Cyclic Voltammetric Behaviour and High-Performance Liquid Chromatography Amperometric Determination of Levamisole
Abstract
1. Introduction
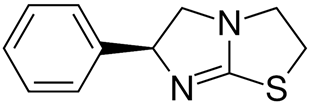
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Artificial Urine
2.3. Cyclic Voltammetry
2.4. High-Performance Liquid Chromatography
2.5. Electrochemical Detection
2.6. Hydrodynamic Voltammetry
2.7. Analytical Application
2.8. Limit of Detection and Limit of Quantification
3. Results
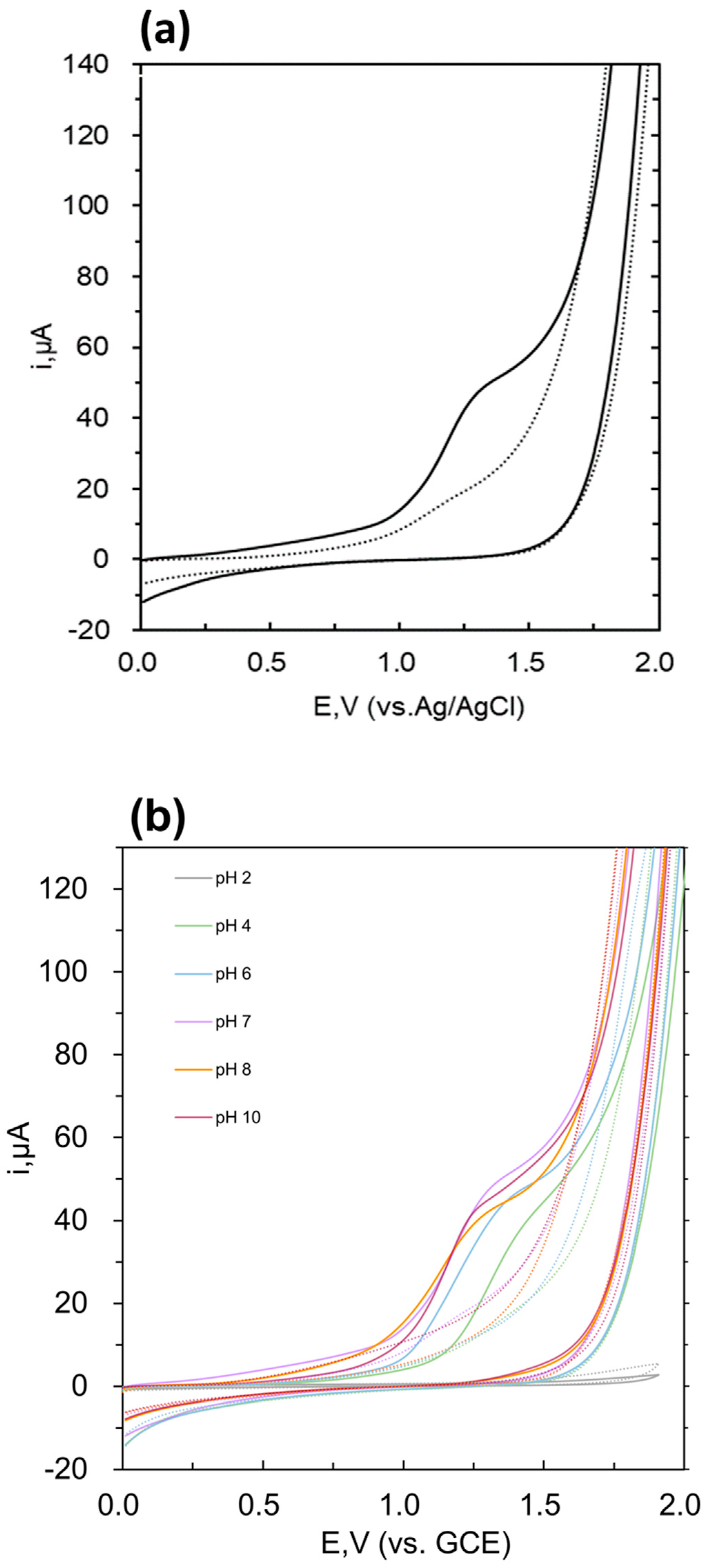
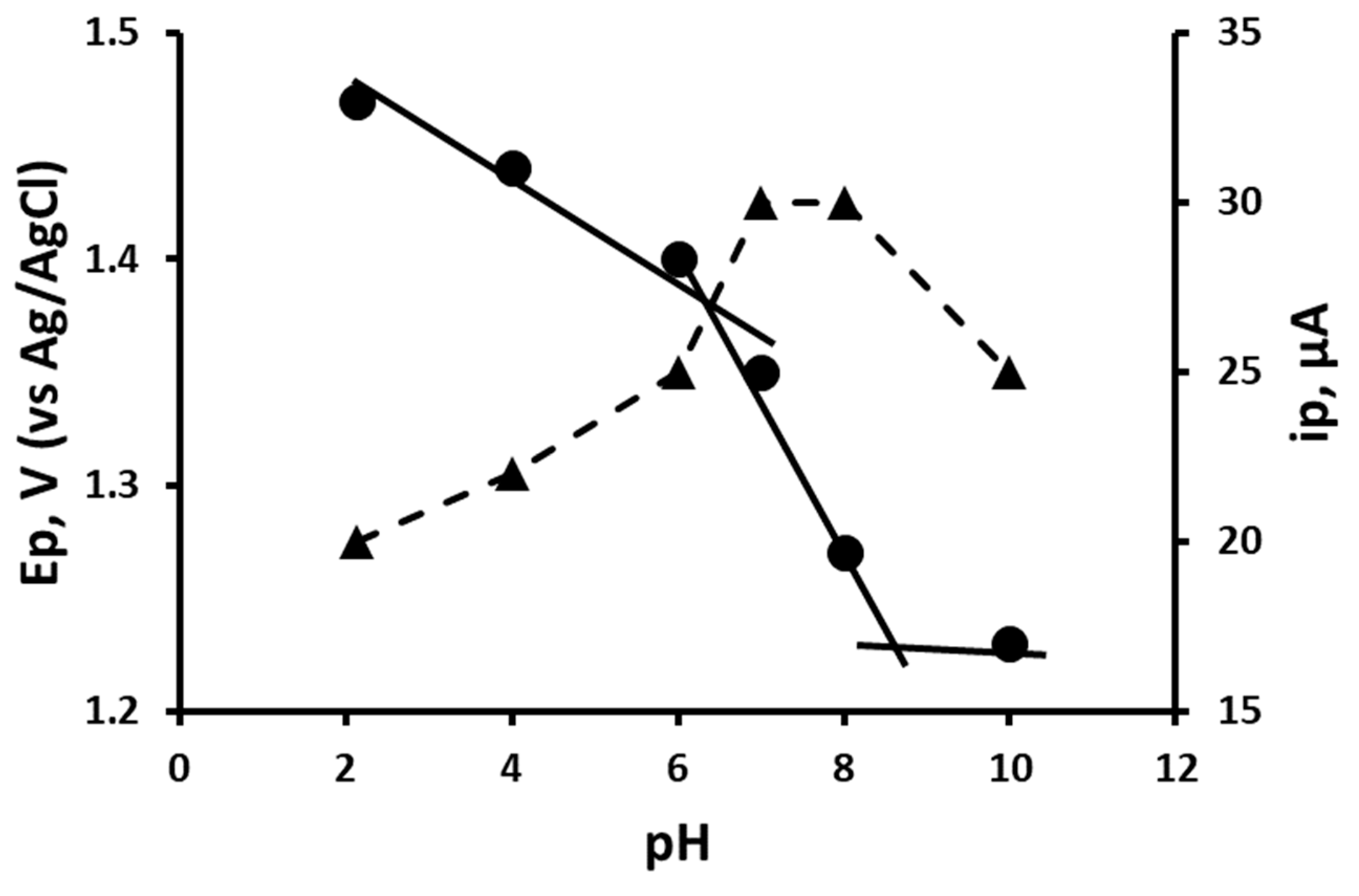
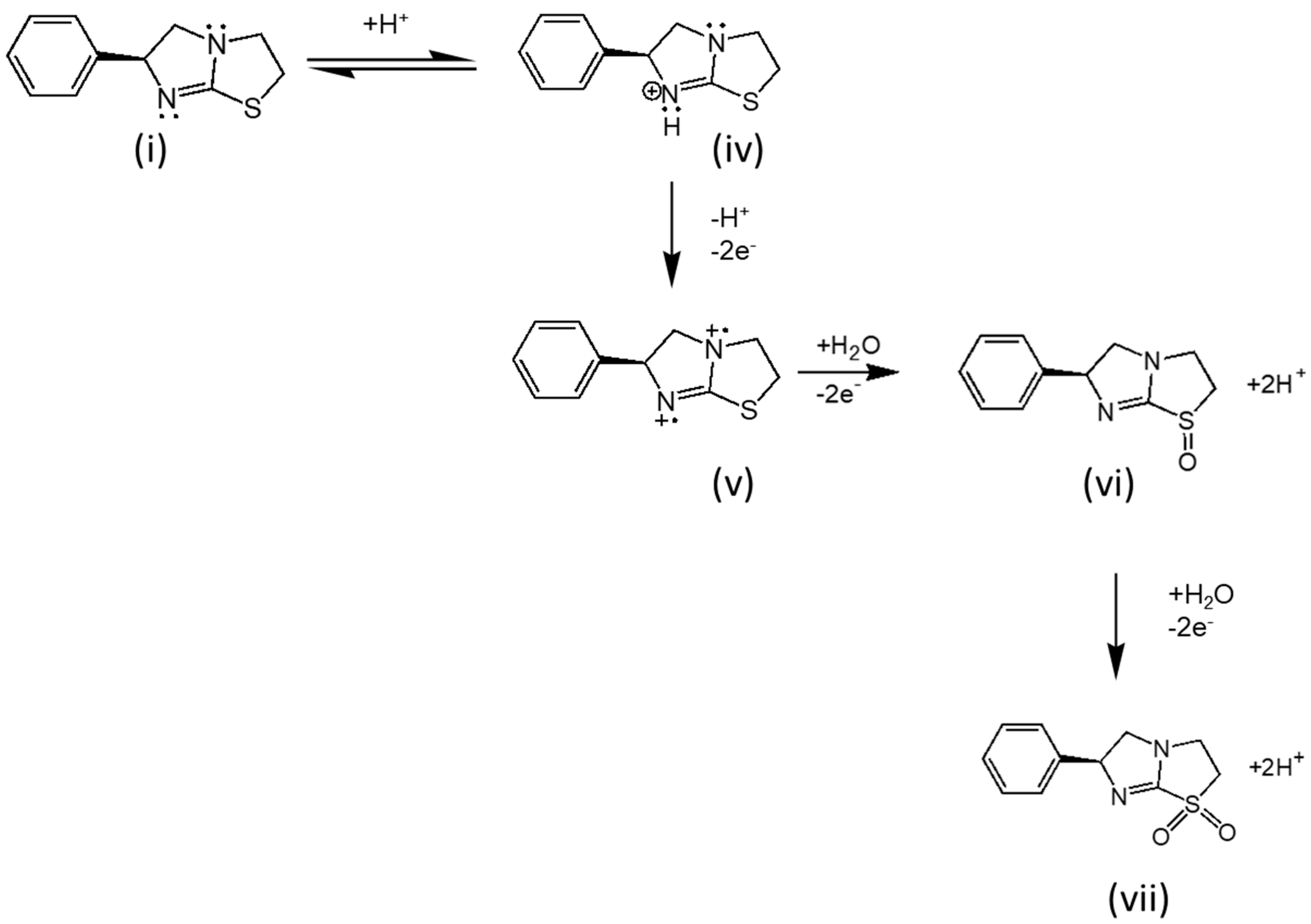
3.1. Cyclic Voltammetric Behaviour of Levamisole
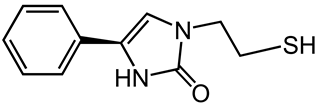
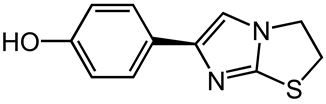
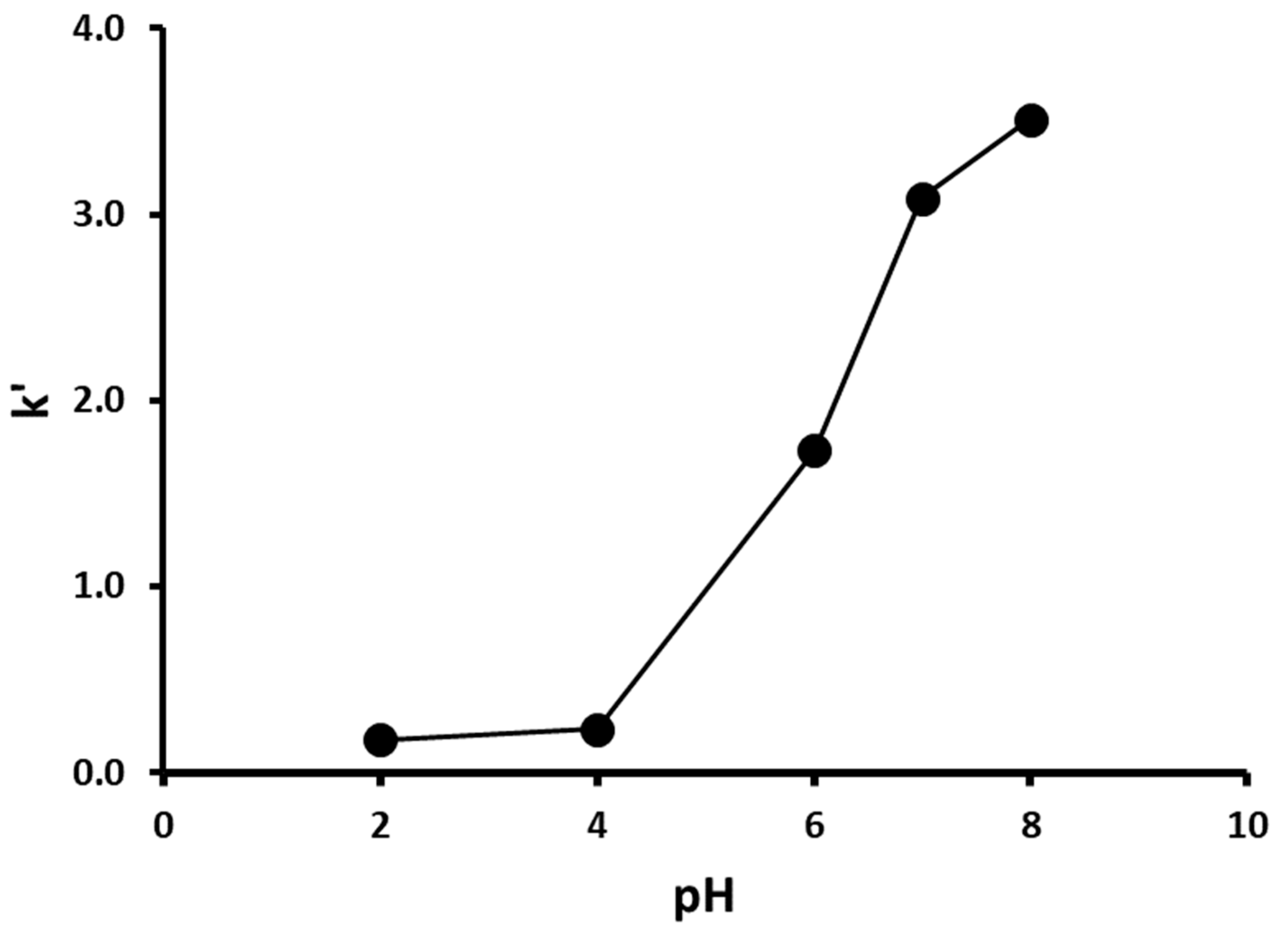
3.2. Effect of pH on the Liquid Chromatographic Separation of Levamisole
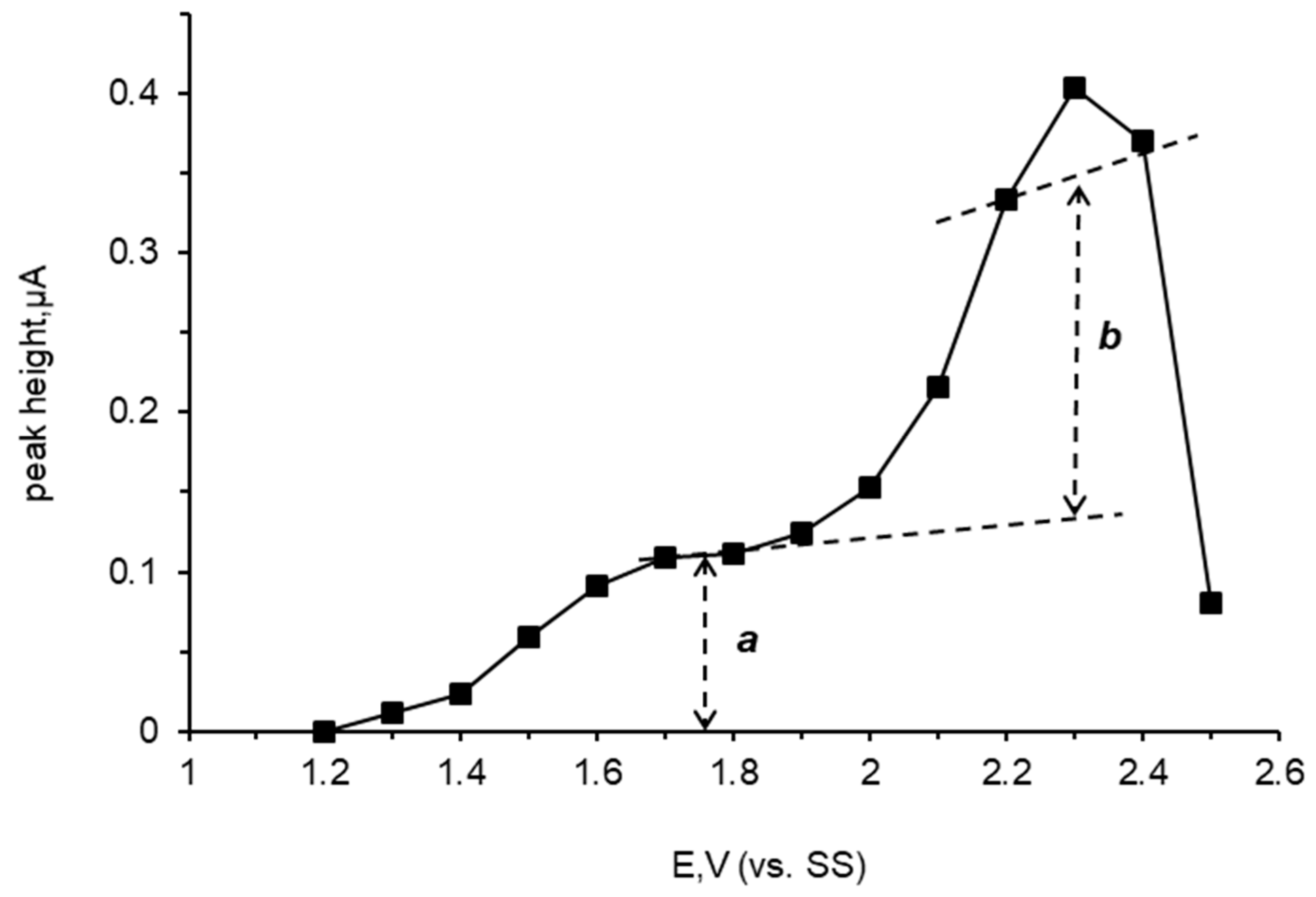
3.3. Hydrodynamic Voltammetry
3.4. Calibration Plot, Limit of Detection, and Precision
3.5. Studies of Possible Interferences
4. Analytical Application
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Keiser, J.; Utzinger, J. Efficacy of current drugs against soil-transmitted helminth infections: Systematic review and meta-analysis. JAMA 2008, 299, 1937–1948. [Google Scholar] [CrossRef]
- World Health Organization. Model List of Essential Medicines, 21st ed.; World Health Organization: Geneva, Switzerland, 2019.
- Janssen, P.A.J. The Levamisole Story. In Progress in Drug Research/Fortschritte der Arzneimittelforschung/Progrés des Recherches Pharmaceutiques; Jucker, E., Ed.; Birkhäuser: Basel, Switzerland, 1976; Volume 20. [Google Scholar] [CrossRef]
- Firozabad, A.R.; Meybodi, Z.A.; Mousavinasab, S.R.; Sahebnasagh, A.; Jelodar, M.G.; Karimzadeh, I.; Habtemariam, S.; Saghafi, F. Efficacy and safety of Levamisole treatment in clinical presentations of non-hospitalized patients with COVID-19: A double-blind, randomized, controlled trial. BMC Infect. Dis. 2021, 21, 297. [Google Scholar] [CrossRef]
- Keim, C.K.; Schwartz, R.A.; Kapila, R. Levamisole-induced and COVID-19-induced retiform purpura: Two overlapping, emerging clinical syndromes. Arch. Dermatol. Res. 2023, 315, 265–273. [Google Scholar] [CrossRef]
- Lee, K.C.; Ladizinski, B.; Federman, D.G. Complications associated with use of levamisole-contaminated cocaine: An emerging public health challenge. Mayo Clin. Proc. 2012, 87, 581–586. [Google Scholar] [CrossRef] [PubMed]
- Vanhoutte, P.M.; Nueten, J.M.V.; Verbeuren, T.J.; Lapuron, P.M. Differential effects of the isomers of tetnamisole on adrenergic neunotransmission in cutaneous veins of dog. J. Pharmacol. Exp. Ther. 1977, 200, 127–140. [Google Scholar]
- Brugmans, J.; Van Lommel, R.; Baro, F. Clinical aspects of tetramisole, an anthelmintic with antidepressant activity. Psychopharmacology 1972, 26, 85. [Google Scholar]
- Bobon, J.; Bobon, D.P.; Bourdouxhe, S.; Pinchard, A. A pilot open trial of dexamisole (R 12.563) in retarded depression. J. Int. Med. Res. 1974, 2, 171–174. [Google Scholar] [CrossRef]
- Gutierrez, J.; Eisenberg, R.L.; Koval, N.J.; Armstrong, E.R.; Tharappel, J.; Hughes, C.G.; Tobin, T. Pemoline and Tetramisole ‘Positives’ in English racehorses following Levamisole administration. Ir. Vet. J. 2010, 63, 498–500. [Google Scholar] [CrossRef]
- Hofmaier, T.; Luf, A.; Seddik, A.; Stockner, T.; Holy, M.; Freissmuth, M.; Ecker, G.F.; Schmid, R.; Sitte, H.H.; Kudlacek, O. Aminorex, a metabolite of the cocaine adulterant levamisole, exerts amphetamine like actions at monoamine transporters. Neurochem. Int. 2014, 73, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Waller, P.J. From discovery to development: Current industry perspectives for the development of novel methods of helminth control in livestock. Vet. Parasitol. 2006, 139, 1–14. [Google Scholar] [CrossRef]
- Knowles, L.; Buxton, J.A.; Skuridina, N.; Achebe, I.; Legatt, D.; Fan, S.; Zhu, N.Y.; Talbot, J. Levamisole Tainted Cocaine Causing Severe Neutropenia in Alberta and British Columbia. Harm Reduct. J. 2009, 6, 30. [Google Scholar] [CrossRef] [PubMed]
- Larocque, A.; Hoffman, R.S. Levamisole in cocaine: Unexpected news from an old acquaintance. Clin. Toxicol. 2012, 50, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Misquith, J.; Dias, A. Validated Ultra Violet Spectroscopic Methods for The Determination of Levamisole Hydrochloride. Inter. J. Pharm. Tech Res. 2012, 4, 1631–1637. [Google Scholar]
- Syed, S.M.; Farooqui, Z.S.; Gawale, K.; Hamde, D. A Validated UV Spectroscopic Method for determination of Levamisole HCl. Int. J. Pharm. Res. Appl. 2020, 5, 97–101. [Google Scholar]
- Sowjanya, S.; Devadasu, C. Development of RP-HPLC Method for the Simultaneous Quantitation of Levamisole and Albendazole: Application to Assay Validation. Int. J. Anal. Chem. 2018, 2018, 5746305. [Google Scholar] [CrossRef]
- Lin, T.-J.; Yen, K.-T.; Chen, C.-F.; Yan, S.-T.; Su, K.-W.; Chiang, Y.-L. Label-Free Uric Acid Estimation of Spot Urine Using Portable Device Based on UV Spectrophotometry. Sensors 2022, 22, 3009. [Google Scholar] [CrossRef]
- de Jong, M.; Sleegers, N.; Kim, J.; Van Durme, F.; Samyn, N.; Wang, J.; DeWael, K. Electrochemical fingerprint of street samples for fast on-site screening of cocaine in seized drug powders. Chem. Sci. 2016, 7, 2364–2370. [Google Scholar] [CrossRef]
- Rocha, R.G.; Stefano, J.S.; Arantes, I.V.S.; Ribeiro, M.M.A.C.; Santana, M.H.P.; Richter, E.M.; Munoz, R.A.A. Simple Strategy for Selective Determination of Levamisole in Seized Cocaine and Pharmaceutical Samples Using Disposable Screen-printed Electrodes. Electroanalysis 2019, 31, 153–159. [Google Scholar] [CrossRef]
- Holbrook, A.; Scales, B. Polarographic determination of tetramisole hydrochloride in extracts of animal tissue. Anal. Biochem. 1967, 18, 46–53. [Google Scholar] [CrossRef]
- Sadeghi, S.; Fathi, F.; Abbasifar, J. Potentiometric sensing of levamisole hydrochloride based on molecularly imprinted polymer. Sens. Actuators B Chem. 2007, 122, 158–164. [Google Scholar] [CrossRef]
- Stefano, J.S.; Dias, A.C.; Arantes, I.V.S.; Costa, B.M.C.; Silva, L.A.J.; Richter, E.M.; Banks, C.E.; Munoz, R.A.A. Batch-injection Amperometric Analysis on Screen-printed Electrodes: Analytical System for High-throughput Determination of Pharmaceutical Molecules. Electroanalysis 2019, 31, 518–526. [Google Scholar] [CrossRef]
- Lourencao, B.C.; Medeiros, R.A.; Thomasi, S.S.; Ferreira, A.G.; Rocha-Filho, R.C.; Fatibello-Filho, O. Amperometric flow-injection determination of the anthelmintic drugs ivermectin and levamisole using electrochemically pretreated boron-doped diamond electrodes. Sens. Actuators B Chem. 2016, 222, 181–189. [Google Scholar] [CrossRef]
- Khan, L.; Read, H.; Ritchie, S.; Proft, T. Artificial Urine for Teaching Urinalysis Concepts and Diagnosis of Urinary Tract Infection in the Medical Microbiology Laboratory. J. Microbiol. Biol. Educ. 2017, 18. [Google Scholar] [CrossRef]
- Peng, X.; Giltrow, D.; Bowdler, P.; Honeychurch, K.C. Liquid Chromatography Electrochemical Determination of Nicotine in Third-Hand Smoke. Electroanalysis 2017, 29, 374–379. [Google Scholar] [CrossRef]
- Monteiro, M.C.; Winiarski, J.P.; Santana, E.R.; Szpoganicz, B.; Vieira, I.C. Ratiometric Electrochemical Sensor for Butralin Determination Using a Quinazoline-Engineered Prussian Blue Analogue. Materials 2023, 16, 1024. [Google Scholar] [CrossRef]
- Tyrpenou, A.E.; Xylouri-Frangiadaki, E.M. Determination of Levamisole in Sheep Muscle Tissue by High-Performance Liquid Chromatography and Photo Diode Array Detection. Chromatographia 2006, 63, 321–326. [Google Scholar] [CrossRef]
- Robertson, S.J.; Martin, R.J. The Activation of Nicotinic Acetylcholine Receptors in the Nematode Parasite Ascaris suum by the Application of Levamisole to the Cytoplasmic Surface of Muscle Membrane. Pestic. Sci. 1993, 37, 293–299. [Google Scholar] [CrossRef]
- Wahyuni, W.T.; Putra, B.R.; Marken, F. Voltammetric detection of vitamin B1 (thiamine) in neutral solution at a glassy carbon electrode via in situ pH modulation. Analyst 2020, 145, 1903–1909. [Google Scholar] [CrossRef] [PubMed]
- El-Kholy, H.; Kemppainen, B.; Ravis, W.; Hoerr, F. Pharmacokinetics of levamisole in broiler breeder chickens. J. Vet. Pharmacol. Ther. 2006, 29, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Robertson, A.P.; Bjorn, H.E.; Martin, R.J. Resistance to levamisole resolved at the single-channel level. FASEB J. 1999, 13, 749–760. [Google Scholar] [CrossRef] [PubMed]
- Zubenia, N.; Kormosh, Z.; Semenyshyn, D.; Kochubei, V.; Korolchuk, S.; Savchuk, T. Design and application of a levamisole-selective membrane sensor. Anal. Bioanal. Electrochem. 2016, 8, 466–477. [Google Scholar]
- Dickinson, N.A.; Hudson, H.E.; Taylor, P.J. Levamisole: Its Stability in Aqueous Solutions at Elevated Temperatures. Analyst 1971, 96, 235–243. [Google Scholar] [CrossRef]
- Wolford, A.; McDonald, T.S.; Eng, H.; Hansel, S.; Chen, Y.; Bauman, J.; Sharma, R.; Kalgutkar, A.S. Immune-mediated agranulocytosis caused by the cocaine adulterant levamisole: A case for reactive metabolite(s) involvement. Drug Metab. Dispos. 2012, 40, 1067–1075. [Google Scholar] [CrossRef]
- Masui, M.; Sayo, H.; Tsuda, Y. Anodic oxidation of amines. Part I. Cyclic voltammetry of aliphatic amines at a stationary glassy-carbon electrode. J. Chem. Soc. B 1968, 1968, 973–976. [Google Scholar] [CrossRef]
- Amjad, U.; Lashin, A.; Abbasi, R.; Rana, U.A.; Al-Arifi, N.; Khan, G.S.; Ali, S.; Kraatz, H.-B.; Shaha, A. pH and Temperature Responsive Electrooxidation of Thiazole Derivatives, and Preliminary Screening of Their Antioxidant Activity. J. Electrochem. Soc. 2016, 163, H350–H358. [Google Scholar] [CrossRef]
- Jordan, P.H.; Hart, J.P. Voltammetric behaviour of morphine at a glassy carbon electrode and its determination in human serum by liquid chromatography with electrochemical detection under basic conditions. Analyst 1991, 116, 991–996. [Google Scholar] [CrossRef]
- Garrido, J.M.P.J.; Delerue-Matos, C.; Borges, F.; Macedo, T.R.A.; Oliveira-Brett, A.M. Voltammetric Oxidation of Drugs of Abuse III. Heroin and Metabolites. Electroanalysis 2004, 16, 1497–1502. [Google Scholar] [CrossRef]
- Teófilo, K.R.; Arantes, L.; Marinho, P.A.; Macedo, A.; Pimentel, D.M.; Rocha, D.P.; de Oliveira, A.C.; Richter, E.; Muñoz, R.; dos Santos, W.T. Electrochemical detection of 3,4-methylenedioxymethamphetamine (ecstasy) using a boron-doped diamond electrode with differential pulse voltammetry: Simple and fast screening method for application in forensic analysis. Microchem. J. 2020, 157, 105088. [Google Scholar] [CrossRef]
- Horwitz, W.; Albert, R. The Horwitz Ratio (HorRat): A Useful Index of Method Performance with Respect to Precision. J. AOAC Int. 2006, 89, 1095–1109. [Google Scholar] [CrossRef] [PubMed]
- Honeychurch, K.C.; Hart, J.P. Voltammetric Behavior of p-Nitrophenol and Its Trace Determination in Human Urine by Liquid Chromatography with a Dual Reductive Mode Electrochemical Detection System. Electroanalysis 2007, 19, 2176–2184. [Google Scholar] [CrossRef]
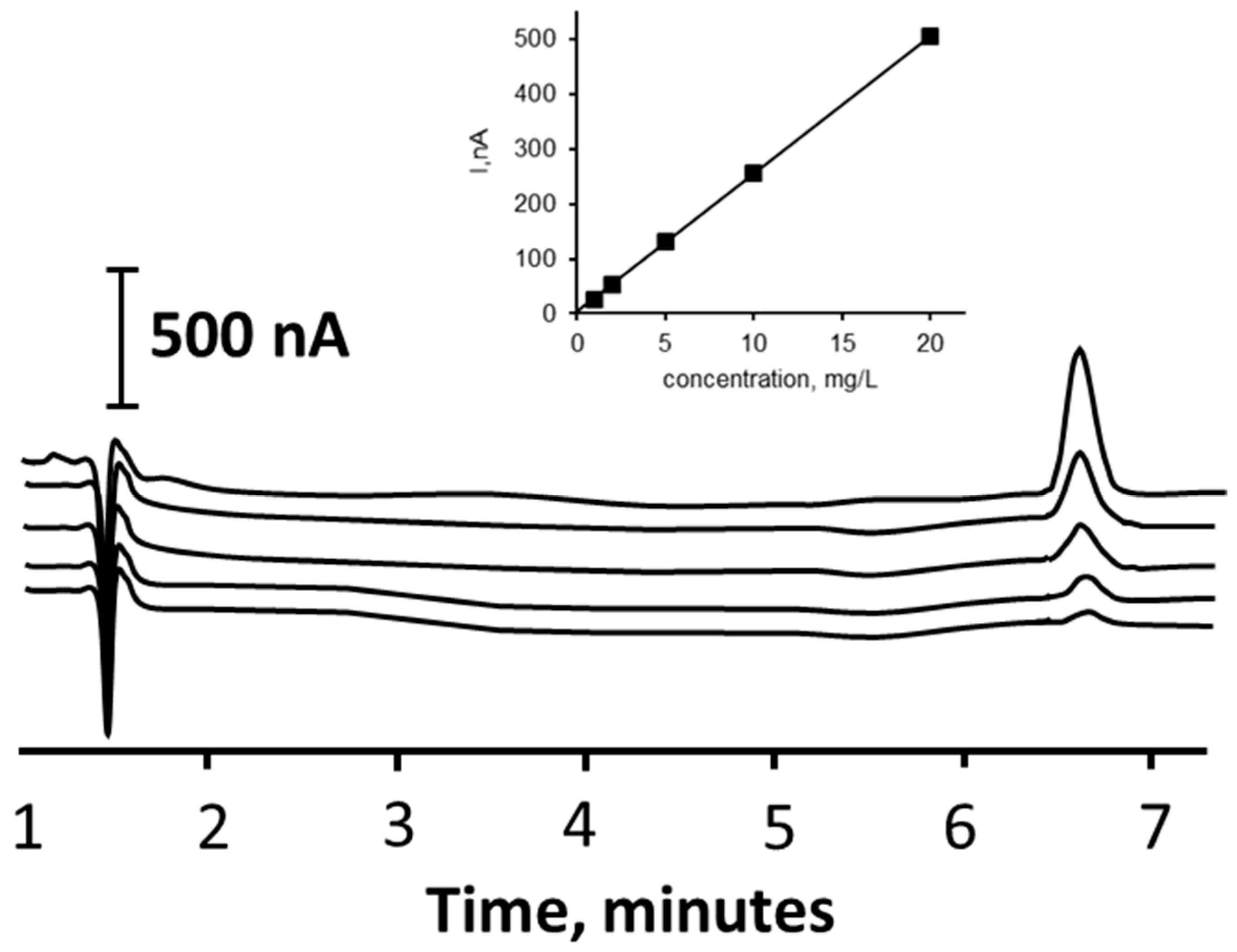
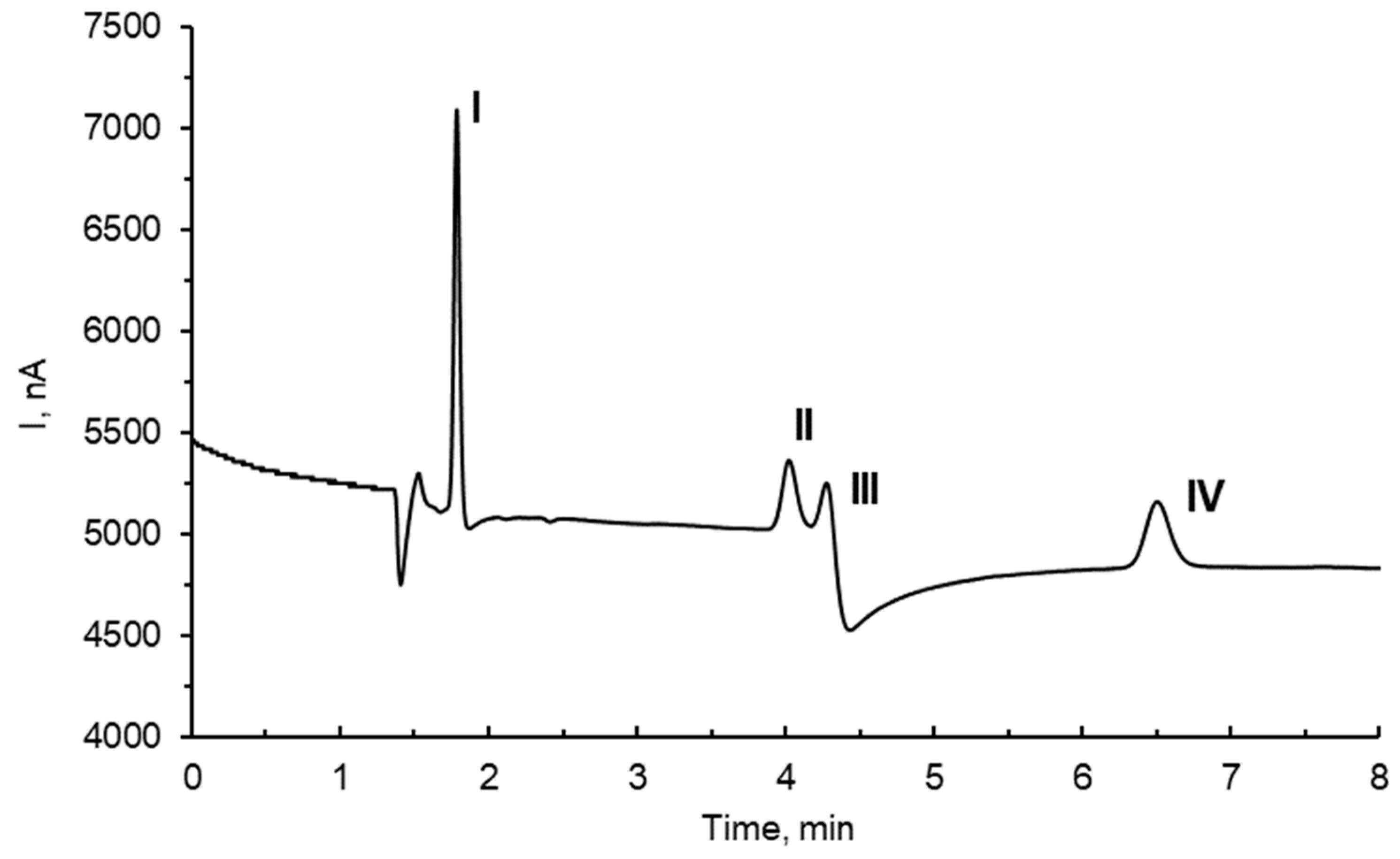
| Sample | Native, mg/L | Added mg/L | %Rec |
|---|---|---|---|
| 1 | ND | 1.25 | 100.3 |
| 2 | ND | 1.25 | 100.3 |
| 3 | ND | 1.25 | 87.5 |
| 4 | ND | 1.25 | 97.1 |
| 5 | ND | 1.25 | 113 |
| Mean | 99.6 | ||
| SD | 8.14 | ||
| %CV | 8.2 | ||
| HorRat | 0.53 | ||
| 6 | ND | 2.33 | 93.0 |
| 7 | ND | 2.13 | 85.1 |
| 8 | ND | 2.17 | 86.7 |
| 9 | ND | 2.84 | 113.7 |
| 10 | ND | 2.37 | 94.6 |
| Mean | 94.6 | ||
| SD | 10.2 | ||
| %CV | 10.8 | ||
| HorRat | 0.78 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chan, T.Y.J.; Honeychurch, K.C. Cyclic Voltammetric Behaviour and High-Performance Liquid Chromatography Amperometric Determination of Levamisole. Sci 2024, 6, 15. https://doi.org/10.3390/sci6010015
Chan TYJ, Honeychurch KC. Cyclic Voltammetric Behaviour and High-Performance Liquid Chromatography Amperometric Determination of Levamisole. Sci. 2024; 6(1):15. https://doi.org/10.3390/sci6010015
Chicago/Turabian StyleChan, Tsz Yan Joyce, and Kevin C. Honeychurch. 2024. "Cyclic Voltammetric Behaviour and High-Performance Liquid Chromatography Amperometric Determination of Levamisole" Sci 6, no. 1: 15. https://doi.org/10.3390/sci6010015
APA StyleChan, T. Y. J., & Honeychurch, K. C. (2024). Cyclic Voltammetric Behaviour and High-Performance Liquid Chromatography Amperometric Determination of Levamisole. Sci, 6(1), 15. https://doi.org/10.3390/sci6010015







