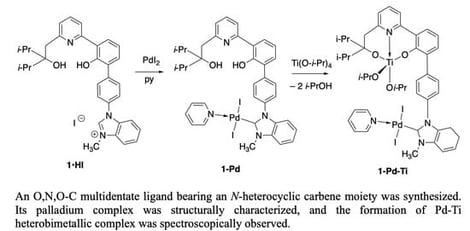
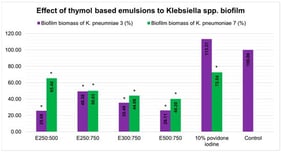
Background: Increasing antimicrobial resistance has directed studies toward investigating the antimicrobial activity of thymol, as well as the antibiofilm and antioxidant potential of its emulsions (with Tween 80) against multidrug-resistant (MDR)
K. pneumoniae isolates. Methods: A microdilution assay was used to estimate thymol’s antibacterial potential against 10 clinical isolates (labeled 1–10). The dynamic light scattering technique was used to measure the particle size diameter (Zavg) of formulated emulsions. The antibiofilm potential of emulsions was assessed in vitro using a crystal violet assay and ex vivo on a surgical drain through a colony-forming unit assay. Antioxidant activity was screened by using the DPPH assay. Results: The MIC values were ≤1.5 mg/mL for strains 1 and 7 and <0.5 mg/mL for the other strains. Emulsions E
250:500, E
250:750, E
300:750, and E
500:750 were stable and homogeneous, with a Zavg of approx. 200 nm (128.4 ± 0.8 nm for E
250:750). These emulsions significantly reduced the biofilm biomass of strains 3 and 7 (50.6–74.32% and 34.60–59.8% of inhibition, respectively), with the strongest activity observed for E
250:500 and E
500:750. Antibiofilm potential was confirmed ex vivo, with E
500:750 showing the highest efficacy (ΔLogCFU 2.60 and 2.68 for strains 3 and 7). E
250:750 demonstrated the highest capacity to neutralize the DPPH• radical. Conclusions: Thymol and its emulsions exhibited antibacterial and antibiofilm activity against MDR
K. pneumoniae isolates, along with the proven antioxidant properties of the emulsions.