Gene Expression of Putative Pathogenicity-Related Genes in Verticillium dahliae in Response to Elicitation with Potato Extracts and during Infection Using Quantitative Real-Time PCR
Abstract
:1. Introduction
2. Results
2.1. Isolate Aggressiveness and Disease Assessment
2.2. Differential Fungal Gene Expression in Response to Elicitation with Potato Extracts and Inoculation
2.2.1. Genes Involved in Cell Differentiation and Proliferation
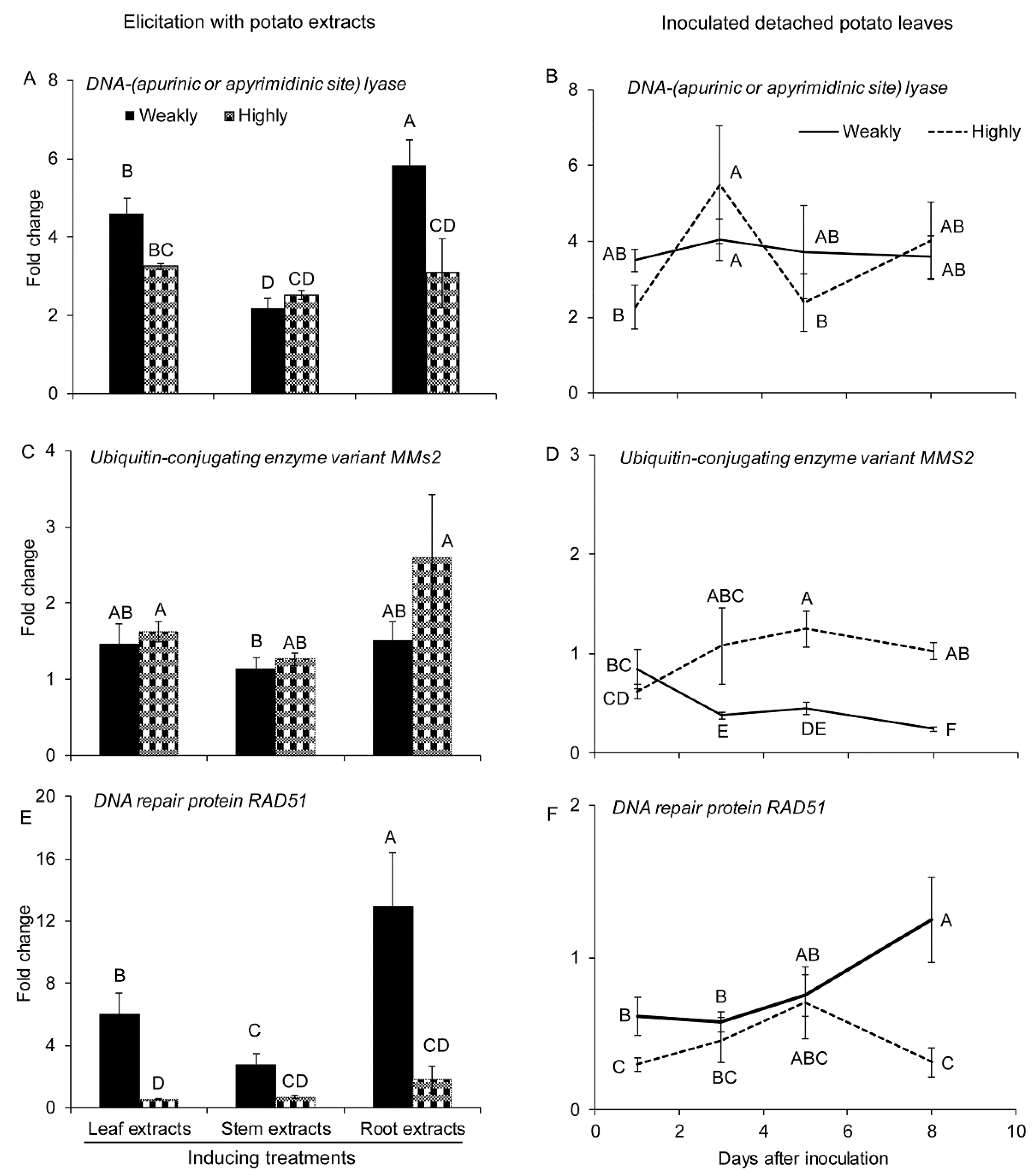
2.2.2. Genes Involved in DNA Repair
2.2.3. Genes Related to ROS Production and Cleavage
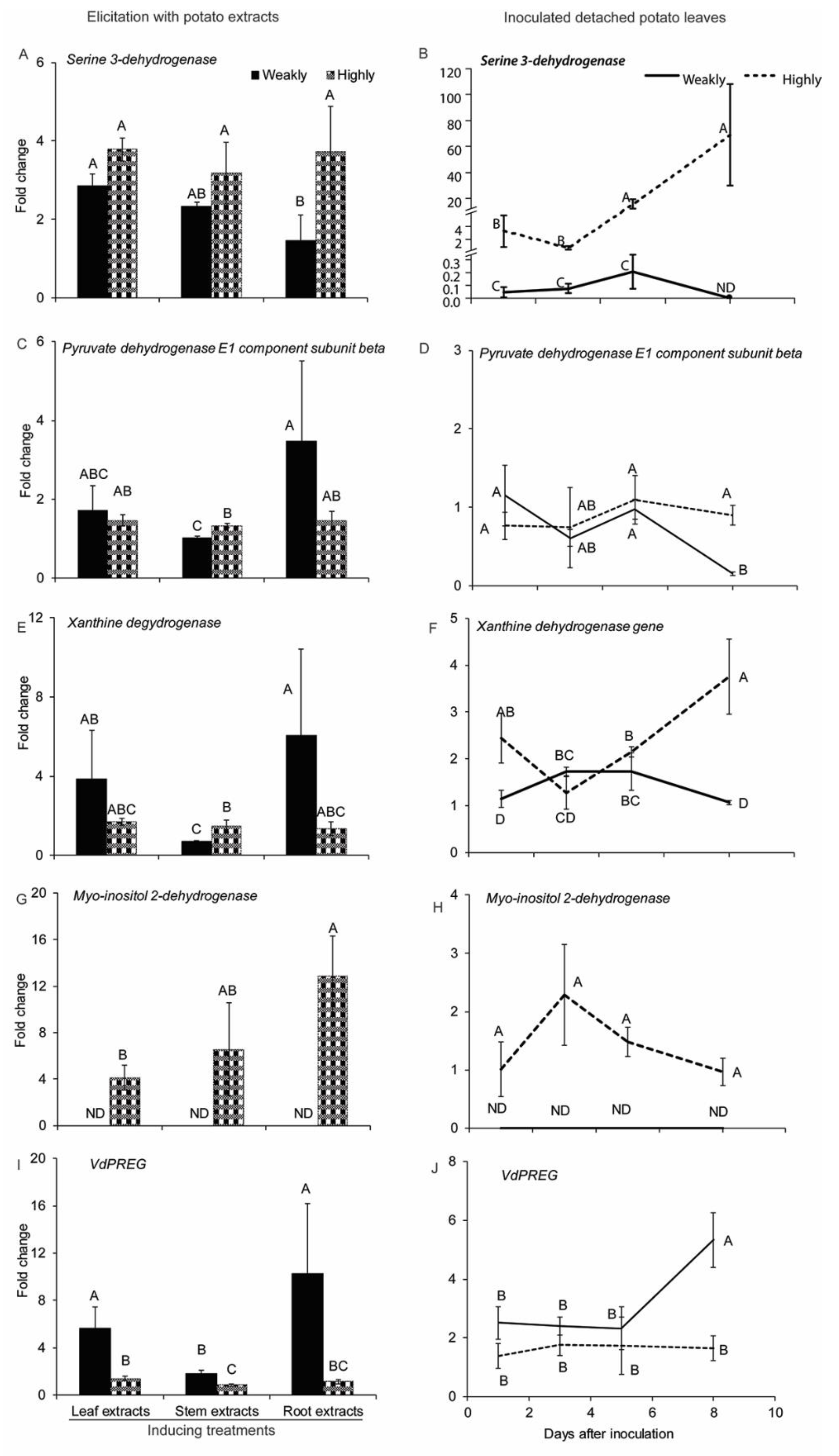
2.2.4. Genes Related to Cellular Metabolism
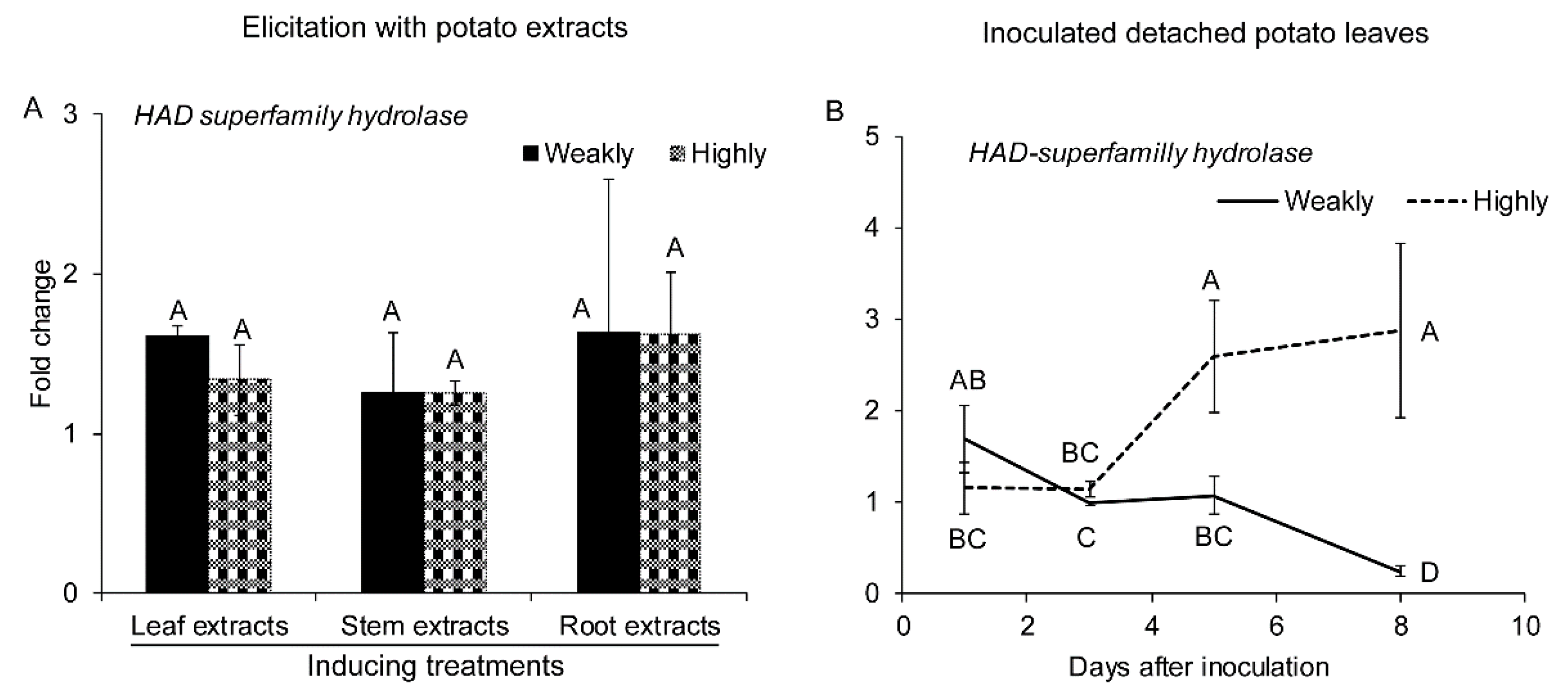
2.2.5. Genes Related to Detoxification
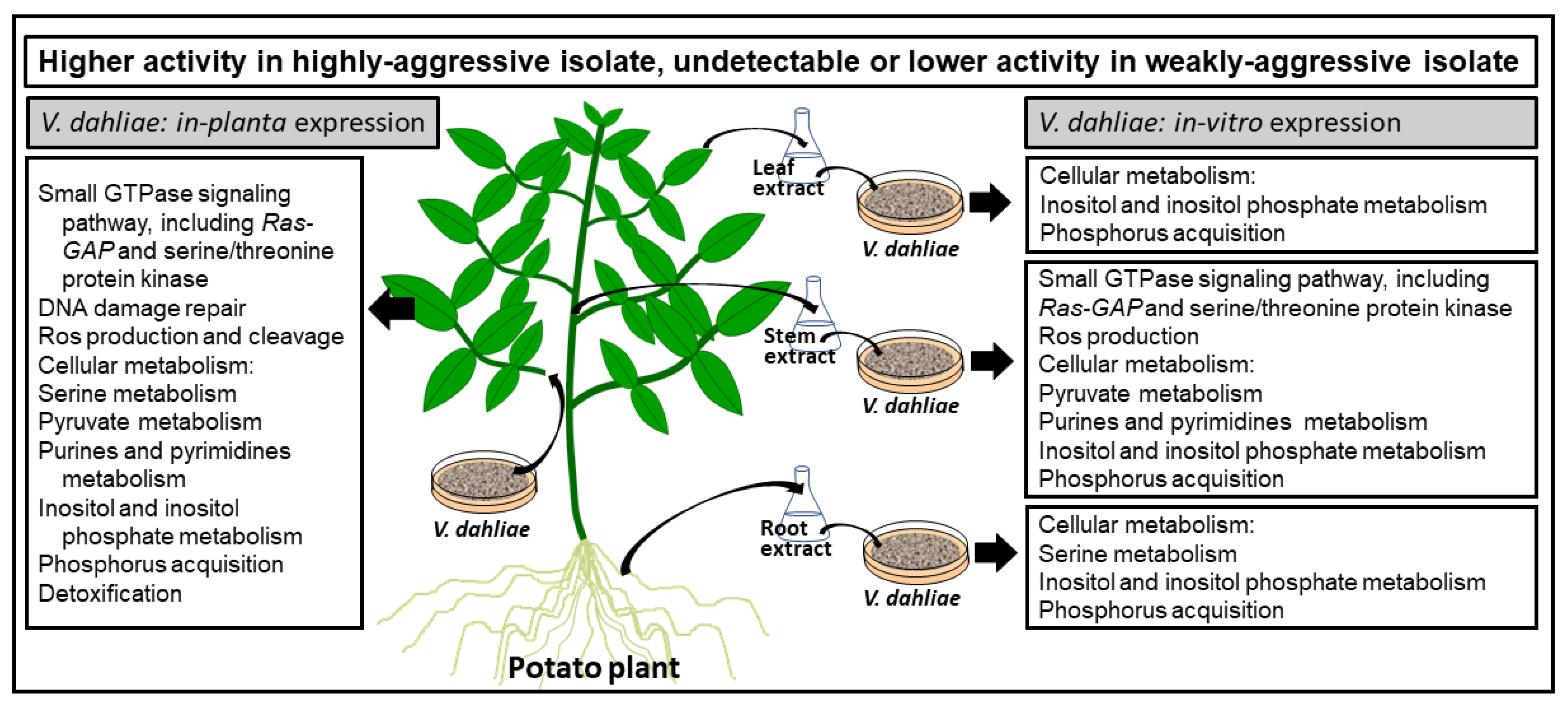
3. Discussion
4. Materials and Methods
4.1. V. dahliae Isolates
4.2. Pathogenicity Tests of V. dahliae Isolates
4.3. Gene Selection and Primers’ Design
4.4. Elicitation and Inoculation Methods
4.5. RNA Extraction and RT-qPCR
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pegg, G.F.; Brady, B.L. Verticillium Wilts; CABI: Wallingford, UK, 2002. [Google Scholar]
- Krikun, J.; Orion, D. Verticillium wilt of potato: Importance and control. Phytoparasitica 1979, 7, 107–116. [Google Scholar] [CrossRef]
- Molina, O.I.; Tenuta, M.; El Hadrami, A.; Buckley, K.; Cavers, C.; Daayf, F. Potato Early Dying and Yield Responses to Compost, Green Manures, Seed Meal and Chemical Treatments. Am. J. Potato Res. 2014, 91, 414–428. [Google Scholar] [CrossRef]
- Rowe, R.C.; Powelson, M.L. Potato Early Dying: Management Challenges in a Changing Production Environment. Plant Dis. 2002, 86, 1184–1193. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nnodu, E.C.; Harrison, M.D. The relationship betweenVerticillium albo-atrum inoculum density and potato yield. Am. J. Potato Res. 1979, 56, 11–25. [Google Scholar] [CrossRef]
- Wilhelm, S. Longevity of the Verticillium wilt fungus in the laboratory and field. Phytopathology 1955, 45, 180–181. [Google Scholar]
- Olsson, S.; Nordbring-Hertz, B. Microsclerotial germination of Verticillium dahliae as affected by rape rhizosphere. FEMS Microbiol. Ecol. 1985, 1, 293–299. [Google Scholar] [CrossRef]
- Fradin, E.F.; Thomma, B.P.H.J. Physiology and molecular aspects of Verticillium wilt diseases caused by V. dahliae and V. albo-atrum. Mol. Plant Pathol. 2006, 7, 71–86. [Google Scholar] [CrossRef]
- Bowers, J. Infection and Colonization of Potato Roots by Verticillium dahliae as Affected by Pratylenchus penetrans and P. crenatus. Phytopathology 1996, 86, 614–621. [Google Scholar] [CrossRef]
- Daayf, F. Verticillium wilts in crop plants: Pathogen invasion and host defence responses. Can. J. Plant Pathol. 2014, 37, 8–20. [Google Scholar] [CrossRef]
- Smit, F.; Dubery, I.A. Cell wall reinforcement in cotton hypocotyls in response to a Verticillium dahliae elicitor. Phytochemistry 1997, 44, 811–815. [Google Scholar] [CrossRef]
- Griffiths, D.A. The development of lignitubers in roots after infection by Verticillium dahliae Kleb. Can. J. Microbiol. 1971, 17, 441–444. [Google Scholar] [CrossRef] [PubMed]
- Gayoso, C.; Pomar, F.; Novo-Uzal, E.; Merino, F.; de Ilárduya, Ó.M. The Ve-mediated resistance response of the tomato to Verticillium dahliae involves H 2 O 2, peroxidase and lignins and drives PALgene expression. BMC Plant Biol. 2010, 10, 232. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Debode, J.; Clewes, E.; De Backer, G.; Höfte, M. Lignin is involved in the reduction of Verticillium dahliae var. longisporum inoculum in soil by crop residue incorporation. Soil Biol. Biochem. 2005, 37, 301–309. [Google Scholar] [CrossRef]
- Daayf, F.; Nicole, M.; Boher, B.; Pando, A.; Geiger, J.P. Early vascular defense reactions of cotton roots infected with a defoliating mutant strain of Verticillium dahliae. Eur. J. Plant Pathol. 1997, 103, 125–136. [Google Scholar] [CrossRef]
- Mace, M.; Stipanovic, R.; Bell, A. Relation between sensitivity to terpenoid phytoalexins and virulence to cotton of Verticillium dahliae strains. Pestic. Biochem. Physiol. 1990, 36, 79–82. [Google Scholar] [CrossRef]
- Mace, M.E.; Stipanovic, R.D.; Bell, A.A. Histochemical localization of desoxyhemigossypol, a phytoalexin in Verticillium dahliae -infected cotton stems. New Phytol. 1989, 111, 229–232. [Google Scholar] [CrossRef] [PubMed]
- Robb, J.; Brisson, J.D.; Busch, L.; Lu, B.C. Ultrastructure of wilt syndrome caused by Verticillium dahliae. VII. Correlated light and transmission electron microscope identification of vessel coatings and tyloses. Can. J. Bot. 1979, 57, 822–834. [Google Scholar] [CrossRef]
- Jiménez-Díaz, R.M.; Cirulli, M.; Bubici, G.; Jiménez-Gasco, M.D.M.; Antoniou, P.P.; Tjamos, E.C. Verticillium Wilt, A Major Threat to Olive Production: Current Status and Future Prospects for its Management. Plant Dis. 2012, 96, 304–329. [Google Scholar] [CrossRef] [Green Version]
- Davis, J.R. Influence of Continuous Cropping of Several Potato Clones on the Epidemiology of Verticillium Wilt of Potato. Phytopathology 1994, 84, 207–214. [Google Scholar] [CrossRef]
- El Hadrami, A.; Islam, M.; Adam, L.R.; Daayf, F. A cupin domain-containing protein with a quercetinase activity (VdQase) regulates Verticillium dahliae’s pathogenicity and contributes to counteracting host defenses. Front. Plant Sci. 2015, 6, 440. [Google Scholar] [CrossRef] [Green Version]
- El-Bebany, A.F.; Rampitsch, C.; Daayf, F. Proteomic analysis of the phytopathogenic soilborne fungus Verticillium dahliae reveals differential protein expression in isolates that differ in aggressiveness. Proteomics 2010, 10, 289–303. [Google Scholar] [CrossRef] [PubMed]
- El-Bebany, A.F.; Henriquez, M.A.; Badawi, M.; Adam, L.R.; El Hadrami, A.; Daayf, F. Induction of putative pathogenicity-related genes in Verticillium dahliae in response to elicitation with potato root extracts. Env.. Exp. Bot. 2011, 72, 251–257. [Google Scholar] [CrossRef]
- Zhu, X.; Soliman, A.; Islam, M.R.; Adam, L.R.; Daayf, F. Verticillium dahliae’s Isochorismatase hydrolase is a virulence factor that contributes to interference with potato’s Salicylate and Jasmonate defense signaling. Front. Plant Sci. 2017, 8, 399. [Google Scholar] [CrossRef] [Green Version]
- Santos, B.; Martín-Cuadrado, A.B.; De Aldana, C.R.V.; Del Rey, F.; Pérez, P. Rho4 GTPase Is Involved in Secretion of Glucanases during Fission Yeast Cytokinesis. Eukaryot. Cell 2005, 4, 1639–1645. [Google Scholar] [CrossRef] [Green Version]
- Dekker, N.; Speijer, D.; Grün, C.H.; Berg, M.V.D.; De Haan, A.; Hochstenbach, F. Role of the α-Glucanase Agn1p in Fission-Yeast Cell Separation. Mol. Biol. Cell 2004, 15, 3903–3914. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- García, I.; Jiménez, D.; Martín, V.; Durán, A.; Sánchez, Y. The α-glucanase Agn1p is required for cell separation in Schizosaccharomyces pombe. Biol. Cell 2005, 97, 569–576. [Google Scholar] [CrossRef]
- Villalobos-Duno, H.; San-Blas, G.; Paulinkevicius, M.; Sánchez-Martín, Y.; Nino-Vega, G. Biochemical Characterization of Paracoccidioides brasiliensis α-1,3-Glucanase Agn1p, and Its Functionality by Heterologous Expression in Schizosaccharomyces pombe. PLoS ONE 2013, 8, e66853. [Google Scholar] [CrossRef] [Green Version]
- Hercyk, B.S.; Onwubiko, U.N.; Das, M.E. Coordinating septum formation and the actomyosin ring during cytokinesis in Schizosaccharomyces pombe. Mol. Microbiol. 2019, 112, 1645–1657. [Google Scholar] [CrossRef] [Green Version]
- Tsui, C.; Raguraj, A.; Pickart, C.M. Ubiquitin binding site of the ubiquitin E2 variant (UEV) protein Mms2 is required for DNA damage tolerance in the yeast RAD6 pathway. J. Biol. Chem. 2005, 280, 19829–19835. [Google Scholar] [CrossRef] [Green Version]
- Tchigvintsev, A.; Singer, A.; Brown, G.; Flick, R.; Evdokimova, E.; Tan, K.; Gonzalez, C.F.; Savchenko, A.; Yakunin, A.F. Biochemical and Structural Studies of Uncharacterized Protein PA0743 from Pseudomonas aeruginosa Revealed NAD+-dependent l-Serine Dehydrogenase. J. Biol. Chem. 2012, 287, 1874–1883. [Google Scholar] [CrossRef] [Green Version]
- Tomazett, M.V.; Baeza, L.C.; Paccez, J.D.; Parente-Rocha, J.A.; Ribeiro-Dias, F.; Soares, C.M.D.A. Identification and characterization of Paracoccidioides lutzii proteins interacting with macrophages. Microbes Infect. 2019, 21, 401–411. [Google Scholar] [CrossRef]
- Galbraith, M.P.; Feng, S.F.; Borneman, J.; Triplett, E.W.; de Bruijn, F.J.; Rossbachl, S. A functional myo-inositol catabolism pathway is essential for rhizopine utilization by Sinorhizobium meliloti. Microbiology 1998, 144, 2915–2924. [Google Scholar] [CrossRef] [Green Version]
- Berman, T.; Magasanik, B. The Pathway of myo-Inositol Degradation in Aerobacter aerogenes Dehydrogenation and Dehydration. J. Biol. Chem. 1966, 241, 800–806. [Google Scholar] [CrossRef]
- Jiang, G.; Krishnan, A.H.; Kim, Y.W.; Wacek, T.J.; Krishnan, H.B. A functional myo-inositol dehydrogenase gene is required for efficient nitrogen fixation and competitiveness of Sinorhizobium fredii USDA191 to nodulate soybean (Glycine max [L.] Merr.). J. Bacteriol. 2001, 183, 2595–2604. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boguski, M.S.; McCormick, F. Proteins regulating Ras and its relatives. Nat. Cell Biol. 1993, 366, 643–654. [Google Scholar] [CrossRef]
- Scheffzek, K.; Ahmadian, M.R.; Kabsch, W.; Wiesmüller, L.; Lautwein, A.; Schmitz, F.; Wittinghofer, A. The Ras-RasGAP complex: Structural basis for GTPase activation and its loss in oncogenic Ras mutants. Science 1997, 277, 333–339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dickman, M.B.; Yarden, O. Serine/threonine protein kinases and phosphatases in filamentious fungi. Fungal Genet. Biol. 1999, 26, 99–117. [Google Scholar] [CrossRef] [Green Version]
- Manser, E.; Leung, T.; Salihuddin, H.; Zhao, Z.-S.; Lim, L. A brain serine/threonine protein kinase activated by Cdc42 and Rac1. Nat. Cell Biol. 1994, 367, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Park, G.; Xue, C.; Zhao, X.; Kim, Y.; Orbach, M.; Xu, J.-R. Multiple Upstream Signals Converge on the Adaptor Protein Mst50 in Magnaporthe grisea. Plant Cell 2006, 18, 2822–2835. [Google Scholar] [CrossRef] [Green Version]
- Ding, S.; Zhou, X.; Zhao, X.; Xu, J.-R. The PMK1 MAP Kinase Pathway and Infection-Related Morphogenesis. In Advances in Genetics, Genomics and Control of Rice Blast Disease; Metzler, J.B., Ed.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 13–21. [Google Scholar]
- Bluhm, B.H.; Zhao, X.; Flaherty, J.E.; Xu, J.-R.; Dunkle, L.D. RAS2 Regulates Growth and Pathogenesis in Fusarium graminearum. Mol. Plant-Microbe Interact. 2007, 20, 627–636. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muñoz, M.J.; Bejarano, E.R.; Daga, R.R.; Jimenez, J. The identification of Wos2, a p23 homologue that interacts with Wee1 and Cdc2 in the mitotic control of fission yeasts. Genetics 1999, 153, 1561–1572. [Google Scholar] [PubMed]
- Seifert, F.; Ciszak, E.; Korotchkina, L.; Golbik, R.; Spinka, M.; Dominiak, P.; Sidhu, S.; Brauer, J.; Patel, M.S.; Tittmann, K. Phosphorylation of serine 264 impedes active site accessibility in the E1 component of the human pyruvate dehydrogenase multienzyme complex. Biochemistry 2007, 46, 6277–6287. [Google Scholar] [CrossRef]
- Lessard, I.; Perham, R.N. Expression in Escherichia coli of genes encoding the E1 alpha and E1 beta subunits of the pyruvate dehydrogenase complex of Bacillus stearothermophilus and assembly of a functional E1 component (alpha 2 beta 2) in vitro. J. Biol. Chem. 1994, 269, 10378–10383. [Google Scholar] [CrossRef]
- Dallo, S.F.; Kannan, T.R.; Blaylock, M.W.; Baseman, J.B. Elongation factor Tu and E1 β subunit of pyruvate dehydrogenase complex act as fibronectin binding proteins in Mycoplasma pneumoniae. Mol. Microbiol. 2002, 4, 1041–1051. [Google Scholar] [CrossRef] [PubMed]
- Kato, M.; Wynn, M.R.; Chuang, L.J.; Tso, C.S.; Machius, M.; Li, J.; Chuang, T.D. Structural basis for inactivation of the human pyruvate dehydrogenase complex by phosphorylation: Role of disordered phosphorylation loops. Structure 2008, 16, 1849–1859. [Google Scholar] [CrossRef] [Green Version]
- Bittner, F.; Oreb, M.; Mendel, R.R. ABA3 is a molybdenum cofactor sulfurase required for activation of aldehyde oxidase and xanthine dehydrogenase in Arabidopsis thaliana. J. Biol. Chem. 2001, 276, 40381–40384. [Google Scholar] [CrossRef] [Green Version]
- Engerson, T.D.; McKelvey, T.G.; Rhyne, D.B.; Boggio, E.B.; Snyder, S.J.; Jones, H.P. Conversion of xanthine dehydrogenase to oxidase in ischemic rat tissues. J. Clin. Investig. 1987, 6, 1564–1570. [Google Scholar] [CrossRef]
- Enroth, C.; Eger, B.T.; Okamoto, K.; Nishino, T.; Nishion, T.; Pai, E.F. Crystal structures of bovine milk xanthine dehydrogenase and xanthine oxidase: Structure-based mechanism of conversion. Proc. Nat. Acad. Sci. USA 2000, 20, 10723–10728. [Google Scholar] [CrossRef] [Green Version]
- Hille, R.; Nishino, T. Xanthine oxidase and xanthine dehydrogenase. FASEB J. 1995, 9, 995–1003. [Google Scholar] [CrossRef] [PubMed]
- Khobragade, C.N.; Bodade, R.G.; Shinde, M.S.; Jaju, D.R.; Bhosle, R.B.; Dawane, B.S. Microbial and xanthine dehydrogenase inhibitory activity of some flavones. J. Enzym. Inhib. Med. Chem. 2008, 23, 341–346. [Google Scholar] [CrossRef] [Green Version]
- Saxton, A. A macro for converting mean separation output to letter groupings in Proc Mixed. In Proceedings of the 23rd SAS Users Group International, Nashville, TN, USA, 22–25 March 1998; pp. 1243–1246. [Google Scholar]
- Armenteros, J.J.A.; Tsirigos, K.D.; Sønderby, C.K.; Petersen, T.N.; Winther, O.; Brunak, S.; Von Heijne, G.; Nielsen, H. SignalP 5.0 improves signal peptide predictions using deep neural networks. Nat. Biotechnol. 2019, 37, 420–423. [Google Scholar] [CrossRef]
- Sperschneider, J.; Dodds, P.N.; Gardiner, D.M.; Singh, K.B.; Taylor, J.M. Improved prediction of fungal effector proteins from secretomes with EffectorP 2.0. Mol. Plant Pathol. 2018, 19, 2094–2110. [Google Scholar] [CrossRef] [Green Version]
- Sperschneider, J.; Gardiner, D.M.; Dodds, P.N.; Tini, F.; Covarelli, L.; Singh, K.B.; Manners, J.M.; Taylor, J.M. EffectorP: Predicting fungal effector proteins from secretomes using machine learning. New Phytol. 2016, 210, 743–761. [Google Scholar] [CrossRef] [Green Version]
- Davies, A.; Masson, J.-Y.; McIlwraith, M.J.; Stasiak, A.Z.; Stasiak, A.; Venkitaraman, A.R.; West, S.C. Role of BRCA2 in Control of the RAD51 Recombination and DNA Repair Protein. Mol. Cell 2001, 7, 273–282. [Google Scholar] [CrossRef]
- Ulrich, H.D.; Jentsch, S. Two RING finger proteins mediate cooperation between ubiquitin-conjugating enzymes in DNA repair. EMBO J. 2000, 19, 3388–3397. [Google Scholar] [CrossRef] [Green Version]
- Hofmann, R.M.; Pickart, C.M. Noncanonical MMS2-Encoded Ubiquitin-Conjugating Enzyme Functions in Assembly of Novel Polyubiquitin Chains for DNA Repair. Cell 1999, 96, 645–653. [Google Scholar] [CrossRef] [Green Version]
- Nakamura, J.; A Swenberg, J. Endogenous apurinic/apyrimidinic sites in genomic DNA of mammalian tissues. Cancer Res. 1999, 59, 2522–2526. [Google Scholar] [PubMed]
- Aburatani, H.; Hippo, Y.; Ishida, T.; Takashima, R.; Matsuba, C.; Kodama, T.; Takao, M.; Yasui, A.; Yamamoto, K.; Asano, M. Cloning and characterization of mammalian 8-hydroxyguanine-specific DNA glycosylase/apurinic, apyrimidinic lyase, a functional mutM homologue. Cancer Res. 1997, 57, 2151–2156. [Google Scholar] [PubMed]
- Peleg, Y.; Addison, R.; Aramayo, R.; Metzenberg, R.L. Translocation ofNeurospora crassaTranscription Factor NUC-1 into the Nucleus Is Induced by Phosphorus Limitation. Fungal Genet. Biol. 1996, 20, 185–191. [Google Scholar] [CrossRef]
- Kang, S. Functional domains of the transcriptional activator NUC-1 in Neurospora crassa. Gene 1993, 130, 259–264. [Google Scholar] [CrossRef]
- Burroughs, A.M.; Allen, K.N.; Dunaway-Mariano, D.; Aravind, L. Evolutionary genomics of the HAD superfamily: Understanding the structural adaptations and catalytic diversity in a superfamily of phosphoesterases and allied enzymes. J. Mol. Biol. 2006, 361, 1003–1034. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koonin, E.V.; Tatusov, R.L. Computer analysis of bacterial haloacid dehalogenases defines a large superfamily of hydrolases with diverse specificity: Application of an iterative approach to database search. J. Mol. Biol. 1994, 244, 125–132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuznetsova, E.; Nocek, B.; Brown, G.; Makarova, K.S.; Flick, R.; Wolf, Y.I.; Khusnutdinova, A.; Evdokimova, E.; Jin, K.; Tan, K.; et al. Functional diversity of haloacid dehalogenase superfamily phosphatases from Saccharomyces cerevisiae biochemical, structural, and evolutionary insights. J. Biol. Chem. 2015, 290, 18678–18698. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuznetsova, E.; Proudfoot, M.; Gonzalez, C.F.; Brown, G.; Omelchenko, M.V.; Borozan, I.; Carmel, L.; Wolf, Y.I.; Mori, H.; Savchenko, A.V.; et al. Genome-wide analysis of substrate specificities of the Escherichia coli haloacid dehalogenase-like phosphatase family. J. Biol. Chem. 2006, 281, 36149–36161. [Google Scholar] [CrossRef] [Green Version]
- Nantel, A.; Dignard, D.; Bachewich, C.; Harcus, D.; Marcil, A.; Bouin, A.-P.; Sensen, C.W.; Hogues, H.; Hoog, M.V.H.; Gordon, P.; et al. Transcription Profiling ofCandida albicansCells Undergoing the Yeast-to-Hyphal Transition. Mol. Biol. Cell 2002, 13, 3452–3465. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cvrcková, F.; Nasmyth, K. Yeast G1 cyclins CLN1 and CLN2 and a GAP-like protein have a role in bud formation. EMBO J. 1993, 12, 5277–5286. [Google Scholar] [CrossRef]
- Harispe, L.; Portela, C.; Scazzocchio, C.; Peñalva, M.A.; Gorfinkiel, L. Ras GTPase-Activating Protein Regulation of Actin Cytoskeleton and Hyphal Polarity in Aspergillus nidulans. Eukaryot. Cell 2007, 7, 141–153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tian, H.; Zhou, L.; Guo, W.; Wang, X. Small GTPase Rac1 and its interaction partner Cla4 regulate polarized growth and pathogenicity in Verticillium dahliae. Fungal Genet. Biol. 2015, 74, 21–31. [Google Scholar] [CrossRef]
- Norton, T.S.; Fortwendel, J.R. Control of Ras-mediated signaling in Aspergillus fumigatus. Mycopathol. 2014, 178, 325–330. [Google Scholar] [CrossRef] [Green Version]
- Fortwendel, J.R.; Fuller, K.K.; Stephens, T.J.; Bacon, W.C.; Askew, D.S.; Rhodes, J.C. Aspergillus fumigatus RasA Regulates Asexual Development and Cell Wall Integrity. Eukaryot. Cell 2008, 7, 1530–1539. [Google Scholar] [CrossRef] [Green Version]
- Tzima, A.; Paplomatas, E.J.; Rauyaree, P.; Kang, S. Roles of the catalytic subunit of cAMP-dependent protein kinase A in virulence and development of the soilborne plant pathogen Verticillium dahliae. Fungal Genet. Biol. 2010, 47, 406–415. [Google Scholar] [CrossRef]
- Chen, Y.; Zhai, S.; Sun, Y.; Li, M.; Dong, Y.; Wang, X.; Zhang, H.; Zheng, X.; Wang, P.; Zhang, Z. MoTup1 is required for growth, conidiogenesis and pathogenicity of Magnaporthe oryzae. Mol. Plant Pathol. 2015, 16, 799–810. [Google Scholar] [CrossRef] [PubMed]
- Eboigbe, L.; Tzima, A.K.; Paplomatas, E.J.; Typas, M.A. Crosstalk between the cAMP-PKA pathway and the β-1, 6-endoglucanase in Verticillium dahliae. Phytopathol. Mediterr. 2019, 58, 587–595. [Google Scholar]
- Sharma, P.; Jha, A.B.; Dubey, R.S.; Pessarakli, M. Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J. Bot. 2012. [Google Scholar] [CrossRef] [Green Version]
- Broomfield, S.; Chow, B.L.; Xiao, W. MMS2, encoding a ubiquitin-conjugating-enzyme-like protein, is a member of the yeast error-free postreplication repair pathway. Proc. Natl. Acad. Sci. USA 1998, 95, 5678–5683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bailly, V.R.; Verly, W.G. AP endonucleases and AP lyases. Nucleic Acids Res. 1989, 17, 3617. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bailly, V.; Lamb, J.; Sung, P.; Prakash, S.; Prakash, L. Specific complex formation between yeast RAD6 and RAD18 proteins: A potential mechanism for targeting RAD6 ubiquitin-conjugating activity to DNA damage sites. Genes Dev. 1994, 8, 811–820. [Google Scholar] [CrossRef] [Green Version]
- Prakash, S.; Sung, P.; Prakash, L. DNA repair genes and proteins of Saccharomyces cerevisiae. Annu. Rev. Genet. 1993, 27, 33–70. [Google Scholar] [CrossRef]
- Morrison, A.; Christensen, R.B.; Alley, J.; Beck, A.K.; Bernstine, E.G.; Lemontt, J.F.; Lawrence, C.W. REV3, a Saccharomyces cerevisiae gene whose function is required for induced mutagenesis, is predicted to encode a nonessential DNA polymerase. J. Bacteriol. 1989, 171, 5659–5667. [Google Scholar] [CrossRef] [Green Version]
- Jentsch, S.; McGrath, J.P.; Varshavsky, A. The yeast DNA repair gene RAD6 encodes a ubiquitin-conjugating enzyme. Nat. Cell Biol. 1987, 329, 131–134. [Google Scholar] [CrossRef]
- Reynolds, P.; Weber, S.; Prakash, L. RAD6 gene of Saccharomyces cerevisiae encodes a protein containing a tract of 13 consecutive aspartates. Proc. Natl. Acad. Sci. USA 1985, 82, 168–172. [Google Scholar] [CrossRef] [Green Version]
- O’Rourke, E.J.; Chevalier, C.; Pinto, A.V.; Thiberge, J.M.; Ielpi, L.; Labigne, A.; Radicella, J.P. Pathogen DNA as target for host-generated oxidative stress: Role for repair of bacterial DNA damage in Helicobacter pylori colonization. Proc. Natl. Acad. Sci. USA 2003, 100, 2789–2794. [Google Scholar] [CrossRef] [Green Version]
- Da Silva, R.P.; Matsumoto, M.T.; Braz, J.D.; Voltan, A.R.; de Oliveira, H.C.; Soares, C.P.; Giannini, M.J.S.M. Differential gene expression analysis of Paracoccidioides brasiliensis during keratinocyte infection. J. Med. Microbiol. 2011, 60, 269–280. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Mogg, C.; Walkowiak, S.; Joshi, M.; Subramaniam, R. Characterization of NADPH oxidase genes NoxA and NoxB in Fusarium graminearum. Can. J. Plant Pathol. 2014, 36, 12–21. [Google Scholar] [CrossRef]
- Malagnac, F.; Lalucque, H.; Lepère, G.; Silar, P. Two NADPH oxidase isoforms are required for sexual reproduction and ascospore germination in the filamentous fungus Podospora anserina. Fungal Genet. Biol. 2004, 41, 982–997. [Google Scholar] [CrossRef]
- Lara-Ortíz, T.; Riveros-Rosas, H.; Aguirre, J. Reactive oxygen species generated by microbial NADPH oxidase NoxA regulate sexual development in Aspergillus nidulans. Mol. Microbiol. 2003, 50, 1241–1255. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, Y.-L.; Zhou, T.-T.; Guo, H.-S. Hyphopodium-Specific VdNoxB/VdPls1-Dependent ROS-Ca2+ Signaling Is Required for Plant Infection by Verticillium dahliae. PLoS Pathog. 2016, 12, e1005793. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heller, J.; Tudzynski, P. Reactive Oxygen Species in Phytopathogenic Fungi: Signaling, Development, and Disease. Annu. Rev. Phytopathol. 2011, 49, 369–390. [Google Scholar] [CrossRef] [PubMed]
- Bedard, K.; Lardy, B.; Krause, K.-H. NOX family NADPH oxidases: Not just in mammals. Biochimie 2007, 89, 1107–1112. [Google Scholar] [CrossRef]
- Bazil, J.N.; Pannala, V.R.; Dash, R.K.; Beard, D.A. Determining the origins of superoxide and hydrogen peroxide in the mammalian NADH:ubiquinone oxidoreductase. Free. Radic. Biol. Med. 2014, 77, 121–129. [Google Scholar] [CrossRef] [Green Version]
- Kussmaul, L.; Hirst, J. The mechanism of superoxide production by NADH:ubiquinone oxidoreductase (complex I) from bovine heart mitochondria. Proc. Natl. Acad. Sci. USA 2006, 103, 7607–7612. [Google Scholar] [CrossRef] [Green Version]
- Brand, M.D.; Affourtit, C.; Esteves, T.C.; Green, K.; Lambert, A.J.; Miwa, S.; Pakay, J.L.; Parker, N. Mitochondrial superoxide: Production, biological effects, and activation of uncoupling proteins. Free Radic. Biol. Med. 2004, 37, 755–767. [Google Scholar] [CrossRef] [PubMed]
- Turrens, J.F. Mitochondrial formation of reactive oxygen species. J. Physiol. 2003, 552, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Zhou, H.J.; Zhang, H.; Huang, Y.; Hinojosa-Kirschenbaum, F.; Fan, P.; Yao, L.; Belardinelli, L.; Tellides, G.; Giordano, F.J.; et al. Thioredoxin-2 inhibits mitochondrial ROS generation and ASK1 activity to maintain cardiac function. Circulation 2015, 131, 1082. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hong, S.M.; Lim, H.W.; Kim, I.H.; Kim, K.; Park, E.H.; Lim, C.J. Stress-dependent regulation of the gene encoding thioredoxin reductase from the fission yeast. FEMS Microbiol. Lett. 2006, 234, 379–385. [Google Scholar] [CrossRef]
- Missall, T.A.; Lodge, J.K. Thioredoxin Reductase Is Essential for Viability in the Fungal Pathogen Cryptococcus neoformans. Eukaryot. Cell 2005, 4, 487–489. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salmon, T.B.; Evert, B.A.; Song, B.; Doetsch, P.W. Biological consequences of oxidative stress-induced DNA damage in Saccharomyces cerevisiae. Nucleic Acids Res. 2004, 32, 3712–3723. [Google Scholar] [CrossRef] [Green Version]
- Uziel, O.; Borovok, I.; Schreiber, R.; Cohen, G.; Aharonowitz, Y. Transcriptional Regulation of the Staphylococcus aureus Thioredoxin and Thioredoxin Reductase Genes in Response to Oxygen and Disulfide Stress. J. Bacteriol. 2004, 186, 326–334. [Google Scholar] [CrossRef] [Green Version]
- Enjalbert, B.; Nantel, A.; Whiteway, M. Stress-induced Gene Expression in Candida albicans: Absence of a General Stress Response. Mol. Biol. Cell 2003, 14, 1460–1467. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fradin, C.; Kretschmar, M.; Nichterlein, T.; Gaillardin, C.; D’Enfert, C.; Hube, B. Stage-specific gene expression of Candida albicans in human blood. Mol. Microbiol. 2003, 47, 1523–1543. [Google Scholar] [CrossRef]
- Godon, C.; Lagniel, G.; Lee, J.; Buhler, J.-M.; Kieffer, S.; Perrot, M.; Boucherie, H.; Toledano, M.B.; Labarre, J. The H2O2 Stimulon in Saccharomyces cerevisiae. J. Biol. Chem. 1998, 273, 22480–22489. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamazawa, R.; Nakajima, Y.; Mushiake, K.; Yoshimoto, T.; Ito, K. Crystal structure of serine dehydrogenase from Escherichia coli: Important role of the C-terminal region for closed-complex formation. J. Biochem. 2011, 149, 701–712. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiong, D.; Wang, Y.; Tian, L.; Tian, C. MADS-Box Transcription Factor VdMcm1 Regulates Conidiation, Microsclerotia Formation, Pathogenicity, and Secondary Metabolism of Verticillium dahliae. Front. Microbiol. 2016, 7, 1192. [Google Scholar] [CrossRef]
- Wang, H.; Chen, B.; Tian, J.; Kong, Z. Verticillium dahliae VdBre1 is required for cotton infection by modulating lipid metabolism and secondary metabolites. Environ. Microbiol. 2020. [Google Scholar] [CrossRef]
- Tang, C.; Li, T.; Klosterman, S.J.; Tian, C.; Wang, Y. The bZIP transcription factor VdAtf1 regulates virulence by mediating nitrogen metabolism in Verticillium dahliae. New Phytol. 2020, 226, 1461–1479. [Google Scholar] [CrossRef]
- Korte-Berwanger, M.; Sakinc, T.; Kline, K.; Nielsen, H.V.; Hultgren, S.; Gatermann, S.G. Significance of the D-serine-deaminase and D-serine metabolism of Staphylococcus saprophyticus for virulence. Infect. Immun. 2013, 81, 4525–4533. [Google Scholar] [CrossRef] [Green Version]
- Venugopal, A.; Bryk, R.; Shi, S.; Rhee, K.; Rath, P.; Schnappinger, D.; Ehrt, S.; Nathan, C. Virulence of Mycobacterium tuberculosis Depends on Lipoamide Dehydrogenase, a Member of Three Multienzyme Complexes. Cell Host Microbe 2011, 9, 21–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kong, L.; Cai, Y.; Huang, W.; Cheng, C.H.; Tan, R. Inhibition of xanthine oxidase by some Chinese medicinal plants used to treat gout. J. Ethnopharmacol. 2000, 73, 199–207. [Google Scholar] [CrossRef]
- Nagao, A.; Seki, M.; Kobayashi, H. Inhibition of Xanthine Oxidase by Flavonoids. Biosci. Biotechnol. Biochem. 1999, 63, 1787–1790. [Google Scholar] [CrossRef]
- Gonzalez, A.G.; Bazzocchi, I.L.; Moujir, L.; Ravelo, Á.G.; Correa, M.D.; Gupta, M.P. Xanthine oxidase inhibitory activity of some Panamanian plants from Celastraceae and Lamiaceae. J. Ethnopharmacol. 1995, 46, 25–29. [Google Scholar] [CrossRef]
- Hayashi, T.; Sawa, K.; Kawasaki, M.; Arisawa, M.; Shimizu, M.; Morita, N. Inhibition of cow’s milk xanthine oxidase by flavonoids. J. Nat. Prod. 1988, 51, 345–348. [Google Scholar] [CrossRef]
- Kang, S.; Metzenberg, R.L. Molecular analysis of nuc-1+, a gene controlling phosphorus acquisition in Neurospora crassa. Mol. Cell. Biol. 1990, 10, 5839–5848. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kang, S.; Metzenberg, R.L. Insertional mutagenesis in Neurospora crassa: Cloning and molecular analysis of the preg+ gene controlling the activity of the transcriptional activator NUC-1. Genetics 1993, 133, 193–202. [Google Scholar] [CrossRef]
- Deng, S.; Wang, C.-Y.; Zhang, X.; Wang, Q.; Lin, L. VdNUC-2, the Key Regulator of Phosphate Responsive Signaling Pathway, Is Required for Verticillium dahliae Infection. PLoS ONE 2015, 10, e0145190. [Google Scholar] [CrossRef]
- Kretschmer, M.; Reiner, E.; Hu, G.; Tam, N.; Oliveira, D.L.; Caza, M.; Yeon, J.H.; Kim, J.; Kastrup, C.J.; Jung, W.H.; et al. Defects in Phosphate Acquisition and Storage Influence Virulence of Cryptococcus neoformans. Infect. Immun. 2014, 82, 2697–2712. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boyce, K.J.; Kretschmer, M.; Kronstad, J.W. The vtc4 Gene Influences Polyphosphate Storage, Morphogenesis, and Virulence in the Maize Pathogen Ustilago maydis. Eukaryot. Cell 2006, 5, 1399–1409. [Google Scholar] [CrossRef] [Green Version]
- Jung, W.-H.; Son, Y.-E.; Oh, S.-H.; Fu, C.; Kim, H.S.; Kwak, J.-H.; Cardenas, M.E.; Heitman, J.; Park, H.-S. Had1 Is Required for Cell Wall Integrity and Fungal Virulence inCryptococcus neoformans. G3 Genes|Genomes|Genet. 2018, 8, 643–652. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lattanzio, V.; Lattanzio, V.M.; Cardinali, A. Role of phenolics in the resistance mechanisms of plants against fungal pathogens and insects. Phytochem. Adv. Res. 2006, 661, 23–67. [Google Scholar]
- Wittstock, U.; Gershenzon, J. Constitutive plant toxins and their role in defense against herbivores and pathogens. Curr. Opin. Plant Biol. 2002, 5, 300–307. [Google Scholar] [CrossRef]
- Ryan, C.A. Protease inhibitors in plants: Genes for improving defenses against insects and pathogens. Annu. Rev. Phytopathol. 1990, 28, 425–449. [Google Scholar] [CrossRef]
- Wink, M. Plant breeding: Importance of plant secondary metabolites for protection against pathogens and herbivores. Theor. Appl. Genet. 1988, 75, 225–233. [Google Scholar] [CrossRef]
- Loschke, D.; Hadwiger, L.; Wagoner, W. Comparison of mRNA populations coding for phenylalanine ammonia lyase and other peptides from pea tissue treated with biotic and abiotic phytoalexin inducers. Physiol. Plant Pathol. 1983, 23, 163–173. [Google Scholar] [CrossRef]
- Kim, Y.; Komoda, E.; Miyashita, M.; Miyagawa, H. Continuous Stimulation of the Plant Immune System by the Peptide Elicitor PIP-1 Is Required for Phytoalexin Biosynthesis in Tobacco Cells. J. Agric. Food Chem. 2014, 62, 5781–5788. [Google Scholar] [CrossRef]
- Alkher, H.; El Hadrami, A.; Rashid, K.Y.; Adam, L.R.; Daayf, F. Cross-pathogenicity of Verticillium dahliae between potato and sunflower. Eur. J. Plant Pathol. 2009, 124, 505–519. [Google Scholar] [CrossRef] [Green Version]
- Uppal, A.K.; El Hadrami, A.; Adam, L.R.; Daayf, F.; Tenuta, M. Pathogenic variability of Verticillium dahliae isolates from potato fields in Manitoba and screening of bacteria for their biocontrol. Can. J. Plant Pathol. 2007, 29, 141–152. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Lund, R.E. Tables for an approximate test for outliers in linear models. Technometrics 1975, 17, 473–476. [Google Scholar] [CrossRef]
- Yao, Z.; Islam, R.; Badawi, M.A.; El-Bebany, A.F.; Daayf, F. Overexpression of StRbohA in Arabidopsis thaliana enhances defence responses against Verticillium dahliae. Physiol. Mol. Plant Pathol. 2015, 90, 105–114. [Google Scholar] [CrossRef]



| Primer’s Name | Primer Sequence | Tm (°C) | Accession Number | Amplification Length (bp) |
|---|---|---|---|---|
| Ras-GAP like protein-F | ACGCTGTCCAACCTTCAC | 53 | VDAG_01012 | 273 |
| Ras-GAP like protein-R | GTTGATCTTGTCCCAGTCG | 52.7 | VDAG_01012 | |
| Pyruvate dehydrogenase E1 component subunit beta-F | CTTCGGCGACAAGAGGGT | 58.1 | VDAG_01642 | 283 |
| Pyruvate dehydrogenase E1 component subunit beta-R | GGGAATGCTGCCATACCAC | 58.2 | VDAG_01642 | |
| DNA-(apurinic or apyrimidinic site) lyase-F | CCGGCTGGGACGTTTA | 55.1 | VDAG_02445 | 152 |
| DNA-(apurinic or apyrimidinic site) lyase-R | GCGGAATCTGGTGGTTG | 54.2 | VDAG_02445 | |
| glucan endo-1,3-alpha-glucosidase agn1-F | GCCTTCGGAAACCTCAAT | 54.6 | VDAG_04101 | 307 |
| glucan endo-1,3-alpha-glucosidase agn1-R | CTCCCATGAACTCATACGC | 53.2 | VDAG_04101 | |
| Thioredoxin (VdTRX)-F | GCTGCTCCTGTTTATGCCTTTCCA | 66.8 | VDAG_04529 | 145 |
| Thioredoxin (VdTRX)-R | GAGGTTATGCGGCTTGTTCGT | 61.8 | VDAG_04529 | |
| serine/threonine protein kinase-F | GGTGGGTGCGGTCAAATA | 56.9 | VDAG_04632 | 272 |
| serine/threonine protein kinase-R | AGGCATCCGTAGCACGAC | 56.2 | VDAG_04632 | |
| Ubiquitin-conjugating enzyme variantMMS2-F | CATCCTCGGTCCTCCTCA | 55.5 | VDAG_05365 | 235 |
| Ubiquitin-conjugating enzyme variantMMS2-R | CGCCATGTACCTCCTGATC | 55.6 | VDAG_05365 | |
| VdPREG-F | GGGAATCTGACTAGGTTTCATT | 54.7 | VDAG_06766 | 227 |
| VdPREG-R | GAGTCGGACAGACCTTTGG | 54.7 | VDAG_06766 | |
| Xanthine dehydrogenase-F | GGCTGCTGCATGGATAAG | 54.4 | VDAG_07735 | 227 |
| Xanthine dehydrogenase-R | CCGACAAATACCGACACG | 54.9 | VDAG_07735 | |
| myo-inositol 2-dehydrogenase-F | AGTCTGGCATCGACAATAAC | 52.3 | VDAG_08205 | 192 |
| myo-inositol 2-dehydrogenase-R | GCAGTCTCAACACGCAAA | 52.6 | VDAG_08205 | |
| HAD-superfamily hydrolase-F | AGCCCGACCCTGCCATCTA | 62.6 | VDAG_08490 | 225 |
| HAD-superfamily hydrolase-R | GGAACTCTTGCCAGTCCTTCA | 59.2 | VDAG_08490 | |
| DNA repair protein RAD51-F | ATGGTGAGGGCGAGATGG | 58.3 | VDAG_08796 | 153 |
| DNA repair protein RAD51-R | GGGTGTAAGCGACGGATT | 55.4 | VDAG_08796 | |
| VdWos2-F | GTCTGCTACCAAGGCAACTCC | 58.7 | VDAG_08865 | 227 |
| VdWos2-R | TCTCCTCCGTGTCAATCTCG | 58.1 | VDAG_08865 | |
| NADH-ubiquinone oxidoreductase-F | ATCGGGGCGGGTCTCATT | 62.1 | VDAG_09026 | 116 |
| NADH-ubiquinone oxidoreductase-R | CCTTCGGCAGGCTTCTCC | 60.3 | VDAG_09026 | |
| Serine 3-dehydrogenase-F | ACTTGGGCATTAAGGTGGTC | 56.5 | VDAG_09532 | 348 |
| Serine 3-dehydrogenase-R | CATCGCAGTCAGTTGTCGTAG | 56.6 | VDAG_09532 | |
| His3-F (Zhu et al., 2017) | ATGGCTCGCACTAAGCAA | 54.8 | VDAG_10035 | 238 |
| His3-R (Zhu et al., 2017) | TGAAGTCCTGGGCAATCT | 52.7 | VDAG_10035 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, X.; Arfaoui, A.; Sayari, M.; Adam, L.R.; Daayf, F. Gene Expression of Putative Pathogenicity-Related Genes in Verticillium dahliae in Response to Elicitation with Potato Extracts and during Infection Using Quantitative Real-Time PCR. Pathogens 2021, 10, 510. https://doi.org/10.3390/pathogens10050510
Zhu X, Arfaoui A, Sayari M, Adam LR, Daayf F. Gene Expression of Putative Pathogenicity-Related Genes in Verticillium dahliae in Response to Elicitation with Potato Extracts and during Infection Using Quantitative Real-Time PCR. Pathogens. 2021; 10(5):510. https://doi.org/10.3390/pathogens10050510
Chicago/Turabian StyleZhu, Xiaohan, Arbia Arfaoui, Mohammad Sayari, Lorne R. Adam, and Fouad Daayf. 2021. "Gene Expression of Putative Pathogenicity-Related Genes in Verticillium dahliae in Response to Elicitation with Potato Extracts and during Infection Using Quantitative Real-Time PCR" Pathogens 10, no. 5: 510. https://doi.org/10.3390/pathogens10050510
APA StyleZhu, X., Arfaoui, A., Sayari, M., Adam, L. R., & Daayf, F. (2021). Gene Expression of Putative Pathogenicity-Related Genes in Verticillium dahliae in Response to Elicitation with Potato Extracts and during Infection Using Quantitative Real-Time PCR. Pathogens, 10(5), 510. https://doi.org/10.3390/pathogens10050510








