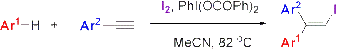4.2. General procedure for the iodoarylation of alkynes
A mixture of an arene (10 mmol), an arylacetylene (1 mmol), I
2 (1.25 mmol), PhI(OCOPh)
2 (3 mmol) and MeCN (6 mL) was placed in a 25 mL round-bottom flask. The reaction mixture was stirred for about 5 min at room temperature and then refluxed at 82 ºC with stirring until the completion of the reaction. The reaction mixture was dissolved in CH
2Cl
2, (20 mL) and water (20 mL) was added to the CH
2Cl
2 solution. The mixture was then washed with 1M aqueous sodium thiosulphate solution to remove the unreacted iodine. The aqueous reaction mixture was extracted with CH
2Cl
2 (4 × 10 mL) and the CH
2Cl
2 extract was dried over anhydrous sodium sulfate. Finally, the solvent was removed under reduced pressure below 40 ºC. Individual pure compounds were isolated from the reaction mixture by column chromatography on silica gel using hexane and CH
2Cl
2 as eluent. Data of 1,1-diaryl-2-iodoethenes
3a-e,
5a-e, and
12a-e were previously reported in the literature [
7].
1-Iodo-2-(pentamethylphenyl)-1,2-diphenylethene (7a): Pale yellow crystalline solid; mp 194.5-197 °C; 1H-NMR δ: 7.44-6.99 (m, 10H, ArH), 2.20 (s, 6H, 2xMe), 2.08 (s, 3H, Me), 2.03 (s, 6H, 2xMe); 13C-NMR δ: 149.34, 144.94, 144.66, 137.66, 133.88, 132.35, 130.48, 129.37, 128.35, 127.64, 127.40, 127.31, 100.50, 18.80, 16.67, 16.49 (one peak overlapped); Anal. calcd. for C25H25I: C, 66.38; H, 5.57. Found: C, 66.34, H, 5.59.
1-Iodo-1,2-diphenyl-2-(2,4,6-trimethylphenyl)ethene (7b): Pale yellow crystalline solid; mp 154-156 °C; 1H-NMR δ: 7.42-7.06 (m, 10H, ArH), 6.64 (s, 2H, ArH), 2.16 (s, 6H, 2x Me), 2.13 (s, 3H, Me); 13C-NMR δ: 147.55, 144.50, 144.19, 137.29, 136.77, 135.40, 129.52, 128.93, 128.39, 127.66, 127.60, 127.56, 127.42, 101.39, 20.92, 20.75; HRMS-EI: m/z calcd. for C23H21I [M]+: 424.0688; found: 424.0689.
1-Iodo-2-(4-methoxyphenyl)-2-(pentamethylphenyl)ethene (9a): Pale yellow crystalline solid; mp 110-111.5 °C; a mixture of E- and Z-isomers (92:8); 1H-NMR δ: 7.42 (d, J = 9.0 Hz, ArH), 7.14 (d, J = 9.0 Hz, ArH), 7.02 (s, =CH), 6.82 (d, J = 9.0 Hz, ArH), 6.76 (d, J = 9.0 Hz, ArH), 6.24 (s, =CH), 3.78 (s, OMe), 3.76 (s, OMe), 2.28 (s, Me), 2.23 (s, 2xMe), 2.19 (s, 2xMe), 2.14 (s, Me), 2.04 (s, 2xMe); 13C- NMR δ: 159.29, 159.01, 152.24, 150.80, 139.69, 138.94, 134.20, 132.68, 132.46, 132.09, 131.34, 130.45, 130.30, 130.18, 129.52, 127.65, 113.84, 113.07, 79.28, 75.80, 55.20, 55.14, 18.06, 17.60, 16.94, 16.89, 16.80, 16.58; HRMS-EI: m/z calcd. for C20H23IO [M]+: 406.0794; found: 406.0791.
1-Iodo-2-(4-methoxyphenyl)-2-(2,4,6-trimethylphenyl)ethene (9b): Pale yellow highly viscous liquid; a mixture of E- and Z-isomers (87:13); 1H-NMR δ: 7.36-6.76 (m, ArH and =CH), 3.77 (s, 2xOMe), 2.33 (s, Me), 2.27 (s, Me), 2.21 (s, 2xMe), 2.06 (s, 2xMe); HRMS-EI: m/z calcd. for C18H19IO [M]+: 378.0481; found: 378.0479.
1-Iodo-2-(4-methoxyphenyl)-2-(2,3,5,6-tetramethylphenyl)ethene (9c): Pale yellow crystalline solid; mp 108-110 °C; a mixture of E- and Z-isomers (51:49); 1H-NMR δ: 7.36-6.77 ( m, ArH and =CH), 3.78 (s, 2xOMe), 2.25 (s, 2xMe), 2.21 (s, 2xMe), 2.14 (s, 2xMe), 1.99 (s, 2xMe); 13C-NMR δ: 159.37, 159.06, 157.94, 151.51, 144.75, 141.42, 134.16, 133.68, 133.40, 131.69, 131.05, 130.91, 130.83, 130.51, 130.15, 127.59, 113.89, 113.12, 79.04, 55.22, 55.16, 20.12, 20.11, 16.47, 15.82; HRMS-EI: m/z calcd. for C19H21IO [M]+: 392.0637; found: 392.0637.
1-(3-Bromo-2,4,6-trimethylphenyl)-2-iodo-1-(4-methoxyphenyl)ethene (9d): Pale yellow highly viscous liquid; a mixture of E- and Z-isomers (58:42); 1H-NMR δ: 7.81-6.68 ( m, ArH and =CH), 3.86 (s, OMe), 3.79 (s, OMe), 2.45 (s, Me), 2.38 (s, Me), 2.37 (s, Me), 2.21 (s, Me), 2.16 (s, Me), 2.01 (s, Me); HRMS-EI: m/z calcd. for C18H18BrIO [M]+: 455.9586; found: 455.9583.
1-(2,5-Dimethylphenyl)-2-iodo-1-(4-methoxyphenyl)ethene (9e): Pale yellow highly viscous liquid; a mixture of E- and Z-isomers (60:40), 1H-NMR δ: 7.76-6.73 (m, ArH and =CH), 3.86 (s, OMe), 3.79 (s, OMe), 2.33 (s, Me), 2.32 (s, Me), 2.21 (s, 2xMe); 13C-NMR δ: 159.08, 157.55, 157.22, 144.44, 143.91, 139.27, 136.44,135.76, 131.30, 130.55, 130.43, 129.98, 129.93, 129.02, 128.78, 128.39, 113.40, 110.02, 85.30, 77.21, 56.33, 55.20, 20.97, 19.14, 19.09, 17.74; HRMS-EI: m/z calcd. for C17H17IO [M]+: 364.0324; found: 364.0327.