. Pectic oligosaccharides: Manufacture and functional properties. Trends Food Sci. Technol. 2013, 30, 153–161. [Google Scholar] [CrossRef]
- Tzounis, X.; Rodriguez-Mateos, A.; Vulevic, J.; Gibson, G.R.; Kwik-Uribe, C.; Spencer, J.P. Prebiotic evaluation of cocoa-derived flavanols in healthy humans by using a randomized, controlled, double-blind, crossover intervention study. Am. J. Clin. Nutr. 2011, 93, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Varzakas, T.; Kandylis, P.; Dimitrellou, D.; Salamoura, C.; Zakynthinos, G.; Proestos, C. Innovative and fortified food: Probiotics, prebiotics, gmos, and superfood. In Preparation and Processing of Religious and Cultural Foods; Elsevier: London, UK, 2018; pp. 67–129. [Google Scholar]
- Al-Sheraji, S.; Ismail, A.; Manap, M.; Mustafa, S.; Yusof, R.; Hassan, F. Prebiotics as functional foods: A review. J. Funct Foods 2013, 5, 1542–1553. [Google Scholar] [CrossRef]
- Panesar, P.S.; Kumari, S.; Panesar, R. Biotechnological approaches for the production of prebiotics and their potential applications. Crit. Rev. Biotechnol. 2013, 33, 345–364. [Google Scholar] [CrossRef] [PubMed]
- Havenaar, R.; Bonnin-Marol, S.; Van Dokkum, W.; Petitet, S.; Schaafsma, G. Inulin: Fermentation and microbial ecology in the intestinal tract. Food Rev. Int. 1999, 15, 109–120. [Google Scholar] [CrossRef]
- Sangeetha, P.; Ramesh, M.; Prapulla, S. Recent trends in the microbial production, analysis and application of fructooligosaccharides. Trends Food Sci. Technol. 2005, 16, 442–457. [Google Scholar] [CrossRef]
- Yun, J.W. Fructooligosaccharides—Occurrence, preparation, and application. Enzym. Microb. Technol. 1996, 19, 107–117. [Google Scholar] [CrossRef]
- Prapulla, S.; Subhaprada, V.; Karanth, N. Microbial production of oligosaccharides: A review. Adv. Appl. Microbiol. 2000, 47, 299–343. [Google Scholar]
- Barreteau, H.; Delattre, C.; Michaud, P. Production of oligosaccharides as promising new food additive generation. Food Technol. Biotechnol. 2006, 44, 323–333. [Google Scholar]
- Caicedo, L.; Silva, E.; Sánchez, O. Semibatch and continuous fructooligosaccharides production by aspergillus sp. N74 in a mechanically agitated airlift reactor. J. Chem. Technol. Biotechnol. 2009, 84, 650–656. [Google Scholar] [CrossRef]
- Sangeetha, P.; Ramesh, M.; Prapulla, S. Production of fructo-oligosaccharides by fructosyl transferase from aspergillus oryzae cfr 202 and aureobasidium pullulans cfr 77. Process Biochem. 2004, 39, 755–760. [Google Scholar] [CrossRef]
- Mohkam, M.; Nezafat, N.; Berenjian, A.; Negahdaripour, M.; Behfar, A.; Ghasemi, Y. Role of bacillus genus in the production of value-added compounds. In Bacilli and Agrobiotechnology; Springer: Basel, Switzerland, 2016; pp. 1–33. [Google Scholar]
- Chen, W.-C.; Liu, C.-H. Production of β-fructofuranosidase by aspergillus japonicus. Enzym. Microb. Technol. 1996, 18, 153–160. [Google Scholar] [CrossRef]
- Prata, M.B.; Mussatto, S.I.; Rodrigues, L.R.; Teixeira, J.A. Fructooligosaccharide production by penicillium expansum. Biotechnol. Lett. 2010, 32, 837–840. [Google Scholar] [CrossRef]
- Maiorano, A.E.; Piccoli, R.M.; Da Silva, E.S.; de Andrade Rodrigues, M.F. Microbial production of fructosyltransferases for synthesis of pre-biotics. Biotechnol. Lett. 2008, 30, 1867–1877. [Google Scholar] [CrossRef]
- Mussatto, S.I.; Aguilar, C.N.; Rodrigues, L.R.; Teixeira, J.A. Fructooligosaccharides and β-fructofuranosidase production by aspergillus japonicus immobilized on lignocellulosic materials. J. Mol. Catal. B Enzym. 2009, 59, 76–81. [Google Scholar] [CrossRef]
- Jong, W.Y.; Seung, K.S. The production of high-content fructo-oligosaccharides from sucrose by the mixed-enzyme system of fructosyltransferase and glucose oxidase. Biotechnol. Lett. 1993, 15, 573–576. [Google Scholar] [CrossRef]
- Sheu, D.C.; Duan, K.J.; Cheng, C.Y.; Bi, J.L.; Chen, J.Y. Continuous production of high-content fructooligosaccharides by a complex cell system. Biotechnol. Prog. 2002, 18, 1282–1286. [Google Scholar] [CrossRef]
- Yun, J.W.; Lee, M.G.; Song, S.K. Batch production of high-content fructo-oligosaccharides from sucrose by the mixed-enzyme system of β-fructofuranosidase and glucose oxidase. J. Ferment. Bioeng. 1994, 77, 159–163. [Google Scholar] [CrossRef]
- Lin, T.-J.; Lee, Y.-C. High-content fructooligosaccharides production using two immobilized microorganisms in an internal-loop airlift bioreactor. J. Chin. Inst. Chem. Eng. 2008, 39, 211–217. [Google Scholar] [CrossRef]
- Nishizawa, K.; Nakajima, M.; Nabetani, H. Kinetic study on transfructosylation by. Beta.-fructofuranosidase from aspergillus niger atcc 20611 and availability of a membrane reactor for fructooligosaccharide production. Food Sci. Technol. Res. 2001, 7, 39–44. [Google Scholar] [CrossRef]
- Crittenden, R.; Playne, M. Purification of food-grade oligosaccharides using immobilised cells of zymomonas mobilis. Appl. Microbiol. Biotechnol. 2002, 58, 297–302. [Google Scholar]
- Goulas, A.; Tzortzis, G.; Gibson, G.R. Development of a process for the production and purification of α-and β-galactooligosaccharides from bifidobacterium bifidum ncimb 41171. Int. Dairy J. 2007, 17, 648–656. [Google Scholar] [CrossRef]
- Hernández, O.; Ruiz-Matute, A.I.; Olano, A.; Moreno, F.J.; Sanz, M.L. Comparison of fractionation techniques to obtain prebiotic galactooligosaccharides. Int. Dairy J. 2009, 19, 531–536. [Google Scholar] [CrossRef] [Green Version]
- Yoon, S.-H.; Mukerjea, R.; Robyt, J.F. Specificity of yeast (saccharomyces cerevisiae) in removing carbohydrates by fermentation. Carbohydr. Res. 2003, 338, 1127–1132. [Google Scholar] [CrossRef]
- Palcic, M.M. Biocatalytic synthesis of oligosaccharides. Curr. Opin. Biotechnol. 1999, 10, 616–624. [Google Scholar] [CrossRef]
- Weijers, C.A.; Franssen, M.C.; Visser, G.M. Glycosyltransferase-catalyzed synthesis of bioactive oligosaccharides. Biotechnol. Adv. 2008, 26, 436–456. [Google Scholar] [CrossRef]
- Koizumi, S.; Endo, T.; Tabata, K.; Ozaki, A. Large-scale production of udp-galactose and globotriose by coupling metabolically engineered bacteria. Nat. Biotechnol. 1998, 16, 847–850. [Google Scholar] [CrossRef]
- Albermann, C.; Piepersberg, W.; Wehmeier, U.F. Synthesis of the milk oligosaccharide 2′-fucosyllactose using recombinant bacterial enzymes. Carbohydr. Res. 2001, 334, 97–103. [Google Scholar] [CrossRef]
- Priem, B.; Gilbert, M.; Wakarchuk, W.W.; Heyraud, A.; Samain, E. A new fermentation process allows large-scale production of human milk oligosaccharides by metabolically engineered bacteria. Glycobiology 2002, 12, 235–240. [Google Scholar] [CrossRef] [Green Version]
- Monsan, P.; Paul, F. Enzymatic synthesis of oligosaccharides. FEMS Microbiol. Rev. 1995, 16, 187–192. [Google Scholar] [CrossRef] [Green Version]
- Osman, A.; Tzortzis, G.; Rastall, R.A.; Charalampopoulos, D. Bbgiv is an important bifidobacterium β-galactosidase for the synthesis of prebiotic galactooligosaccharides at high temperatures. J. Agric. Food Chem. 2012, 60, 740–748. [Google Scholar] [CrossRef]
- Prenosil, J.; Stuker, E.; Bourne, J. Formation of oligosaccharides during enzymatic lactose: Part i: State of art. Biotechnol. Bioeng. 1987, 30, 1019–1025. [Google Scholar] [CrossRef]
- Rabiu, B.A.; Jay, A.J.; Gibson, G.R.; Rastall, R.A. Synthesis and fermentation properties of novel galacto-oligosaccharides by β-galactosidases frombifidobacterium species. Appl. Environ. Microbiol. 2001, 67, 2526–2530. [Google Scholar] [CrossRef]
- Zarate, S.; Lopez-Leiva, M. Oligosaccharide formation during enzymatic lactose hydrolysis: A literature review. J. Food Prot. 1990, 53, 262–274. [Google Scholar] [CrossRef]
- Neri, D.F.; Balcão, V.M.; Costa, R.S.; Rocha, I.C.; Ferreira, E.M.; Torres, D.P.; Rodrigues, L.R.; Carvalho, L.B.; Teixeira, J.A. Galacto-oligosaccharides production during lactose hydrolysis by free aspergillus oryzae β-galactosidase and immobilized on magnetic polysiloxane-polyvinyl alcohol. Food Chem. 2009, 115, 92–99. [Google Scholar] [CrossRef]
- Iqbal, S.; Nguyen, T.-H.; Nguyen, T.T.; Maischberger, T.; Haltrich, D. Β-galactosidase from lactobacillus plantarum wcfs1: Biochemical characterization and formation of prebiotic galacto-oligosaccharides. Carbohydr. Res. 2010, 345, 1408–1416. [Google Scholar] [CrossRef]
- Iqbal, S.; Nguyen, T.-H.; Nguyen, H.A.; Nguyen, T.T.; Maischberger, T.; Kittl, R.; Haltrich, D. Characterization of a heterodimeric gh2 β-galactosidase from lactobacillus sakei lb790 and formation of prebiotic galacto-oligosaccharides. J. Agric. Food Chem. 2011, 59, 3803–3811. [Google Scholar] [CrossRef]
- Yi, S.H.; Alli, I.; Park, K.H.; Lee, B. Overexpression and characterization of a novel transgalactosylic and hydrolytic β-galactosidase from a human isolate bifidobacterium breve b24. New Biotechnol. 2011, 28, 806–813. [Google Scholar] [CrossRef]
- Fukuda, H.; Hama, S.; Tamalampudi, S.; Noda, H. Whole-cell biocatalysts for biodiesel fuel production. Trends Biotechnol. 2008, 26, 668–673. [Google Scholar] [CrossRef]
- Burton, S.G.; Cowan, D.A.; Woodley, J.M. The search for the ideal biocatalyst. Nat. Biotechnol. 2002, 20, 37–45. [Google Scholar] [CrossRef] [Green Version]
- Schmid, A.; Dordick, J.; Hauer, B.; Kiener, A.; Wubbolts, M.; Witholt, B. Industrial biocatalysis today and tomorrow. Nature 2001, 409, 258–268. [Google Scholar] [CrossRef] [PubMed]
- Onishi, N.; Yokozeki, K. Gluco-oligosaccharide and galacto-oligosaccharide production by rhodotorula minuta ifo879. J. Ferment. Bioeng. 1996, 82, 124–127. [Google Scholar] [CrossRef]
- Onishi, N.; Yamashiro, A.; Yokozeki, K. Production of galacto-oligosaccharide from lactose by sterigmatomyces elviae cbs8119. Appl. Environ. Microbiol. 1995, 61, 4022–4025. [Google Scholar] [PubMed]
- Onishi, N.; Kira, I.; Yokozeki, K. Galacto-oligosaccharide production from lactose by sirobasidium magnum cbs6803. Lett. Appl. Microbiol. 1996, 23, 253–256. [Google Scholar] [CrossRef] [PubMed]
- Onishi, N.; Tanaka, T. Purification and properties of a galacto-and gluco-oligosaccharide-producing β-glycosidase from rhodotorula minuta ifo879. J. Ferment. Bioeng. 1996, 82, 439–443. [Google Scholar] [CrossRef]
- Onishi, N.; Tanaka, T. Purification and characterization of galacto-oligosaccharide-producing β-galactosidase from sirobasidium magnum. Lett. Appl. Microbiol. 1997, 24, 82–86. [Google Scholar] [CrossRef]
- Li, Y.; Lu, L.; Wang, H.; Xu, X.; Xiao, M. Cell surface engineering of a β-galactosidase for galactooligosaccharide synthesis. Appl. Environ. Microbiol. 2009, 75, 5938–5942. [Google Scholar] [CrossRef] [PubMed]
- Osman, A.; Tzortzis, G.; Rastall, R.A.; Charalampopoulos, D. A comprehensive investigation of the synthesis of prebiotic galactooligosaccharides by whole cells of bifidobacterium bifidum ncimb 41171. J. Biotechnol. 2010, 150, 140–148. [Google Scholar] [CrossRef] [PubMed]
- Ji, E.-S.; Park, N.-H.; Oh, D.-K. Galacto-oligosaccharide production by a thermostable recombinant β-galactosidase from thermotoga maritima. World J. Microbiol. Biotechnol. 2005, 21, 759–764. [Google Scholar] [CrossRef]
- Terpe, K. Overview of bacterial expression systems for heterologous protein production: From molecular and biochemical fundamentals to commercial systems. Appl. Microbiol. Biotechnol. 2006, 72, 211–222. [Google Scholar] [CrossRef]
- Demain, A.L.; Vaishnav, P. Production of recombinant proteins by microbes and higher organisms. Biotechnol. Adv. 2009, 27, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Li, G.; Ren, X.; Herrler, G. Select what you need: A comparative evaluation of the advantages and limitations of frequently used expression systems for foreign genes. J. Biotechnol. 2007, 127, 335–347. [Google Scholar] [CrossRef]
- Porro, D.; Sauer, M.; Branduardi, P.; Mattanovich, D. Recombinant protein production in yeasts. Mol. Biotechnol. 2005, 31, 245–259. [Google Scholar] [CrossRef]
- Buckholz, R.G.; Gleeson, M.A. Yeast systems for the commercial production of heterologous proteins. Nat. Biotechnol. 1991, 9, 1067–1072. [Google Scholar] [CrossRef]
- Flint, H.J.; Duncan, S.H.; Scott, K.P.; Louis, P. Interactions and competition within the microbial community of the human colon: Links between diet and health. Environ. Microbiol. 2007, 9, 1101–1111. [Google Scholar] [CrossRef] [PubMed]
- Cecchini, D.A.; Laville, E.; Laguerre, S.; Robe, P.; Leclerc, M.; Doré, J.; Henrissat, B.; Remaud-Siméon, M.; Monsan, P.; Potocki-Véronèse, G. Functional metagenomics reveals novel pathways of prebiotic breakdown by human gut bacteria. PLoS ONE 2013, 8, e72766. [Google Scholar] [CrossRef] [PubMed]
- Belenguer, A.; Duncan, S.H.; Calder, A.G.; Holtrop, G.; Louis, P.; Lobley, G.E.; Flint, H.J. Two routes of metabolic cross-feeding between bifidobacterium adolescentis and butyrate-producing anaerobes from the human gut. Appl. Environ. Microbiol. 2006, 72, 3593–3599. [Google Scholar] [CrossRef]
- Ryan, S.M.; Fitzgerald, G.F.; van Sinderen, D. Screening for and identification of starch-, amylopectin-, and pullulan-degrading activities in bifidobacterial strains. Appl. Environ. Microbiol. 2006, 72, 5289–5296. [Google Scholar] [CrossRef]
- Rossi, M.; Corradini, C.; Amaretti, A.; Nicolini, M.; Pompei, A.; Zanoni, S.; Matteuzzi, D. Fermentation of fructooligosaccharides and inulin by bifidobacteria: A comparative study of pure and fecal cultures. Appl. Environ. Microbiol. 2005, 71, 6150–6158. [Google Scholar] [CrossRef]
- Falony, G.; Vlachou, A.; Verbrugghe, K.; De Vuyst, L. Cross-feeding between bifidobacterium longum bb536 and acetate-converting, butyrate-producing colon bacteria during growth on oligofructose. Appl. Environ. Microbiol. 2006, 72, 7835–7841. [Google Scholar] [CrossRef]
- Walker, A.W.; Duncan, S.H.; Leitch, E.C.M.; Child, M.W.; Flint, H.J. Ph and peptide supply can radically alter bacterial populations and short-chain fatty acid ratios within microbial communities from the human colon. Appl. Environ. Microbiol. 2005, 71, 3692–3700. [Google Scholar] [CrossRef]
- Duncan, S.H.; Louis, P.; Thomson, J.M.; Flint, H.J. The role of ph in determining the species composition of the human colonic microbiota. Environ. Microbiol. 2009, 11, 2112–2122. [Google Scholar] [CrossRef] [PubMed]
- Wilson, B.; Whelan, K. Prebiotic inulin-type fructans and galacto-oligosaccharides: Definition, specificity, function, and application in gastrointestinal disorders. J. Gastroenterol. Hepatol. 2017, 32, 64–68. [Google Scholar] [CrossRef] [PubMed]
- Hunter, J.; Tuffnell, Q.; Lee, A. Controlled trial of oligofructose in the management of irritable bowel syndrome. J. Nutr. 1999, 129, 1451S–1453S. [Google Scholar] [CrossRef] [PubMed]
- Olesen, M.; Gudmand-Høyer, E. Efficacy, safety, and tolerability of fructooligosaccharides in the treatment of irritable bowel syndrome. Am. J. Clin. Nutr. 2000, 72, 1570–1575. [Google Scholar] [CrossRef] [PubMed]
- Paineau, D.; Payen, F.; Panserieu, S.; Coulombier, G.; Sobaszek, A.; Lartigau, I.; Brabet, M.; Galmiche, J.-P.; Tripodi, D.; Sacher-Huvelin, S.; et al. The effects of regular consumption of short-chain fructo-oligosaccharides on digestive comfort of subjects with minor functional bowel disorders. Br. J. Nutr. 2008, 99, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Silk, D.; Davis, A.; Vulevic, J.; Tzortzis, G.; Gibson, G. Clinical trial: The effects of a trans-galactooligosaccharide prebiotic on faecal microbiota and symptoms in irritable bowel syndrome. Aliment. Pharmacol. Ther. 2009, 29, 508–518. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, J.O.; Whelan, K.; Stagg, A.J.; Gobin, P.; Al-Hassi, H.O.; Rayment, N.; Kamm, M.; Knight, S.C.; Forbes, A. Clinical, microbiological, and immunological effects of fructo-oligosaccharide in patients with crohn’s disease. Gut 2006, 55, 348–355. [Google Scholar] [CrossRef]
- Benjamin, J.L.; Hedin, C.R.; Koutsoumpas, A.; Ng, S.C.; McCarthy, N.E.; Hart, A.L.; Kamm, M.A.; Sanderson, J.D.; Knight, S.C.; Forbes, A.; et al. Randomised, double-blind, placebo-controlled trial of fructo-oligosaccharides in active crohn’s disease. Gut 2011, 60, 923–929. [Google Scholar] [CrossRef]
- Joossens, M.; De Preter, V.; Ballet, V.; Verbeke, K.; Rutgeerts, P.; Vermeire, S. Effect of oligofructose-enriched inulin (of-in) on bacterial composition and disease activity of patients with crohn’s disease: Results from a double-blinded randomised controlled trial. Gut 2011, 61, 958. [Google Scholar] [CrossRef]
- Candela, M.; Guidotti, M.; Fabbri, A.; Brigidi, P.; Franceschi, C.; Fiorentini, C. Human intestinal microbiota: Cross-talk with the host and its potential role in colorectal cancer. Crit. Rev. Microbiol. 2011, 37, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Louis, P.; Flint, H.J. Diversity, metabolism and microbial ecology of butyrate-producing bacteria from the human large intestine. FEMS Microbiol. Lett. 2009, 294, 1–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davis, C.D.; Milner, J.A. Gastrointestinal microflora, food components and colon cancer prevention. J. Nutr. Biochem. 2009, 20, 743–752. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pool-Zobel, B.L. Inulin-type fructans and reduction in colon cancer risk: Review of experimental and human data. Br. J. Nutr. 2005, 93, S73–S90. [Google Scholar] [CrossRef]
- Rafter, J.; Bennett, M.; Caderni, G.; Clune, Y.; Hughes, R.; Karlsson, P.C.; Klinder, A.; O’Riordan, M.; O’Sullivan, G.C.; Pool-Zobel, B.; et al. Dietary synbiotics reduce cancer risk factors in polypectomized and colon cancer patients. Am. J. Clin. Nutr. 2007, 85, 488–496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patel, R.M.; Denning, P.W. Therapeutic use of prebiotics, probiotics, and postbiotics to prevent necrotizing enterocolitis: What is the current evidence? Clin. Perinatol. 2013, 40, 11–25. [Google Scholar] [CrossRef] [PubMed]
- Knol, J.; Boehm, G.; Lidestri, M.; Negretti, F.; Jelinek, J.; Agosti, M.; Stahl, B.; Marini, A.; Mosca, F. Increase of faecal bifidobacteria due to dietary oligosaccharides induces a reduction of clinically relevant pathogen germs in the faeces of formula-fed preterm infants. Acta Paediatr. 2005, 94, 31–33. [Google Scholar] [CrossRef]
- Boehm, G.; Lidestri, M.; Casetta, P.; Jelinek, J.; Negretti, F.; Stahl, B.; Marini, A. Supplementation of a bovine milk formula with an oligosaccharide mixture increases counts of faecal bifidobacteria in preterm infants. Arch. Dis. Childhood-Fetal Neonatal Ed. 2002, 86, F178–F181. [Google Scholar] [CrossRef]
- Kapiki, A.; Costalos, C.; Oikonomidou, C.; Triantafyllidou, A.; Loukatou, E.; Pertrohilou, V. The effect of a fructo-oligosaccharide supplemented formula on gut flora of preterm infants. Early Hum. Dev. 2007, 83, 335–339. [Google Scholar] [CrossRef]
- Indrio, F.; Riezzo, G.; Raimondi, F.; Bisceglia, M.; Cavallo, L.; Francavilla, R. Effects of probiotic and prebiotic on gastrointestinal motility in newborns. J. Physiol. Pharmacol. 2009, 60, 27–31. [Google Scholar]
- Indrio, F.; Riezzo, G.; Raimondi, F.; Francavilla, R.; Montagna, O.; Valenzano, M.L.; Cavallo, L.; Boehm, G. Prebiotics improve gastric motility and gastric electrical activity in preterm newborns. J. Pediatr. Gastroenterol. Nutr. 2009, 49, 258–261. [Google Scholar] [CrossRef] [PubMed]
- Labayen, I.; Forga, L.; Gonzalez, A.; Lenoir-Wijnkoop, I.; Martinez, J.A. Relationship between lactose digestion, gastrointestinal transit time and symptoms in lactose malabsorbers after dairy consumption. Aliment. Pharmacol. Ther. 2001, 15, 543–549. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Srinivasjois, R.; Rao, S.; Patole, S. Prebiotic supplementation of formula in preterm neonates: A systematic review and meta-analysis of randomised controlled trials. Clin. Nutr. 2009, 28, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Klinder, A.; Gietl, E.; Hughes, R.; Jonkers, N.; Karlsson, P.; McGlyn, H.; Pistoli, S.; Tuohy, K.; Rafter, J.; Rowland, I.; et al. Gut fermentation products of insulin-derived prebiotics beneficially modulate markers of tumour progression in human colon tumour cells. Int. J. Cancer Prev. 2004, 1, 19–32. [Google Scholar]
- Verghese, M.; Rao, D.; Chawan, C.; Shackelford, L. Dietary inulin suppresses azoxymethane-induced preneoplastic aberrant crypt foci in mature fisher 344 rats. J. Nutr. 2002, 132, 2804–2808. [Google Scholar] [CrossRef] [PubMed]
- Denji, K.A.; Mansour, M.R.; Akrami, R.; Ghobadi, S.; Jafarpour, S.; Mirbeygi, S. Effect of dietary prebiotic mannan oligosaccharide (mos) on growth performance, intestinal microflora, body composition, haematological and blood serum biochemical parameters of rainbow trout (oncorhynchus mykiss) juveniles. J. Fish. Aquat. Sci. 2015, 10, 255. [Google Scholar]
- Klatt, N.R.; Canary, L.A.; Sun, X.; Vinton, C.L.; Funderburg, N.T.; Morcock, D.R.; Quiñones, M.; Deming, C.B.; Perkins, M.; Hazuda, D.J.; et al. Probiotic/prebiotic supplementation of antiretrovirals improves gastrointestinal immunity in siv-infected macaques. J. Clin. Investig. 2013, 123, 903–907. [Google Scholar] [CrossRef]
- Langen, L.V.; Mirjam, A.; Dieleman, L.A. Prebiotics in chronic intestinal inflammation. Inflamm. Bowel Dis. 2009, 15, 454–462. [Google Scholar] [CrossRef]
- Steed, H.; Macfarlane, S. Mechanisms of prebiotic impact on health. In Prebiotics and Probiotics Science and Technology; Springer: New York, NY, USA, 2009; pp. 135–161. [Google Scholar]
- Oyofo, B.; DeLoach, J.; Corrier, D.; Norman, J.; Ziprin, R.; Mollenhauer, H. Prevention of salmonella typhimurium colonization of broilers with d-mannose. Poult. Sci. 1989, 68, 1357–1360. [Google Scholar] [CrossRef]
- Thorburn, A.N.; Macia, L.; Mackay, C.R. Diet, metabolites, and “western-lifestyle” inflammatory diseases. Immunity 2014, 40, 833–842. [Google Scholar] [CrossRef]
- Fujiwara, R.; Takemura, N.; Watanabe, J.; Sonoyama, K. Maternal consumption of fructo-oligosaccharide diminishes the severity of skin inflammation in offspring of nc/nga mice. Br. J. Nutr. 2010, 103, 530–538. [Google Scholar] [CrossRef]
- Shadid, R.; Haarman, M.; Knol, J.; Theis, W.; Beermann, C.; Rjosk-Dendorfer, D.; Schendel, D.J.; Koletzko, B.V.; Krauss-Etschmann, S. Effects of galactooligosaccharide and long-chain fructooligosaccharide supplementation during pregnancy on maternal and neonatal microbiota and immunity—A randomized, double-blind, placebo-controlled study. Am. J. Clin. Nutr. 2007, 86, 1426–1437. [Google Scholar] [CrossRef] [PubMed]
- Firmansyah, A.; Pramita, G.; Carrie Fassler, A.; Haschke, F.; Link-Amster, H. Improved humoral immune response to measles vaccine in infants receiving infant cereal with fructooligosaccharides. J. Pediatr. Gastroenterol. Nutr. 2001, 31, A521. [Google Scholar]
- Langkamp-Henken, B.; Bender, B.S.; Gardner, E.M.; Herrlinger-Garcia, K.A.; Kelley, M.J.; Murasko, D.M.; Schaller, J.P.; Stechmiller, J.K.; Thomas, D.J.; Wood, S.M. Nutritional formula enhanced immune function and reduced days of symptoms of upper respiratory tract infection in seniors. J. Am. Geriatr. Soc. 2004, 52, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Lomax, A.R.; Cheung, L.V.; Tuohy, K.M.; Noakes, P.S.; Miles, E.A.; Calder, P.C. Β2-1 fructans have a bifidogenic effect in healthy middle-aged human subjects but do not alter immune responses examined in the absence of an in vivo immune challenge: Results from a randomised controlled trial. Br. J. Nutr. 2012, 108, 1818–1828. [Google Scholar] [CrossRef] [PubMed]
- Saavedra, J.; Tschernia, A.; Moore, N.; Abi-Hanna, A.; Coletta, F.; Emenhiser, C.; Yolken, R. Gastro-intestinal function in infants consuming a weaning food supplemented with oligofructose, a prebiotic. J. Pediatr. Gastroenterol. Nutr. 1999, 29, 513. [Google Scholar] [CrossRef]
- Tschernia, A.; Moore, N.; Abi-Hanna, A.; Yolken, R.; Coletta, F.; Emenhiser, C.; Saavedra, J. Effects of long-term consumption of a weaning food supplemented with oligofructose, a prebiotic, on general infant health status. J. Pediatr. Gastroenterol. Nutr. 1999, 29, 503. [Google Scholar] [CrossRef]
- Clarke, S.; Green-Johnson, J.; Brooks, S.; Ramdath, D.; Bercik, P.; Avila, C.; Inglis, G.; Green, J.; Yanke, L.; Selinger, L. Β2-1 fructan supplementation alters host immune responses in a manner consistent with increased exposure to microbial components: Results from a double-blinded, randomised, cross-over study in healthy adults. Br. J. Nutr. 2016, 115, 1748–1759. [Google Scholar] [CrossRef]
- Lomax, A.R.; Cheung, L.V.; Noakes, P.S.; Miles, E.A.; Calder, P.C. Inulin-type β2-1 fructans have some effect on the antibody response to seasonal influenza vaccination in healthy middle-aged humans. Front. Immunol. 2015, 6, 490. [Google Scholar] [CrossRef]
- Grüber, C.; van Stuijvenberg, M.; Mosca, F.; Moro, G.; Chirico, G.; Braegger, C.P.; Riedler, J.; Boehm, G.; Wahn, U.; MIPS 1 Working Group. Reduced occurrence of early atopic dermatitis because of immunoactive prebiotics among low-atopy-risk infants. J. Allergy Clin. Immunol. 2010, 126, 791–797. [Google Scholar]
- Moro, G.; Arslanoglu, S.; Stahl, B.; Jelinek, J.; Wahn, U.; Boehm, G. A mixture of prebiotic oligosaccharides reduces the incidence of atopic dermatitis during the first six months of age. Arch. Dis. Childhood 2006, 91, 814–819. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guigoz, Y.; Rochat, F.; Perruisseau-Carrier, G.; Rochat, I.; Schiffrin, E. Effects of oligosaccharide on the faecal flora and non-specific immune system in elderly people. Nutr. Res. 2002, 22, 13–25. [Google Scholar] [CrossRef]
- Vulevic, J.; Drakoularakou, A.; Yaqoob, P.; Tzortzis, G.; Gibson, G.R. Modulation of the fecal microflora profile and immune function by a novel trans-galactooligosaccharide mixture (b-gos) in healthy elderly volunteers. Am. J. Clin. Nutr. 2008, 88, 1438–1446. [Google Scholar] [PubMed]
- Vulevic, J.; Juric, A.; Walton, G.E.; Claus, S.P.; Tzortzis, G.; Toward, R.E.; Gibson, G.R. Influence of galacto-oligosaccharide mixture (b-gos) on gut microbiota, immune parameters and metabonomics in elderly persons. Br. J. Nutr. 2015, 114, 586–595. [Google Scholar] [CrossRef] [PubMed]
- Kukkonen, K.; Savilahti, E.; Haahtela, T.; Juntunen-Backman, K.; Korpela, R.; Poussa, T.; Tuure, T.; Kuitunen, M. Probiotics and prebiotic galacto-oligosaccharides in the prevention of allergic diseases: A randomized, double-blind, placebo-controlled trial. J. Allergy Clin. Immunol. 2007, 119, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Gaman, A.; Kuo, B. Neuromodulatory processes of the brain–gut axis. Neuromodulation 2008, 11, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Mudd, A.T.; Alexander, L.S.; Berding, K.; Waworuntu, R.V.; Berg, B.M.; Donovan, S.M.; Dilger, R.N. Dietary prebiotics, milk fat globule membrane, and lactoferrin affects structural neurodevelopment in the young piglet. Front. Pediatr. 2016, 4, 4. [Google Scholar] [CrossRef]
- Grenham, S.; Clarke, G.; Cryan, J.F.; Dinan, T.G. Brain–gut–microbe communication in health and disease. Front. Physiol. 2011, 2, 94. [Google Scholar] [CrossRef]
- Liu, X.; Cao, S.; Zhang, X. Modulation of gut microbiota–brain axis by probiotics, prebiotics, and diet. J. Agric. Food Chem. 2015, 63, 7885–7895. [Google Scholar] [CrossRef]
- Forsythe, P.; Bienenstock, J.; Kunze, W.A. Vagal pathways for microbiome-brain-gut axis communication. In Microbial Endocrinology: The Microbiota-Gut-Brain Axis in Health and Disease; Springer: New York, NY, USA, 2014; pp. 115–133. [Google Scholar]
- Savignac, H.M.; Corona, G.; Mills, H.; Chen, L.; Spencer, J.P.; Tzortzis, G.; Burnet, P.W. Prebiotic feeding elevates central brain derived neurotrophic factor, n-methyl-d-aspartate receptor subunits and d-serine. Neurochem. Int. 2013, 63, 756–764. [Google Scholar] [CrossRef]
- Williams, S.; Chen, L.; Savignac, H.M.; Tzortzis, G.; Anthony, D.C.; Burnet, P.W. Neonatal prebiotic (bgos) supplementation increases the levels of synaptophysin, glun2a-subunits and bdnf proteins in the adult rat hippocampus. Synapse 2016, 70, 121–124. [Google Scholar] [CrossRef] [PubMed]
- Sudo, N.; Chida, Y.; Aiba, Y.; Sonoda, J.; Oyama, N.; Yu, X.N.; Kubo, C.; Koga, Y. Postnatal microbial colonization programs the hypothalamic–pituitary–adrenal system for stress response in mice. J. Physiol. 2004, 558, 263–275. [Google Scholar] [CrossRef] [PubMed]
- Heijtz, R.D.; Wang, S.; Anuar, F.; Qian, Y.; Björkholm, B.; Samuelsson, A.; Hibberd, M.L.; Forssberg, H.; Pettersson, S. Normal gut microbiota modulates brain development and behavior. Proc. Natl. Acad. Sci. USA 2011, 108, 3047–3052. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neufeld, K.; Kang, N.; Bienenstock, J.; Foster, J. Reduced anxiety-like behavior and central neurochemical change in germ-free mice. Neurogastroenterol. Motil. 2011, 23, 255-e119. [Google Scholar] [CrossRef] [PubMed]
- Nelson, E.D.; Ramberg, J.E.; Best, T.; Sinnott, R.A. Neurologic effects of exogenous saccharides: A review of controlled human, animal, and in vitro studies. Nutr. Neurosci. 2012, 15, 149–162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Best, T.; Howe, P.; Bryan, J.; Buckley, J.; Scholey, A. Acute effects of a dietary non-starch polysaccharide supplement on cognitive performance in healthy middle-aged adults. Nutr. Neurosci. 2015, 18, 76–86. [Google Scholar] [CrossRef] [PubMed]
- Best, T.; Kemps, E.; Bryan, J. Saccharide effects on cognition and well-being in middle-aged adults: A randomized controlled trial. Dev. Neuropsychol. 2009, 35, 66–80. [Google Scholar] [CrossRef]
- Han, H.S.; Jang, J.-H.; Jang, J.H.; Choi, J.S.; Kim, Y.J.; Lee, C.; Lim, S.H.; Lee, H.-K.; Lee, J. Water extract of triticum aestivum l. And its components demonstrate protective effect in a model of vascular dementia. J. Med. Food 2010, 13, 572–578. [Google Scholar] [CrossRef]
- Schmidt, K.; Cowen, P.J.; Harmer, C.J.; Tzortzis, G.; Errington, S.; Burnet, P.W. Prebiotic intake reduces the waking cortisol response and alters emotional bias in healthy volunteers. Psychopharmacology 2015, 232, 1793–1801. [Google Scholar] [CrossRef]
- van den Berg, J.P.; Westerbeek, E.; Bröring-Starre, T.; Garssen, J.; van Elburg, R.M. Neurodevelopment of preterm infants at 24 month after neonatal supplementation of a prebiotic mix: A randomized trial. J. Pediatr. Gastroenterol. Nutr. 2016, 63, 270–276. [Google Scholar] [CrossRef]
- Smith, A.P.; Sutherland, D.; Hewlett, P. An investigation of the acute effects of oligofructose-enriched inulin on subjective wellbeing, mood and cognitive performance. Nutrients 2015, 7, 8887–8896. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.P. The concept of well-being: Relevance to nutrition research. Br. J. Nutr. 2005, 93, S1–S5. [Google Scholar] [CrossRef] [PubMed]
- Waworuntu, R.; Hain, H.; Chang, Q.; Thiede, L.; Hanania, T.; Berg, B. Dietary prebiotics improve memory and social interactions while reducing anxiety when provided early in life to normally developing rodents (637.5). FASEB J. 2014, 28, 637.5. [Google Scholar]
- Messaoudi, M.; Rozan, P.; Nejdi, A.; Hidalgo, S.; Desor, D. Behavioural and cognitive effects of oligofructose-enriched inulin in rats. Br. J. Nutr. 2005, 93, S27–S30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buie, T.; Campbell, D.B.; Fuchs, G.J.; Furuta, G.T.; Levy, J.; VandeWater, J.; Whitaker, A.H.; Atkins, D.; Bauman, M.L.; Beaudet, A.L.; et al. Evaluation, diagnosis, and treatment of gastrointestinal disorders in individuals with asds: A consensus report. Pediatrics 2010, 125, S1–S8. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.B.; Johansen, L.J.; Powell, L.D.; Quig, D.; Rubin, R.A. Gastrointestinal flora and gastrointestinal status in children with autism–comparisons to typical children and correlation with autism severity. BMC Gastroenterol. 2011, 11, 22. [Google Scholar] [CrossRef] [PubMed]
- Navarro, F.; Liu, Y.; Rhoads, J.M. Can probiotics benefit children with autism spectrum disorders? World J. Gastroenterol. 2016, 22, 10093. [Google Scholar] [CrossRef]
- De Angelis, M.; Piccolo, M.; Vannini, L.; Siragusa, S.; De Giacomo, A.; Serrazzanetti, D.I.; Cristofori, F.; Guerzoni, M.E.; Gobbetti, M.; Francavilla, R. Fecal microbiota and metabolome of children with autism and pervasive developmental disorder not otherwise specified. PLoS ONE 2013, 8, e76993. [Google Scholar] [CrossRef]
- Lefranc-Millot, C.; Guérin-Deremaux, L.; Wils, D.; Neut, C.; Miller, L.; Saniez-Degrave, M. Impact of a resistant dextrin on intestinal ecology: How altering the digestive ecosystem with nutriose®, a soluble fibre with prebiotic properties, may be beneficial for health. J. Int. Med. Res. 2012, 40, 211–224. [Google Scholar] [CrossRef]
- Nankova, B.B.; Agarwal, R.; MacFabe, D.F.; La Gamma, E.F. Enteric bacterial metabolites propionic and butyric acid modulate gene expression, including creb-dependent catecholaminergic neurotransmission, in pc12 cells-possible relevance to autism spectrum disorders. PLoS ONE 2014, 9, e103740. [Google Scholar] [CrossRef]
- Müller, J.B.; Guggenheim, P.; Haemmerli, U. Treatment of chronic portal-systemic encephalopathy with lactulose. Lancet 1966, 287, 890–893. [Google Scholar] [CrossRef]
- Prasad, S.; Dhiman, R.K.; Duseja, A.; Chawla, Y.K.; Sharma, A.; Agarwal, R. Lactulose improves cognitive functions and health-related quality of life in patients with cirrhosis who have minimal hepatic encephalopathy. Hepatology 2007, 45, 549–559. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shukla, S.; Shukla, A.; Mehboob, S.; Guha, S. Meta-analysis: The effects of gut flora modulation using prebiotics, probiotics and synbiotics on minimal hepatic encephalopathy. Aliment. Pharmacol. Ther. 2011, 33, 662–671. [Google Scholar] [CrossRef] [PubMed]
- Elkington, S.; Floch, M.; Conn, H. Lactulose in the treatment of chronic portal-systemic encephalopathy: A double-blind clinical trial. N. Engl. J. Med. 1969, 281, 408–412. [Google Scholar] [CrossRef] [PubMed]
- Blanc, P.; Daures, J.P.; Rouillon, J.M.; Peray, P.; Pierrugues, R.; Larrey, D.; Gremy, F.; Michel, H. Lactitol or lactulose in the treatment of chronic hepatic encephalopathy: Results of a meta-analysis. Hepatology 1992, 15, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Cammà, C.; Fiorello, F.; Tinè, F.; Marchesini, G.; Fabbri, A.; Pagliaro, L. Lactitol in treatment of chronic hepatic encephalopathy. Digest. Dis. Sci. 1993, 38, 916–922. [Google Scholar] [CrossRef] [PubMed]
- Weber, F., Jr. Lactulose and combination therapy of hepatic encephalopathy: The role of the intestinal microflora. Digest. Dis. 1996, 14, 53–63. [Google Scholar] [CrossRef]
- Hong, K.-B.; Jeong, M.; Han, K.S.; Hwan Kim, J.; Park, Y.; Suh, H.J. Photoprotective effects of galacto-oligosaccharide and/or bifidobacterium longum supplementation against skin damage induced by ultraviolet irradiation in hairless mice. Int. J. Food Sci. Nutr. 2015, 66, 923–930. [Google Scholar] [CrossRef]
- Kawakami, K.; Makino, I.; Asahara, T.; Kato, I.; Onoue, M. Dietary galacto-oligosaccharides mixture can suppress serum phenol and p-cresol levels in rats fed tyrosine diet. J. Nutr. Sci. Vitaminol. 2005, 51, 182–186. [Google Scholar] [CrossRef]
- Mitsuyoshi, K.; Masuoka, N.; Chiaki, K.; Sugimoto, S.; Iizuka, R.; Manabe, K.; Toshiro, S.; Kazutoshi, O.; Nonaka, C.; Miyazaki, K.; et al. Consecutive intake of fermented milk containing bifidobacterium breve strain yakult and galacto-oligosaccharides benefits skin condition in healthy adult women. Biosci. Microbiot. Food Health 2013, 32, 33–39. [Google Scholar]
- Miyazaki, K.; Masuoka, N.; Kano, M.; Iizuka, R. Bifidobacterium fermented milk and galacto-oligosaccharides lead to improved skin health by decreasing phenols production by gut microbiota. Benef. Microbes 2013, 5, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Mohkam, M.; Nezafat, N.; Berenjian, A.; Mobasher, M.A.; Ghasemi, Y. Identification of bacillus probiotics isolated from soil rhizosphere using 16s rrna, reca, rpob gene sequencing and rapd-pcr. Probiot. Antimicrob. Proteins 2016, 8, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Mohkam, M.; Rasoul-Amini, S.; Shokri, D.; Berenjian, A.; Rahimi, F.; Sadraeian, M.; Khalvati, B.; Gholami, A.; Ghasemi, Y. Characterization and in vitro probiotic assessment of potential indigenous bacillus strains isolated from soil rhizosphere. Minerva Biotechnol. 2016, 28, 19–28. [Google Scholar]
- Mozaffarian, D.; Benjamin, E.; Go, A.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; Das, S.R.; Sarah de Ferranti, M.; Després, J.-P.; Fullerton, H.J.; et al. Aha statistical update. Heart Dis. Stroke 2015, 131, 29–322. [Google Scholar]
- Letexier, D.; Diraison, F.; Beylot, M. Addition of inulin to a moderately high-carbohydrate diet reduces hepatic lipogenesis and plasma triacylglycerol concentrations in humans. Am. J. Clin. Nutr. 2003, 77, 559–564. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Russo, F.; Chimienti, G.; Riezzo, G.; Pepe, G.; Petrosillo, G.; Chiloiro, M.; Marconi, E. Inulin-enriched pasta affects lipid profile and lp (a) concentrations in italian young healthy male volunteers. Eur. J. Nutr. 2008, 47, 453–459. [Google Scholar] [CrossRef]
- Forcheron, F.; Beylot, M. Long-term administration of inulin-type fructans has no significant lipid-lowering effect in normolipidemic humans. Metabolism 2007, 56, 1093–1098. [Google Scholar] [CrossRef]
- Vogt, J.A.; Ishii-Schrade, K.B.; Pencharz, P.B.; Jones, P.J.; Wolever, T.M. L-rhamnose and lactulose decrease serum triacylglycerols and their rates of synthesis, but do not affect serum cholesterol concentrations in men. J. Nutr. 2006, 136, 2160–2166. [Google Scholar] [CrossRef]
- Jenkins, D.; Wolever, T.; Jenkins, A.; Brighenti, F.; Vuksan, V.; Rao, A.V.; Cunnane, S.C.; Ocana, A.; Corey, P.; Vezina, C. Specific types of colonic fermentation may raise low-density-lipoprotein-cholesterol concentrations. Am. J. Clin. Nutr. 1991, 54, 141–147. [Google Scholar] [CrossRef]
- Vulevic, J.; Juric, A.; Tzortzis, G.; Gibson, G.R. A mixture of trans-galactooligosaccharides reduces markers of metabolic syndrome and modulates the fecal microbiota and immune function of overweight adults. J. Nutr. 2013, 143, 324–331. [Google Scholar] [CrossRef]
- Tiwari, U.; Cummins, E. Meta-analysis of the effect of β-glucan intake on blood cholesterol and glucose levels. Nutrition 2011, 27, 1008–1016. [Google Scholar] [CrossRef] [PubMed]
- Brighenti, F. Dietary fructans and serum triacylglycerols: A meta-analysis of randomized controlled trials. J. Nutr. 2007, 137, 2552S–2556S. [Google Scholar] [CrossRef] [PubMed]
- Beynen, A.C.; Buechler, K.F.; Van der Molen, A.J.; Geelen, M.J. The effects of lactate and acetate on fatty acid and cholesterol biosynthesis by isolated rat hepatocytes. Int. J. Biochem. 1982, 14, 165–169. [Google Scholar] [CrossRef]
- Wolever, T.; Brighenti, F.; Royall, D.; Jenkins, A.L.; Jenkins, D.J. Effect of rectal infusion of short chain fatty acids in human subjects. Am. J. Gastroenterol. 1989, 84, 1027–1033. [Google Scholar] [PubMed]
- Wolever, T.; Spadafora, P.J.; Cunnane, S.C.; Pencharz, P.B. Propionate inhibits incorporation of colonic [1, 2–13c] acetate into plasma lipids in humans. Am. J. Clin. Nutr. 1995, 61, 1241–1247. [Google Scholar] [CrossRef] [PubMed]
- Mortensen, P.B.; Holtug, K.; Rasmussen, H.S. Short-chain fatty acid production from mono-and disaccharides in a fecal incubation system: Implications for colonic fermentation of dietary fiber in humans. J. Nutr. 1988, 118, 321–325. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.E.; Parnell, J.A.; Eksteen, B.; Raman, M.; Bomhof, M.R.; Rioux, K.P.; Madsen, K.L.; Reimer, R.A. Gut microbiota manipulation with prebiotics in patients with non-alcoholic fatty liver disease: A randomized controlled trial protocol. BMC Gastroenterol. 2015, 15, 169. [Google Scholar] [CrossRef] [PubMed]
- Tarantino, G.; Finelli, C. Systematic review on intervention with prebiotics/probiotics in patients with obesity-related nonalcoholic fatty liver disease. Future Microbiol. 2015, 10, 889–902. [Google Scholar] [CrossRef]
- Cashman, K. Calcium intake, calcium bioavailability and bone health. Br. J. Nutr. 2002, 87, S169–S177. [Google Scholar] [CrossRef] [Green Version]
- Carlson, J.L.; Erickson, J.M.; Lloyd, B.B.; Slavin, J.L. Health effects and sources of prebiotic dietary fiber. Curr. Dev. Nutr. 2018, 2, nzy005. [Google Scholar] [CrossRef]
- Ellegård, L.; Andersson, H.; Bosaeus, I. Inulin and oligofructose do not influence the absorption of cholesterol, or the excretion of cholesterol, ca, mg, zn, fe, or bile acids but increases energy excretion in ileostomy subjects. Eur. J. Clin. Nutr. 1997, 51, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Van den Heuvel, E.; Schaafsma, G.; Muys, T.; van Dokkum, W. Nondigestible oligosaccharides do not interfere with calcium and nonheme-iron absorption in young, healthy men. Am. J. Clin. Nutr. 1998, 67, 445–451. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tahiri, M.; Tressol, J.C.; Arnaud, J.; Bornet, F.R.; Bouteloup-Demange, C.; Feillet-Coudray, C.; Brandolini, M.; Ducros, V.; Pépin, D.; Brouns, F.; et al. Effect of short-chain fructooligosaccharides on intestinal calcium absorption and calcium status in postmenopausal women: A stable-isotope study. Am. J. Clin. Nutr. 2003, 77, 449–457. [Google Scholar] [CrossRef] [PubMed]
- López-Huertas, E.; Teucher, B.; Boza, J.J.; Martínez-Férez, A.; Majsak-Newman, G.; Baró, L.; Carrero, J.J.; González-Santiago, M.; Fonollá, J.; Fairweather-Tait, S. Absorption of calcium from milks enriched with fructo-oligosaccharides, caseinophosphopeptides, tricalcium phosphate, and milk solids. Am. J. Clin. Nutr. 2006, 83, 310–316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Den Heuvel, E.G.; Muijs, T.; Van Dokkum, W.; Schaafsma, G. Lactulose stimulates calcium absorption in postmenopausal women. J. Bone Miner. Res. 1999, 14, 1211–1216. [Google Scholar] [CrossRef] [PubMed]
- van den Heuvel, E.G.; Schoterman, M.H.; Muijs, T. Transgalactooligosaccharides stimulate calcium absorption in postmenopausal women. J. Nutr. 2000, 130, 2938–2942. [Google Scholar] [CrossRef] [PubMed]
- Abrams, S.A.; Griffin, I.J.; Hawthorne, K.M.; Liang, L.; Gunn, S.K.; Darlington, G.; Ellis, K.J. A combination of prebiotic short-and long-chain inulin-type fructans enhances calcium absorption and bone mineralization in young adolescents. Am. J. Clin. Nutr. 2005, 82, 471–476. [Google Scholar] [CrossRef]
- Griffin, I.; Davila, P.; Abrams, S. Non-digestible oligosaccharides and calcium absorption in girls with adequate calcium intakes. Br. J. Nutr. 2002, 87, S187–S191. [Google Scholar] [CrossRef] [Green Version]
- Svensson, U.; Håkansson, J. Safety of Food and Beverages: Safety of Probiotics and Prebiotics; Elsevier: New York, NY, USA, 2014. [Google Scholar]
- Garg, B.D.; Balasubramanian, H.; Kabra, N.S. Physiological effects of prebiotics and its role in prevention of necrotizing enterocolitis in preterm neonates. J. Matern.-Fetal Neonatal Med. 2018, 31, 2071–2078. [Google Scholar] [CrossRef]
- Tsai, Y.-L.; Lin, T.-L.; Chang, C.-J.; Wu, T.-R.; Lai, W.-F.; Lu, C.-C.; Lai, H.-C. Probiotics, prebiotics and amelioration of diseases. J. Biomed. Sci. 2019, 26, 3. [Google Scholar] [CrossRef]


| Prebiotic | Dose | Subjects | Main Results | Reference |
|---|---|---|---|---|
| FOS | 6 g/day for 4 weeks | Patients with IBS | No therapeutic effect. | [99] |
| 20 g/day for 12 weeks | Patients with IBS | No therapeutic effect. | [100] | |
| 5 g/day for 6 weeks | Patients with IBS | Improvement in IBS syndromes. | [102] | |
| 15 g/day for 3 weeks | Patients with active ileocolonic Crohn’s disease | Crohn’s disease improvement. | [103] | |
| 15 g/day for 4 weeks | Patients with Crohn’s disease | No clinical improvement in Crohn’s disease. | [104] | |
| GOS | 3.5 g/day for 12 weeks | Patients with IBS | Improvement in IBS syndromes. | [102] |
| Mixture of FOS and GOS | 0.8 g/dL of a mixture of GOS and FOS, ratio 9:1 for 30 days | Healthy newborns | Improvement in gastric emptying and bowel motility. | [115] |
| 0.8 g/dL of a mixture of GOS and FOS, ratio 9:1 for 15 days | Healthy newborns | Improvement in gastric emptying and bowel motility. | [116] | |
| Inulin-enriched FOS | 20 g/day for 4 weeks | Patients with inactive and mild to moderately active Crohn’s disease | No clinical Improvement in Crohn’s disease. | [105] |
| Raftilose® Synergy 1 + Bifidobacterium lactis Bb12, Lactobacillus rhamnosus GG | HT29 or CaCo-2 cells | Cell growth inhibition. As a result, this mixture can decrease the progression of colorectal cancer. | [119] | |
| Different doses | Rats with colon carcinogen | Long-chain inulin effects are dose-dependent on colorectal cancer. | [120] | |
| Synergy 1 + Bifidobacterium lactis Bb12, Lactobacillus rhamnosus GG | Colon cancer patients and polypectomized patients | Decrease in the progression of colorectal cancer. | [110] | |
| Lactose | 25 g daily for 15 days | Lactose malabsorbers | Improvement in lactose digestion. | [117] |
| Prebiotic | Dose | Subjects | Main Results | Reference |
|---|---|---|---|---|
| FOS | 8 oz/day of an experimental formula containing FOS for 183 days | Adults aged 65 and older | Antibody responses toward viral vaccines improved. Hospitalization due to influenza and side effects of influenza vaccines decreased. | [130] |
| 8 g/day Orafti® Synergy1 for 8 weeks | Adults aged 45–63 years | Immune responses toward influenza vaccines improved. | [135] | |
| 0.55 g FOS per 15 g of cereal for 6 months | Non-breast-feeding infants aged 4–24 months | Diarrhea associated fever, febrile seizure incident, antibiotics usage, and duration of infectious disease decreased. | [133] | |
| 3 × 5 g/day FOS consisted of two 28 day treatments separated by a 14-day washout | Healthy volunteers | IL-4 in serum, CD282+/TLR2+ myeloid dendritic cells, and toll-like receptor 2-mediated immune response were up-regulated. | [134] | |
| Not exactly defined | Infants | Risk of some immune diseases, such as atopic dermatitis, reduced. | [136,137] | |
| 2 × 4 g/day for 3 weeks | Elderly nursing home patients | IL-6 expression and phagocytosis in monocytes and granulocytes decreased. | [138] | |
| 8 g/day Orafti® Synergy1 for 4 weeks | Adults aged 45–65 years | Salivary IgA, immune cells in serum, activation, and proliferation of T and NK cell not changed. | [131] | |
| GOS | 5.5 g/day for 10 weeks | Elderly subjects | Phagocytosis, NK cell activity, and IL-10 (an anti-inflammatory cytokine) level increased. Pro-inflammatory cytokines, such as IL-6, IL-1β, and tumor necrosis factor-α, levels decreased. | [139] |
| 5.5 g/day consisted of two 10 weeks of treatment separated by 4 weeks of washout | Elderly subjects | IL-10, IL-8, C-reactive protein, and NK cell activity elevated. IL-1β level decreased. | [140] | |
| Not exactly defined | Infants | Risk of some immune diseases, such as atopic dermatitis, reduced. | [136] | |
| 0.8 g/100 mL | Infants | [137] | ||
| 0.8 g/day for 6 months | Newborn infants | [141] | ||
| AOS | Not exactly defined | Infants | Atopic dermatitis in low-risk infants reduced. | [136] |
| Oligofructose and inulin mixture | Oligofructose (70%) and inulin (30%) with a concentration of 1 g per 25 g of dry weight cereal during 4 weeks prior to measles vaccination | Infants aged 7–9 months | Antibody responses toward viral vaccines improved. | [129] |
| Prebiotic | Dose | Subjects | Main Results | Reference |
|---|---|---|---|---|
| Non-starch polysaccharides (NSPs) | 4 g of NSPs (Ambrotose®) | Middle-aged healthy adults | Recognition and working memory performance improved. | [153] |
| 3.6 g/day for 12 weeks | Middle-aged healthy adults | Cognitive function and well-being optimized. | [154] | |
| Mixture of FOS, GOS, and AOS | Supplementation between day 3 and 30 of life, and the results measured during 24 months | Preterm infants | Neurodevelopment did not improve significantly. | [157] |
| Inulin-enriched oligofructose | 5 g, the results measured after 4 h | 19–30 years old healthy individuals | Mood, recognition, immediate memory, and recall enhanced. | [158] |
| 10 g/day of Synergy® 1, the results measured after 43 days | 19–64 years old healthy individuals | Long-term memory did not change significantly. | [159] | |
| Mixture of GOS and polydextrose | 2.4 and 7 g/L of polydextrose and GOS | Male piglets | They may have neurodevelopment effect in human infants. | [143] |
| 7 g/kg prebiotics mixture | Rats | Memory and social behaviors improved, and anxiety-like behaviors reduced. | [160] | |
| 15 g/kg prebiotics mixture | Mice | |||
| Water extract of Triticum aestivum composed of arabinoxylan, β-glucan, and arabinose | - | Rats | Arabinoxylan, β-glucan, and arabinose had preserved cognition effects against vascular dementia. | [155] |
| GOS | 5.5 g/day for 3 weeks | 18–45 years old healthy volunteers | Salivary cortisol awakening response was decreased, attentional vigilance to negative versus positive information reduced, and the concentration improved. | [156] |
| Lactulose | Lactoferrin (0.6 g/L) and Milk fat globule membrane (MFGM) (5.0 g/L) | Male piglets | Lactulose appeared to have neurodevelopment effect in human infants. | [143] |
| Duphalac® 90–150 mL/d | Patients with chronic portal-systemic encephalopathy (PSE) | Blood ammonia levels decreased. | [168] | |
| 30–60 mL of lactulose in 2 or 3 divided doses for 3 months | Patients with cirrhosis | Cognitive function and health-related quality of life improved. | [169] | |
| Meta-analysis | Patients with subclinical hepatic encephalopathy | Lactulose had the most beneficial influence among prebiotics and probiotics. | [170] | |
| 67 mg/day for long-term therapy (1 to 10 months) | Patients with chronic PSE | The lower intestinal tract was acidified, and lactulose had a beneficial effect on chronic PSE. | [171] |
| Prebiotic | Dose | Subjects | Main Results | Reference |
|---|---|---|---|---|
| AOS | Not exactly defined | Infants | Formula supplementation with a specific mixture of oligosaccharides was effective in preventing atopic dermatitis in low-risk infants. | [136] |
| GOS | Not exactly defined | Infants | Risk of some immune diseases, such as atopic dermatitis, reduced. | [136] |
| 0.8 g/100 mL | Infants | [137] | ||
| 0.8 g/day for 6 months | Newborn infants | [141] | ||
| GOS with or without probiotics | 100 mg of GOS daily for 12 weeks | Hairless mice exposed to the UV | Water retention enhanced, and erythema reduced. | [175] |
| 600 mg of GOS for 4 weeks | Adult healthy women | Water and keratin reduction caused by phenols decreased. | [177] |
| Prebiotic | Dose | Subjects | Main Results | Reference |
|---|---|---|---|---|
| Inulin-enriched pasta | 2-weeks run-in period, a baseline assessment, two 5-weeks study periods (11% inulin-enriched or control pasta) | Healthy individuals | HDL-cholesterol level elevated; total cholesterol/HDL-cholesterol ratio, triglycerides, and lipoprotein A levels reduced. | [183] |
| Inulin | 10 g/day for 3 weeks | Healthy individuals | Hepatic lipogenesis and plasma triacylglycerol concentrations reduced. | [182] |
| Mixture of inulin and oligofructose | 10 g/day for 6 months | Healthy individuals | Plasma triacylglycerol concentrations and hepatic lipogenesis were not changed. A non-significant decreasing trend in plasma total and low-density lipoprotein cholesterol levels were observed, and high-density lipoprotein cholesterol concentration increased. | [184] |
| L-rhamnose | 25 g/day for 4 weeks | Healthy adults | Triacylglycerol (TAG) and net TAG-fatty acid (TAGFA) synthesis decreased. | [185] |
| Lactulose | 25 g/day for 4 weeks | Healthy adults | Triacylglycerol (TAG) and net TAG-fatty acid (TAGFA) synthesis decreased. | [185] |
| 18–25 g/day for 2 weeks | Healthy individuals | Free fatty acid concentrations were reduced by increasing the absorbed acetate from the colon. | [186] | |
| GOS | Administrating Bi2muno (B-GOS) for 2 six weeks | Overweight subjects with ≥3 risk factors of metabolic syndrome | Circulating cholesterol, TAG, and total:HDL cholesterol ratio decreased. | [187] |
| Prebiotic | Dose | Subjects | Main Results | Reference |
|---|---|---|---|---|
| Inulin or oligofructose | 17 g of inulin or oligofructose and 7 g for three experimental periods of three days each. | Patients with conventional ileostomy because of ulcerative colitis | No significant effect on calcium, magnesium, zinc, and iron absorption. | [198] |
| FOS or GOS | 15 g/day for 3 weeks | Healthy, nonanemic, male | No significant effect on calcium and iron absorption. | [199] |
| Short chain FOS | 10 g/day for 5 weeks | Healthy, postmenopausal women | No significant effect on calcium absorption. | [200] |
| FOS enriched milk | 5 g FOS/L with light breakfast | Healthy adults | No significant effect on calcium absorption. | [201] |
| Lactulose | 5 or 10 g per day for two 9 days with 19-day washout in between | Post-menopausal women | Calcium absorption increased in a dose-response way. | [202] |
| Trans-galacto-oligosaccharides | 20 g for two 9 days with 19-day washout in between | Post-menopausal women | Calcium absorption increased. | [203] |
| A mixed short and long degree of polymerized inulin-type fructan product | 8 g/day for 8 weeks or 1 year | Calcium absorption increased significantly. | [204] | |
| The mixture of inulin + oligofructose | 8 g/day for two 3 weeks, separated by a 2-week washout period | Girls at or near menarche. | Calcium absorption increased. | [205] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Davani-Davari, D.; Negahdaripour, M.; Karimzadeh, I.; Seifan, M.; Mohkam, M.; Masoumi, S.J.; Berenjian, A.; Ghasemi, Y. Prebiotics: Definition, Types, Sources, Mechanisms, and Clinical Applications. Foods 2019, 8, 92. https://doi.org/10.3390/foods8030092
Davani-Davari D, Negahdaripour M, Karimzadeh I, Seifan M, Mohkam M, Masoumi SJ, Berenjian A, Ghasemi Y. Prebiotics: Definition, Types, Sources, Mechanisms, and Clinical Applications. Foods. 2019; 8(3):92. https://doi.org/10.3390/foods8030092
Chicago/Turabian StyleDavani-Davari, Dorna, Manica Negahdaripour, Iman Karimzadeh, Mostafa Seifan, Milad Mohkam, Seyed Jalil Masoumi, Aydin Berenjian, and Younes Ghasemi. 2019. "Prebiotics: Definition, Types, Sources, Mechanisms, and Clinical Applications" Foods 8, no. 3: 92. https://doi.org/10.3390/foods8030092