Journal Description
Molbank
Molbank
is an international, peer-reviewed, open access journal comprised of a unique collection of one-compound-per-paper short notes on synthetic compounds and natural products published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, ESCI (Web of Science), Reaxys, CAPlus / SciFinder, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 14.7 days after submission; acceptance to publication is undertaken in 2.8 days (median values for papers published in this journal in the first half of 2025).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
0.4 (2024)
Latest Articles
Two Cocrystals of Phenazine with Different Phenylboronic Acids
Molbank 2025, 2025(3), M2036; https://doi.org/10.3390/M2036 (registering DOI) - 14 Jul 2025
Abstract
►
Show Figures
Boronic acids are an important class of molecules diversely used in organic synthesis, catalysis, medicinal chemistry, and for the design of functional materials. Particularly, aryl boronic acids in the solid state are known to exhibit pharmaceutical and photoluminescent properties for antimicrobial, sensing, and
[...] Read more.
Boronic acids are an important class of molecules diversely used in organic synthesis, catalysis, medicinal chemistry, and for the design of functional materials. Particularly, aryl boronic acids in the solid state are known to exhibit pharmaceutical and photoluminescent properties for antimicrobial, sensing, and drug delivery applications. Furthermore, the phenazine molecule is known for its diverse pharmacological properties, including antibiotic activity. In the case of molecular crystalline solids, it is well established that understanding noncovalent interactions remains key to designing or engineering their functional properties. While both aryl boronic acids and phenazine molecules individually represent an important class of compounds, their co-assembly in the crystalline state is of interest within the context of supramolecular chemistry and crystal engineering. Herein, we report the supramolecular features of two newly synthesized cocrystals, which are composed of para-F/CF3-substituted phenylboronic acids, respectively, and phenazine, as demonstrated by structure analysis by single-crystal X-ray diffraction.
Full article
Open AccessShort Note
A Pyrene-Anchored Nickel N-Heterocyclic Carbene–Isoquinoline Complex Promotes CO2 Reduction
by
Xue Chen, Li-Li Yu, Shu-Ying Chen, Tong Wang and Quan Zhou
Molbank 2025, 2025(3), M2035; https://doi.org/10.3390/M2035 - 8 Jul 2025
Abstract
In this study, on the basis of a previous report, a pyrene-anchored nickel complex was designed and synthesized via five steps. The NMR spectra of the synthesized complex were found to exhibit significant proton and carbon chemical shift anisotropy. Cyclic voltammetry spectra showed
[...] Read more.
In this study, on the basis of a previous report, a pyrene-anchored nickel complex was designed and synthesized via five steps. The NMR spectra of the synthesized complex were found to exhibit significant proton and carbon chemical shift anisotropy. Cyclic voltammetry spectra showed that the introduction of pyrene slightly influenced the onset potential of CO2 reduction. Lastly, controlled-potential electrolysis experiments disclosed that a pyrene-anchored nickel carbene–isoquinoline (Ni−2) complex selectively converted CO2 into CH4 with a TON value of 2.3 h−1.
Full article
(This article belongs to the Topic Heterocyclic Carbene Catalysis)
►▼
Show Figures

Figure 1
Open AccessShort Note
3′H-Spiro[dibenzo[c,h]xanthene-7,1′-isobenzofuran]-3′-one
by
Brian A. Chalmers, David B. Cordes, Aidan P. McKay, Iain L. J. Patterson, Nadiia Vladymyrova and Iain A. Smellie
Molbank 2025, 2025(3), M2033; https://doi.org/10.3390/M2033 - 7 Jul 2025
Abstract
Target compound 3′H-spiro[dibenzo[c,h]xanthene-7,1′-isobenzofuran]-3′-one (1) has long been known to be a by-product obtained from the preparation of naphtholphthalein. The structure of compound 1 was elucidated in the early 20th century; however, this compound has not
[...] Read more.
Target compound 3′H-spiro[dibenzo[c,h]xanthene-7,1′-isobenzofuran]-3′-one (1) has long been known to be a by-product obtained from the preparation of naphtholphthalein. The structure of compound 1 was elucidated in the early 20th century; however, this compound has not previously been fully characterized using modern techniques. In this report, 1H NMR and 13C NMR spectra are provided. X-ray crystallography is also used to characterize the title compound for the first time.
Full article
(This article belongs to the Section Structure Determination)
►▼
Show Figures

Figure 1
Open AccessShort Note
(5R,7R,11bR)-9-(di(1H-Indol-3-yl)methyl)-4,4,7,11b-tetramethyl-1,2,3,4,4a,5,6,6a,7,11,11a,11b-dodecahydrophenanthro[3,2-b]furan-5-yl Acetate
by
Jessica A. Perez-Rangel, Gabriela Servín-García, Atilano Gutiérrez-Carrillo, Alejandro Islas-Jácome, Luis Chacón-García, Rosa E. del Río and Carlos J. Cortés-García
Molbank 2025, 2025(3), M2034; https://doi.org/10.3390/M2034 - 7 Jul 2025
Abstract
►▼
Show Figures
The semi-synthesis of the (5R,7R,11bR)-9-(di(1H-indol-3-yl)methyl)-4,4,7,11b-tetramethyl-1,2,3,4,4a,5,6,6a,7,11,11a,11b-dodecahydrophenanthro[3,2-b]furan-5-yl acetate was performed via a pseudo-multicomponent reaction involving a double Friedel–Crafts alkylation between the natural product-derived aldehyde 6β-acetoxyvouacapane and the corresponding indole. The transformation was carried
[...] Read more.
The semi-synthesis of the (5R,7R,11bR)-9-(di(1H-indol-3-yl)methyl)-4,4,7,11b-tetramethyl-1,2,3,4,4a,5,6,6a,7,11,11a,11b-dodecahydrophenanthro[3,2-b]furan-5-yl acetate was performed via a pseudo-multicomponent reaction involving a double Friedel–Crafts alkylation between the natural product-derived aldehyde 6β-acetoxyvouacapane and the corresponding indole. The transformation was carried out under solvent-free mechanochemical conditions using mortar and pestle grinding, with ZnCl2 as the catalyst. Structural elucidation of the target compound was accomplished using 1D and 2D NMR spectroscopy (1H, 13C, COSY, HSQC, and HMBC), FT-IR, and high-resolution mass spectrometry (HRMS).
Full article

Graphical abstract
Open AccessShort Note
Diethyl 3-(4-Bromobenzoyl)-7-(4-pyridyl)indolizine-1,2-dicarboxylate
by
Mihaela Cristea, Mihai Răducă, Maria Gdaniec, Sergiu Shova, Nicoleta Doriana Banu and Florea Dumitrascu
Molbank 2025, 2025(3), M2032; https://doi.org/10.3390/M2032 - 7 Jul 2025
Abstract
►▼
Show Figures
The title compound, C26H21BrN2O5 (Compound 4), was obtained via our previously described procedure with modifications, i.e., via a facile one-pot three component reaction starting from commercially available materials. Compound 4 was crystallized from nitromethane. It
[...] Read more.
The title compound, C26H21BrN2O5 (Compound 4), was obtained via our previously described procedure with modifications, i.e., via a facile one-pot three component reaction starting from commercially available materials. Compound 4 was crystallized from nitromethane. It crystalized in a triclinic crystal system, in the P-

Graphical abstract
Open AccessShort Note
N,N,N′-Tris(trimethylsilyl)-2-pyridinecarboximidamide
by
Mukaila A. Ibrahim, Kathryn E. Preuss and René T. Boeré
Molbank 2025, 2025(3), M2031; https://doi.org/10.3390/M2031 - 3 Jul 2025
Abstract
N,N,N′-tris(trimethylsilyl)-carboximidamides are effective reagents in synthetic chemistry in reactions with both non-metal and metal halides, because the side product is the mild and volatile ClSi(CH3)3 rather than corrosive HCl. The title compound inserts the 2-pyridylamidinate
[...] Read more.
N,N,N′-tris(trimethylsilyl)-carboximidamides are effective reagents in synthetic chemistry in reactions with both non-metal and metal halides, because the side product is the mild and volatile ClSi(CH3)3 rather than corrosive HCl. The title compound inserts the 2-pyridylamidinate fragment into several non-metal systems, including custom chelating radical ligands. The single-crystal X-ray diffraction structure was determined and modeled by Hirshfeld atom refinement using custom aspherical atomic scattering factors. Excellent data quality led to a model with enhanced precision of all interatomic distances and free refinement of H-atom positions and anisotropic displacement ellipsoids. This structure model is compared to the four previously published analogous structures.
Full article
(This article belongs to the Section Structure Determination)
►▼
Show Figures

Figure 1
Open AccessShort Note
[1,2,5]Thiadiazolo[3,4-b]pyrazine-5,6(4H,7H)-dione
by
Lidia S. Konstantinova, Natalia V. Obruchnikova and Oleg A. Rakitin
Molbank 2025, 2025(3), M2030; https://doi.org/10.3390/M2030 - 1 Jul 2025
Abstract
Fused 1,2,5-chalcogenadiazoles are often used as biologically active compounds and organic optovoltaic materials. [1,2,5]Thiadiazolo[3,4-b]pyrazines are much less studied due to difficulties in their preparation. In this communication, [1,2,5]thiadiazolo[3,4-b]pyrazine-5,6(4H,7H)-dione, a key precursor for the synthesis of
[...] Read more.
Fused 1,2,5-chalcogenadiazoles are often used as biologically active compounds and organic optovoltaic materials. [1,2,5]Thiadiazolo[3,4-b]pyrazines are much less studied due to difficulties in their preparation. In this communication, [1,2,5]thiadiazolo[3,4-b]pyrazine-5,6(4H,7H)-dione, a key precursor for the synthesis of 5,6-dihalo-[1,2,5]thiadiazolo[3,4-b]pyrazines, was prepared via condensation of 1,2,5-thiadiazole-3,4-diamine with oxalic acid or oxalyl chloride. The structure of the newly synthesized compound was established by elemental analysis, high-resolution mass spectrometry, 1H and 13C NMR, IR spectroscopy, and mass spectrometry.
Full article
(This article belongs to the Collection Heterocycle Reactions)
►▼
Show Figures
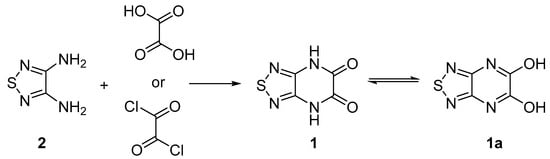
Scheme 1
Open AccessShort Note
4-(4-Formyl-3,5-dimethoxyphenoxy)butyric Acid (BAL)
by
Alex Lovstedt, Tracy R. Thompson and George Barany
Molbank 2025, 2025(3), M2029; https://doi.org/10.3390/M2029 - 26 Jun 2025
Abstract
The title compound, 4-(4-formyl-3,5-dimethoxyphenoxy)butyric acid (BAL), is an important “handle” for solid-phase synthesis of peptides and related compounds. Reported here is an X-ray single crystal structural analysis of BAL. The molecule is almost entirely flat, and the crystal is held together by π-stacking
[...] Read more.
The title compound, 4-(4-formyl-3,5-dimethoxyphenoxy)butyric acid (BAL), is an important “handle” for solid-phase synthesis of peptides and related compounds. Reported here is an X-ray single crystal structural analysis of BAL. The molecule is almost entirely flat, and the crystal is held together by π-stacking and hydrogen bonding.
Full article
(This article belongs to the Section Structure Determination)
►▼
Show Figures

Figure 1
Open AccessShort Note
4-([1,1′:3′,1′′-terphenyl]-2′-yloxy)-5-chlorophthalonitrile
by
Dmitry Erzunov, Vyacheslav Baklagin, Igor Abramov, Vladimir Maizlish, Roman Rumyantsev and Arthur Vashurin
Molbank 2025, 2025(3), M2028; https://doi.org/10.3390/M2028 - 24 Jun 2025
Abstract
The study focuses on the synthesis and detailed crystal structure analysis of 4-([1,1′:3′,1′′-terphenyl]-2′-yloxy)-5-chlorophthalonitrile. Using X-ray diffraction methods, the authors achieved the precise refinement of the atomic arrangement, revealing the specific spatial organization of molecules within the crystal lattice. The manuscript thoroughly discusses the
[...] Read more.
The study focuses on the synthesis and detailed crystal structure analysis of 4-([1,1′:3′,1′′-terphenyl]-2′-yloxy)-5-chlorophthalonitrile. Using X-ray diffraction methods, the authors achieved the precise refinement of the atomic arrangement, revealing the specific spatial organization of molecules within the crystal lattice. The manuscript thoroughly discusses the key intermolecular interactions—such as hydrogen bonding and π-π stacking—that govern the crystal packing. These interactions play a crucial role in stabilizing the structure and have a direct impact on the material’s physical and chemical properties, including its thermal stability and optical characteristics.
Full article
(This article belongs to the Section Structure Determination)
►▼
Show Figures
Figure 1
Open AccessCommunication
The Absolute Configuration Determination of Patagonic Acid
by
Eva E. Soto-Guzmán, Hilda J. Pagaza-Ibarra, Antonio J. Oliveros-Ortiz, Gabriela Rodríguez-García, Yliana López, Brenda Y. Bedolla-García, Carlos M. Cerda-García-Rojas, Christine Thomassigny, Mario A. Gómez-Hurtado, Armando Talavera-Alemán and Rosa E. del Río
Molbank 2025, 2025(3), M2027; https://doi.org/10.3390/M2027 - 23 Jun 2025
Abstract
(−)-Patagonic acid (1) is a clerodane diterpene isolated from several plants from the Alismataceae, Asteraceae, Euphorbiaceae, Fabaceae, Lamiaceae, Salicaceae, Sapindaceae, and Velloziaceae families, and its biological potential as an inhibitor of butyrylcholinesterase (BChE) and acetylcholinesterase (AChE) and as an anti-inflammatory compound
[...] Read more.
(−)-Patagonic acid (1) is a clerodane diterpene isolated from several plants from the Alismataceae, Asteraceae, Euphorbiaceae, Fabaceae, Lamiaceae, Salicaceae, Sapindaceae, and Velloziaceae families, and its biological potential as an inhibitor of butyrylcholinesterase (BChE) and acetylcholinesterase (AChE) and as an anti-inflammatory compound has been described. Furthermore, the enantiomer (+)-1 is also described in Fabaceae and Verbenaceae. A lack of formal studies about the absolute configuration (AC) determination of 1 is emphasized. Thus, the present manuscript describes the AC determination of patagonic acid (1). The chemical correlation of (−)-1 from (−)-hardwickiic acid (2) was achieved by a simplistic oxidative process. The specific rotation value and electronic circular dichroism (ECD) analysis allowed for the AC determination of (−)-1 as (5R,8R,9S,10R)-(−)-patagonic acid. ECD revealed a positive exciton chirality (EC) phenomenon in both (−)-1 and (−)-2, which is directly associated with their configuration and conformational preferences, which were assessed by DFT calculations at the B3LYP/DGDZVP level of theory. Since the NMR data of (+)-1 are fully coincident with those from its enantiomer studied herein, the chirality of (5S,8S,9R,10S)-(+)-patagonic acid could also be determined. These experimental conclusions deeply complement the literature related to clerodane compounds biosynthesized in several families of plants of scientific interest.
Full article
(This article belongs to the Section Natural Product Chemistry)
►▼
Show Figures

Graphical abstract
Open AccessShort Note
2-(2-(Benzylamino)-2-Oxoethyl)-1-Methyl-1H-Pyrrole-3-Carboxylic Acid
by
Monika Fryc, Beata Gryzło, Pravin Kumar and Agnieszka Zagórska
Molbank 2025, 2025(3), M2026; https://doi.org/10.3390/M2026 - 20 Jun 2025
Abstract
►▼
Show Figures
Here, 2-(2-(Benzylamino)-2-oxoethyl)-1-methyl-1H-pyrrole-3-carboxylic acid was efficiently synthesised in good yield via an amide coupling reaction between 2-carboxymethyl-1-methyl-1H-pyrrole-3-carboxylic acid and benzylamine, employing TBTU as the coupling reagent and DIPEA as the base. The reaction was carried out in dichloromethane at room
[...] Read more.
Here, 2-(2-(Benzylamino)-2-oxoethyl)-1-methyl-1H-pyrrole-3-carboxylic acid was efficiently synthesised in good yield via an amide coupling reaction between 2-carboxymethyl-1-methyl-1H-pyrrole-3-carboxylic acid and benzylamine, employing TBTU as the coupling reagent and DIPEA as the base. The reaction was carried out in dichloromethane at room temperature. The compound was characterised by melting point determination, 1H and 13C NMR, IR spectroscopy, and mass spectrometry. The combined analytical data confirm the target molecule’s successful synthesis and structural integrity.
Full article

Figure 1
Open AccessShort Note
N-(2-((2-(1H-indol-3-yl)ethyl)carbamoyl)phenyl)furan-2-carboxamide
by
Diyana Dimitrova, Iliyan Ivanov, Stanimir Manolov and Dimitar Bojilov
Molbank 2025, 2025(3), M2025; https://doi.org/10.3390/M2025 - 20 Jun 2025
Abstract
In the present study, we describe the synthesis of N-(2-((2-(1H-indol-3-yl)ethyl)carbamoyl)phenyl)furan-2-carboxamide via a two-step reaction sequence. Initially, isatoic anhydride was reacted with tryptamine to afford the corresponding intermediate, which was subsequently subjected to acylation using furan-2-carbonyl chloride. The final product was
[...] Read more.
In the present study, we describe the synthesis of N-(2-((2-(1H-indol-3-yl)ethyl)carbamoyl)phenyl)furan-2-carboxamide via a two-step reaction sequence. Initially, isatoic anhydride was reacted with tryptamine to afford the corresponding intermediate, which was subsequently subjected to acylation using furan-2-carbonyl chloride. The final product was comprehensively characterized by melting point analysis, 1H and 13C NMR, HSQC, IR, and MS spectrometry. The combined spectroscopic and analytical data unequivocally confirm the successful synthesis and structural integrity of the target compound.
Full article
(This article belongs to the Section Structure Determination)
►▼
Show Figures
Figure 1
Open AccessShort Note
bis(2-Phenylpyridinato)-[4,4′-bis(iodoethynyl)-2,2′-bipyridine]-iridium(III) Hexafluorophosphate
by
Patrick Endres, Nishi Singh, Andreas Winter, Helmar Görls and Ulrich S. Schubert
Molbank 2025, 2025(2), M2024; https://doi.org/10.3390/M2024 - 18 Jun 2025
Abstract
►▼
Show Figures
This work presents the synthesis and structural characterization of a novel type of biscyclometalated Ir(III) complex, which is equipped with two iodoethynyl moieties on its 2,2′-bipyridine (bpy) ligand. Iodoethynyl moieties represent prominent donor systems for the formation of supramolecular structures via halogen bonding
[...] Read more.
This work presents the synthesis and structural characterization of a novel type of biscyclometalated Ir(III) complex, which is equipped with two iodoethynyl moieties on its 2,2′-bipyridine (bpy) ligand. Iodoethynyl moieties represent prominent donor systems for the formation of supramolecular structures via halogen bonding (X-bonding). The synthesis of bis(2-phenylpyridinato)-[4,4′-bis(iodoethynyl)-2,2′-bipyridine]iridium(III) hexafluorophosphate, (2)(PF6), is straightforward and involves post-complexation iodination, thus expanding the already rich toolbox for performing “chemistry on the complex”. The formation of the iodoethynyl moieties was unequivocally proven by 1H-NMR spectroscopy, ESI-TOF mass spectrometry, and single-crystal XRD analysis.
Full article

Figure 1
Open AccessShort Note
(Z)-2-(Bromomethyl)-3-(hydroxymethylene)-7-methoxy-5-methyl-2-(tribromomethyl)-4-chromanone
by
Chein-Chung Lee and Yen-Ku Wu
Molbank 2025, 2025(2), M2023; https://doi.org/10.3390/M2023 - 16 Jun 2025
Abstract
►▼
Show Figures
An attempt to achieve the deprotonative bromination of a 2-methyl-3-formyl-chromenone with tetrabromomethane led to an unexpected bromo-tribromomethylation product. We report the synthesis and characterization of the title compound.
Full article

Figure 1
Open AccessShort Note
4-Methyl-N-(1-benzyl)-N’-(1-benzylidene)benzenesulfonohydrazide
by
Leticia Almazán-Sánchez, Marco A. García-Eleno, Diego Martínez-Otero and Erick Cuevas-Yañez
Molbank 2025, 2025(2), M2022; https://doi.org/10.3390/M2022 - 13 Jun 2025
Abstract
►▼
Show Figures
4-Methyl-N-(1-benzyl)-N’-(1-benzylidene)benzenesulfonohydrazide is formed through a direct, solventless reaction between benzaldehyde tosylhydrazone and potassium carbonate, which is carried out using an eco-friendly grinding method. The NMR spectra of the compound are here described. The structure was unequivocally determined by X-ray
[...] Read more.
4-Methyl-N-(1-benzyl)-N’-(1-benzylidene)benzenesulfonohydrazide is formed through a direct, solventless reaction between benzaldehyde tosylhydrazone and potassium carbonate, which is carried out using an eco-friendly grinding method. The NMR spectra of the compound are here described. The structure was unequivocally determined by X-ray analysis. As suggested by Hirshfeld surface analysis, the predominant intermolecular H-O interactions in this molecule are involved in crystal packing.
Full article
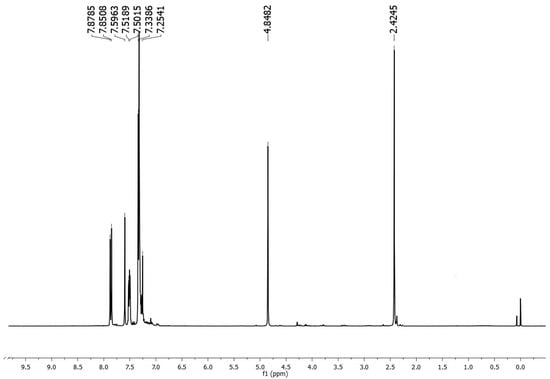
Figure 1
Open AccessShort Note
4,4-Dichloro-1,3-dithietane-2-one
by
Tracy R. Thompson, William W. Brennessel, Erik S. Goebel, Matthew J. Turcotte and George Barany
Molbank 2025, 2025(2), M2021; https://doi.org/10.3390/M2021 - 13 Jun 2025
Abstract
The title compound, 4,4-dichloro-1,3-dithietane-2-one, was encountered when opening a commercial capped amber bottle labeled “thiophosgene” that had been stored in a cold room (4 °C) for decades without any special precautions. Treating it as an unknown, the structure was established by single crystal
[...] Read more.
The title compound, 4,4-dichloro-1,3-dithietane-2-one, was encountered when opening a commercial capped amber bottle labeled “thiophosgene” that had been stored in a cold room (4 °C) for decades without any special precautions. Treating it as an unknown, the structure was established by single crystal X-ray analysis, and confirmed by 13C NMR, FTIR, melting behavior, and elemental analysis; its behavior under several mass spectrometric conditions was also examined. The dithietane appears to be a spontaneously formed cyclodimer of thiophosgene in which exactly one (not zero, not both) of the dichloromethylene moieties has been hydrolyzed to a carbonyl function. The relative long-term stability of the hydrolyzed dimer, along with a pathway back to thiophosgene, suggests that it might serve as a storage vehicle for toxic thiophosgene. Furthermore, as noted elsewhere, the title compound reacts with nucleophiles under mild aqueous conditions, suggesting that it may be a useful probe in chemical biology.
Full article
(This article belongs to the Collection Molecules from Side Reactions)
►▼
Show Figures

Figure 1
Open AccessCommunication
[Palladium-decabismuth(4+)]-tetrakis(tetrachloridoaluminate) Cluster Compound, [Pd@Bi10][AlCl4]4: Synthesis, Crystal Structure, and Electronic Structure
by
S. M. Gayomi K. Samarakoon and Sviatoslav Baranets
Molbank 2025, 2025(2), M2020; https://doi.org/10.3390/M2020 - 9 Jun 2025
Abstract
Black, needle-like single crystals of [Pd@Bi10][AlCl4]4 were synthesized in a one-pot reaction between PdCl2, Bi, and BiCl3 at 180 °C in the Lewis acidic ionic liquid (LAIL) medium [BMIm]Cl∙4.2AlCl4 (BMIm = 1-n-butyl-3-methylimidazolium).
[...] Read more.
Black, needle-like single crystals of [Pd@Bi10][AlCl4]4 were synthesized in a one-pot reaction between PdCl2, Bi, and BiCl3 at 180 °C in the Lewis acidic ionic liquid (LAIL) medium [BMIm]Cl∙4.2AlCl4 (BMIm = 1-n-butyl-3-methylimidazolium). Single-crystal X-ray diffraction revealed that the compound crystallizes in the triclinic space group P
(This article belongs to the Section Structure Determination)
►▼
Show Figures

Figure 1
Open AccessShort Note
5,5′-Di((E)-buta-1,3-dien-1-yl)-2,2′,3,3′-tetramethoxy-1,1′-biphenyl
by
Maria Antonietta Dettori, Davide Fabbri, Roberto Dallocchio and Paola Carta
Molbank 2025, 2025(2), M2018; https://doi.org/10.3390/M2018 - 6 Jun 2025
Abstract
Phenylbutanoids, commonly found in various medicinal plants, have attracted significant attention due to their remarkable biological activities, including antioxidant, anti-inflammatory, and neuroprotective effects, as well as for their versatility as starting materials in organic synthesis. Among phenylbutanoids, phenyl-1,3-butadienes represent a unique class of
[...] Read more.
Phenylbutanoids, commonly found in various medicinal plants, have attracted significant attention due to their remarkable biological activities, including antioxidant, anti-inflammatory, and neuroprotective effects, as well as for their versatility as starting materials in organic synthesis. Among phenylbutanoids, phenyl-1,3-butadienes represent a unique class of conjugated dienes, characterized by a phenyl (C6H5) group attached to a 1,3-butadiene (-CH=CH-CH=CH2) backbone. In this study, we synthesized the hydroxylated biphenyl 5,5′-di((E)-buta-1,3-dien-1-yl)-2,2′,3,3′-tetramethoxy-1,1′-biphenyl 1, closely related to its corresponding monomer 2, which is known for its broad range of pharmacological activities. The synthesis was carried out using microwave-assisted technologies. The structure of the synthesized compound was confirmed through elemental analysis, 13C-NMR, 1H-NMR, and ESI-MS spectrometry. Furthermore, we computed this novel compound’s conformational energy profile (CEP), evaluating how its energy varies with changes in the dihedral bond angle.
Full article
(This article belongs to the Section Organic Synthesis and Biosynthesis)
►▼
Show Figures

Graphical abstract
Open AccessCommunication
A Neutral Heteroleptic Cu(I) Complex with Diimine and Diphosphine Ligands
by
Xiaojuan Sun, Ruilong Sheng, Marijana Petkovic, Jolanta Jaśkowska and Zhiqiang Wang
Molbank 2025, 2025(2), M2019; https://doi.org/10.3390/M2019 - 6 Jun 2025
Abstract
►▼
Show Figures
Developing organic luminescent materials with the advantages of low cost, high thermal stability, and strong emission performance is incredibly desirable. In this work, we synthesized a new neutral heteroleptic Cu(I) complex characterized by single-crystal X-ray diffraction, FT-IR, NMR, and MALDI-TOF-MS. The neutral heteroleptic
[...] Read more.
Developing organic luminescent materials with the advantages of low cost, high thermal stability, and strong emission performance is incredibly desirable. In this work, we synthesized a new neutral heteroleptic Cu(I) complex characterized by single-crystal X-ray diffraction, FT-IR, NMR, and MALDI-TOF-MS. The neutral heteroleptic Cu(I) complex has a typical distorted tetrahedral configuration, and the complex molecules are connected into 1D chains via C-H···π interactions in crystal.
Full article

Graphical abstract
attachment
Supplementary material:
Supplementary File 1 (PDF, 1197 KiB)
Supplementary File 2 (MOL, 3 KiB)
Supplementary File 3 (INCHI, 724 B)
Supplementary File 4 (MOL, 3 KiB)
Supplementary File 5 (MOL, 3 KiB)
Supplementary File 6 (INCHI, 780 B)
Supplementary File 7 (MOL, 3 KiB)
Supplementary File 8 (MOL, 2 KiB)
Supplementary File 9 (INCHI, 686 B)
Supplementary File 10 (MOL, 2 KiB)
Supplementary File 11 (MOL, 2 KiB)
Supplementary File 12 (INCHI, 659 B)
Supplementary File 13 (MOL, 2 KiB)
Supplementary File 1 (PDF, 1197 KiB)
Supplementary File 2 (MOL, 3 KiB)
Supplementary File 3 (INCHI, 724 B)
Supplementary File 4 (MOL, 3 KiB)
Supplementary File 5 (MOL, 3 KiB)
Supplementary File 6 (INCHI, 780 B)
Supplementary File 7 (MOL, 3 KiB)
Supplementary File 8 (MOL, 2 KiB)
Supplementary File 9 (INCHI, 686 B)
Supplementary File 10 (MOL, 2 KiB)
Supplementary File 11 (MOL, 2 KiB)
Supplementary File 12 (INCHI, 659 B)
Supplementary File 13 (MOL, 2 KiB)
Open AccessCommunication
Novel Acid-Catalyzed Transformation of 1-Benzyl-3-Chloro-5-Hydroxy-4-[(4-Methylphenyl)Sulfanyl]-1,5-Dihydro-2H-Pyrrol-2-One
by
Liliya S. Kosolapova, Elena Sh. Saigitbatalova, Liliya Z. Latypova, Marat F. Valiev, Darya P. Gerasimova and Almira R. Kurbangalieva
Molbank 2025, 2025(2), M2017; https://doi.org/10.3390/M2017 - 4 Jun 2025
Abstract
Nitrogen-containing heterocycles of 3-pyrrolin-2-one series are widely represented in natural and synthetic compounds, with a broad spectrum of pharmacological activity and considerable potential in medicinal and synthetic organic chemistry. In this communication, we report the previously unknown acid-catalyzed transformation of a N-substituted
[...] Read more.
Nitrogen-containing heterocycles of 3-pyrrolin-2-one series are widely represented in natural and synthetic compounds, with a broad spectrum of pharmacological activity and considerable potential in medicinal and synthetic organic chemistry. In this communication, we report the previously unknown acid-catalyzed transformation of a N-substituted derivative of 3-pyrrolin-2-one that generates two types of heterocyclic moieties. The reflux of 1-benzyl-3-chloro-5-hydroxy-4-[(4-methylphenyl)sulfanyl]-1,5-dihydro-2H-pyrrol-2-one in toluene in the presence of catalytic amounts of H2SO4 resulted in the formation of a mixture of 1-benzyl-3-[(4-methylphenyl)sulfanyl]-1H-pyrrole-2,5-dione and 1-benzyl-7-methyl-1H-benzo[4,5]thieno[3,2-b]pyrrole-2,3-dione. The structures of four novel nitrogen-containing heterocycles were elucidated through IR, NMR spectroscopy and HRMS spectrometry. A new derivative of the fused tricyclic compounds, possessing benzo[b]thiophene and pyrrole-1,2-dione fragments, was also characterized by single-crystal X-ray diffraction.
Full article
(This article belongs to the Collection Heterocycle Reactions)
►▼
Show Figures

Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Catalysts, Chemistry, Molbank, Molecules, Sustainable Chemistry
Towards the Sustainable Synthesis of Biologically Active Molecules in Green Solvents
Topic Editors: Antonio Salomone, Serena PerroneDeadline: 31 December 2025

Conferences
Special Issues
Special Issue in
Molbank
Carbonylation Chemistry in the Synthesis of High Value Added Compounds
Guest Editors: Bartolo Gabriele, Raffaella MancusoDeadline: 20 April 2026
Topical Collections
Topical Collection in
Molbank
Molecules from Catalytic Processes
Collection Editor: Nicola Della Ca’






![4-[2-(Chlorodiphenylstannyl)phenyl]-4-hydroxybutan-2-one](https://pub.mdpi-res.com/title_story/title_story_1751895266425.jpg?1752235493)






