Urban Parks Are Related to Functional and Phylogenetic Filtering of Raptor Assemblages in the Austral Pampas, Argentina
Abstract
Simple Summary
Abstract
1. Introduction
2. Methods
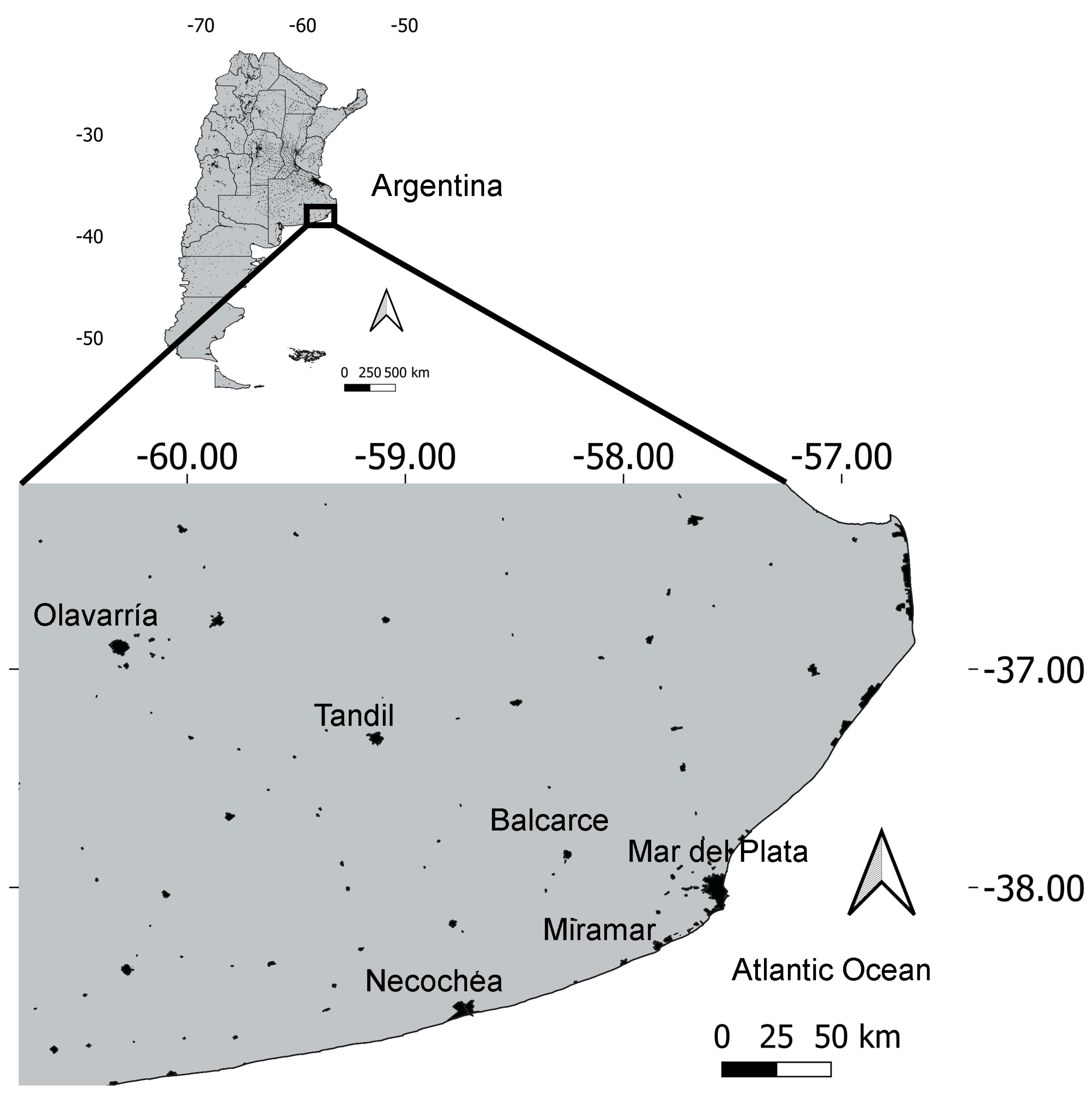
2.1. Study Area
2.2. Regional Pool of Species
2.3. Sample Coverage
2.4. Raptor Traits
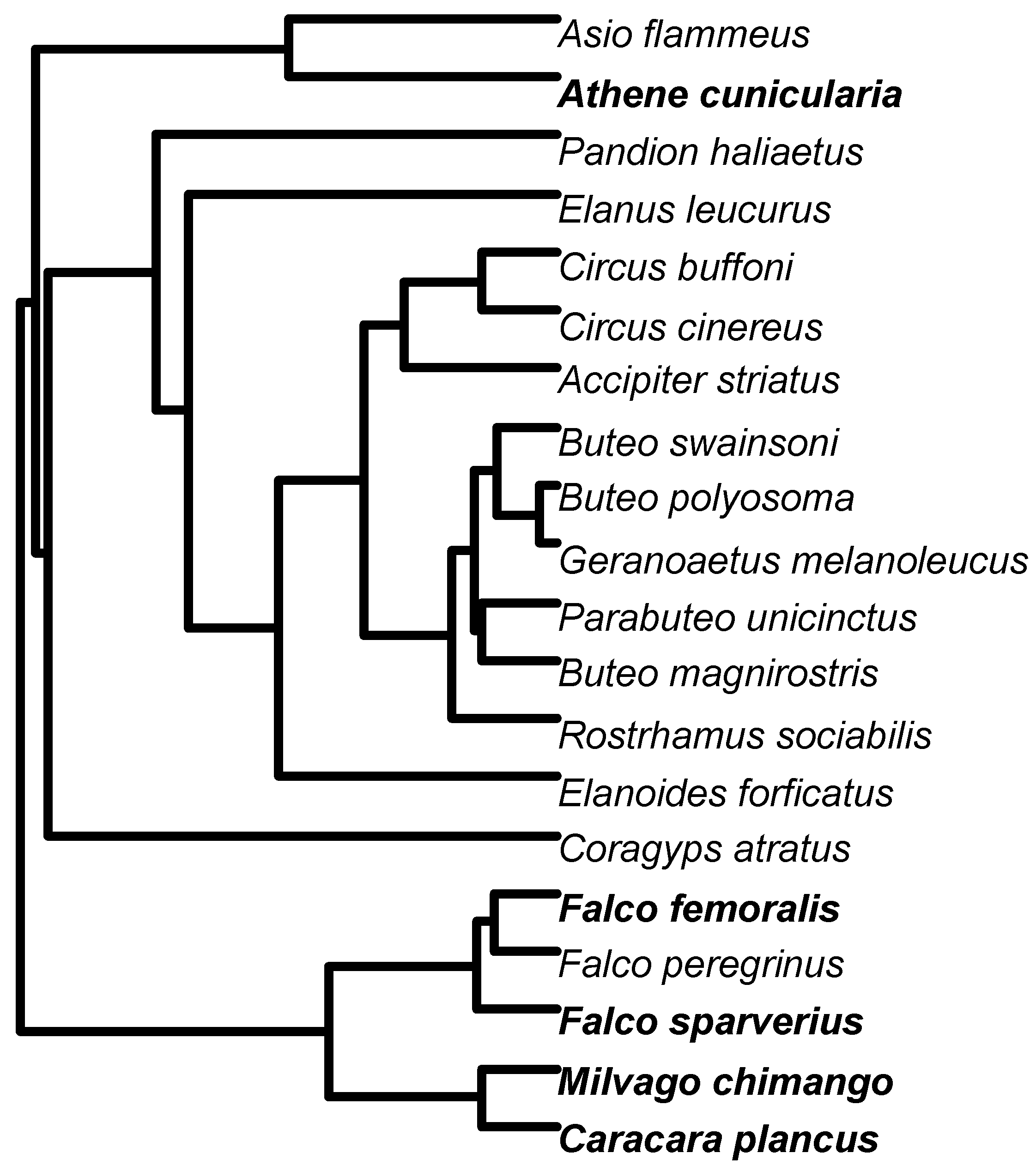
2.5. Raptor Phylogeny
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
6. Limitations of the Study
Supplementary Materials
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Sekercioglu, C.H. Increasing awareness of avian ecological function. Trends Ecol. Evol. 2006, 21, 464–471. [Google Scholar] [CrossRef]
- Donázar, J.A.; Cortés-Avizanda, A.; Fargallo, J.A.; Margalida, A.; Moleón, M.; Morales-Reyes, Z.; Moreno-Opo, R.; Pérez-García, J.M.; Sánchez-Zapata, J.A.; Zuberogoitia, I.; et al. Roles of raptors in a changing world: From flagships to providers of key ecosystem services. Ardeola 2016, 63, 181–234. [Google Scholar] [CrossRef]
- Kumar, N.; Jhala, Y.V.; Qureshi, Q.; Gosler, A.G.; Sergio, F. Human-attacks by an urban raptor are tied to human subsidies and religious practices. Sci. Rep. 2019, 9, 2545. [Google Scholar] [CrossRef] [PubMed]
- Bosakowski, T.; Smith, D.G. Distribution and species richness of a forest raptor community in relation to urbanization. J. Raptor Res. 1997, 31, 26–33. [Google Scholar]
- Hogg, J.R.; Nilon, C.H. Habitat associations of birds of prey in urban business parks. Urban Ecosyst. 2015, 18, 267–284. [Google Scholar] [CrossRef][Green Version]
- Solaro, C. Costs and benefits of urban living in raptors. In Birds of Prey; Sarasola, J.H., Garnde, J.M., Negro, J.J., Eds.; Springer: Cham, Switzerland, 2018; pp. 177–196. [Google Scholar]
- Dwyer, J.F.; Hindmarch, S.; Kratz, G.E. Raptor mortality in urban landscapes. In Urban Raptors; Island Press: Washington, DC, USA, 2018; pp. 199–213. [Google Scholar]
- Croci, S.; Butet, A.; Clergeau, P. Does urbanization filter birds on the basis of their biological traits. Condor 2008, 110, 223–240. [Google Scholar] [CrossRef]
- Patankar, S.; Jambhekar, R.; Suryawanshi, K.R.; Nagendra, H. Which traits influence bird survival in the city? A review. Land 2021, 10, 92. [Google Scholar] [CrossRef]
- Cooper, D.S.; Shultz, A.J.; Şekercioğlu, Ç.H.; Osborn, F.M.; Blumstein, D.T. Community science data suggest the most common raptors (Accipitridae) in urban centres are smaller, habitat-generalist species. Ibis 2022, 164, 771–784. [Google Scholar] [CrossRef]
- Headland, T.; Colombelli-Négrel, D.; Callaghan, C.T.; Sumasgutner, S.C.; Kleindorfer, S.; Sumasgutner, P. Smaller Australian raptors have greater urban tolerance. Sci. Rep. 2023, 13, 11559. [Google Scholar] [CrossRef]
- Boal, C.W. Urban raptor communities: Why some raptors and not others occupy urban environments. In Urban Raptors; Boal, C.W., Ed.; Island Press: Washington, DC, USA, 2018; pp. 36–50. [Google Scholar]
- Leveau, L.M. Big cities with small green areas hold a lower species richness and proportion of migrant birds: A global analysis. Urban For. Urban Green. 2021, 57, 126953. [Google Scholar] [CrossRef]
- Leveau, L.M.; Gorleri, F.C.; Roesler, I.; González-Táboas, F. What makes an urban raptor? Ibis 2022, 164, 1213–1226. [Google Scholar] [CrossRef]
- Sol, D.; González-Lagos, C.; Moreira, D.; Maspons, J.; Lapiedra, O. Urbanisation tolerance and the loss of avian diversity. Ecol. Lett. 2014, 17, 942–950. [Google Scholar] [CrossRef]
- Winchell, K.M.; Schliep, K.P.; Mahler, D.L.; Revell, L.J. Phylogenetic signal and evolutionary correlates of urban tolerance in a widespread neotropical lizard clade. Evolution 2020, 74, 1274–1288. [Google Scholar] [CrossRef]
- Nielsen, A.B.; Van Den Bosch, M.; Maruthaveeran, S.; van den Bosch, C.K. Species richness in urban parks and its drivers: A review of empirical evidence. Urban Ecosyst. 2014, 17, 305–327. [Google Scholar] [CrossRef]
- Gilbert, O. The Ecology of Urban Habitats; Chapman and Hall: London, UK, 1989. [Google Scholar]
- Mannan, R.W.; Boal, C.W. Home range characteristics of male Cooper’s hawks in an urban environment. Wilson Bull. 2000, 112, 21–27. [Google Scholar] [CrossRef]
- Schütz, C.; Schulze, C.H. Park size and prey density limit occurrence of Eurasian Sparrowhawks in urban parks during winter. Avian Res. 2018, 9, 30. [Google Scholar] [CrossRef]
- Zúñiga-Vega, J.J.; Gutiérrez-García, M.; Suárez-Rodríguez, M.; Morales-Salcedo, V.D.; Palencia-Martínez, M.; Espinosa-Lucas, A.; Ramírez-Cruz, G.A.; Pérez-García, R.D.; Saleh-Subaie, N.; Solano-Zavaleta, I. Raptors in the city: Site occupancy and abundance of a top predator inhabiting urban green spaces within a megacity. Landsc. Urban Plan. 2023, 234, 104725. [Google Scholar] [CrossRef]
- Jokimäki, J.; Huhta, E. Artificial nest predation and abundance of birds along an urban gradient. Condor 2000, 102, 838–847. [Google Scholar] [CrossRef]
- Jokimäki, J.; Kaisanlahti-Jokimäki, M.L.; Sorace, A.; Fernández-Juricic, E.; Rodriguez-Prieto, I.; Jimenez, M.D. Evaluation of the “safe nesting zone” hypothesis across an urban gradient: A multi-scale study. Ecography 2005, 28, 59–70. [Google Scholar] [CrossRef]
- Vincze, E.; Seress, G.; Lagisz, M.; Nakagawa, S.; Dingemanse, N.J.; Sprau, P. Does urbanization affect predation of bird nests? A meta-analysis. Front. Ecol. Evol. 2017, 5, 29. [Google Scholar] [CrossRef]
- Leveau, L.M. Large-scale variations of raptor communities in urban green spaces of neotropical cities. Perspect. Ecol. Conserv. 2022, 20, 352–359. [Google Scholar] [CrossRef]
- Poulin, R.G.; Todd, L.D.; Haug, E.A.; Millsap, B.A.; Martell, M.S. Burrowing Owl (Athene cunicularia), version 1.0. In Birds of the World; Poole, A.F., Ed.; Cornell Lab of Ornithology: Ithaca, NY, USA, 2020. [Google Scholar] [CrossRef]
- Wiggins, D.A.; Holt, D.W.; Leasure, S.M. Short-eared Owl (Asio flammeus), version 1.0. In Birds of the World; Billerman, S.M., Ed.; Cornell Lab of Ornithology: Ithaca, NY, USA, 2020. [Google Scholar] [CrossRef]
- Fuller, M.R.; Mosher, J. Raptor survey techniques. In Raptor Management Techniques Manual; Science Technical Service No., 10, Pendleton, B.A.G., Millsap, B.A., Cline, K.W., Bird, D.M., Eds.; National Wildlife Federation: Washington, DC, USA, 1987; pp. 37–65. [Google Scholar]
- Palomino, D.; Carrascal, L.M. Habitat associations of a raptor community in a mosaic landscape of Central Spain under urban development. Landsc. Urban Plan. 2007, 83, 268–274. [Google Scholar] [CrossRef]
- Kumar, S.; Sohil, A.; Kichloo, M.A.; Sharma, N. Landscape heterogeneity affects diurnal raptor communities in a sub-tropical region of northwestern Himalayas, India. PLoS ONE 2022, 17, e0246555. [Google Scholar] [CrossRef] [PubMed]
- Keddy, P.A.; Laughlin, D.C. A Framework for Community Ecology: Species Pools, Filters and Traits; Cambridge University Press: Cambridge, UK, 2021. [Google Scholar]
- Pärtel, M.; Zobel, M.; Zobel, K.; van der Maarel, E. The species pool and its relation to species richness: Evidence from Estonian plant communities. Oikos 1996, 75, 111–117. [Google Scholar] [CrossRef]
- Narosky, T.; Di Giacomo, A. Las Aves de la Provincia de Buenos Aires: Distribución y Estatus; Vazquez Mazzini Editores, L.O.L.A., Ed.; Asociación Ornitológica del Plata: Buenos Aires, Argentina, 1993. [Google Scholar]
- Leveau, C.M.; Leveau, L.M. Conteos de aves rapaces en el sudeste de la provincia de Buenos Aires, Argentina. Nuestras Aves 2001, 41, 9. [Google Scholar] [CrossRef]
- Isacch, J.P.; Bó, M.S.; Vega, L.E.; Favero, M.; Baladrón, A.V.; Pretelli, M.G.; Stellatelli, O.A.; Cardoni, D.A.; Copello, S.; Block, C.; et al. Diversidad de Tetrápodos en un mosaico de ambientes del sudeste de la ecorregión Pampeana como herramienta para planificar en conservación. Rev. Mus. Argent. Cienc. Nat. 2016, 18, 211–233. [Google Scholar]
- Hsieh, T.C.; Ma, K.H.; Chao, A. iNEXT: An R package for rarefaction and extrapolation of species diversity (H ill numbers). Methods Ecol. Evol. 2016, 7, 1451–1456. [Google Scholar] [CrossRef]
- Wilman, H.; Belmaker, J.; Simpson, J.; de la Rosa, C.; Rivadeneira, M.M.; Jetz, W. EltonTraits 1.0: Species-level foraging attributes of the world’s birds and mammals: Ecological Archives E095–178. Ecology 2014, 95, 2027. [Google Scholar] [CrossRef]
- Jetz, W.; Thomas, G.H.; Joy, J.B.; Redding, D.W.; Hartmann, K.; Mooers, A.O. Global distribution and conservation of evolutionary distinctness in birds. Curr. Biol. 2014, 24, 919–930. [Google Scholar] [CrossRef]
- Ericson, P.G.; Zuccon, D.; Ohlson, J.I.; Johansson, U.S.; Alvarenga, H.; Prum, R.O. Higher-level phylogeny and morphological evolution of tyrant flycatchers, cotingas, manakins, and their allies (Aves: Tyrannida). Mol. Phylogenetics Evol. 2006, 40, 471–483. [Google Scholar] [CrossRef]
- Rambaut, A.; Drummond, A.J. (2002–2018). TreeAnnotator v 1.8.2. Available online: http://beast.bio.ed.ac.uk/ (accessed on 25 October 2018).
- Paradis, E.; Claude, J.; Strimmer, K. APE: Analyses of phylogenetics and evolution in R language. Bioinformatics 2004, 20, 289–290. [Google Scholar] [CrossRef]
- Kembel, S.W.; Cowan, P.D.; Helmus, M.R.; Cornwell, W.K.; Morlon, H.; Ackerly, D.D.; Blomberg, S.P.; Webb, C.O. Picante: R tools for integrating phylogenies and ecology. Bioinformatics 2010, 26, 1463–1464. [Google Scholar] [CrossRef]
- Petchey, O.L.; Evans, K.L.; Fishburn, I.S.; Gaston, K.J. Low functional diversity and no redundancy in British avian assemblages. J. Anim. Ecol. 2007, 76, 977–985. [Google Scholar] [CrossRef] [PubMed]
- Laliberté, E.; Legendre, P.; Shipley, B. FD: Measuring Functional Diversity from Multiple Traits, and Other Tools for Functional Ecology. R package, Version 1.0-11. 2014. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing. Version 3.4.0. R Foundation for Statistical Computing: Vienna, Austria, 2017. [Google Scholar]
- Bierregaard, R.O.; Kirwan, G.M.; Boesman, P.F.D. Chimango Caracara (Milvago chimango), version 1.0. In Birds of the World; (del Hoyo, J., Elliott, A., Sargatal, J., Christie, D.A., de Juana, E., Eds.; Cornell Lab of Ornithology: Ithaca, NY, USA, 2020. [Google Scholar] [CrossRef]
- Morrison, J.L.; Dwyer, J.F. Crested Caracara (Caracara plancus), version 1.0. In Birds of the World; Poole, A.F., Ed.; Cornell Lab of Ornithology: Ithaca, NY, USA, 2021. [Google Scholar] [CrossRef]
- Leveau, L.M.; Leveau, C.M.; Pardiñas, U.F. Dieta del Milano Blanco (Elanus leucurus) en Argentina. Ornitol. Neotrop. 2002, 13, 307–311. [Google Scholar]
- Pedrana, J.; Isacch, J.P.; Bó, M.S. Habitat relationships of diurnal raptors at local and landscape scales in southern temperate grasslands of Argentina. Emu-Austral Ornithol. 2008, 108, 301–310. [Google Scholar] [CrossRef]
- Pearman, M.; Areta, J.I. Field Guide to the birds of Argentina and the Southwest Atlantic; Bloomsbury Publishing: London, UK, 2020. [Google Scholar]
- Leveau, L.M.; Leveau, C.M. Uso de hábitat por aves rapaces en un agroecosistema pampeano. Hornero 2002, 17, 9–15. [Google Scholar] [CrossRef]
- de la Peña, M.R. Nidos y Reproducción de las Aves Argentinas; Ediciones Biológica. Serie Naturaleza, Conservación y Sociedad N° 8; Edciones Biológica: Santa Fe, Argentina, 2013; 590p. [Google Scholar]
- Leveau, L.M.; Vaccaro, A.S.; Bellocq, M.I. Urbanization and bird diversity: Does the relationship change in deserts and subtropical forests? Urban Ecosyst. 2022, 25, 1891–1900. [Google Scholar] [CrossRef]
- Neate-Clegg, M.H.; Tonelli, B.A.; Youngflesh, C.; Wu, J.X.; Montgomery, G.A.; Şekercioğlu, Ç.H.; Tingley, M.W. Traits shaping urban tolerance in birds differ around the world. Curr. Biol. 2023, 33, 1677–1688. [Google Scholar] [CrossRef]
- Travaini, A.; Donazar, J.A.; Rodriguez, A.; Bustamante, J.; Delibes, M.; Hiraldo, F.; Ceballos, O.; Funes, M. Nest-site characteristics of four raptor species in the Argentinian Patagonia. Wilson Bull. 1994, 106, 753–757. [Google Scholar]
- Seipke, S.H. First record of Southern Caracaras (Caracara plancus) nesting on a human-made object. J. Raptor Res. 2012, 46, 228–230. [Google Scholar] [CrossRef]
- Dwyer, J.F.; Dalla Rosa, J.P. Use of anthropogenic nest substrates by Crested Caracaras. Southeast. Nat. 2015, 14, N10–N15. [Google Scholar] [CrossRef]
- Peery, M.Z. Factors affecting interspecies variation in home-range size of raptors. Auk 2000, 117, 511–517. [Google Scholar] [CrossRef]
- Evans, K.L.; Chamberlain, D.E.; Hatchwell, B.J.; Gregory, R.D.; Gaston, K.J. What makes an urban bird? Glob. Chang. Biol. 2011, 17, 32–44. [Google Scholar] [CrossRef]
- Travaini, A.; Rodriguez, A.; Ceballos, O.; Donazar, J.A.; Hiraldo, F. Roadside raptor surveys in central Argentina. El Hornero 1995, 14, 064–066. [Google Scholar] [CrossRef]
- MacGregor-Fors, I.; García-Arroyo, M.; Quesada, J. Keys to the city: An integrative conceptual framework on avian urban filtering. J. Urban Ecol. 2022, 8, juac026. [Google Scholar] [CrossRef]
- Cavalli, M.; Isacch, J.P.; Baladrón, A.V.; Biondi, L.M.; Bó, M.S. Differing nest-defence behaviour in urban and rural populations of breeding Burrowing Owls. Emu 2016, 116, 428–434. [Google Scholar] [CrossRef]
- Biondi, L.M.; Fuentes, G.M.; Córdoba, R.S.; Bó, M.S.; Cavalli, M.; Paterlini, C.A.; Castano, M.V.; García, G.O. Variation in boldness and novelty response between rural and urban predatory birds: The Chimango Caracara, Milvago chimango as study case. Behav. Process. 2020, 173, 104064. [Google Scholar] [CrossRef]
- Lima, H.S.; Mariz, D.; da Silva Costa, B.M.; de FViturino, L.; Couto, G.; Naka, L.N. Adaptation of Crested Caracaras (Caracara plancus) to Urban Environments: First Report of a Nest Made of Human-Made Materials. J. Raptor Res. 2022, 56, 365–370. [Google Scholar] [CrossRef]
- Romero-Vidal, P.; Blanco, G.; Hiraldo, F.; Díaz-Luque, J.A.; Luna, Á.; Lera, D.; Zalba, S.; Carrete, M.; Tella, J.L. Nesting innovations in neotropical parrots associated to anthropogenic environmental changes. Ecol. Evol. 2023, 13, e10462. [Google Scholar] [CrossRef]
- Zuñiga-Palacios, J.; Zuria, I.; Moreno, C.E.; Almazán-Núñez, R.C.; González-Ledesma, M. Can small vacant lots become important reservoirs for birds in urban areas? A case study for a Latin American city. Urban For. Urban Green. 2020, 47, 126551. [Google Scholar] [CrossRef]
- Zilio, F.; Bolzan, A.; de Mendonça-Lima, A.; da Silva, C.O.; Verrastro, L.; Borges-Martins, M. Raptor assemblages in grasslands of Southern Brazil: Species richness and abundance and the influence of the survey method. Zool. Stud. 2013, 52, 27. [Google Scholar] [CrossRef][Green Version]

| English Name | Species | Nest | Diet | Habitat | Mass (g) | Foraging | Migration |
|---|---|---|---|---|---|---|---|
| Black Vulture | Coragyps atratus | rock | scavenger | 1 | 1881.69 | flight | 1 |
| Osprey | Pandion haliaetus | tree | reptile/fish | 2 | 1483.2 | flight | 3 |
| Black-chested Buzzard-Eagle | Geranoaetus melanoleucus | rock | mammals | 1 | 2378.62 | perch | 1 |
| Variable Hawk | Geranoaetus polyosoma | tree | mammals | 1 | 782.61 | both | 3 |
| White-tailed Hawk | Elanus leucurus | tree | mammals | 2 | 346 | flight | 1 |
| Swallow-tailed Kite | Elanoides forficatus | tree | invertebrates | 3 | 416.24 | flight | 3 |
| Harris’s Hawk | Parabuteo unicinctus | tree | generalist | 2 | 850.28 | perch | 1 |
| Snail Kite | Rostrhamus sociabilis | ground | invertebrates | 1 | 366.94 | flight | 2 |
| Roadside Hawk | Rupornis magnirostris | tree | generalist | 2 | 269 | perch | 1 |
| Swainson’s Hawk | Buteo swainsoni | tree | invertebrates | 2 | 946.61 | both | 3 |
| Sharp-shinned Hawk | Accipiter striatus | tree | birds | 3 | 130.59 | both | 1 |
| Cinereous Harrier | Circus cinereus | ground | birds | 1 | 399.67 | flight | 2 |
| Long-winged Harrier | Circus buffoni | ground | birds | 1 | 507.4 | flight | 2 |
| Short-eared Owl | Asio flammeus | ground | mammals | 1 | 322.61 | flight | 2 |
| Burrowing Owl | Athene cunicularia | ground | generalist | 1 | 150.61 | both | 1 |
| Chimango Caracara | Milvago chimango | tree | generalist | 2 | 296 | both | 1 |
| Crested Caracara | Caracara plancus | tree | generalist | 2 | 1078.62 | both | 1 |
| Aplomado Falcon | Falco femoralis | tree | birds | 2 | 335.76 | both | 1 |
| American Kestrel | Falco sparverius | tree | invertebrates | 2 | 114.61 | perch | 1 |
| Peregrine Falcon | Falco peregrinus | rock | birds | 1 | 759.95 | flight | 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leveau, L.M. Urban Parks Are Related to Functional and Phylogenetic Filtering of Raptor Assemblages in the Austral Pampas, Argentina. Birds 2024, 5, 38-47. https://doi.org/10.3390/birds5010003
Leveau LM. Urban Parks Are Related to Functional and Phylogenetic Filtering of Raptor Assemblages in the Austral Pampas, Argentina. Birds. 2024; 5(1):38-47. https://doi.org/10.3390/birds5010003
Chicago/Turabian StyleLeveau, Lucas M. 2024. "Urban Parks Are Related to Functional and Phylogenetic Filtering of Raptor Assemblages in the Austral Pampas, Argentina" Birds 5, no. 1: 38-47. https://doi.org/10.3390/birds5010003
APA StyleLeveau, L. M. (2024). Urban Parks Are Related to Functional and Phylogenetic Filtering of Raptor Assemblages in the Austral Pampas, Argentina. Birds, 5(1), 38-47. https://doi.org/10.3390/birds5010003





