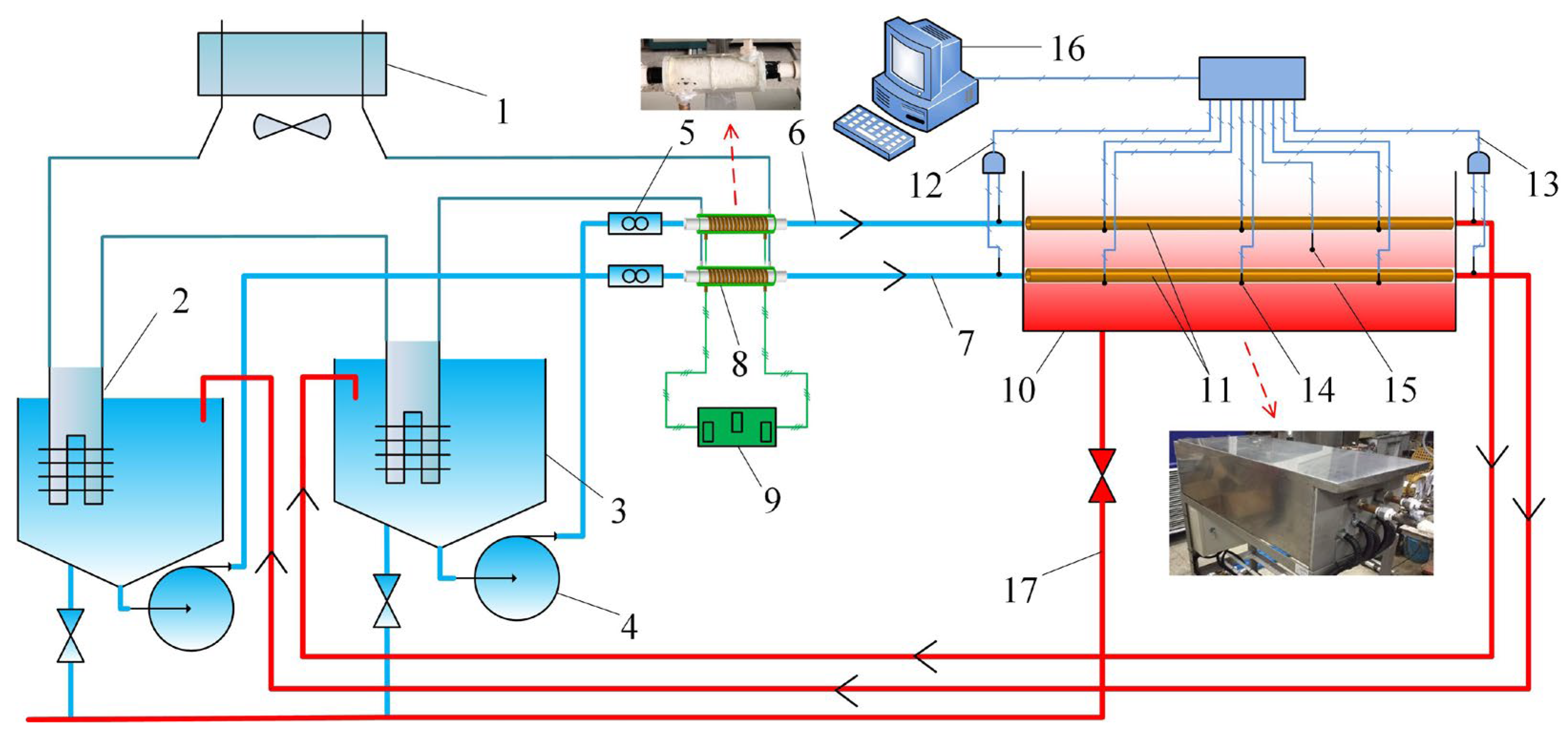
3.1. Formation of Crystallization Fouling
Figure 3 shows the formation process of crystallization fouling on the heat exchange surface. A decrease in solution temperature will result in a reduction in solubility for salts with normal solubility. Due to supersaturation, the dissolved salt will crystallize and precipitate on the cooling surface. However, for salts with inverse solubility, the solubility decreases with increasing solution temperature, leading to supersaturation and the subsequent precipitation of the dissolved salts on heating surfaces. When the solution flows through the heat exchange surface, a boundary layer is generated with a temperature gradient. The CaCO
3 dissolved in the solution is an insoluble salt with inverse solubility. It will be supersaturated at high temperatures, accelerate crystallization, and adsorb on the heat exchange surface, thus forming crystallization fouling, which can also be called water scale. In water-cooling systems, the most common fouling components are CaCO
3, MgCO
3, CaSO
4, MgSO
4, etc.
Although supersaturation is a necessary condition for the formation of crystallization fouling, it is not a sufficient condition. In fact, crystal growth requires a decrease in Gibbs free energy as the crystal size increases. If the crystal is smaller than its critical size, its Gibbs free energy will increase with increasing crystal size, and the crystal will tend to be redissolved in solution. Therefore, the solid boundary, such as the heat exchange surface, or suspended particles, like embryos, often serve as crystallization sites. As a result, there are two ways of forming crystallization fouling. The first way is that the ions, such as Ca
2+ and CO
32−, form crystal nuclei in a solution and then grow into crystals (bulk crystallization). These crystal particles are then transported and attached to the heat exchange surface, which is called homogeneous precipitation, as shown in
Figure 3a. The second way is that the ions are directly transported to the nearby heat exchange surface by the fluid. They make contact with the heat exchange surface and nucleate and grow into crystals, thus forming crystallization fouling (surface crystallization). This process is called heterogeneous precipitation, as shown in
Figure 3b. During the process of heat transfer, both homogeneous and heterogeneous precipitation occur simultaneously. Therefore, the main factor affecting crystallization fouling is solution supersaturation. The higher the solution supersaturation, the stronger the driving force of crystallization. The heat exchanger surface is more likely to form crystallization fouling.
3.2. Fouling Resistance
In order to validate the accuracy of the data obtained from the experimental rig, two groups of experiments were performed under the same conditions. The inlet and thermostatic bath temperatures were 40 °C and 60 °C, respectively. The concentration of CaCO
3 was 1000 mg/L, while the volume flow rate was 0.3 m
3/h. No AEFTD was used to treat the experimental solution. A comparison of the experimental results between the two groups is presented in
Figure 4. Fouling resistance was a function of time. The results showed that there was no significant difference between the two fouling resistance curves in the growth stage. After 150 min, the asymptotic values of fouling resistance tended to be consistent, with a discrepancy of only 3.23% between the two experimental groups, thus demonstrating the reproducibility of the results obtained from the experimental rig.
Figure 5 shows six fouling resistance curves under different electromagnetic frequencies, while
Table 1 and
Table 2 present the specific experimental conditions. As the experimental fluid flowed through the heat exchange surface that was clean and without pollutants, the CaCO
3 precipitated and gradually deposited on the tube side of the heat exchanger, thus resulting in fouling resistance. It can be observed from
Figure 5 that, over the whole experimental period, the fouling resistance curves showed the same trend, rising first and then gradually stabilizing. Therefore, they can be divided into the rising stage and the stable stage. During the rising stage, the CaCO
3 continually crystallized and adhered to the tube side [
24]. The fouling resistance of the heat exchange surface rapidly increased, during which the deposition rate (
) was higher than the removal rate (
). Thereafter, as
was equivalent to
, the fouling resistance reached the stable stage and ceased to increase. The fouling resistance curves show obvious differences under different electromagnetic frequencies. When compared to the control group, the fouling resistances with the AEFTD were greatly reduced, indicating that the application of the AEFTD could effectively inhibit CaCO
3 fouling formation on the tube side of the heat exchanger. Xing [
25] reached the same conclusion.
Figure 6 shows the fouling resistances at 50, 100, and 150 min under different electromagnetic frequencies. By comparing the fouling resistances that have the same electromagnetic frequency at different moments, it can be found that the fouling resistance gradually increased as the experiment proceeded. However, different electromagnetic frequencies corresponded to varying degrees of fouling resistance increments. The fouling resistance increment in the control group was the highest, and 1.5 kHz had the smallest increment. In addition, at three specific time points, fouling resistance initially decreased and subsequently increased and reached its minimum at 1.5 kHz with increasing electromagnetic frequency.
The asymptotic value of fouling resistance can serve as an indicator for the final state of fouling accumulation on the tube side of a heat exchanger when the experimental system is in equilibrium. Achieving a smaller asymptotic value for fouling resistance is a desirable result when using an AEFTD.
Table 3 shows the asymptotic values for the fouling resistance and anti-fouling rates under different experimental groups. The maximum asymptotic value for fouling resistance in the control group was 10.15 × 10
−5 m
2·K/W. The anti-fouling rate was between 36.65% and 71.13%, corresponding to different electromagnetic frequencies. When the electromagnetic frequency was 1.5 kHz, the asymptotic value of fouling resistance reached the minimum, which was 2.93 × 10
−5 m
2·K/W, with an anti-fouling rate of 71.13%. It can be found that the treatment effects of the axial-electromagnetic field are different under different electromagnetic frequencies. The effectiveness of the AEFTD is contingent upon the selection of the appropriate electromagnetic parameters. This study revealed an extreme character in the treatment effect of an axial-electromagnetic field, with the optimal performance achieved at the electromagnetic frequency of 1.5 kHz.
3.3. Growth Rate of CaCO3 Fouling
It is conceivable that during the fouling formation period, the fouling components will be deposited on the heat exchange surface, leading to an increase in fouling resistance. However, there is a phenomenon through which the fouling components are removed by fluid, thus resulting in a reduction in fouling resistance. Therefore, changes in fouling resistance over time are influenced by both the deposition and removal factors. The fouling formation process can be described using the following differential equation:
where
is the mass of fouling deposition per unit area (kg/m
2),
is the deposition rate (kg/(m
2·s)), and
is the removal rate (kg/(m
2·s)).
Assuming that the characteristics of the fouling components and the thickness of the fouling layer are evenly distributed along the heat exchange surface, the fouling resistance can be defined as follows:
where
is the density of the fouling components (kg/m
3), and
is the coefficient of thermal conductivity (W/(m·K)).
The differential of Equation (14) can be obtained as follows:
Thereafter, Equation (15) is substituted into Equation (13), and the result is as follows:
where
is the deposition rate (m·K/N), and
is the removal rate (m·K/N).
Equation (16) indicates that the growth rate of fouling resistance per unit area can be described as the difference between the deposition rate (
) and removal rate (
), which is termed the Kern–Seaton model [
22].
As shown in
Figure 7, by taking the experimental data from the control group as an example, it can be found that the entire period of the fouling resistance curve can be divided into two stages: the rising and stable stages. The experimental data were fitted using the Boltzmann function, and the R
2 of the fitting curve marked in red was greater than 0.96, which indicated that the fitting effect was excellent and the fitting curve could correctly reflect the variation in the fouling resistance during the experiment. In addition, the R
2s of the six fitting curves in the experimental groups were greater than 0.94.
The fitting curve marked in red in
Figure 7 can be expressed as the following equation:
where
is the fouling resistance (m
2·K/W),
t is the running time of the experimental rig (min), and
A1 = −9.95 × 10
−5,
A2 = 1.03 × 10
−4,
A3 = 26.71, and
t0 = −2.16.
The growth rate curve of fouling resistance, which is marked with a green dotted line, can be expressed by the differential equation of the fitting curve:
The fouling resistance of the heat exchange surface can characterize the accumulation state of CaCO
3 fouling. Therefore, the growth rates of fouling resistance and CaCO
3 fouling were considered equivalent. According to the above analysis,
td in the control group (
Figure 7) was taken as the deadline, and the average growth rates for CaCO
3 fouling on the tube side of the heat exchanger under different electromagnetic frequencies were determined using Equation (18), as shown in
Figure 8. The average growth rates showed a large difference with and without the AEFTD. When comparing the experimental results of the asymptotic value for the fouling resistances in
Figure 6, both exhibit a similar trend with increasing electromagnetic frequency, indicating that the average growth rate was proportional to the asymptotic value of fouling resistance. In other words, the slower the growth rate, the smaller the fouling resistance asymptotic value. The average growth rate without the AEFTD was 6.95 × 10
−7. After the application of the AEFTD, the average growth rates dropped by 37.12% to 68.20%, corresponding to different electromagnetic frequencies, indicating that CaCO
3 fouling formation on the tube side was retarded. A minimum average growth rate of 2.21 × 10
−7 was achieved at the electromagnetic frequency of 1.5 kHz. The reduced growth rate using the AEFTD is the desired result.
3.4. Outlet Temperature of Experimental Fluid
During the experiment, the inlet temperature and volume flow rate of the experimental fluid and other working parameters remained constant. When the experimental fluid passed through the heat exchange surface, the heat exchange behavior resulted in an increase in the temperature of the experimental fluid. As the experiment proceeded, fouling accumulation on the heat exchange surface could reduce heat transfer efficiency, which led to a decrease in the outlet temperature of the experimental fluid. At the beginning of the experiment, the average outlet temperature of the experimental fluid was 32.7
0.15 °C, indicating that the temperature of the experimental fluid had increased by 2.7 °C after passing through the shell-and-tube heat exchanger. Subsequently, the outlet temperature of the experimental fluid gradually dropped and then stabilized due to fouling.
Figure 9 shows the outlet temperatures of the experimental fluid at 50, 100, and 150 min under different electromagnetic frequencies. The experimental results illustrated that the outlet temperatures of the experimental fluid with the AEFTD were higher than that of the control group. In addition, the outlet temperature of the experimental fluid at 1.5 kHz was significantly higher than that of the other experimental conditions. Except for the results of the control group and the 1.5 kHz condition, there was no significant difference in outlet temperatures among other experimental conditions.
Therefore, by combining the conclusions from
Figure 5, the experimental results of the control group and 1.5 kHz condition were taken as examples for analysis. The asymptotic value of fouling resistance in the control group was the maximum: 10.15 × 10
−5 m
2·K/W, indicating that fouling accumulation on the heat exchange surface was more serious and obviously hindered heat transfer and the asymptotic value for fouling resistance at 1.5 kHz was the minimum: 2.93 × 10
−5 m
2·K/W. Meanwhile, it can be seen from
Figure 9 that the outlet temperatures of the experimental fluid corresponding to the former were the lowest at each moment, while the outlet temperatures corresponding to the latter were the highest. When the experiment was carried out for 150 min and was compared with the initial outlet temperature, the outlet temperature of the control group decreased by 1.04 °C, and the outlet temperature of the 1.5 kHz condition decreased by 0.34 °C, indicating that the application of the AEFTD at 1.5 kHz increased the outlet temperature by 0.7 °C. The above analysis results show that the trend change was the opposite between fouling resistance and the outlet temperature of the experimental fluid, which indicated that the application of the AEFTD enhanced heat transfer.
3.5. SEM of CaCO3 Fouling
Crystallization fouling is affinitive to the nucleation and growth of crystals. Therefore, variation in crystal morphology is also important for investigating the influence of the AEFTD on the crystallization fouling of a heat exchange surface. The three main types of CaCO
3 crystal morphologies are vaterite, aragonite, and calcite [
26], which belong to hexagonal, trigonal, and orthorhombic systems, respectively. Regarding the stability of the crystals, vaterite, with a spherical structure, is the least stable, followed by metastable aragonite with an acicular structure, whereas calcite, with a rhombohedral structure, forms the most stable crystal morphology and is widely found in nature. Prior to commencing the experiment, a red copper plate with a thickness of 0.5 mm, length of 20 mm, and width of 20 mm was bent and placed close to the tube side of the heat exchanger. The fouling samples on the red copper plate were dried and analyzed using SEM during the experiment. SEM micrographs (×2000) under different electromagnetic frequencies are shown in
Figure 10.
According to the SEM micrographs in
Figure 10, it can be found that the AEFTD has a significant impact on the morphology of CaCO
3 crystals. The CaCO
3 without the use of the AEFTD (
Figure 10a) was found to be regular calcite with a rhombohedral structure and clustered together, which preferably formed dense hard fouling on the tube side of the heat exchanger and could not be easily removed. However, when compared to the control group, the morphology of the CaCO
3 crystals was different from that of the axial-electromagnetic field. In addition to calcite, vaterite with a spherical structure was observed (
Figure 10b–f). Vaterite is a nonpolar crystal, which does not easily aggregate into clusters and is suspended in the fluid to form muddy fouling that is carried away by turbulent flow, as opposed to accumulating on the tube side of the heat exchanger. Therefore, the appearance of vaterite crystals is a good phenomenon for inhibiting fouling formation on the tube side of the heat exchanger.
The proportion and size of vaterite in
Figure 10b (0.5 kHz) were relatively small because the unstable crystal morphology grew further and recrystallized into calcite. However, with increasing electromagnetic frequency, the proportion of vaterite and crystal size increased. When the electromagnetic frequency was 1.5 kHz (
Figure 10d), the CaCO
3 remained vaterite in its morphology (with a large size) and did not transform into calcite. Vaterite was the major crystalline phase formed during this period. Furthermore, the proportion and size of vaterite slightly decreased when compared with those in
Figure 10d (1.5 kHz), as the electromagnetic frequency continued to increase to 2.5 kHz (
Figure 10f). These results indicate that the axial-electromagnetic field promoted CaCO
3 crystallization in vaterite, as opposed to calcite. When combined with the experimental results obtained in
Section 3.2, it was found that the formation of vaterite, which did not easily adhere to the tube side of the heat exchanger under the axial-electromagnetic field, was the main reason for the reduction in fouling on the heat exchange surface.
3.6. Anti-Fouling Mechanism
The anti-fouling mechanism of the AEFTD for crystallization fouling can be explained from two perspectives.
On the one hand, calcium carbonate is a kind of typical crystallization fouling. From the perspective of crystal growth, unstable vaterite was formed in the first stage through spheroidal crystal growth during CaCO
3 crystal formation. This was followed by the conversion of vaterite to stable calcite in the second stage through dissolution and recrystallization. Crystal growth is accompanied by a decrease in the energy of the system, which is termed the Gibbs free energy [
27]. Unstable crystalline phases first appeared and were gradually transformed into stable products. The system achieved minimum energy when the reaction reached equilibrium. Crystal nucleation is the first step of crystal growth, and its driving force is determined by the difference in volume-free energy between CaCO
3 crystals and the experimental fluid. This force is proportional to the degree of supersaturation in the experimental fluid. The resistance to nucleation is the interfacial energy between the CaCO
3 crystals and the experimental fluid. Therefore, there is a critical radius for the crystal nucleus,
. When
, the embryonic crystal can form a crystal nucleus. The relationship between the variation in free energy and the radius of a crystal nucleus is shown in
Figure 11.
The free energy variation can be calculated as follows:
where
is the free energy variation (J),
is the volume of free-energy difference between the liquid and solid phases in the system (J), and
is the interfacial energy in the system (J).
and
can be calculated as follows:
where
is the free energy difference per unit volume (J/m
3), and
is the surface energy per unit area (J/m
2), i.e., the specific surface energy.
When combined with the axial-electromagnetic field, there are many particles associated with fouling in the experimental fluid, such as with Ca
2+, CO
32−, and CaCO
3 crystals, and they have their own natural frequencies. When the electromagnetic frequency is close to or consistent with their natural frequencies, the particles resonate, absorbing more energy from the electromagnetic field, transforming this into internal energy in the fluid. Different frequencies correspond to different energies absorbed by the experimental fluid. When
, the energy provided by the electromagnetic field will increase
so as to reach the nucleation barrier
quickly and promote the growth of the embryonic crystal to the critical radius,
r. When
, the volume-free energy becomes dominant, resulting in a decrease in
, promoting further crystal growth. However, the energy from the electromagnetic field will inhibit the decrease in the
, which can prevent the crystal size from continuing to grow and maintain the crystal in a metastable state. As a result, the decrease in Gibbs free energy was suppressed [
28], thus impeding the transformation of vaterite into calcite. SEM micrographs of the fouling samples show the crystal morphology of vaterite. The greater the amount of energy absorbed by the fluid, the better the effect of inhibiting the transformation of vaterite to calcite. Fouling formation on the tube side of the heat exchanger surface was inhibited. In this study, the experimental fluid absorbed the most energy from the axial-electromagnetic field at 1.5 kHz.
On the other hand, water is the most common carrier of crystallization fouling in nature. Deionized water was used as the experimental fluid in this study. The angle between the two hydrogen-oxygen bonds in a water molecule is 104.5°. As a result, the center of oxygen and hydrogen atoms do not coincide, and water molecules are polar, which causes a type of electrostatic attraction force between the positive and negative charges of the two water molecules, namely the hydrogen bond. Due to the existence of hydrogen bonds, multiple individual water molecules will be associated with water molecular clusters [
29]. Because the charges at each end of the hydrogen bond have opposite electrical properties. When the experimental fluid passes through the AEFTD, the Lorentz force from the axial-electromagnetic field will change some of the properties of the water body. The equation is shown as follows:
where
q is the quantity of the charge (C),
is its velocity vector (m/s), and
is the magnetic induction intensity vector (T).
The mass center of the molecular water cluster is on the central oxygen atom, and the whole water molecular cluster moves around this oxygen atom, around which the whole cluster moves and rotates. The water molecular clusters are in equilibrium without the axial-alternating magnetic field, and under the action of the axial-alternating magnetic field, when the rotation axis of the water cluster is in line with the direction of the axial-alternating magnetic field, the water molecules will also rotate around the central axis. The energy of the hydrogen bond is very sensitive to the distance between the two charges. As can be seen from Equation (23), the axial-electromagnetic field causes positive and negative charges (linked by hydrogen bonds) to move in opposite directions periodically, which breaks or distorts the hydrogen bond, hence weakening or destroying the hydrogen bond network in the molecular water clusters. This results in the formation of smaller clusters, water molecule dimers, or individual water molecules.
The spectroscopy results also showed that the angle of the hydrogen bond in a water molecule decreased from 104.5° to 103.0° under the action of the magnetic field, indicating the distortion of the hydrogen bond [
30]. Toledo et al. [
31] obtained a similar conclusion through theoretical calculations, where a magnetic field weakened the hydrogen bonds between the water molecules and destroyed larger molecular water clusters, which formed more and smaller molecular water clusters with stronger hydrogen bonds.
According to the Stokes–Einstein equation [
32], the self-diffusion coefficient of a particle is inversely proportional to its radius, which is defined as follows:
where
is the radius of the particle (m),
k is the Boltzmann constant (J/K),
T is the thermodynamic temperature (K),
is the viscosity (Pa·s), and
D is the self-diffusion coefficient (m
2/s).
Therefore, the axial-electromagnetic field enhances the diffusivity and activity of water molecules. When combined with the experimental conditions in this study, the experimental fluid contains molecular water clusters, Ca2+, CO32−, and CaCO3 crystals. Ca2+, CO32−, and CaCO3 crystals have a layer of water molecules arranged in order. The former two are formed by hydration, which is called hydration ions. These peripheral water molecules provide a barrier to the interaction between Ca2+ and CO32− and to the adsorption of Ca2+ and CO32− on the surface of the CaCO3 crystals. Under the action of an axial-electromagnetic field of 1.5 kHz, the diffusion capacity of water molecules is significantly enhanced, and the peripheral water molecules will break free, thus promoting the reaction between Ca2+ and CO32− and the homogeneous nucleation of CaCO3 crystals in bulk, which reduces the probability of CaCO3 adhering to the tube side of the heat exchanger.