Novel Environmentally Responsive Polyvinyl Polyamine Hydrogels Capable of Phase Transformation with Temperature for Applications in Reservoir Profile Control
Abstract
:1. Introduction
2. Results and Discussion
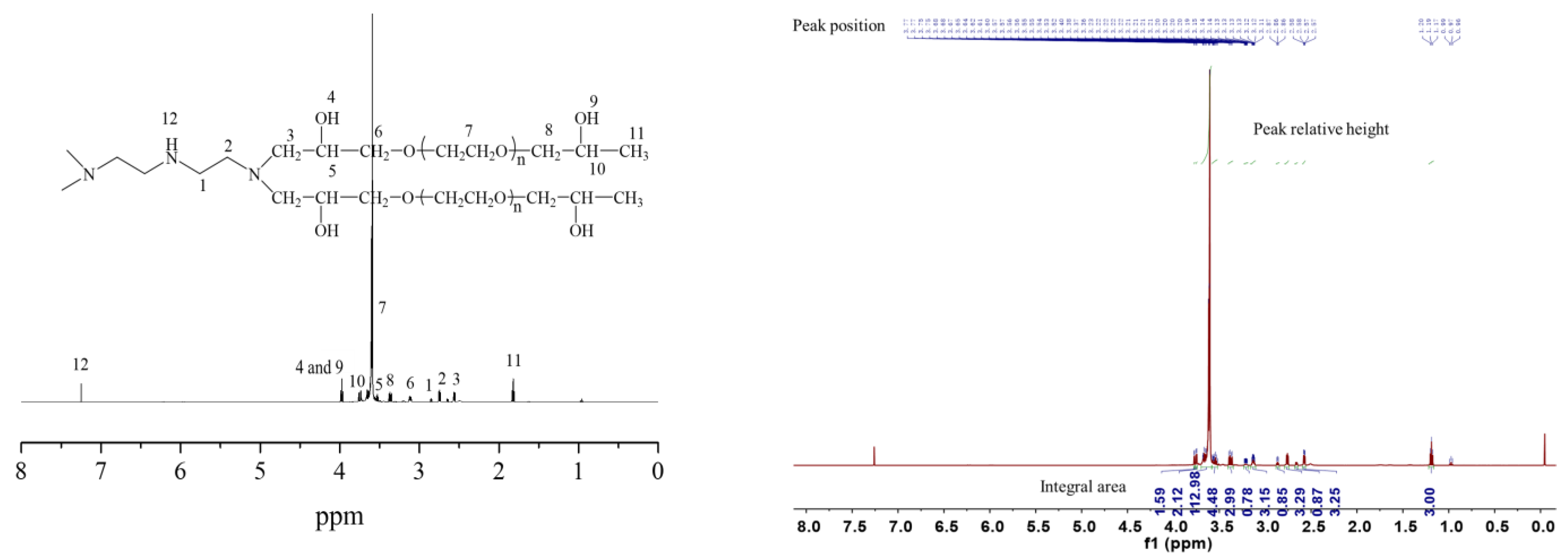
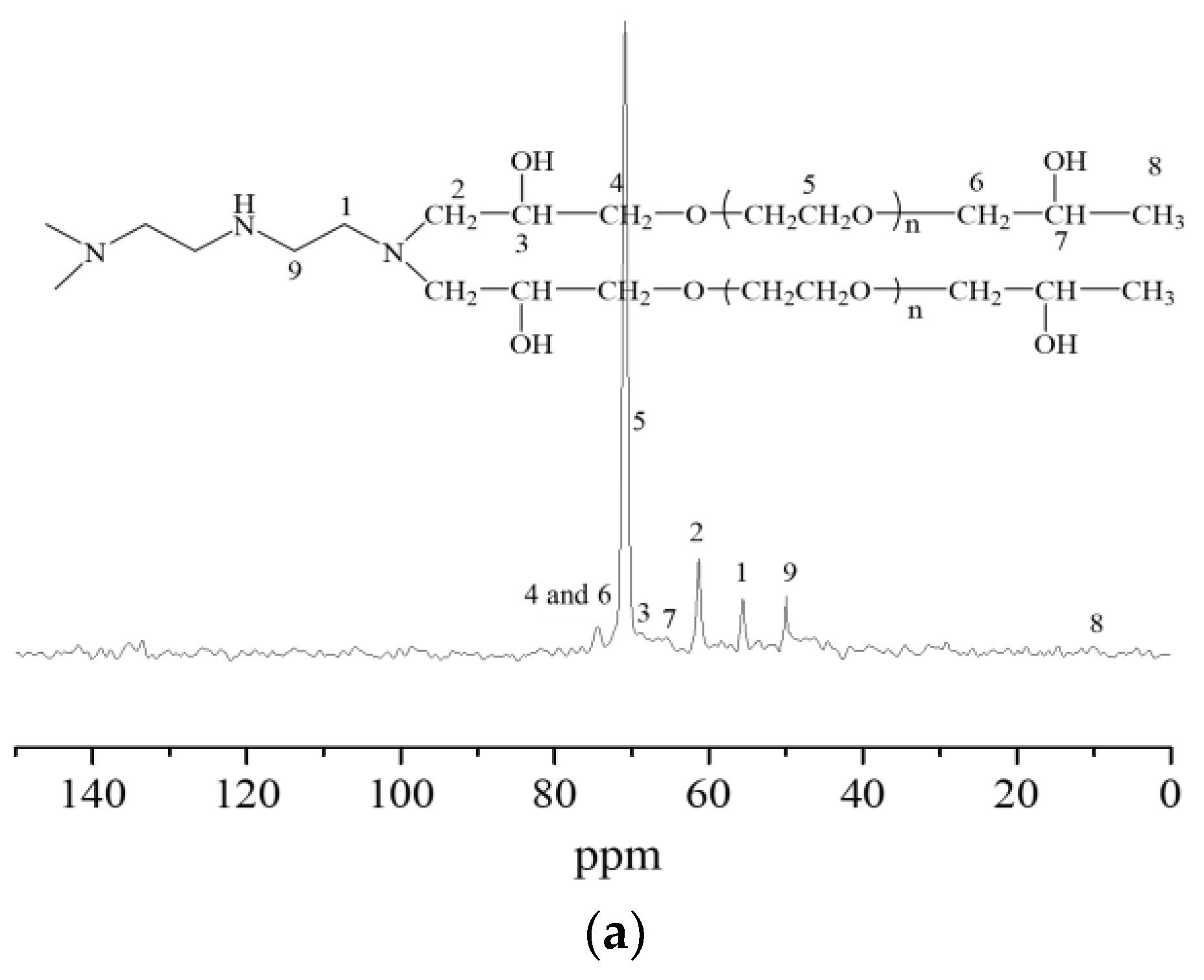
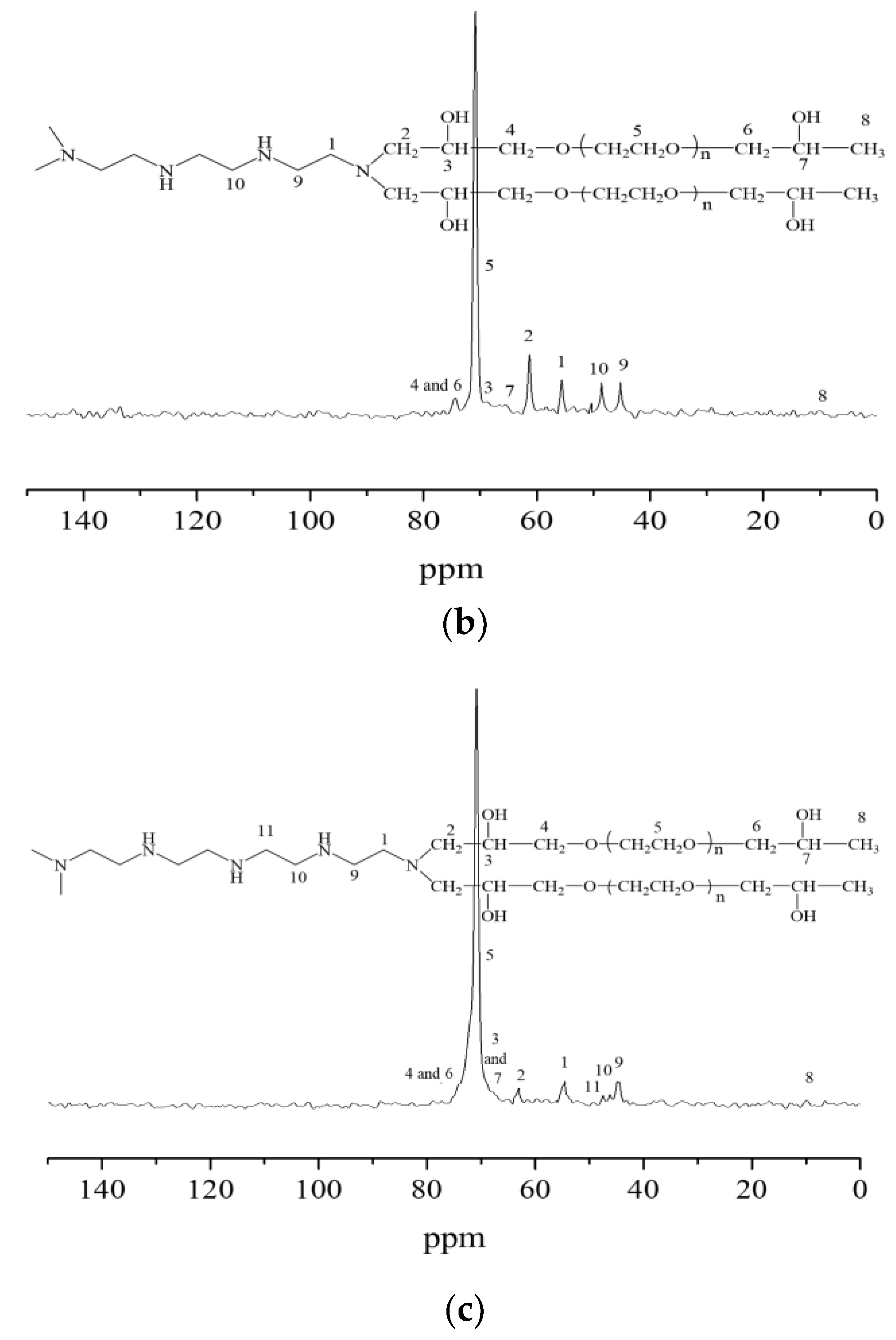
2.1. Characterizations of the Synthesized Hydrogels
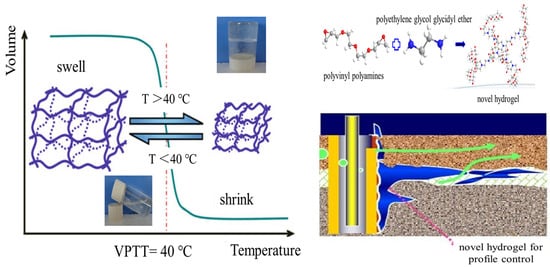
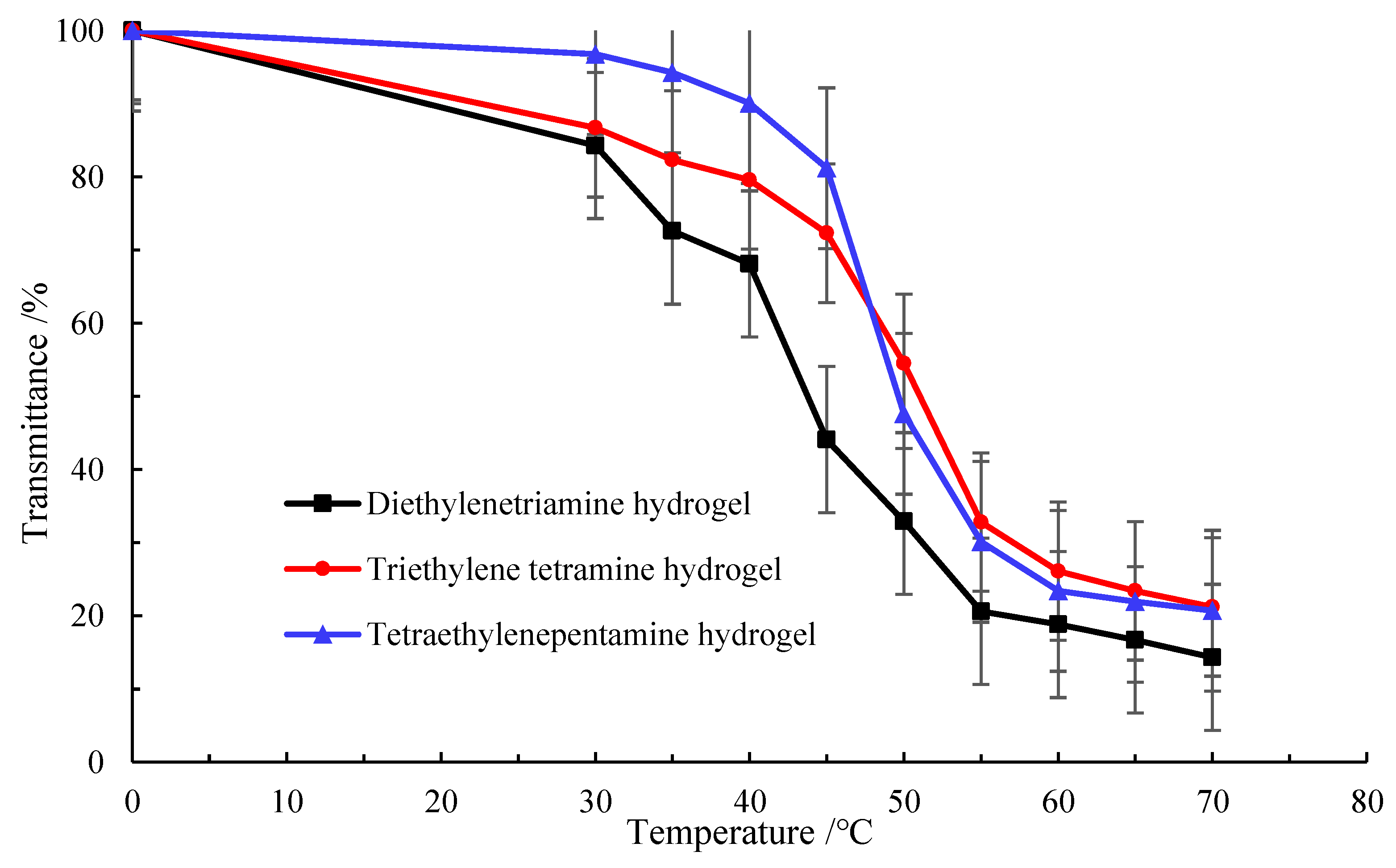
2.2. Effect of Temperature on Phase Transformation and Environmental Responsiveness
2.3. Profile Improvement Mechanism of Polyvinyl Polyamine Hydrogels
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Synthesis of Polyvinyl Polyamine Hydrogels
4.3. Structural Characterization of Polyvinyl Polyamine Hydrogel
4.4. Temperature Sensitivity Test of Polyvinyl Polyamine Hydrogel
4.5. Profile Control Performance of Polyvinyl Polyamine Hydrogel
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shaheen, S.; Saeed, Z.; Ahmad, A.; Pervaiz, M.; Younas, U.; Mahmood Khan, R.R.; Luque, R.; Rajendran, S. Green synthesis of graphene-based metal nanocomposite for electro and photocatalytic activity; recent advancement and future prospective. Chemosphere 2023, 311, 136982. [Google Scholar] [CrossRef] [PubMed]
- McGarry, L.F.; El-Zubir, O.; Waddell, P.G.; Cucinotta, F.; Houlton, A.; Horrocks, B.R. Vesicles, fibres, films and crystals: A low-molecular-weight-gelator [Au(6-thioguanosine)2]Cl which exhibits a co-operative anion-induced transition from vesicles to a fibrous metallo-hydrogel. Soft Matter 2023, 19, 8386–8402. [Google Scholar] [CrossRef]
- Cheng, Q.; Cao, G.; Wang, P.; Bu, R.; Bai, Y. CO2 diffusive characteristics and influencing factors in the porous medium with saturated polyacrylamide solutions. Arab. J. Chem. 2023, 16, 105106. [Google Scholar] [CrossRef]
- Nguyen, A.K.; Molley, T.G.; Kardia, E.; Ganda, S.; Chakraborty, S.; Wong, S.L.; Ruan, J.; Yee, B.E.; Mata, J.; Vijayan, A.; et al. Hierarchical assembly of tryptophan zipper peptides into stress-relaxing bioactive hydrogels. Nat. Commun. 2023, 14, 6604. [Google Scholar] [CrossRef] [PubMed]
- Madhan, A.; Kun, G.; Zhixin, Y. A State-of-the-Art Review of Nanoparticles Application in Petroleum with a Focus on Enhanced Oil Recovery. Appl. Sci. 2018, 8, 871. [Google Scholar] [CrossRef]
- Siddiqui, Z.; Sarkar, B.; Kim, K.K.; Kumar, A.; Paul, R.; Mahajan, A.; Grasman, J.M.; Yang, J.; Kumar, V.A. Self-assembling peptide hydrogels facilitate vascularization in two-component scaffolds. Chem. Eng. J. 2021, 422, 130145. [Google Scholar] [CrossRef]
- Zhao, X.; Luo, J.; Huang, Y.; Mu, L.; Chen, J.; Liang, Z.; Yin, Z.; Chu, D.; Han, Y.; Guo, B. Injectable Antiswelling and High-Strength Bioactive Hydrogels with a Wet Adhesion and Rapid Gelling Process to Promote Sutureless Wound Closure and Scar-free Repair of Infectious Wounds. ACS Nano 2023, 17, 22015–22034. [Google Scholar] [CrossRef]
- Al-Shajalee, F.; Arif, M.; Machale, J.; Verrall, M.; Almobarak, M.; Iglauer, S.; Wood, C. A multiscale investigation of cross-linked polymer gel injection in sandstone gas reservoirs: Implications for water shutoff treatment. Energy Fuels 2020, 34, 14046–14057. [Google Scholar] [CrossRef]
- Ye, Z.; He, E.; Xie, S.; Han, L.; Chen, H.; Luo, P.; Shu, Z.; Shi, L.; Lai, N. The mechanism study of disproportionate permeability reduction by hydrophobically associating water-soluble polymer gel. J. Pet. Sci. Eng. 2010, 72, 64–66. [Google Scholar] [CrossRef]
- Kawara, S.; Cunningham, B.; Bezer, J.; KC, N.; Zhu, J.; Tang, M.X.; Ishihara, J.; Choi, J.J.; Au, S.H. Capillary-Scale Hydrogel Microchannel Networks by Wire Templating. Small 2023, 19, 2370345. [Google Scholar] [CrossRef]
- Tanaka, T. Collapse of gels and the critical endpoint. Phys. Rev. Lett. 1978, 40, 820–823. [Google Scholar] [CrossRef]
- Song, X.; Zhang, Z.; Zhu, J. Thermoresponsive hydrogel induced by dual supramolecular assemblies and its controlled release property for enhanced anticancer drug delivery. Biomacromolecules 2020, 21, 1516–1527. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Bai, S.; Li, Z.; Gu, G.; Wang, H.; Wei, L.; Sun, X.; Xuan, F.; Guo, X.; Wang, Y. Light-triggered reversible phase transition of non-photoresponsive PNIPAm for remote-controlled smart objects. Mater. Sci. Eng. C-Mater. Biol. Appl. 2021, 118, 111352. [Google Scholar] [CrossRef]
- Cseri, L.; Hardian, R.; Anan, S.; Vovusha, H.; Schwingenschlögl, U.; Budd, P.M.; Sada, K.; Kokado, K.; Szekely, G. Bridging the interfacial gap in mixed-matrix membranes by nature-inspired design: Precise molecular sieving with polymer-grafted metal-organic frameworks. J. Mater. Chem. A 2021, 9, 42. [Google Scholar] [CrossRef]
- Han, Y.-L.; Kim, H.-R.; Kim, H.-K.; Park, J.-W. Thermosensitive poly(N-isopropylacrylamide)-grafted magnetic-cored dendrimers for benzene uptake. Chemosphere 2022, 307, 135988. [Google Scholar] [CrossRef] [PubMed]
- Manjula, B.; Varaprasad, K.; Sadiku, R.; Ramam, K.; Reddy, G.V.S.; Raju, K.M. Development of microbial resistant thermosensitive Ag nanocomposite (gelatin) hydrogels via green process. J. Biomed. Mater. Res. Part A 2014, 102, 928–934. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Chen, L.; Shen, T.; Wu, D. Preparation and properties of a novel thermo-sensitive hydrogel based on chitosan/hydroxypropyl methylcellulose/glycerol. Int. J. Biol. Macromol. 2016, 93, 775–782. [Google Scholar] [CrossRef]
- Zhang, T.; Chen, S.; Dou, H.; Liu, Q.; Shu, G.; Lin, J.; Zhang, W.; Peng, G.; Zhong, Z.; Fu, H. Novel glucosamine-loaded thermosensitive hydrogels based on poloxamers for osteoarthritis therapy by intra-articular injection. Mater. Sci. Eng. C-Mater. Biol. Appl. 2021, 118, 111352. [Google Scholar] [CrossRef]
- Anghelache, A.; Teodorescu, M.; Stǎnescu, P.O.; Drǎghici, C.; Vuluga, D.M. Novel crosslinked thermoresponsive hydrogels with controlled poly (ethylene glycol)-poly (propylene glycol) multiblock copolymer structure. Colloid Polym. Sci. 2014, 292, 829–838. [Google Scholar] [CrossRef]
- Bai, Y.; Ban, C.; He, S.; Zhao, J.; Zhang, H. Temperature-responsive self-lubricating hydrogel from dynamic Diels-Alder crosslinking for reservoir in-depth profile control. J. Mol. Liq. 2021, 323, 114595. [Google Scholar] [CrossRef]
- Wang, Z.; Wu, X.; Dong, J.; Yang, X.; He, F.; Peng, S.; Li, Y. Porifera-inspired cost-effective and scalable “porous hydrogel sponge” for durable and highly efficient solar-driven desalination. Chem. Eng. J. 2022, 427, 130905. [Google Scholar] [CrossRef]
- Li, L.; Li, W.; Tong, K.; Jiu, J.; Suganuma, K. Intense pulsed light-induced structure-transformed ultrathin Ni shell for improving the chemical, thermal, and electrical reliability of metal nanowire electrodes without transmittance loss. Chem. Eng. J. 2020, 390, 124517. [Google Scholar] [CrossRef]
- Aranburu, N.; Eguiazábal, J.I.; Guerrica-Echevarria, G. The effects of the location of organoclay on the structure and mechanical properties of compatibilized polypropylene/polyamide-12 ternary nanocomposites. Polym. Eng. Sci. 2018, 58, 830–838. [Google Scholar] [CrossRef]
- Gu, C.; Lv, Y.; Fan, X.; Zhao, C.; Dai, C.; Zhao, G. Study on rheology and microstructure of phenolic resin cross-linked nonionic polyacrylamide (NPAM) gel for profile control and water shutoff treatments. J. Pet. Sci. Eng. 2018, 169, 546–552. [Google Scholar] [CrossRef]
- Liu, R.; Gou, R.; Pu, W.-f.; Ren, H.; Du, D.-j.; Chen, P.; Mei, Z.-l. Visual Laminations Combined with Nuclear Magnetic Reso-nance to Study the Micro-Unrecovered Oil Distribution and Displacement Behavior of Chemical Flooding in a Complex Con-glomerate. Energy Fuels 2019, 33, 4041–4052. [Google Scholar] [CrossRef]
- Varel, F.T.; Dai, C.; Shaikh, A.; Li, J.; Sun, N.; Yang, N.; Zhao, G. Chromatography and oil displacement mechanism of a dis-persed particle gel strengthened Alkali/Surfactant/Polymer combination flooding system for enhanced oil recovery. Colloids Surf. A 2021, 610, 125642. [Google Scholar] [CrossRef]
- Wang, A.; Du, M.; Ni, J.; Liu, D.; Pan, Y.; Liang, X.; Liu, D.; Ma, J.; Wang, J.; Wang, W. Enhanced and synergistic catalytic activation by photoexcitation driven S−scheme heterojunction hydrogel interface electric field. Nat. Commun. 2023, 14, 6733. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Q.; Cao, G.; Bai, Y.; Zhu, Z.; Zhang, N.; Li, D. Probing the demulsification mechanism of emulsion with SPAN series based on the effect of solid phase particles. Molecules 2023, 28, 3261. [Google Scholar] [CrossRef]
- Shin, M.; Shin, S.H.; Lee, M.; Kim, H.J.; Jeong, J.H.; Choi, Y.H.; Oh, D.X.; Park, J.; Jeon, H.; Eom, Y. Rheological criteria for distinguishing self-healing and non-self-healing hydrogels. Polymer 2021, 229, 123969. [Google Scholar] [CrossRef]
- Sánchez-Cid Bueno, P.; Jiménez-Rosado, M.; Alonso González, M.; Romero García, A.; Pérez-Puyana, V.M. Applied Rheology as Tool for the Assessment of Chitosan Hydrogels for Regenerative Medicine. Polymers 2021, 13, 2189. [Google Scholar] [CrossRef]
- Liu, X.; Wang, S.; Shi, W.; Liu, H. Thermo-/pH-Dual-Sensitive PEG/PAMAM Nanogel: Reaction and Dynamics and Plugging Application of CO2 Channeling. Gels. 2022, 8, 683. [Google Scholar] [CrossRef] [PubMed]
- Guvendiren, M.; Heiney, P.A.; Yang, S. Precipitated Calcium Carbonate Hybrid Hydrogels: Structural and Mechanical Prop-erties. Macromolecules 2009, 42, 6606–6613. [Google Scholar] [CrossRef]
- Yang, H.; Zhang, H.; Zheng, W.; Li, X.; Wang, F.; Li, X.; Zhang, D.; Turtabayev, S.; Kang, W. Research on synthesis and salt thickening behavior of a binary copolymer amphiphilic polymer. J. Petrol. Sci. Eng. 2021, 204, 108713. [Google Scholar] [CrossRef]
- Liao, K.L.; Wei, M.; Fu, L.P.; Ma, Q.L.; An, J.N.; Bai, J.M.; Wang, M.L.; He, Y.F. Study on the Relationship between the Relative Molecular Mass of a Polymer Clay Stabilizer and the Permeability of a Tight Reservoir. ACS Omega 2022, 7, 25751–25759. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhu, Z.; Jiang, T.; Liu, J.; Dong, Y.; Wu, Y.; Zhao, M.; Dai, C.; Li, L. Probing the effect of Young’s modulus on the reservoir regulation abilities of dispersed particle gels. Gels 2023, 9, 402. [Google Scholar] [CrossRef] [PubMed]
- Shamlooh, M.; Hamza, A.; Hussein, I.A.; Nasser, M.S.; Magzoub, M.; Salehi, S. Investigation of the Rheological Properties of Nanosilica-Reinforced PAM /PEI Gels for Wellbore Strengthening at High Reservoir Temperatures. Energy Fuels 2019, 3, 6829–6836. [Google Scholar] [CrossRef]
- Dai, C.; Liu, Y.; Zou, C.; You, Q.; Yang, S.; Zhao, M.; Zhao, G.; Wu, Y.; Sun, Y. Investigation on matching relationship be-tween dispersed particle gel (DPG) and reservoir pore-throats for in-depth profile control. Fuel 2017, 207, 109–120. [Google Scholar] [CrossRef]
- Briggs, M.A.; Wang, C.; Day-Lewis, F.D.; Williams, K.H.; Dong, W.; Lane, J.W. Return flows from beaver ponds enhance floodplain-to-river metals exchange in alluvial mountain catchments. Sci. Total Environ. 2019, 685, 357–369. [Google Scholar] [CrossRef]
- Ding, F.; Dai, C.; Sun, Y.; Zhao, G.; You, Q.; Liu, Y. Gelling Behavior of PAM/Phenolic Crosslinked Gel and Its Profile Control in a Low-Temperature and High-Salinity Reservoir. Gels 2022, 8, 433. [Google Scholar] [CrossRef]







| System | Temperature/°C | kwi/ μm2 | kwt/ μm2 | Plugging Rates/% | Standard Deviation/% |
|---|---|---|---|---|---|
| Tetraethylenepentamine hydrogel | 20 | 16.98 | 14.11 | 16.8 | 2.7 |
| 30 | 13.79 | 18.79 | 3.9 | ||
| 40 | 2.24 | 87.81 | 3.1 | ||
| 50 | 1.98 | 88.34 | 1.8 | ||
| 60 | 2.09 | 87.69 | 5.2 | ||
| 70 | 9.37 | 44.82 | 4.5 |
| System | Core Type | Initial Permeability | Shunting Rate (%) | Profile Improvement Rate (%) | |
|---|---|---|---|---|---|
| Before Injecting Hydrogel | After Injecting Hydrogel | ||||
| Diethylenetriamine hydrogel | Low permeability | 16.98 | 12 | 62.68 | 91.88 |
| High permeability | 50.94 | 88 | 37.32 | ||
| Triethylenetetramine hydrogel | Low permeability | 16.98 | 13 | 71.24 | 93.97 |
| High permeability | 50.94 | 87 | 28.76 | ||
| Tetraethylenepentamine hydrogel | Low permeability | 16.98 | 13 | 64.72 | 91.86 |
| High permeability | 50.94 | 87 | 35.28 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meng, J.; Mao, G.; Zhu, Z.; Li, Q.; Lin, X.; Wang, L.; Li, Y.; Huang, Y. Novel Environmentally Responsive Polyvinyl Polyamine Hydrogels Capable of Phase Transformation with Temperature for Applications in Reservoir Profile Control. Gels 2023, 9, 950. https://doi.org/10.3390/gels9120950
Meng J, Mao G, Zhu Z, Li Q, Lin X, Wang L, Li Y, Huang Y. Novel Environmentally Responsive Polyvinyl Polyamine Hydrogels Capable of Phase Transformation with Temperature for Applications in Reservoir Profile Control. Gels. 2023; 9(12):950. https://doi.org/10.3390/gels9120950
Chicago/Turabian StyleMeng, Jianxun, Guoliang Mao, Zhixuan Zhu, Qingsong Li, Xuesong Lin, Lichao Wang, Yiran Li, and Yue Huang. 2023. "Novel Environmentally Responsive Polyvinyl Polyamine Hydrogels Capable of Phase Transformation with Temperature for Applications in Reservoir Profile Control" Gels 9, no. 12: 950. https://doi.org/10.3390/gels9120950