1. Introduction
Tumors or cancerous cells are a classical sign of inflammation and can be benign or malignant (cancerous). In America, nearly 606,880 people were anticipated to die from cancer in 2019, which translated to about 1660 deaths each day. Cancer is the second most common cause of death in the U.S., exceeded only by heart disease [
1]. Therefore, the study of tumor treatment is required to save human lives in the world. Several researchers [
2,
3,
4,
5] studied the therapeutic treatment of bioheat transfer in skin tissue with the help of mathematical modeling. Mathematical modeling of heat transfer in biological systems has been a broad field of study for various biologists, physicians, mathematicians, and engineers [
6]. An efficient clarification of the physiological relation between the vascular system and tissue is necessary in medical science for treating fatal diseases like tumors. Currently, mathematical models are commonly used to describe the process of hyperthermia, cryosurgery, and many other techniques for the treatment of tumors. It is mandatory to know the thermal effect in skin tissue during the hyperthermia treatment process. The size, shape, and location of tumors are important factors for the treatment process [
7].
Several bioheat transfer-based models have assumed the physiological properties of human beings to be constant, which are not described accurately for hyperthermia treatment of tumors or cancer. However, because the inner structure of the human body is inhomogeneous, the physiological parameters depend on local tissue temperature. Some researchers [
8,
9,
10] considered the perfusion term to be a function of the temperature in local tissue. Similarly, some authors [
11,
12] assumed a metabolic heat source in their model, and this was also a function of the temperature in local tissue. The perfusion term and metabolic heat source are both considered a function of temperature in local tissue, i.e., a realistic-type function, and the external heat source is taken as electromagnetic radiation [
13]. However, the location and shape parameters are not derived very well.
Modeling of the tumor treatment is done by the study of the heat transfer in the biological system. The treatment of tumors has been broadly studied in pre-clinical models with human clinical trials [
14]. The treatment techniques such as hyperthermia, thermal ablation, cryoablation, and cryosurgery are used for selectively destroying the tumor in living skin tissue. Thermal therapy is an ideal modality for the treatment of infected cells using different types of external heat sources like electromagnetic irradiation [
6,
10,
12,
13], magnetic nanoparticles (MNPs) [
15,
16], etc.
Andreozzi et al. (2019) [
17] performed a sensitivity analysis of the hyperthermia effects on a typical transient percolation process in a tumor. In this process, the temperature was raised in a tumor region according to different categories of thermal therapy. Bioheat transfer was analyzed by Andreozzi (2019) [
18], who took many mechanisms into account, such as thermal conduction in tissues, convection and blood perfusion, metabolic heat generation, vascular structure, and the change of tissue properties depending on the physiological condition. A numerically investigated bioheat transfer model has been used for hyperthermia treatment with the convection term instead of the perfusion term in the energy conservation equation for tissue and blood [
19]. Wang et al. (2015) [
20] studied the temperature distribution within biological organs for therapeutic aspects related to hyperthermia treatments such as radiofrequency ablation. The accuracy of temperature-based treatment depends on accurate prediction and control of the temperature in skin tissue [
12]. A quantitative analysis of the relationship between arterial blood and tissue temperature was done by Pennes (1948) [
21]. There are many bioheat transfer models for studying the heat transfer in skin tissue in the existing literature, and it was found that the commonly used bioheat transfer model for analyzing the temperature distribution is the Pennes bioheat transfer model [
21], which is based on the classical constitutive relation that was introduced by Fourier, i.e.,
Penne’s bioheat transfer (PBHT) model predicts the temperature with the infinite speed of propagation, which is incompatible in the real domain. To unify this, consecutively, Cattaneo [
22] and Vernotte [
23] introduced it in 1958, independently; so, the heat flux and temperature gradient are combined with a constitutive relation, which is given as:
Equation (
2) is known as the single-phase-lag (SPL) constitutive relation. Relaxation time
indicates the lag time due to heat flux. In 1995, Tzou [
24,
25] introduced his concept in the generalization of the SPL model by assuming thermalization time due to the temperature gradient, called the dual-phase-lag (DPL) constitutive relation, i.e.,
where
is known as the thermalization time, and the combination of the DPL constitutive relation and energy balance equation is known as the DPLBHT model. For the study of micro-scale responses in time and to capture the micro-scale responses in space, the DPL bioheat transfer model has been used. Therefore, in the existing literature, the DPL bioheat transfer model is the most realistic in comparison with others [
26].
Thermal-probes and cryoprobes are used for tumor or cancer treatment. In the DPLBHT model, the thermal correlation between the cylindrical cryoprobe and skin tissues was studied by Mochnaki and Machrzak [
27]. A relation was developed between the heat transfer in perfused skin tissue with the thermal-probe and a local symmetric component of the vascular system [
28]. Many authors [
8,
29,
30,
31,
32,
33] have assumed a non-linear PBHT model with a physiological property such as perfusion rate for finding the temperature in skin tissues. In reality, These types of models do not give realistic data of the temperature in skin tissue because they do not consider the relaxation and thermalization time in the heat flux and temperature gradient, respectively. This is a drawback of this type of nonlinear bioheat transfer model.
The thermal behaviors of a perfused tissue with two co-current and counter-current vascular networks were investigated numerically under an interstitial hyperthermia process using both local thermal equilibrium (LTE) and local thermal nonequilibrium (LTNE) assumptions [
34]. Zhang et al. [
30] analyzed the PBHT model under the steady-state condition with the perfusion rate in skin tissues varying linearly, quadratically, and exponentially with local skin tissue temperature and solved it using the boundary reciprocity method. The smoothed particle hydrodynamic method was used for the results of the nonlinear PBHT model with space coordinate-dependent thermal conductivity. Several researchers studied the temperature distribution in skin tissues using the DPL bioheat transfer model, undertaking different types of volumetric heat sources. They solved the DPLBHT model using the finite element Legendre wavelet Galerkin method [
15] and used the FERK (4,5) method [
10,
12,
13] and finite difference-decomposition method [
6]. The development of the reconfigurable distributed multiple-input multiple-output technique in a practical communication environment was proposed by Do and Haas [
35].
In this paper, we propose the highly non-linear DPLBHT equation under the constant boundary condition, which consists of temperature-dependent metabolism and blood perfusion heat generation, as well as a Gaussian heat source. This model is very useful for hyperthermia treatment because the physiological properties of biological skin tissue are considered as a realistic function of local tissue temperature. These types of physiological properties with a Gaussian heat source have been used in the DPLBHT model till now. Due to the lower computational complexity and less data storage, combined with the high accuracy, the problem is converted into a system of ordinary differential equations with initial conditions using the finite difference scheme. This system of ordinary differential equations with initial conditions is solved using the RK (4,5) scheme. All parameters such as the location parameter, regional parameter, and relaxation and thermalization time provide a better understanding of the control temperature in the hyperthermia condition. The metabolic heat source, associated metabolic heat source, and external heat source increase as the local skin tissue temperature increases. The perfusion rate, associated with the perfusion heat source parameters, decreases as the local skin tissue temperature increases as well.
This paper is organized into seven sections. In the first section, the introduction of the bioheat transfer models, some methods, and also the nomenclature are given, which support our proposed work. Our mathematical problem is formulated in
Section 2. In the third section, our mathematically formulated problem is converted into a dimensionless form.
Section 4 describes the solution of the proposed problem using the FERK (4,5) method. In
Section 5, we propose the exact solution of our problem for a particular case to verify the FERK (4,5) method. The results and discussion are given in
Section 6. The last section consists of the conclusions of the proposed work.
2. Formulation of the Problem
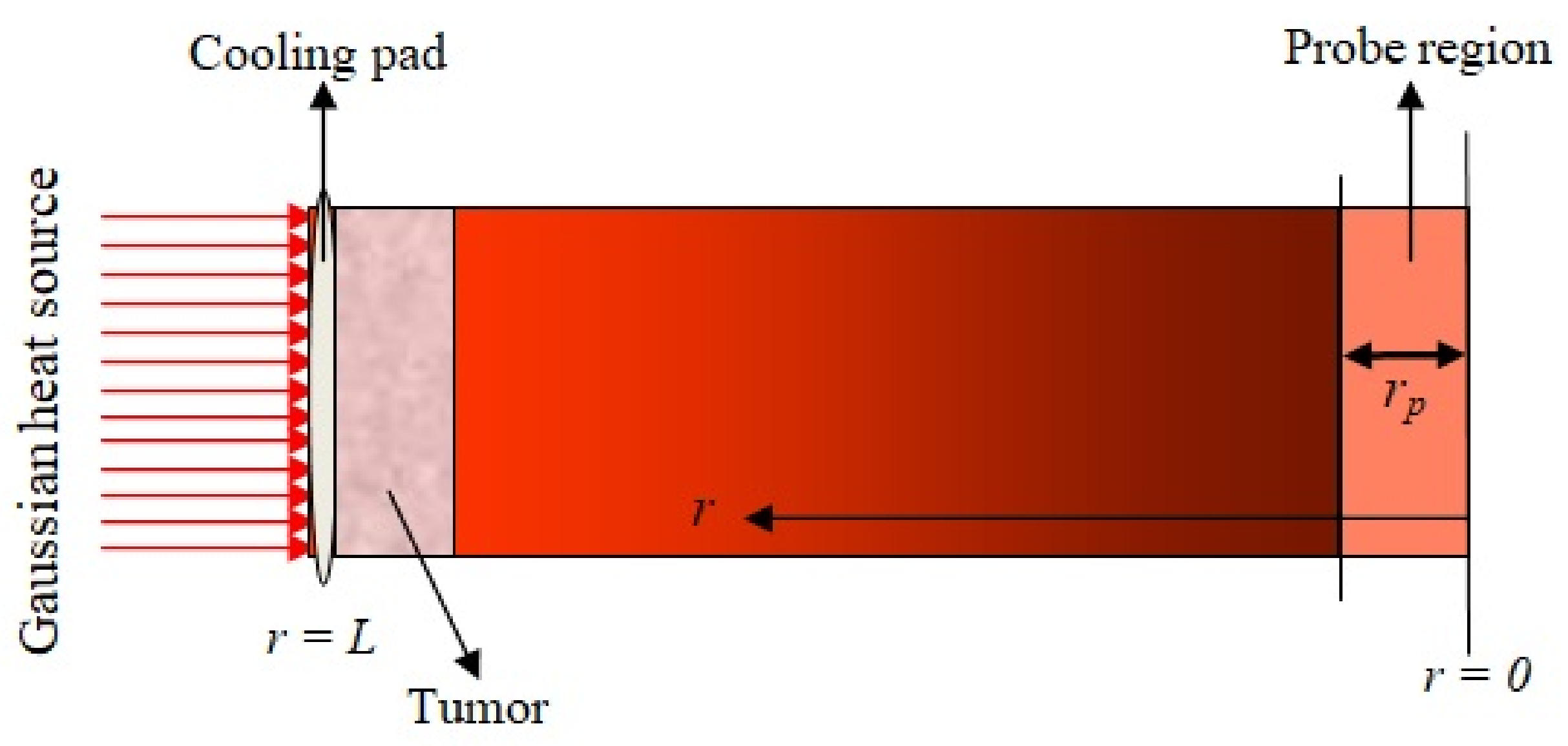
Hyperthermia is a treatment process of tumors and cancer. In this treatment process, the temperature of the tumor region is kept between 41 °C and 46 °C, with approximately a time period of 15 to 60 min [
36]. The outer surface of the skin tissue is kept at a fixed temperature
= 37 °C, initially, during hyperthermia treatment. The outer surface is heated with a Gaussian heat source externally. The inner surface of the skin, i.e.,
, is insulated, and the temperature of the outer surface is maintained constant with the help of a cooling pad, which is shown in
Figure 1. The tumor region is indicated by the schematic geometry of the skin tissue, which depends on the probe region parameter
. If this parameter increases or decreases, then the location of the tumor in skin tissue changes. This is seen in the external heat source. We used a heated metal disc with temperature control and approximated by the one-dimensional non-linear DPLBHT model in the Cartesian coordinate system under the first kind (constant) boundary condition. This is a combination of the DPL constitutive relation and also the one-dimensional energy balance equation. The energy balance equation in one-dimensional form is written as [
21]:
where the left-hand side denotes the conduction term in the skin tissue and the first term of the right-hand side denotes the convection term in the skin tissue.
,
, and
are the perfusion heat source, the metabolic heat source, and the externally applied heat source term, which is taken as a Gaussian-type heat source. The metabolic heat source is generated in the body by the intake of food, and the perfusion heat rate is the heat source that is spent in blood circulation. Still, the Gaussian heat source is externally applied on the outer surface.
The blood perfusion
indicates convection in the blood. This term removes heat due to the flow of blood. It was defined by several researchers [
10,
13,
30].
The perfusion coefficient
depends on the tissue temperature due to the anatomical structure of the skin tissue containing blood vessels.
was taken from Zhang et al. [
30]:
where
and
a are the assumed blood perfusion coefficient and associated blood perfusion coefficient initially.
In Equation (
4),
indicates the metabolism of the human body. It is the factor that increases the local tissue temperature. The realistic function
of the local tissue temperature was taken from Mitchell et al. [
11]:
where
and
are the initial metabolic heat source coefficient and the associated metabolic constant, respectively.
T and
are the local skin temperature and the initial temperature of the skin tissue, respectively.
A Gaussian heat source-type expression is curve fitted based on the experimental measurements of the specific absorption rate distribution in the target region [
37]. The selection of a Gaussian distribution source termed as a spatial heating source helps to determine the hyperthermia position. Therefore, that Gaussian-type heat source expression is applied on the outer surface, and it was taken from Kumar et al. [
13]:
where
is the reference value of external heat generation in the tissue;
is the measured length from the outer surface;
L is the length of the tissue temperature.
is the length of the probe region.
Now, the energy balanced equation is combined with the approximation of the DPL constitutive relation of the first order in one-dimensional form, then we obtain the non-linear DPLBHT equation [
13], i.e.,
initially subjected to:
Physically, the heating/cooling condition at the outer surface
was given in [
15], i.e.,
and the inner surface is adiabatic [
10,
12], so that it is defined as:
In the existing literature, the DPLBHT model with a Gaussian heat source in the presence of a realistic function of the metabolic and perfusion heat generation terms has not been studied till now. We considered the Gaussian heat source because it helps with the control of temperature during hyperthermia treatment.