Comparing Conventional and Supercritical Extraction of (−)-Mammea A/BB and the Antioxidant Activity of Calophyllum brasiliense Extracts
Abstract
:1. Introduction
2. Results and Discussion
2.1. Overall Yield of Extraction
| Supercritical fluid extraction | Soxhlet extraction | |||||
|---|---|---|---|---|---|---|
| Operational conditions | Yield (%) a | Solvent | Yield (%) a | |||
| T (K) | Pressure (MPa) | CO2 density (g/cm³) | Mesh 30 | Mesh 50 | ||
| 313 | 10.92 | 0.6813 | 1.4 ± 0.02 | 1.6 ± 0.01 | ||
| 313 | 15.00 | 0.7811 | 2.0 ± 0.06 | 2.2 ± 0.04 | Dichloromethane | 4.3 ± 0.03 |
| 333 | 17.67 | 0.6813 | 2.2 ± 0.04 | 2.3 ± 0.05 | Hexane | 3.9 ± 0.01 |
| 333 | 24.41 | 0.7811 | 2.9 ± 0.02 | 2.9 ± 0.06 | ||
| 313 | 25.00 | 0.8802 | 2.8 ± 0.02 | 2.8 ± 0.04 | ||


2.2. (-) Mammea A/BB Content
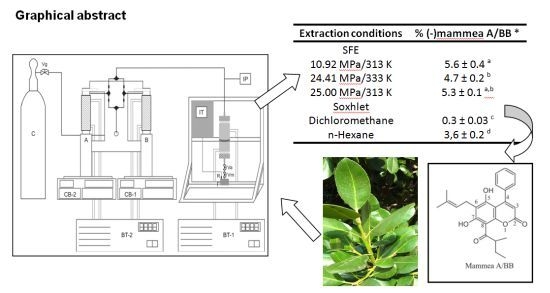
| Extraction conditions | % (−)-mammea A/BB * |
|---|---|
| SFE | |
| 10.92 MPa/313 K | 5.6 ± 0.4 a |
| 24.41 MPa/333 K | 4.7 ± 0.2 b |
| 25.00 MPa/313 K | 5.3 ± 0.1 a,b |
| Soxhlet | |
| Dichloromethane | 0.3 ± 0.03 c |
| n-Hexane | 3.6 ± 0.2 d |
2.3. Total Phenols
| Extraction condition | Mean Absorbance | Total phenols (mg of GAE/g of extract) |
|---|---|---|
| SFE | ||
| 10.92 MPa/313 K | 0.126 ± 0.01 | 15.06 ± 1.75 |
| 24.41 MPa/333 K | 0.230 ± 0.02 | 26.98 ± 2.90 |
| 25.00MPa/313 K | 0.285 ± 0.01 | 33.29 ± 1.75 |
| Soxhlet | ||
| Dichloromethane | 0.360 ± 0.01 | 41.89 ± 1.75 |
| n-Hexane | 0.159 ± 0.01 | 18.84 ± 1.75 |
2.4. Antioxidant Activity- DPPH Method
| Antioxidant Acitivity Percentage (AA%) | IC50 | ||||||
|---|---|---|---|---|---|---|---|
| Concentration of the extracts (µg/mL) | (µg/mL) | ||||||
| Extraction conditions | 25 | 33.33 | 50 | 150 | 250 | 350 | |
| SFE | |||||||
| 10.92 MPa/313 K | 15.40 | 18.48 | 31.22 | 50.12 | 69.03 | 79.54 | 149.35 a |
| 24.41 MPa/333 K | 12.44 | 17.30 | 21.59 | 50.09 | 66.08 | 78.08 | 149.13 a |
| 25.00MPa/313 K | 13.60 | 17.29 | 28.95 | 54.91 | 68.17 | 76.18 | 131.73 a |
| Soxhlet | |||||||
| Dichloromethane | 3.94 | 9.00 | 20.66 | 40.41 | 58.52 | 75.60 | 206.58 b |
| n-Hexane | 8.02 | 9.39 | 18.21 | 39.90 | 52.34 | 60.46 | 242.84 b |
2.5. Mathematical Modeling
| Parameters | Extraction condition | ||||
|---|---|---|---|---|---|
| P = 10.92 MPa | P = 15.00 MPa | P = 17.67 MPa | P = 24.41 MPa | P = 25.00 MPa | |
| T = 313 K | T = 313 K | T = 333 K | T = 333 K | T = 313 K | |
| Qf (mL/min) | 3 | 3 | 3 | 3 | 3 |
| m (g) | 20.0681 | 20.0057 | 20.0136 | 20.0035 | 20.0028 |
| ε | 0.87 | 0.85 | 0.85 | 0.85 | 0.85 |
| ρCO2 (g/mL) | 0.6813 | 0.7811 | 0.6813 | 0.7811 | 0.8802 |
| ρbed (g/mL) | 0.1169 | 0.1165 | 0.1166 | 0.1165 | 0.1165 |
| u (cm/min) | 0.5405 | 0.5533 | 0.5533 | 0.5533 | 0.5533 |
| Ceq (g/mL) | 7.768 × 10−4 | 1.271 × 10−3 | 1.385 × 10−3 | 2.007 × 10−3 | 2.739 × 10−3 |
| k (mL/g min) | 3.59 | 3.27 | 3.73 | 6.12 | 3.19 |

3. Experimental
3.1. Pre-treatment of the Vegetal Matrix
3.2. Extraction- Organic Solvent Extraction
3.3. Extraction- Supercritical Fluid Extraction (SFE)

| Pressure (MPa) | Temperature (k) | Density (g/mL) | Flow (mL/min) |
|---|---|---|---|
| 10.92 | 313 | 0.6813 | 3 |
| 15.00 | 313 | 0.7811 | 3 |
| 17.67 | 333 | 0.6813 | 3 |
| 24.41 | 333 | 0.7811 | 3 |
| 25.00 | 313 | 0.8802 | 3 |
3.4. Quantification of (−)-Mammea A/BB
3.5. Total Phenol Contents
3.6. Antioxidant Activity
3.7. Mathematical Modeling





 is the calculated extracted mass,
is the calculated extracted mass,  is the mass experimentally obtained, n exp is the number of experimental data of the kinetic curve.
is the mass experimentally obtained, n exp is the number of experimental data of the kinetic curve.4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Stevens, P.F. A revision of the Old World species of Calophyllum (Guttiferae). J. Arnold Arbor. 1980, 61, 117–699. [Google Scholar]
- Cronquist, A. An Integrated System of Classification of Flowering Plants; Columbia University Press: New York, NY, USA, 1981; p. 337. [Google Scholar]
- Corrêa, M.P. Dicionário das plantas úteis do Brasil e das exóticas cultivadas; Imprensa Nacional: Rio de Janeiro, Brazil, 1978; Volume 61, pp. 117–699. [Google Scholar]
- Reyes-Chilpa, R.; Jimenez-Estrada, M.; Estrada-Muñiz, E. Antifungal Xanthones from Calophyllum brasiliensis Heartwood. J. Chem. Ecol. 1997, 23, 1901–1911. [Google Scholar] [CrossRef]
- Mckee, T.C.; Covington, C.; Fuller, R.W.; Bokesch, L.R.; Young, S.; Cardellina, J.H.; Kadushin, M.R.; Soejarto, D.D.; Stevens, P.F.; Cragg, G.M.; Boyd, M.R. Pyranocoumarins from Tropical species of the genus Calophyllum: A chemotaxonomic study of extracts in the Natural Cancer Institute Collection. J. Nat. Prod. 1998, 61, 1252–1256. [Google Scholar] [CrossRef]
- Ali, M.S.; Mahmud, S.; Perveen, S.V.; Rizwani, G. Epimeers from the leaves of Calophylluminophyllum. Phytochemistry 1999, 50, 1385–1389. [Google Scholar] [CrossRef]
- Morel, C.; Séraphin, D.; Oger, J.M.; Litaudon, M.; Sévenet, T.; Richomme, P.; Bruneton, J. New xanthones from Calophyllumcaledonicum. J. Nat. Prod. 2000, 63, 1471–1474. [Google Scholar] [CrossRef]
- Silva, K.L.; Santos, A.R.S.; Mattos, P.E.O.; Yunes, R.A.; Dellemonache, F.; Cechinel-Filho, V. Chemical composition and analgesic activity of Calophyllumbrasiliense. Therapie 2001, 56, 431–434. [Google Scholar]
- Sartori, N.T.; Canapelle, D.; Sousa, P.T., Jr.; Martins, D.T.O. Gastroprotective effect from Calophyllumbrasiliense Camb. bark on experimental gastric lesions in rats and mice. J. Ethnopharmacol. 1999, 67, 149–156. [Google Scholar] [CrossRef]
- Lewis, W.H. Medical Botany – Plants Affecting Man’s Health; John Wiley & Sons: New York, NY, USA, 1977. [Google Scholar]
- Duke, J.A.; Martinez, R.V. Amazonian Ethnobotanical Dictionary; CRC Press: Boca Raton, MD, USA, 1994. [Google Scholar]
- Vasquez, M.R. Useful plants of Amazonian Peru; Second Draft; Filled with USDA’s National Agricultural Library: Beltsville, MD, USA, 1990. [Google Scholar]
- Rutter, R.A. Catalogo de plantas utiles de Ia Amazonia Peruana; Instituto Lingüístico de Verano: Yarinacocha, Peru, 1990. [Google Scholar]
- Cottiglia, F.; Dhanapal, B.; Sticher, O.; Heilmann, J. New Chromanone acids with Antibacterial activity from Calophyllum brasiliense. J. Nat. Prod. 2004, 67, 537–541. [Google Scholar] [CrossRef]
- Pretto, J.B.; Cechinel-Filho, V.; Noldin, V.F.; Sartori, M.R.K.; Isaias, D.E.B.; Cruz, A.B. Antimicrobial Activity of Fractions and Compounds from Calophyllum brasiliense (Clusiaceae/Guttiferae). Z. Naturforsch. 2004, 59, 657–662. [Google Scholar]
- Reyes-Chilpa, R.; Estrada-Muñiz, E.; Apan, T.R.; Amekraz, B.; Aumelas, A.; Jankowski, C.K.; Vázquez-Torres, M. Cytotoxic effects of mammea type coumarins from Calophyllum brasiliense. Life Sci. 2004, 75, 1635–1647. [Google Scholar] [CrossRef]
- Ito, C.; Itoigawa, M.; Mishina, Y.; Cechinel-Filho, V.; Enjo, F.; Tokuda, H.; Nishino, H.; Furukawa, H. Structure of three new coumarins and cancer chemopreventive activity of 4- substituted coumarins. J. Nat. Prod. 2003, 66, 368–371. [Google Scholar] [CrossRef]
- Huerta-Reyes, M.; Basualdo, M.C.; Abe, F.; Jimenez-Estrada, M.; Soler, C.; Reyes-Chilpa, R. HIV-1 inhibitory compounds from Calophyllumbrasiliense leaves. Biol. Pharm. Bull. 2004, 27, 1471–1475. [Google Scholar] [CrossRef]
- Brenzan, M.A.; Nakamura, C.V.; Filho, B.P.D.; Ueda-Nakamura, T.; Young, M.A.M.; Cortez, D.A.G. Antileishmanial activity of crude extract and coumarin from Calophyllumbrasiliensis leaves against Leishmaniaamazonensis. Parasitol. Res. 2007, 101, 715–722. [Google Scholar] [CrossRef]
- Honda, P.A.; Ferreira, I.C.P.; Cortez, D.A.G.; Amado, C.A.B.; Silveira, T.G.V.; Brenzan, M.V.C. Efficacy of components from leaves of Calophyllumbrasiliense against Leishmania (Leishmania) amazonensis. Phytomedicine 2010, 17, 333–338. [Google Scholar] [CrossRef]
- Ito, C.; Itoigawa, M.; Mishina, Y.; Cechinel-Filho, V.; Mukaianaka, T.; Tokuda, H.; Nishino, H.; Furukawa, H.J. Chemical constituents of Calophyllumbrasiliensis: Structure elucidation of seven new xanthones and their cancer chemopreventive activity. J. Nat. Prod. 2002, 65, 267–272. [Google Scholar] [CrossRef]
- Cavero, S.; García-Risco, M.R.; Marín, F.R.; Jaime, L.; Santoyo, S.; Señoráns, F.V.; Reglero, G.; Ibañez, E. Supercritical fluid extraction of antioxidant compounds from oregano. J. Supercrit. Fluids 2006, 38, 62–69. [Google Scholar] [CrossRef]
- Danh, L.T.; Mammucari, R.; Truong, P.; Fostera, N. Response surface method applied to supercritical carbon dioxide extraction of Vetiveriazizanioides essential oil. Chem. Eng. J. 2009, 155, 617–626. [Google Scholar] [CrossRef]
- Casas, L.; Mantell, C.; Rodríguez, M.; Torres, A.; Macías, F.A. Extraction of natural compounds with biological activity from sunflower leaves using supercritical carbon dioxide. Chem. Eng. J. 2009, 152, 301–306. [Google Scholar] [CrossRef]
- Macías-Sánchez, M.D.; Serrano, C.M.; Rodríguez, M.R.; Ossa, E.M. Kinetics of the supercritical fluid extraction of carotenoids from microalgae with CO2 and ethanol as cosolvent. Chem. Eng. J. 2009, 150, 104–113. [Google Scholar] [CrossRef]
- Lin, M.C.; Tsai, M.J.; Wen, K.C. Supercritical fluid extraction of flavonoids from Scutellariae Radix. J. Chrom. A 1999, 830, 387–395. [Google Scholar] [CrossRef]
- Noldin, V.F.; Isaias, D.B.; Cechinel-Filho, V. Gênero Calophyllum: importância química e farmacológica. Quím. Nova. 29, 549–554.
- Ruiz-Marcial, C.; Reyes-Chilpa, R.; Estrada, E.; Reyes-Esparza, J.; Fariña, G.G.; Rodriguez-Fragoso, L. Antiproliferative, cytotoxic and antitumour activity of coumarins isolated from Calophyllumbrasiliense. J. Pharm. Pharmacol. 2007, 59, 719–725. [Google Scholar] [CrossRef]
- Gasparotto-Júnior, A.; Brenzan, M.A.; Piloto, I.C.; Cortez, D.A.G. Estudo fitoquímico e avaliação da atividade moluscicida do Calophyllum brasiliense CAMB (CLUSIACEAE). Quim. Nova 2005, 28, 575–578. [Google Scholar] [CrossRef]
- Reyes-Chilpa, R.; Estrada-Muñiz, E.; Vega-Ávila, E.; Abe, F.; Kinjo, J.; Hernandez-Ortega, S. Trypanocidal constituents in plants: 7. Mammea-type coumarins. Memórias do Instituto Oswaldo Cruz. 2008, 103, 431–436. [Google Scholar] [CrossRef]
- Benellia, P.; Riehlb, C.A.S.; Smania Junior, A.; Smaniac, E.F.A.; Ferreira, S.R.S. Bioactive extracts of orange (Citrus sinensis L. Osbeck) pomace obtained by SFE and low pressure techniques: Mathematical modeling and extract composition. J. Supercrit. Fluids 2010, 55, 132–141. [Google Scholar] [CrossRef]
- dos Santos Freitas, L.; Oliveira, J.V.; Dariva, C.; Jacques, R.A.; Caramão, E.B. Extraction of grape seed oil using compressed carbon dioxide and propane: Extraction yields and characterization of free glycerol compounds. J. Agric. Food Chem. 2008, 56, 2558–2564. [Google Scholar] [CrossRef]
- Rodrigues, M.R.A.; Krause, L.C.; Caramão, E.B.; dos Santos, J.G.; Dariva, C.; de Oliveira, J.V. Chemical composition and extraction yield of the extract of Origanumvulgare obtained from sub and supercritical CO2. J. Agric. Food Chem. 2004, 52, 3042–3047. [Google Scholar] [CrossRef]
- Reynertson, K.A.; Basile, M.J.; Kennelly, E.J. Antioxidant potential of seven myrtaceous fruits. Ethnobot. Res. Applic. 2005, 3, 25–35. [Google Scholar]
- Instituto Adolf Lutz, Normas Analíticas do Instituto Adolfo Lutz: Métodos químicos e físicos para análise de alimentos; IAL: São Paulo, Brazil, 2000; p. 118.
- Díaz-Reinoso, B.; Moure, A.; Domínguez, H.; Parajó, J.C. Supercritical CO2 Extraction and Purification of Compounds with Antioxidant Activity. J. Agric. Food Chem. 2006, 54, 2441–2469. [Google Scholar] [CrossRef]
- Zancan, K.C.; Marques, M.O.M.; Petenate, A.J.; Meireles, M.A.A. Extraction of ginger (Zingiberofficinale Roscoe) oleoresin with CO2 and cosolvents: A study of the antioxidant action of the extracts. J. Supercrit. Fluids 2002, 24, 57–76. [Google Scholar] [CrossRef]
- Brenzan, M.A.; Nakamura, C.V.; Filho, B.P.D.; Ueda-Nakamura, T.; Young, M.C.M.; Mitsui, M.L.; Cortez, D.A.G. Quantitative and qualitative analysis of (-) mammea a/bb coumarin in extracts of Calophyllumbrasiliense Cambess (clusiaceae) by HPLC. J. Liq. Chrom. Relat. Technol. 2010, 33, 283–295. [Google Scholar]
- Meda, A.; Lamien, C.E.; Romito, M.; Millogo, J.; Nacoulma, O.G. Determination of the total phenolic, flavonoid and proline contents in Burkina Fasan honey, as well as their radical scavenging activit. Food Chem. 2005, 91, 571–577. [Google Scholar] [CrossRef]
- Blois, M.S. Antioxidant determinations by the use of a stable free radical. Nature 1958, 18, 1199–1200. [Google Scholar] [CrossRef]
- Brand-Willians, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. Lebensm.-Wiss. u.-Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Corso, M.P.; Fagundes-Klen, M.R.; Silva, E.A.; Cardozo-Filho, L.; Santos, J.N.; Freitas, L.S.; Dariva, C. Extraction of sesame seed (Sesamunindicum L.) oil using compressed propane and supercritical carbon dioxide. J. Supercrit. Fluid. 2010, 52, 56–61. [Google Scholar] [CrossRef]
- De Souza, A.T.; Benazzi, T.L.; Grings, M.B.; Cabral, V.; da Silva, E.A.; Cardozo-Filho, L.; Antunes, O.A.C. Supercritical extraction process and phase equilibrium of Candeia (Eremanthuserythropappus) oil using supercritical carbon. J. Supercrit. Fluid. 2008, 47, 182–187. [Google Scholar] [CrossRef]
- Sample Availability: Sample of the compound (−)-mammea A/BB is available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gonçalves, R.M.; Lemos, C.O.T.; Leal, I.C.R.; Nakamura, C.V.; Cortez, D.A.G.; Da Silva, E.A.; Cabral, V.F.; Cardozo-Filho, L. Comparing Conventional and Supercritical Extraction of (−)-Mammea A/BB and the Antioxidant Activity of Calophyllum brasiliense Extracts. Molecules 2013, 18, 6215-6229. https://doi.org/10.3390/molecules18066215
Gonçalves RM, Lemos COT, Leal ICR, Nakamura CV, Cortez DAG, Da Silva EA, Cabral VF, Cardozo-Filho L. Comparing Conventional and Supercritical Extraction of (−)-Mammea A/BB and the Antioxidant Activity of Calophyllum brasiliense Extracts. Molecules. 2013; 18(6):6215-6229. https://doi.org/10.3390/molecules18066215
Chicago/Turabian StyleGonçalves, Renata Menoci, Caroline Ortega Terra Lemos, Ivana Correa Ramos Leal, Celso Vataru Nakamura, Diógenes Aparício Garcia Cortez, Edson Antonio Da Silva, Vladimir Ferreira Cabral, and Lúcio Cardozo-Filho. 2013. "Comparing Conventional and Supercritical Extraction of (−)-Mammea A/BB and the Antioxidant Activity of Calophyllum brasiliense Extracts" Molecules 18, no. 6: 6215-6229. https://doi.org/10.3390/molecules18066215
APA StyleGonçalves, R. M., Lemos, C. O. T., Leal, I. C. R., Nakamura, C. V., Cortez, D. A. G., Da Silva, E. A., Cabral, V. F., & Cardozo-Filho, L. (2013). Comparing Conventional and Supercritical Extraction of (−)-Mammea A/BB and the Antioxidant Activity of Calophyllum brasiliense Extracts. Molecules, 18(6), 6215-6229. https://doi.org/10.3390/molecules18066215