Microwave Assisted Synthesis of 2,2'-Arylene-substituted Bis(4H-3,1-Benzoxazin-4-one) Derivatives Using the Complex Cyanuric Chloride/N,N-Dimethylformamide
Abstract
:1. Introduction
2. Results and Discussion
3. Experimental
3.1. General
3.2. Preparation of 2,2′-(1,4-Phenylene)bis(4H-3,1-benzoxazin-4-one) (A1)
3.2.1. Step 1: Preparation of 2,2′-[1,4-Phenylene-bis(carbonylimino)]dibenzoic Acid
3.2.2. Step 2: Reaction of 2,2′-[1,4-Phenylene-bis(carbonylimino)]dibenzoic Acid with CC/DMF
3.3. Preparation of 2,2′-(1,4-Naphthylene)bis(4H-3,1-benzoxazin-4-one) (A2)
3.3.1. Step 1: Preparation of 2,2′-[1,4-Naphthylene-bis (carbonylimino)] Dibenzoic Acid
3.3.2. Step 2: Reaction of 2,2′-[1,4-Naphthylene-bis (carbonylimino)] Dibenzoic Acid with CC/DMF
3.4. Preparation of 2,2′-(4,4′-Biphenylene)bis(4H-3,1-benzoxazin-4-one) (A3)
3.4.1. Step 1: Preparation of 2,2′-[4,4′-Biphenylene-bis(carbonylimino)]dibenzoic Acid
3.4.2. Step 2: Reaction of 2,2′-[4,4′-Biphenylene-bis(carbonylimino)]dibenzoic Acid with CC/DMF
3.5. Preparation of 2,2′-(2,7-Naphthylene)bis(4H-3,1-benzoxazin-4-one) (A4)
3.5.1. Step1: Preparation of 2,2′-[2,7-Naphthylene-bis(carbonylimino)]dibenzoic Acid
3.5.2. Step 2: Reaction of 2,2′-[2,7-Naphthylene-bis(carbonylimino)]dibenzoic Acid with CC/DMF
3.6. Preparation of 2,2′-(1,4-Phenylene)bis(5,7-dichloro-4H-3,1-benzoxazin-4-one) (A5)
3.6.1. Step 1: Preparation of 2,2′-[1,4-Phenylene-bis (carbonylimino)]bis(4,6-dichlorobenzoic acid)
3.6.2. Step 2: Reaction of 2,2′-[1,4-Phenylene-bis (carbonylimino)]bis(4,6-dichlorobenzoic acid) with CC/DMF
4. Conclusions
Acknowledgments
References
- Pang, H.; Zhang, W.G.; Zhao, S.M. The Electroluminescent Devices Based on Benzoxazinone as Strong Fluorescent Material. Adv. Mater. Res. 2011, 380, 339–343. [Google Scholar] [CrossRef]
- Pang, H.; Zhang, W.G.; Zhao, S.M. Non-Dopant Electroluminescent Devices Based on Benzoxazinone Derivative as Strong Fluorescence in Solid Film. Adv. Mater. Res. 2011, 399–401, 1151–1155. [Google Scholar] [CrossRef]
- Magno, J.N.; Khayrullin, I. Method and System for Fabricating an OLED. U.S. Patent 0132228 A1, 2004. [Google Scholar]
- Potrawa, T.; Magno, J.; Khayrullin, I.; Hoyer, S. Benzoxazinone and Quinazolinone Derivatives. U.S. Patent 0219390 A1, 2004. [Google Scholar]
- Sarkar, A.K. Low Color, Low Sodium Benzoxazinone UV Absorbers and Process for Making Same. U.S. Patent 6,774,232 B2, 2004. [Google Scholar]
- Rad-Moghadam, K.; Montazeri, N. Efficient synthesis of 2-substituted 4H-3,1-benzoxazin-4-ones under microwave irradiation. Asian J. Chem. 2007, 19, 2467–2469. [Google Scholar]
- Dandia, A.; Singh, R.; Sarawgi, P. Green chemical multi-component one-pot synthesis of fluorinated 2,3-disubstituted quinazolin-4(3H)-ones under solvent-free conditions and their anti-fungal activity. J. Fluor. Chem. 2005, 126, 307–312. [Google Scholar] [CrossRef]
- Kurihara, M.; Saito, H.; Nukada, K.; Yoda, N. Cyclopolycondensation. XII. Polymerization mechanism of polybenzoxazinones in polyphosphoric acid medium. J. Polym. Sci. Part A-1 1969, 7, 2897–2914. [Google Scholar] [CrossRef]
- Rabilloud, G.; Sillion, B. Synthesis of 4H-3,1-benzoxazin-4-ones and 4-(3H)quinazolinones from anthranilic acids and their derivatives by the use of triphenyl phosphite and pyridine. J. Heterocycl. Chem. 1980, 17, 1065–1068. [Google Scholar] [CrossRef]
- Khajavi Mohammad, S.; Shariat Seyed, M. A New Synthesis of 2-Substituted 4H-3,1-Benzoxazin-4-ones by Cyanuric Chloride Cyclodehydration of N-Benzoyl- and N-Acylanthranilic Acids. Heterocycles 2005, 65, 1159–1165. [Google Scholar] [CrossRef]
- Shariat, M.; Abdollahi, S. Synthesis of Benzoxazinone Derivatives: A New Route to 2 (N Phthaloylmethyl)-4H-3,1-benzoxazin-4-one. Molecules 2004, 9, 705–712. [Google Scholar] [CrossRef] [PubMed]
- Hullio, A.A.; Mastoi, G.M. Application of multipurpose dimethyl formamide-like task specific ionic liquid as a recyclable reagent for direct iodination of alcohols. Iran. J. Catal. 2011, 1, 79–86. [Google Scholar]
- De Luca, L.; Giacomelli, G.; Porcheddu, A. An Efficient Route to Alkyl Chlorides from Alcohols Using the Complex TCT/DMF. Org. Lett. 2002, 4, 553–555. [Google Scholar] [CrossRef] [PubMed]
- De Luca, L.; Giacomelli, G.; Porcheddu, A. Beckmann Rearrangement of Oximes under Very Mild Conditions. J. Org. Chem. 2002, 67, 6272–6274. [Google Scholar] [CrossRef] [PubMed]
- Kappe, C.O. Controlled Microwave Heating in Modern Organic Synthesis. Angew. Chem. Int. Ed. Engl. 2004, 43, 6250–6284. [Google Scholar] [CrossRef] [PubMed]
- Fink, J.K. Handbook of Engineering and Specialty Thermoplastics. Polyolefins and Styrenics; John Wiley & Sons, Inc: Hoboken, NJ, USA, 2010; p. 311. [Google Scholar]
Sample Availability: Samples of the compounds A1–5 and B1–5 are available from the authors. |





| Name and Structure | Product | Yield (%) | Time (min) | M.p. (°C) |
|---|---|---|---|---|
 | A1 | 88 | 1 | 314–316 |
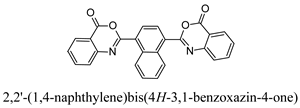 | A2 | 85 | 1.5 | 268–270 |
 | A3 | 85 | 1.5 | 376–378 |
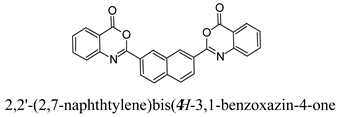 | A4 | 79 | 2 | 278–280 |
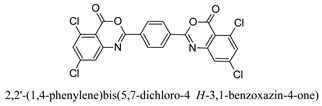 | A5 | 77 | 2 | 325–327 |
© 2012 by the authors. licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Shariat, M.; Samsudin, M.W.; Zakaria, Z. Microwave Assisted Synthesis of 2,2'-Arylene-substituted Bis(4H-3,1-Benzoxazin-4-one) Derivatives Using the Complex Cyanuric Chloride/N,N-Dimethylformamide. Molecules 2012, 17, 11607-11615. https://doi.org/10.3390/molecules171011607
Shariat M, Samsudin MW, Zakaria Z. Microwave Assisted Synthesis of 2,2'-Arylene-substituted Bis(4H-3,1-Benzoxazin-4-one) Derivatives Using the Complex Cyanuric Chloride/N,N-Dimethylformamide. Molecules. 2012; 17(10):11607-11615. https://doi.org/10.3390/molecules171011607
Chicago/Turabian StyleShariat, Mehdi, Mohd Wahid Samsudin, and Zuriati Zakaria. 2012. "Microwave Assisted Synthesis of 2,2'-Arylene-substituted Bis(4H-3,1-Benzoxazin-4-one) Derivatives Using the Complex Cyanuric Chloride/N,N-Dimethylformamide" Molecules 17, no. 10: 11607-11615. https://doi.org/10.3390/molecules171011607
APA StyleShariat, M., Samsudin, M. W., & Zakaria, Z. (2012). Microwave Assisted Synthesis of 2,2'-Arylene-substituted Bis(4H-3,1-Benzoxazin-4-one) Derivatives Using the Complex Cyanuric Chloride/N,N-Dimethylformamide. Molecules, 17(10), 11607-11615. https://doi.org/10.3390/molecules171011607




