Abstract
In this study we report the synthesis of C5/C6-fused uridine phosphonates that are structurally related to earlier reported allosteric P2Y2 receptor ligands. A silyl-Hilbert-Johnson reaction of six quinazoline-2,4-(1H,3H)-dione-like base moieties with a suitable ribofuranosephosphonate afforded the desired analogues after full deprotection. In contrast to the parent 5-(4-fluoropheny)uridine phosphonate, the present extended-base uridine phosphonates essentially failed to modulate the P2Y2 receptor.
1. Introduction
Amongst the many physiological functions of nucleotides, their capacity to mediate signal transmission via cell-surface receptors was discovered in 1929 [1]. Purinergic receptors are subdivided into two families: metabotropic P2Y receptors (P2YRs) and fast acting P2X ion channels [2]. The P2X receptors generally respond to the native agonist ATP, whereas P2YRs can be activated by ATP, UTP, UDP and UDP-glucose. The P2YRs family consist of eight members, which can be divided in two subgroups based on receptor structure and second messenger system: P2Y1,2,4,6,11 and P2Y12,13,14 [3]. Ligand preferences of the eight P2YRs have been well established: P2Y1 (ADP), P2Y2 (UTP=ATP), P2Y4 (UTP), P2Y6 (UDP), P2Y11 (ATP, NAD+), P2Y12 (ADP), P2Y13 (ADP), and P2Y14 (UDP, UDP nucleotide sugars) [4].
The P2Y2 receptor (P2Y2R) is responsible for many physiological functions and therefore attracts considerable attention for developing therapeutics. For example, the P2Y2R agonist diquafosol, which stimulates the secretion of chloride and water by epithelial cells, is approved in Japan for treatment of dry eye disease [5]. P2Y2R activation protects cardiomyocytes from hypoxia in vitro and reduces post-ischemic myocardial damage in vivo [2]. Furthermore, the P2Y2R is a promising target for the treatment of neurodegenerative diseases, including Alzheimer’s disease [6]. Recently, ATP activation of the P2Y2R was also shown to retard bone mineralization [1].
The development of selective P2Y2R agonists and antagonists is a challenging task. Introduction of a 2'-amino and a 2-thio modification in UTP afforded analogue 1 (Figure 1) with enhanced potency and selectivity (versus P2Y4R) [7]. Unfortunately, most P2Y2R agonists contain a polyphosphate group, which is easily hydrolyzed by ecto-nucleotidases [4]. We recently reported a series of stable 5-substituted uridine phosphonate analogues, some of which (e.g., 2, Figure 1) showed promising allosteric partial agonistic activity at P2Y2R [8]. Given the interesting profile of the 5-aryluridine analogues we envisaged modifying this nucleobase to a more rigid structure as a way to potentially enhance affinity for the P2Y2R. In this light we opted for a ring transformation from a 5-aryluracil to a quinazoline-2,4-dione motif, analogous to the biphenyl-naphthalene ring transformations often encountered in medicinal chemistry [9]. The first series of 5,6-annulated uridine analogues were based on the aromatic Topliss-scheme [6-H (3a), Cl (3c), OMe (3e) and Me (3d)] [10]. Additionally the 6-fluorine derivative 3b was chosen based on the notable potency of parent compound 2 which bears a fluorine atom. Finally two nucleobases involving substitution of the benzene ring for a thiophene (3f) or a naphthalene core (3g) were included to gauge the effect of bioisosteric replacement or ring expansion of the nucleobase on the affinity for the P2Y2R.
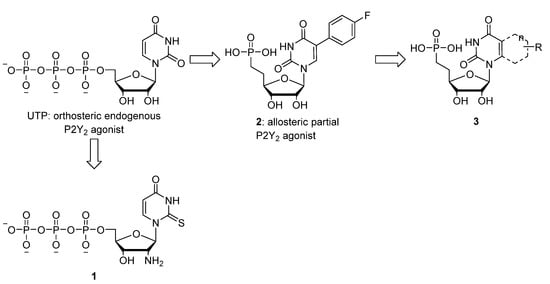
Figure 1.
Structure of UTP, the selective P2Y2 agonist 1, the allosteric partial agonist 2 and the target nucleoside phosphonates 3.
Figure 1.
Structure of UTP, the selective P2Y2 agonist 1, the allosteric partial agonist 2 and the target nucleoside phosphonates 3.
2. Results and Discussion
2.1. Synthesis
To gain access to the target C5/C6-fused uridine phosphonates we first prepared the required “base” moieties 4a–g by condensation of the appropriate ortho-amino aromatic carboxylic acids and urea at high temperature [11,12,13].
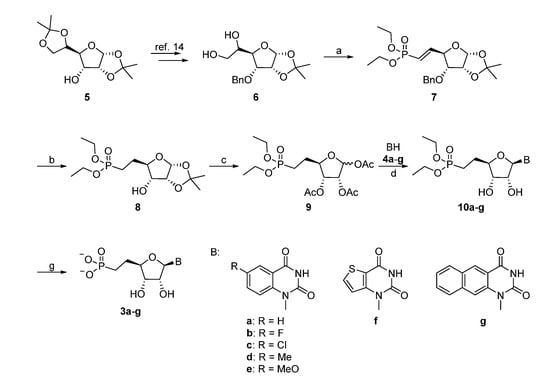
The synthesis of the appropriate glycosyl donor started from the known diol 6, which was obtained in two steps from the commercial 1,2:5,6-O-diisopropylidene-α-d-allofuranose (5) following a known procedure (Scheme 1) [14]. Cleavage of the resulting vicinal diol with sodium periodate gave the aldehyde [15], which was used without purification in the subsequent Wittig-type reaction. During formation of the diethylvinylphosphonate 7, C-4 may be prone to epimerization, which could be limited by performing the reaction at lower temperature. The yield for the conversion of 6 to 7 was significantly increased by using dichloromethane instead of ethanol for the periodate reaction. Hydrogenation with 10% Pd/C at atmospheric pressure allowed to simultaneous reduction of the double bond and removal of the benzyl group. One-pot deprotection of the isopropylidene group and acetylation of 8 afforded 9 (with an anomeric ratio of ca. 4:1 in favor of the α anomer), which was used in a silyl-Hilbert-Johnson reaction with the silylated benzo- or thieno[3,2-d]pyrimidine-2,4-diones to give compounds 10a–g [16]. When HMDS was used as the silylating agent, careful removal of the silylating agents prior to the addition of the base and trimethylsilyltrifluoromethane sulfonate was required. Hence, the use of BSTFA for in situ silylation was preferred, despite the fact that it generally gave lower yields. Final deacetylation, followed by treatment with TMSBr/DCM afforded the desired nucleoside phosphonate analogues 3a–g.
Scheme 1.
Synthesis of the Target Extended Uridine-5'-methylenephosphonates 3a–g.
Scheme 1.
Synthesis of the Target Extended Uridine-5'-methylenephosphonates 3a–g.
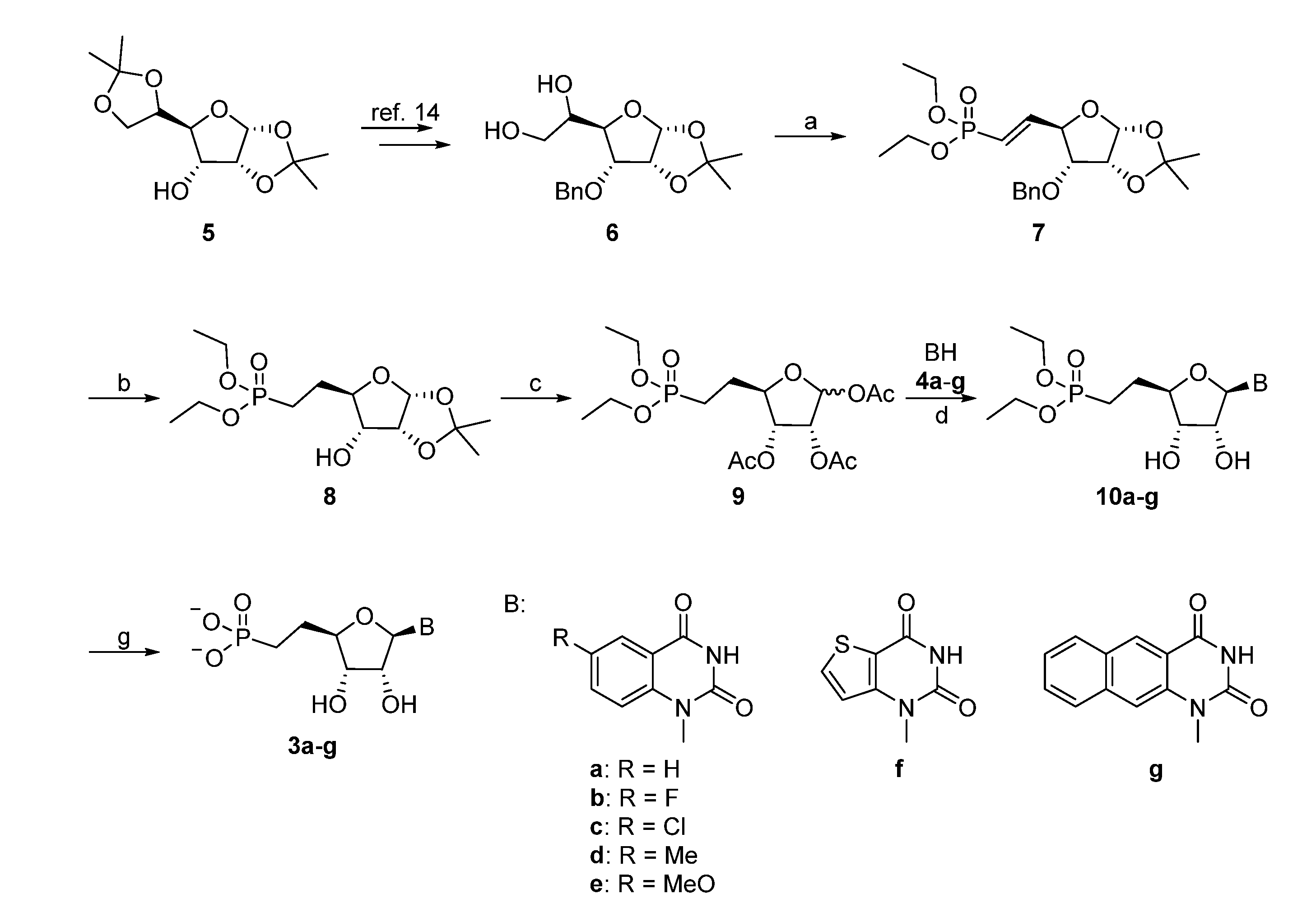
Reagents and conditions: (a) (i) NaIO4, DCM, H2O, rt; (ii) tetraethyl methylenediphosphonate, NaH, THF, 0 °C → rt; (b) H2, 10% Pd/C, rt; (c) (i) 60% AcOH, 85 °C, overnight; (ii) Ac2O, pyridine, overnight; (d) (i) BSTFA, ACN, 65 °C, 2 h; (ii) TMSOTf, CH3CN; (iii) NaOMe/MeOH, rt; (e) TMSBr, DCM, rt.
2.2. Pharmacological Evaluation
Agonist and antagonist activities of the C5/C6-fused uridine phosphonates at the P2Y2 receptor were determined measuring PLC-dependent phosphoinositide hydrolysis in 1321N1 human astrocytoma cells stably expressing the human P2Y2 receptor (1321N1-P2Y2 cells) [17,18] (Figure 2).
Compared to compound 2, the current extended uridine phosphonates were found to stimulate the P2Y2 receptor to a lesser extent, indicating that rigidifying the base moiety is unfavorable for binding to a presumed allosteric binding site. It can be concluded that structural flexibility of the 5-substituent of the earlier discovered series of allosteric P2Y2 agonists is crucial for allosteric agonist activity.
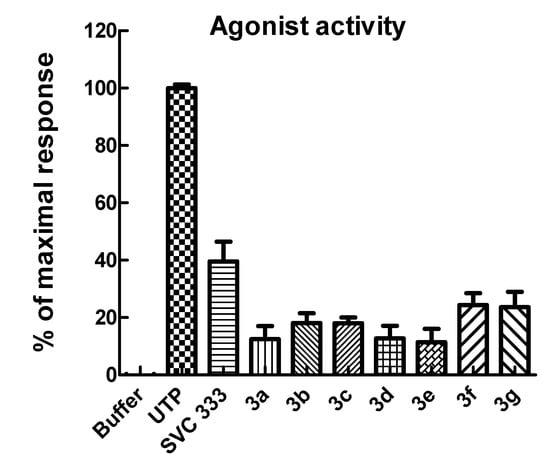
Figure 2.
Inositol phosphate production in 1321N1 cells stably expressing the human P2Y2 receptor. After labeling with [3H]myo-inositol overnight, the cells were treated with agonists for 30 min at 37 °C in the presence of 10 mM LiCl, and inositol phosphate accumulation was quantified. The concentration of all phosphonate analagues was 30 μM and of UTP was 3 μM. The data are presented as the mean ± S.E.M. and are representative of results obtained in three separate experiments.
Figure 2.
Inositol phosphate production in 1321N1 cells stably expressing the human P2Y2 receptor. After labeling with [3H]myo-inositol overnight, the cells were treated with agonists for 30 min at 37 °C in the presence of 10 mM LiCl, and inositol phosphate accumulation was quantified. The concentration of all phosphonate analagues was 30 μM and of UTP was 3 μM. The data are presented as the mean ± S.E.M. and are representative of results obtained in three separate experiments.
The possibility that the modified uridine 5'-phosphonate analogues could inhibit the effect of UTP by displacing it from the orthosteric site was examined by assessing their capacity at high concentrations to inhibit the effect of a near-maximal concentration of UTP (Figure 3). None of the phosphonate analogues gave an inhibitory effect on the action of UTP.
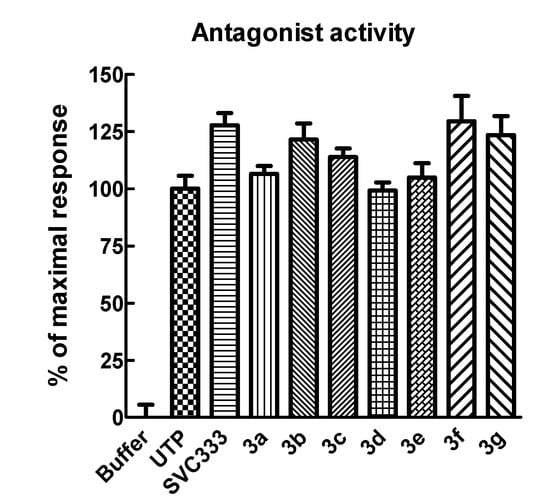
Figure 3.
Effect of high concentrations of phosphonate analogues 3a–g on the P2Y2 receptor-mediated activity of UTP. The effects of 30 μM concentrations of the indicated phosphonates were tested in the presence of 100 μM UTP in 1321N1-P2Y2 cells.
Figure 3.
Effect of high concentrations of phosphonate analogues 3a–g on the P2Y2 receptor-mediated activity of UTP. The effects of 30 μM concentrations of the indicated phosphonates were tested in the presence of 100 μM UTP in 1321N1-P2Y2 cells.
3. Experimental
3.1. General Methods and Materials
All reagents were from standard commercial sources and of analytic grade. All reaction were performed under argon atmosphere except specified otherwise. Dry solvents were directly acquired from commercial sources. Precoated Merck Silica Gel F254 plates were used for TLC. Spots were examined under ultraviolet light at 254 nm and further visualized by sulfuric acid-anisaldehyde spray. Column chromatography was performed on silica gel (200–400 mm, 60 Å). 1H, 13C, and 31P-NMR spectra were recorded in CDCl3, DMSO-d6, or D2O on a Varian Mercury 300 MHz spectrometer (Varian, Palo Alto, CA, USA). Chemical shifts are given in parts per million (ppm δ), δ relative to residual solvent peak or TMS for 1H and 13C and to external D3PO4 for 31P. Structural assignment was confirmed with COSY, HSQC and HMBC. Exact mass measurements were performed on a LCT Premier XE orthogonal time-of flight spectrometer with API-ES source (Waters, Zellik, Belgium) Waters LCT Premier XETM Time of flight (TOF) instrument. Samples were infused in a CH3CN/H2O (1:1) mixture at 10 mL/min.
Diethyl ((E)-2-((3aR,5R,6R,6aR)-6-(benzyloxy)-2,2-dimethyltetrahydrofuro[2,3-d][1,3]dioxol-5-yl)-vinyl)phosphonate (7): NaIO4 (0.52 g, 2.22 mmol) was added to a stirred solution of compound 6 in CH2Cl2 (15 mL) and H2O (10 mL) and the reaction mixture was stirred for 0.5 h at rt. The mixture was then filtrated and the filtrate was extracted with CH2Cl2. The combined organic layers were concentrated in vacuo to give the crude aldehyde, which was used in the next step without further purification. To a stirred suspension of NaH (0.31 g, 7.81 mmol) in dry THF (10 mL) was added tetraethyl methylene bisphosphonate (1.41 g, 4.88 mmol) under argon. After 0.5 h the mixture was cooled in an ice bath and a solution of the crude aldehyde in THF (10 mL) was added. The reaction mixture was allowed to warm to rt and stirred overnight. The crude mixture was diluted with water (20 mL) and extracted with ethyl acetate (3 × 20 mL). The combined organic layers were dried (Na2SO4) and concentrated in vacuo. Silica gel column chromatography (CH2Cl2/MeOH 97:3) afforded compound 7 as a colorless oil (0.77 g, 84.2%). 1H-NMR (300 MHz, CDCl3) δ: 1.28–1.35 (m, 6H, OCH2CH3), 1.36, 1.60 (2s, 6H, C(CH3)2), 3.53 (dd, J = 9.2, 4.2 Hz, 1H, H-3'), 3.98–4.16 (m, 4H, OCH2CH3), 4.55–4.66 (m, 3H, H-2',H-4', OCH2Ph), 4.70–4.77 (m, 1H, OCH2Ph), 5.77 (d, J = 3.8 Hz, 1H, H-1'), 5.96–6.12 (m, 1H, CH=CH), 6.74–6.90 (m, 1H, CH=CH), 7.28–7.42 (m, 5H, Ph). 13C-NMR (75 MHz, CDCl3) δ: 16.47, 16.56 (OCH2CH3), 26.63, 26.93 (C(CH3)2), 61.97 (t, J = 4.7 Hz, OCH2CH3), 72.57 (OCH2Ph), 77.68 (d, J = 9.4 Hz, C-4'), 78.04 (C-2'), 81.93 (d, J = 1.7 Hz, C-3'), 104.15 (C-1'), 113.48 (C(CH3)2), 118.45 (d, J = 186.0 Hz, C-6'), 128.18 , 128.30, 128.68, 137.19 (Ph), 148.00 (d, J = 6.1 Hz, C-5'). 31P-NMR (CDCl3) δ: 17.49. HRMS (ESI): Calculated for C20H30O7P [(M+H)+], 413.1724; found, 413.1754.
Diethyl (2-((3aR,5R,6R,6aR)-6-hydroxy-2,2-dimethyltetrahydrofuro[2,3-d][1,3]dioxol-5-yl)ethyl) phosphonate (8): To a solution of compound 7 (0.91 g, 2.22 mmol) in MeOH was added 10% Pd/C (0.5 g). The reaction mixture was stirred for 2 h under hydrogen atmosphere at rt and filtered. The filtrate was evaporated under vacuum and the residue purified by silica gel column chromatography (CH2Cl2/MeOH 97:3) to yield compound 8 as a colorless oil (0.58 g, 81.5%). 1H-NMR (300 MHz, CDCl3) δ: 1.29–1.35 (m, 6H, OCH2CH3), 1.36, 1.56 (2s, 6H, OC(CH3)2), 1.79–2.12 (m, 4H, H-5'a, H-5'b, H-6'a and H-6'b), 3.62 (ddd, J = 10.3, 8.7, 5.3 Hz, 1H, H-3'), 3.77 (ddd, J = 8.6, 6.7, 4.2 Hz, 1H, H-4'), 4.0–4.23 (m, 4H, OCH2CH3), 4.56 (dd, J = 5.0, 4.1 Hz, 1H, H-2'), 5.77 (d, J = 4.1 Hz, 1H, H-1); 13C-NMR (75 MHz, CDCl3) δ: 16.43, 16.50 (OCH2CH3), 20.61 (d, J = 141.7 Hz, C-6'), 24.99 (d, J = 5.0 Hz, C-5'), 26.40, 26.52 (C(CH3)2), 61.67 (d, J = 6.63 Hz, OCH2CH3), 75.55 (C-3'), 78.76 (C-2'), 79.49 (d, J = 15.8 Hz, C-4'), 103.65 (C-1'), 112.52 (C(CH3)2); 31P-NMR (CDCl3) δ: 31.66. HRMS (ESI): calculated for C13H26O7P [(M+H)+], 325.1411; found, 325.1429.
(3R,4R,5R)-5-(2-(Diethoxyphosphoryl)ethyl)tetrahydrofuran-2,3,4-triyl triacetate (9): A solution of compound 8 (0.45 g, 1.38 mmol) and 60% AcOH (10 mL) was heated at 80 °C under argon for 16 h. When the starting material was consumed completely, the reaction mixture was evaporated in vacuo and co-evaporated twice with AcCN. The residue was dissolved in dry pyridine (12 mL) at 0 °C under argon followed by dropwise addition of acetic anhydride (1.56 mL, 16.52 mmol). The reaction mixture was slowly warmed to rt and stirred overnight. Then it was concentrated and co-evaporated with toluene. The residue was purified by silica gel column chromatography (CH2Cl2/MeOH 95:5) to give 9 (0.47 g, 83.2%) as a low melting solid (α:β anomeric ratio = 41:9). α anomer: 1H-NMR (300 MHz, CDCl3) δ: 1.22–1.30 (m, 6H, OCH2CH3), 1.63–1.94 (m, 4H, H-5'a, H-5′b, H-6'a and H-6'b), 1.99, 2.02, 2.04 (s, 9H, 3COCH3), 3.96–4.08 (m, 4H, OCH2CH3), 4.08–4.17 (m, 1H, H-4'), 5.09 (dd, J = 7.0, 4.9 Hz, 1H, H-3'), 5.25 (d, J = 4.9 Hz, 1H, H-2'), 6.06 (s, 1H, H-1'). 13C-NMR (75 MHz, CDCl3) δ: 16.49, 16.57 (OCH2CH3), 20.58, 20.60 (COCH3), 21.80 (d, J = 143.3 Hz, C-6'), 21.15 (COCH3), 27.27 (d, J = 4.4 Hz, C-5'), 61.73, 61.82 (OCH2CH3), 73.27 (C-3'), 74.65 (C-2'), 81.27 (d, J = 18.2 Hz, C-4'), 98.24 (C-1'), 169.19, 169.52, 169.87 (CO). 31P-NMR (CDCl3) δ: 30.82. β anomer: 1H-NMR (300 MHz, CDCl3) δ: 1.32 (t, J = 7.0 Hz, 6H, OCH2CH3), 1.72–1.98 (m, 4H, H-5'a, H-5'b, H-6'a and H-6'b), 2.06, 2.10, 2.11 (s, 9H, 3 COCH3), 4.01–4.17 (m, 4H, OCH2CH3), 4.20–4.29 (m, 1H, H-4'), 5.02 (dd, J = 6.8, 3.7 Hz, 1H, H-3'), 5.19 (dd, J = 6.8, 4.6 Hz, 1H, H-2'), 6.36 (d, J = 4.6 Hz, 1H, H-1'). 13C-NMR (75 MHz, CDCl3) δ: 16.54, 16.62 (OCH2CH3), 20.43, 20.76, 21.17 (COCH3), 21.81 (d, J = 141.83 Hz, C-6'), 26.72 (d, J = 4.4 Hz, C-5'), 61.81, 61.88 (OCH2CH3), 69.95 (C-2′), 72.02 (C-3'), 83.06 (d, J = 17.1 Hz, C-4'), 93.95 (C-1'), 169.46, 169.80, 170.17 (CH3CO). 31P-NMR (CDCl3) δ: 30.57. ESI-HRMS for [C16H27O10P+H]+ calcd, 411.1420; found, 411.1436. HRMS (ESI): calculated for C16H28O10P [(M+H)+], 411.1415; found 411.1436.
3.2. General Procedures for the Silyl-Hilbert-Johnson Reaction to Synthesize Products 10a to 10g
Method 1: To a suspension of the appropriate quinazoline-2,4-(1H,3H)-dione (1.5 eq.) in dry acetonitrile (50 eq.) was added BSTFA (4 eq.) under argon. The solution was heated at 65 °C for 2 h. After cooling, a solution of triacetate 9 (1 eq.) in dry acetonitrile (50 eq.) and trimethylsilyl triflate (1.5 eq.) were added. The solution was stirred for 2 h at rt. The reaction was quenched with saturated aqueous NaHCO3 (50 mL for 1 mmol triacetate 9) and extracted with dichloromethane (50 mL for 1 mmol triacetate 9). The combined organic layer was washed with saturated aqueous NaHCO3 (3 × 50 mL for 1 mmol triacetate 9), dried over anhydrous MgSO4 and evaporated. The crude mixture was dissolved in MeOH (50 eq.) and 5.4 N sodium methoxide (8 eq.) in MeOH was added dropwise. After 1 h, the mixture was neutralized with acetic acid (10 eq.) and evaporated. Purification of the residue by silica-gel column chromatography (CH2Cl2/MeOH 97:3→95:5) gave the pure product as a white foam.
Method 2: To a suspension of appropriate quinazoline-2,4-(1H,3H)-dione (1.5 eq.) in hexamethyl-disilazane (50 eq.) was added trimethylsilyl chloride (0.7 eq.) and pyridine (10 eq.) under argon. The mixture was stirred at 130 °C overnight. The reaction mixture was evaporated to dryness under high vacuum. To the obtained residue a solution of triacetate 9 (1 eq.) in dry acetonitrile (50 eq.) was added under nitrogen followed by trimethylsilyl triflate (1.5 eq.). The solution was stirred for 2 h at rt. and the work up was similar as described in method 1.
Diethyl-(2-((2R,3S,4R,5R)-5-(2,4-dioxo-3,4-dihydroquinazolin-1(2H)-yl)-3,4-dihydroxy tetrahydrofuran-2-yl)ethyl)phosphonate (10a): Following Method 1 reaction between 9 (0.47 g, 1.14 mmol) and 4a (0.28 g, 1.71 mmol) yielded compound 10a (0.22 g, 50%). 1H-NMR (300 MHz, CDCl3) δ: 1.27 (dt, J = 18.3, 7.1 Hz, 6H, OCH2CH3), 1.79–2.15 (m, 4H, H-5'a, H-5'b, H-6'a and H-6'b), 2.15–2.36 (m), 4.45 (br s), 4.97 (br s, 2OH, NH), 3.88–3.97 (m, 1H, H-4'), 3.97–4.15 (m, 4H, OCH2CH3), 4.59 (t, J = 6.7 Hz, 1H, H-3'), 4.80–4.89 (m, 1H, H-2'), 5.96 (d, J = 2.0 Hz, 1H, H-1'), 7.19–7.27 (m, 1H), 7.45 (d, J = 8.7 Hz, 1H), 7.59–7.67 (m, 1H), 8.14 (d, J = 7.7 Hz, 1H, Ph). 13C-NMR (75 MHz, CDCl3) δ: 15.99–16.20 (m, OCH2CH3), 20.69 (d, J = 141.8 Hz, C-6'), 25.02 (d, J = 4.4 Hz, C-5'), 61.66 (dd, J = 12.2, 6.6 Hz, OCH2CH3), 71.95 (C-3'), 72.50 (C-2'), 82.32 (d, J = 17.7 Hz, C-4'), 91.68 (C-1'), 114.27,115.92, 123.41, 128.37, 135.12 and 140.98 (Ph), 149.74 (C-2), 162.04 (C-4). 31P-NMR (CDCl3) δ: 32.08. HRMS (ESI): calculated for C18H24N2O8P [(M–H)−], 427.1276; found, 427.1284.
Diethyl-(2-((2R,3S,4R,5R)-5-(6-fluoro-2,4-dioxo-3,4-dihydroquinazolin-1(2H)-yl)-3,4-dihydroxy-tetrahydrofuran-2-yl)ethyl)phosphonate (10b): Following Method 2 reaction between 9 (0.34 g, 0.84 mmol) and 4b (0.18 g, 1.01 mmol) yielded compound 10b (133.3 mg, 61.3%). 1H-NMR (300 MHz, CDCl3) δ: 1.29 (dt, J = 17.9, 7.1 Hz, 6H, OCH2CH3), 1.80–2.15 (m, 4H, H-5'a, H-5'b, H-6'a and H-6'b), 3.89–3.98 (m, 1H, H-4'), 3.98–4.16 (m, 4H, OCH2CH3), 4.29, 5.01, 10.31 (br s, 2OH, NH), 4.60 (t, J = 6.2 Hz, 1H, H-3'), 4.83 (dd, J = 6.2, 2.3 Hz, 1H, H-2'), 5.90 (d, J = 2.6 Hz, 1H, H-1'), 7.33–7.42 (m, 1H, Ph), 7.43–7.50 (m, 1H, Ph), 7.78–7.85 (m,1H, Ph). 13C-NMR (75 MHz, CDCl3) δ: 16.57 (d, J = 5.53 Hz, OCH2CH3), 21.14 (d, J = 140.1 Hz, C-6'), 25.52 (t, J = 5.3 Hz, C-5'), 62.14 (dd, J = 13.82, 6.6 Hz, OCH2CH3), 72.36 (C-3'), 73.01 (C-2'), 82.87 (d, J = 17.1 Hz, C-4'), 92.40 (C-1'), 114.10–114.57 (m, Ph), 116.86 (d, J = 7.2 Hz, Ph), 117.68–117.98 (m, Ph), 122.86–123.38 (m, Ph), 137.76 (Ph), 149.83 (C-2), 156.99–160.41 (m, Ph), 161.39 (C-4). 31P-NMR (CDCl3) δ: 32.71. HRMS (ESI): Calculated for C18H23FN2O8P [(M–H)−], 445.1182; found, 445.1188.
Diethyl-(2-((2R,3S,4R,5R)-5-(6-chloro-2,4-dioxo-3,4-dihydroquinazolin-1(2H)-yl)-3,4-dihydroxytetra- hydrofuran-2-yl)ethyl)phosphonate (10c): The reaction between 9 (0.34 g, 0.84 mmol) and 4c (0.25 g, 1.26 mmol) as described for method 1 to yield compound 10c (0.18 g, 45.1%). 1H-NMR (300 MHz, CDCl3) δ:1.28 (dt, J = 16.69, 7.03 Hz, 6H, OCH2CH3), 1.79–2.14 (m, 4H, H-5'a, H-5'b, H-6'a and H-6'b), 3.92 (q, J = 5.8 Hz, 1H, H-4'), 3.97–4.15 (m, 4H, OCH2CH3), 4.55 (t, J = 6.7 Hz, 1H, H-3'), 4.85 (dd, J = 6.15, 2.3 Hz, 1H, H-2'), 4.96, 10.49 (br s OH, NH), 5.89 (d, J = 2.6 Hz, 1H, H-1'), 7.40 (d, J = 9.4 Hz, 1H, Ph), 7.57 (dd, J = 9.1, 2.6 Hz, 1H, Ph), 8.04 (d, J = 2.3 Hz, 1H, Ph). 13C-NMR (75 MHz, CDCl3) δ: 16.44–16.63 (m, OCH2CH3), 21.13 (d, J = 141.0 Hz, C-6'), 25.48 (d, J = 4.4 Hz, C-5'), 62.13 (dd, J = 11.6, 6.6 Hz, OCH2CH3), 72.42 (C-3'), 72.80 (C-2'), 82.84 (d, J = 17.1 Hz, C-4'), 92.25 (C-1'), 116.51, 117.55, 128.08, 129.76, 135.46 and 139.87 (Ph), 149.85 (C-2), 161.33 (C-4). 31P-NMR (CDCl3) δ: 32.68. HRMS (ESI): calculated for C18H23ClN2O8P [(M−H)−], 461.0886; found, 461.0899.
Diethyl-(2-((2R,3S,4R,5R)-5-(6-methyl-2,4-dioxo-3,4-dihydroquinazolin-1(2H)-yl)-3,4-dihydroxytetra- hydrofuran-2-yl)ethyl)phosphonate (10d): The reaction between 9 (0.25 g, 0.61 mmol) and 4d (0.16 g, 0.91 mmol) as described for method 1 to yield compound 10d (125.2 mg, 46.5%). 1H-NMR (300 MHz, CDCl3) δ:1.28 (dt, J = 19.0, 7.1 Hz, 6H, OCH2CH3), 1.81–2.18 (m, 4H, H-5'a, H-5′b, H-6'a and H-6'b), 2.37 (s, 3H, CH3), 3.88–3.97 (m, 1H, H-4'), 3.97–4.16 (m, 4H, OCH2CH3), 4.28 (br s, 1H, OH), 4.56–4.68 (m, 1H, H-3'), 4.80–4.88 (m, 1H, H-2'), 4.98 (d, J = 5.0 Hz, 1H, OH), 5.94 (d, J = 2.3 Hz, 1H, H-1'), 7.34 (d, J = 8.8 Hz, 1H, Ph), 7.40–7.48 (m, 1H, Ph), 7.94 (s, 1H, Ph), 10.28 (s, 1H, NH). 13C-NMR (75 MHz, CDCl3) δ: 15.78–16.15 (m, OCH2CH3), 20.51 (d, J = 141.0 Hz, C-6'), 19.95 (CH3), 24.83 (d, J = 4.4 Hz, C-5'), 61.26–61.77 (m, OCH2CH3), 71.76 (C-3'), 72.55 (C-2'), 82.18 (d, J = 17.1 Hz, C-4'), 91.52 (C-1'), 113.98, 115.56, 127.99, 133.17, 135.95 and 138.69 (Ph), 149.56 (C-2), 161.96 (C-4). 31P-NMR (CDCl3) δ: 32.84. HRMS (ESI): calculated for C19H26N2O8P [(M−H)−], 441.1432; found, 441.1427.
Diethyl-(2-((2R,3S,4R,5R)-5-(6-methoxy-2,4-dioxo-3,4-dihydroquinazolin-1(2H)-yl)-3,4-dihydroxy- tetrahydrofuran-2-yl)ethyl)phosphonate (10e): The reaction between 9 (0.25 g, 0.61 mmol) and 4e (0.18 g, 0.91 mmol) as described for method 1 to yield compound 10e (122.7 mg, 44.0%). 1H-NMR (300 MHz, CDCl3) δ:1.21–1.35 (m, 6H, OCH2CH3), 1.79–2.17 (m, 4H, H-5'a, H-5'b, H-6'a and H-6'b), 3.86 (s, 3H, OCH3), 3.89–3.97 (m, 1H, H-4'), 3.98–4.15 (m, 4H, OCH2CH3), 4.63 (t, J = 6.7 Hz, 1H, H-3'), 4.85 (dd, J = 6.0, 2.5 Hz, 1H, H-2'), 5.04 (br s, OH), 5.92 (d, J = 2.3 Hz, 1H, H-1'), 7.20–7.25 (m, 1H, Ph), 7.39 (d, J = 9.7 Hz, 1H, Ph), 7.59 (d, J = 3.2 Hz, 1H, Ph), 10.27 (br s, 1H, NH). 13C-NMR (75 MHz, CDCl3) δ: 16.42–16.64 (m, OCH2CH3), 21.09 (d, J = 141.0 Hz, C-6'), 25.42 (d, J = 4.4 Hz, C-5'), 56.02 (OCH3), 62.08 (dd, J = 14.9, 6.6 Hz, OCH2CH3), 72.33 (C-3'), 73.08 (C-2'), 82.78 (d, J = 17.1 Hz, C-4'), 92.19 (C-1'), 109.63, 116.27, 117.12, 124.35 and 135.41 (Ph), 149.94 (C-2), 155.95 (Ph), 162.30 (C-4). 31P-NMR (CDCl3) δ: 32.82. HRMS (ESI): calculated for C19H26N2O9P [(M−H)−], 457.1381; found, 457.1390.
Diethyl-(2-((2R,3S,4R,5R)-5-(2,4-dioxo-3,4-dihydrothieno[3,2-d]pyrimidin-1(2H)-yl)-3,4-dihydroxy- tetrahydrofuran-2-yl)ethyl)phosphonate (10f): The reaction between 9 (0.25 g, 0.61 mmol) and 4f (0.15 g, 0.91 mmol) as described for method 1 to yield compound 10f (198.8 mg, 75.2%). 1H-NMR (300 MHz, CDCl3) δ:1.20–1.35 (m, 6H, OCH2CH3), 1.79–2.15 (m, 4H, H-5'a, H-5'b, H-6'a and H-6'b), 3.92 (q, J = 6.1 Hz, 1H, H-4'), 3.97–4.14 (m, 4H, OCH2CH3), 4.50 (t, J = 6.3 Hz, 2H, H-3', OH), 4.80 (dd, J = 5.9, 2.9 Hz, 1H, H-2'), 4.95 (br s, 1H, OH), 5.81 (d, J = 3.2 Hz, 1H, H-1'), 7.13 (d, J = 5.3 Hz, 1H, H-6), 7.73 (d, J = 5.6 Hz, 1H, H-7), 10.33 (br s, 1H, NH). 13C-NMR (75 MHz, CDCl3) δ: 16.43–16.62 (m, OCH2CH3), 21.15 (d, J = 141.0 Hz, C-6'), 25.54 (d, J = 4.4 Hz, C-5'), 62.12 (dd, J = 14.4, 6.6 Hz, OCH2CH3), 72.39 (C-3'), 72.71 (C-2'), 83.14 (d, J = 17.1 Hz, C-4'), 93.87 (C-1'), 114.38 (C-4a), 116.94 (C-6), 135.47 (C-7), 146.87 (C-7a), 151.09 (C-2), 158.53 (C-4). 31P-NMR (CDCl3) δ: 32.63. HRMS (ESI): calculated for C16H22N2O8PS [(M−H)−], 433.0840; found, 433.0838.
Diethyl-(2-((2R,3S,4R,5R)-5-(2,4-dioxo-3,4-dihydrobenzo[g]quinazolin-1(2H)-yl)-3,4-dihydroxytetra- hydrofuran-2-yl)ethyl)phosphonate (10g): The reaction between 9 (0.25 g, 0.61 mmol) and 4g (0.19 g, 0.91 mmol) as described for method 1 to yield compound 10g (125.0 mg, 43.1%). 1H-NMR (300 MHz, CDCl3) δ:1.22 (t, J = 7.0 Hz, 6H, OCH2CH3), 1.77–2.07 (m, 4H, H-5'a, H-5'b, H-6'a and H-6'b), 3.81 (d, J = 4.4 Hz, 1H, H-4'), 3.98 (quin, J = 7.1 Hz, 4H, 2 × OCH2CH3), 4.13–4.26 (m, 1H, H-3'), 4.65 (br s, 1H, H-2'), 5.05 (d, J = 6.2 Hz, 1H, OH), 5.21–5.32 (m, 1H, OH), 6.11 (d, J = 4.1 Hz, 1H, H-1'), 7.54 (t, J = 7.3 Hz, 1H, Ph), 7.66 (t, J = 7.3 Hz, 1H, Ph), 7.91 (s, 1H, Ph), 8.06 (d, J = 8.2 Hz, 1H, Ph), 8.14 (d, J = 8.2 Hz, 1H, Ph), 8.75 (s, 1H, Ph), 11.68 (br s, 1H, NH). 13C-NMR (75 MHz, CDCl3) δ: 16.24, 16.31 (OCH2CH3), 20.74 (d, J = 140.1 Hz, C-6'), 25.63 (d, J = 3.9 Hz, C-5'), 60.92 (dd, J = 6.1, 2.8 Hz, OCH2CH3), 70.12 (C-2'), 72.01 (C-3'), 82.07 (d, J = 17.1 Hz, C-4'), 91.20 (C-1'), 111.46, 116.35, 125.85, 127.59, 128.34, 129.05, 129.36, 129.52, 135.99 and 136.04 (Ph), 149.80 (C-2), 161.65 (C-4). 31P-NMR (CDCl3) δ: 32.08. HRMS (ESI): calculated for C22H26N2O8P [(M−H)−], 477.1432; found, 477.1419.
3.3. General Procedure for the Deprotection of 10a–g to 3a–g
The appropriate phosphonic ester 10 (1 eq.) was dissolved in dry CH2Cl2 (50 eq.) and treated with TMSBr (20 eq.) at 0 °C under argon. After stirring overnight at rt, the mixture was quenched with 7 N NH3/MeOH (20 eq.) and evaporated. The residue was dissolved in H2O (15 mL for 0.1 mmol phosphonic ester) and washed with CH2Cl2 (3 × 10 mL for 0.1 mmol phosphonic ester). The aqueous layer was evaporated and purified with RP high-performance liquid chromatography (HPLC, Phenomenex Luna C-18, H2O/0.1% HCOOH in CH3CN, 90:10→0:100 in 23 min, flow 17.5 mL/min) to give the desired product after lyophilization of the appropriate fractions.
(2-((2R,3S,4R,5R)-5-(2,4-Dioxo-3,4-dihydroquinazolin-1(2H)-yl)-3,4-dihydroxytetrahydrofuran-2-yl)-ethyl) phosphonic acid (3a): Deprotection of compound 10a (0.13 g, 0.30 mmol) with TMSBr (0.91 g, 5.95 mmol) according to the general procedure yielded compound 3a as a white power (32.2 mg, 29.1%). 1H-NMR (300 MHz, D2O) δ: 1.79–2.21 (m, 4H, H-5'a, H-5'b, H-6'a and H-6'b), 3.93–4.06 (m, 1H, H-4'), 4.34 (t, J = 6.9 Hz, 1H, H-3'), 4.86 (dd, J = 6.2, 4.4 Hz, 1H, H-2'), 6.09 (d, J = 3.8 Hz, 1H, H-1'), 7.35 (t, J = 7.5 Hz, 1H, Ph), 7.48 (d, J = 8.5 Hz, 1H, Ph), 7.71–7.81 (m, 1H, Ph), 8.00 (d, J = 7.9 Hz, 1H, Ph). 13C-NMR (75 MHz, D2O) δ: 22.89 (d, J = 135.8 Hz, C-6'), 25.82 (d, J = 3.9 Hz, C-5'), 71.04 (C-2'), 72.22 (C-3'), 82.47 (d, J = 17.7 Hz, C-4'), 90.85 (C-1'), 115.11, 115.72, 124.53, 128.05, 136.31 and 140.37 (Ph) 150.95 (C-2), 164.12 (C-4). 31P-NMR (D2O) δ: 29.02. HRMS (ESI): calculated for C14H16N2O8P [(M−H)−], 371.0650; found, 371.0651.
(2-((2R,3S,4R,5R)-5-(6-fluoro-2,4-dioxo-3,4-dihydroquinazolin-1(2H)-yl)-3,4-dihydroxytetrahydro-furan-2-yl)ethyl)phosphonic acid (3b): The reaction between compound 10b (286.4 mg, 0.54 mmol) and TMSBr (1.65 g, 10.79 mmol) as described above, yielded compound 3b as a white powder (15.7 mg, 14.8%). 1H-NMR (300 MHz, DMSO-d6) δ:1.50–2.04 (m, 4H, H-5'a, H-5'b, H-6'a and H-6'b), 3.65–3.76 (m, 1H, H-4'), 4.04 (t, J = 6.7 Hz, 1H, H-3'), 4.49 (dd, J = 6.4, 4.7 Hz, 1H, H-2'), 5.98 (d, J = 4.4 Hz, 1H, H-1'), 6.66 (br s, 4H, 4OH), 7.47–7.55 (m, 1H, Ph), 7.55–7.64 (m, 1H, Ph), 7.71 (dd, J = 8.2, 2.9 Hz, 1H, Ph), 11.79 (br s, 1H, NH). 13C-NMR (75 MHz, DMSO-d6) δ: 23.81 (d, J = 137.3 Hz, C-6'), 26.41 (C-5'), 69.88 (C-2'), 72.11 (C-3'), 82.49 (d, J = 17.7 Hz, C-4'), 90.77 (C-1′'), 112.63–113.19 (m, C-5), 117.91 (d, J = 7.7 Hz, C-4a), 118.12 (d, J = 7.7 Hz, C-8), 122.31–122.85 (m, C-7), 136.87 (d, J = 1.7 Hz, C-8a), 149.73 (C-2), 156.06–159.37 (m, C-6), 160.85 (d, J = 2.2 Hz, C-4). 31P-NMR (CDCl3) δ: 26.92. HRMS (ESI): calculated for C14H15FN2O8P [(M−H)−], 389.0556; found, 389.0560.
(2-((2R,3S,4R,5R)-5-(6-chloro-2,4-dioxo-3,4-dihydroquinazolin-1(2H)-yl)-3,4-dihydroxytetrahydro-furan-2-yl)ethyl)phosphonic acid (3c): The reaction between compound 10c (0.14 g, 0.31 mmol) and TMSBr (0.94 g, 6.11mmol) as described above, yielded compound 3c as a white powder (49.0 mg, 39.5%). 1H-NMR (300 MHz, D2O) δ:1.80–2.17 (m, 4H, H-5'a, H-5'b, H-6'a and H-6'b), 3.87–3.98 (m, 1H, H-4'), 4.28 (t, J = 7.0 Hz, 1H, H-3'), 4.76 (d, J = 3.8 Hz, 1H, H-2'), 5.88 (d, J = 3.8 Hz, 1H, H-1'), 7.25 (d, J = 9.4 Hz, 1H, Ph), 7.50 (dd, J = 9.1, 2.3 Hz, 1H, Ph), 7.56–7.61 (m, 1H, Ph). 13C-NMR (75 MHz, D2O) δ: 22.69 (d, J = 135.8 Hz, C-6'), 25.63 (d, J = 3.3 Hz, C-5'), 71.29 (C-2'), 72.21 (C-3'), 82.29 (d, J = 18.3 Hz, C-4'), 90.98 (C-1'), 116.62, 116.96, 126.91, 129.32, 135.69 and 138.75 (Ph), 150.20 (C-2), 162.11 (C-4). 31P-NMR (D2O) δ: 30.04. HRMS (ESI): calculated for [C14H15ClN2O8P [(M−H)−], 405.0260; found, 405.0266.
(2-((2R,3S,4R,5R)-3,4-dihydroxy-5-(6-methyl-2,4-dioxo-3,4-dihydroquinazolin-1(2H)-yl)tetrahydro-furan-2-yl)ethyl)phosphonic acid (3d): The reaction between compound 10d (0.11 g, 0.25 mmol) and TMSBr (0.77 g, 5.01mmol) as described above, yielded compound 3d as a white powder (39.6 mg, 41.0%). 1H-NMR (300 MHz, D2O) δ:1.82–2.13 (m, 4H, H-5'a, H-5′b, H-6'a and H-6'b), 2.20 (s, 3H, CH3), 3.86–3.97 (m, 1H, H-4'), 4.28 (t, J = 7.0 Hz, 1H, H-3'), 4.73 (m, 1H, H-2'), 5.86 (d, J = 3.2 Hz, 1H, H-1'), 7.12 (d, J = 8.5 Hz, 1H, Ph), 7.31–7.44 (m, 2H, Ph). 13C-NMR (75 MHz, D2O) δ: 19.62 (CH3), 22.76 (d, J = 133.5 Hz, C-6'), 25.66 (d, J = 2.8 Hz, C-5′), 71.32 (C-2'), 72.26 (C-3'), 82.13 (d, J = 17.7 Hz, C-4'), 90.82 (C-1′), 114.83, 114.96, 127.30, 134.51, 136.96 and 137.79 (Ph), 150.46 (C-2), 163.38 (C-4). 31P-NMR (D2O) δ: 29.86. HRMS (ESI): calculated for C15H18N2O8P [(M−H)−], 385.0806; found, 385.0819.
(2-((2R,3S,4R,5R)-3,4-dihydroxy-5-(6-methoxy-2,4-dioxo-3,4-dihydroquinazolin-1(2H)-yl)tetrahydro-furan-2-yl)ethyl)phosphonic acid (3e): The reaction between compound 10e (0.11 g, 0.23 mmol) and TMSBr (0.70 g, 4.58 mmol) as described above, yielded compound 3e as a white powder (48.9 mg, 53.1%). 1H-NMR (300 MHz, D2O) δ:1.78–2.18 (m, 4H, H-5'a, H-5'b, H-6'a and H-6'b), 3.86 (s, 3H, CH3), 3.93–4.01 (m, 1H, H-4'), 4.32 (t, J = 6.8 Hz, H-3'), 4.84 (dd, J = 6.6, 4.2 Hz, 1H, H-2'), 6.04 (d, J = 4.0 Hz, 1H, H-1'), 7.29–7.35 (m, 1H, Ph), 7.37–7.43 (m, 2H, Ph). 13C-NMR (75 MHz, D2O) δ: 22.99 (d, J = 126.1 Hz, C-6'), 25.51 (C-5'), 55.90 (CH3), 70.98 (C-2'), 72.21 (C-3'), 82.46 (d, J = 18.2 Hz, C-4'), 90.83 (C-1'), 109.87, 116.65, 116.92, 123.80, 134.38 and 155.46 (Ph), 150.68 (C-2), 163.60 (C-4). 31P-NMR (D2O) δ: 28.81. HRMS (ESI): calculated for C15H18N2O9P [(M−H)−], 401.0755; found, 401.0755.
(2-((2R,3S,4R,5R)-5-(2,4-dioxo-3,4-dihydrothieno[3,2-d]pyrimidin-1(2H)-yl)-3,4-dihydroxytetra- hydrofuran-2-yl)ethyl)phosphonic acid (3f): The reaction between compound 10f (0.18 g, 0.41 mmol) and TMSBr (1.24 g, 8.13 mmol) as described above, yielded compound 3f as a white powder (75.6 mg, 49.2%). 1H-NMR (300 MHz, DMSO-d6) δ:1.48–2.04 (m, 4H, H-5'a, H-5'b, H-6'a and H-6'b), 3.67–3.77 (m, 1H, H-4'), 4.01 (t, J = 6.4 Hz, 1H, H-3'), 4.41 (dd, J = 6.4, 5.3 Hz, 1H, H-2'), 5.91 (d, J = 5.0 Hz, 1H, H-1'), 6.53 (br s, 2H, 2OH), 7.25 (d, J = 5.6 Hz, 1H, H-6), 8.12 (d, J = 5.3 Hz, 1H, H-7), 11.62 (s, 1H, NH). 13C-NMR (75 MHz, DMSO-d6) δ: 23.78 (d, J = 135.8 Hz, C-6', 26.48 (C-5'), 70.41 (C-2'), 71.94 (C-3'), 83.00 (d, J = 17.7 Hz, C-4'), 90.78 (C-1′), 113.85 (C-4a), 118.02 (C-6), 135.77 (C-7), 145.17 (C-7a), 150.77 (C-2), 157.91 (C-4). 31P-NMR (DMSO-d6) δ: 28.07. HRMS (ESI): calculated for C12H14N2O8PS [(M−H)−], 377.0214; found, 377.0197.
(2-((2R,3S,4R,5R)-5-(2,4-dioxo-3,4-dihydrobenzo[g]quinazolin-1(2H)-yl)-3,4-dihydroxytetrahydro-furan-2-yl)ethyl)phosphonic acid (3g): The reaction between compound 10g (0.32 g, 0.56 mmol) and TMSBr (1.73 g, 11.30 mmol) as described above. The working up as follows: After stirring overnight, the reaction mixture was evaporated and co-distilled with toluene. The residue was dissolved with 7 N NH3/MeOH (10 mL) and stirred at room temperature for 3 h. The reaction mixture was concentrated and purified with RP high-performance liquid chromatography (HPLC, Phenomenex Luna C-18, H2O/0.1% HCOOH in CH3CN, 90:10→0:100 in 23 min, flow 17.5 mL/min) to give the compound 3g as a white power (50.0 mg, 21.0%). 1H-NMR (300 MHz, DMSO-d6) δ:1.52–2.14 (m, 4H, H-5'a, H-5'b, H-6'a and H-6'b), 3.75–3.85 (m, 1H, H-4'), 4.16 (t, J = 6.6 Hz, 1H, H-3'), 4.63 (dd, J = 6.4, 4.1 Hz, 1H, H-2'), 5.21 (br s, 2H, 2OH), 6.10 (d, J = 4.1 Hz, 1H, H-1'), 7.49–7.58 (m, 1H, Ph), 7.61–7.70 (m, 1H, Ph), 7.93 (s, 1H, Ph), 8.05 (d, J = 8.2 Hz, 1H, Ph), 8.13 (d, J = 8.2 Hz, 1H, Ph), 8.74 (s, 1H, Ph), 11.67 (s, 1H, NH). 13C-NMR (75 MHz, DMSO-d6) δ: 23.54 (d, J = 137.1 Hz, C-6'), 26.14–26.32 (m, C-5'), 69.83 (C-2'), 71.82 (C-3'), 82.23 (d, J = 17.4 Hz, C-4'), 90.86 (C-1'), 111.25, 116.09, 125.56, 127.35, 128.06, 128.75, 129.11, 129.22, 135.70 and 135.74 (Ph), 149.54 (C-2), 161.38 (C-4). 31P-NMR (DMSO-d6) δ: 26.39. HRMS (ESI): calculated for C18H18N2O9P [(M−H)−], 421.0806; found, 421.0795.
3.4. Procedures for Phospholipase C assay
Stable cell lines for study of the human (h) P2Y2, were generated by retroviral expression of the receptor in 1321N1 human astrocytoma cells, which do not natively express P2YRs. Agonist-induced [3H]inositol phosphate production was measured in cells plated at 20,000 cells/well on 96-well plates two days prior to assay. Sixteen hours before the assay, the inositol lipid pool of the cells was radiolabeled by incubation in 100 μL of serum-free inositol-free Dulbecco’s modified Eagle’s medium, containing 1.0 μCi of [3H]myo-inositol. No changes of medium were made subsequent to the addition of [3H]inositol. On the day of the assay, cells were challenged with 25 μL of a five-fold concentrated solution of receptor agonists in 100 mM Hepes (N-(2-hydroxyethyl)-piperazine-N''-2-ethanesulfonic acid), pH 7.3 in HBSS, containing 50 mM LiCl for 30 min at 37 °C. Incubations were terminated by aspiration of the drug-containing medium and addition of 90 μL of ice-cold 50 mM formic acid. After 30 min, supernatants were neutralized with 30 μL of 150 mM NH4OH and applied to Dowex AG1-X8 anion exchange columns. Total inositol phosphates were eluted and radioactivity was measured using a liquid scintillation counter. Antagonist activities were conducted in a similar manner using a maximally efficatious dose of agonist with variable amount of antagonist.
4. Conclusions
Based on the earlier identification a 5-aryl-substituted uridine phosphonate analogue as a promising allosteric partial agonist of the P2Y2R, we prepared a small collection of a novel C5/C6-fused uridine phosphonates. Pharmacological evaluation revealed that the target compounds exhibit much weaker agonistic activity than the parent compound.
Author Contributions
L.S. synthesized the target phosphonates, which were conceptualized by M.D.P.R. M.O.B. and K.A.B. performed the pharmacological evaluation under the supervision of T.K.H. I.K. contributed to the characterization of the compounds. L.S. and S.V.C. wrote the manuscript. S.V.C. coordinated this study.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Boeynaems, J.M.; Communi, D.; Robaye, B. Overview of the pharmacology and physiological roles of P2Y receptors. Membr. Transp. Signal. 2012, 1, 581–588. [Google Scholar] [CrossRef]
- Hochhauser, E.; Cohen, R.; Waldman, M.; Maksin, A.; Isak, A.; Aravot, D.; Jayasekara, P.S.; Müller, C.E.; Jacobson, K.A.; Shainberg, A. P2Y2 receptor agonist with enhanced stability protects the heart from ischemic damage in vitro and in vivo. Purinergic Signal. 2013, 9, 633–642. [Google Scholar] [CrossRef]
- El-Tayeb, A.; Qi, A.D.; Nicholas, R.A.; Müller, C.E. Structural modifications of UMP, UDP, and UTP leading to subtype-selective agonists for P2Y2, P2Y4, and P2Y6 receptors. J. Med. Chem. 2011, 54, 2878–2890. [Google Scholar] [CrossRef]
- Zimmermann, H.; Zebisch, M.; Sträter, N. Cellular function and molecular structure of ecto-nucleotidases. Purinergic Signal. 2012, 8, 437–502. [Google Scholar] [CrossRef]
- Jacobson, K.A.; Boeynaems, J.M. P2Y nucleotide receptors: Promise of therapeutic applications. Drug Discov. Today 2010, 15, 570–578. [Google Scholar] [CrossRef]
- Weisman, G.A.; Ajit, D.; Garrad, R.; Peterson, T.S.; Woods, L.T.; Thebeau, C.; Camden, J.M.; Erb, L. Neuroprotective roles of the P2Y2 receptor. Purinergic Signal. 2012, 8, 559–578. [Google Scholar] [CrossRef]
- Ivanov, A.A.; Ko, H.; Cosyn, L.; Maddileti, S.; Besada, P.; Fricks, I.; Costanzi, S.; Harden, T.K.; van Calenbergh, S.; Jacobson, K.A. Molecular modeling of the human P2Y2 receptor and design of a selective agonist, 2'-amino-2'-deoxy-2-thiouridine 5'-triphosphate. J. Med. Chem. 2007, 50, 1166–1176. [Google Scholar] [CrossRef]
- Van Poecke, S.; Barrett, M.O.; Kumar, T.S.; Sinnaeve, D.; Martins, J.C.; Jacobson, K.A.; Harden, T.K.; van Calenbergh, S. Synthesis and P2Y2 receptor agonist activities of uridine 5'-phosphonate analogues. Bioorg. Med. Chem. 2012, 20, 2304–2315. [Google Scholar] [CrossRef]
- For a systematic overview on ring transformations in medicinal chemistry see: C. Morice and C.G.Wermuth Ring Transformations in: Wermuth, C.G. The Practice of Medicinal Chemistry, 3rd ed.; Academic Press: Amsterdam, The Netherlands, 2008; pp. 343–362.
- Topliss, J.G. Utilization of Operational Schemes for Analog Synthesis in Drug Design. J. Med. Chem. 1972, 15, 1006–1011. [Google Scholar] [CrossRef]
- Li, P.F.; Zhan, C.Y.; Zhang, S.L.; Ding, X.L.; Guo, F.Q.; He, S.G.; Yao, J.N. Alkali metal cations control over nucleophilic substitutions on aromatic fused pyrimidine-2,4-[1h,3h]-diones: Towards new PNA monomers. Tetrahedron 2012, 68, 8908–8915. [Google Scholar]
- Feng, J.; Zhang, Z.Y.; Wallace, M.B.; Stafford, J.A.; Kaldor, S.W.; Kassel, D.B.; Navre, M.; Shi, L.H.; Skene, R.J.; Asakawa, T.; et al. Discovery of alogliptin: A potent, selective, bioavailable, and efficacious inhibitor of dipeptidyl peptidase IV. J. Med. Chem. 2007, 50, 2297–2300. [Google Scholar] [CrossRef]
- Tian, Q.P.; Cheng, Z.G.; Yajima, H.M.; Savage, S.J.; Green, K.L.; Humphries, T.; Reynolds, M.E.; Babu, S.; Gosselin, F.; Askin, D.; et al. A practical synthesis of a PI3K inhibitor under noncryogenic conditions via functionalization of a lithium triarylmagnesiate intermediate. Org. Process Res. Dev. 2013, 17, 97–107. [Google Scholar] [CrossRef]
- Ingall, A.H.; Moore, P.R.; Roberts, S.M. The Use of Free Radical Cyclization in the Synthesis of Compounds related to the Mannostatins. Tetrahedron-Asymmetry 1994, 5, 2155–2162. [Google Scholar] [CrossRef]
- Yoshioka, T.; Shimizu, H.; Ohrui, H. Synthesis of (3R,4S)-3,4,5-Trihydroxy-4-methylpentylphosphonic acid as a potential inhibitor of the nonmevalonate pathway. Biosci. Biotechnol. Biochem. 2004, 68, 1259–1264. [Google Scholar] [CrossRef]
- Iaroshenko, V.O.; Wang, Y.; Zhang, B.; Volochnyuk, D.; Sosnovskikh, V.Y. Facile synthesis of fluorinated benzofuro- and benzothieno[2,3-b]pyridines, α-carbolines and nucleosides containing the α-carboline framework. Synthesis 2009, 14, 2393–2402. [Google Scholar]
- Lazarowski, E.R.; Watt, W.C.; Stutts, M.J.; Boucher, R.C.; Harden, T.K. Pharmacological selectivity of the cloned human P2U-purinoceptor: Potent activation by diadenosine tetraphosphate. Brit. J. Pharmacol. 1995, 116, 1619–1627. [Google Scholar] [CrossRef]
- Nicholas, R.A.; Watt, W.C.; Lazarowski, E.R.; Li, Q.; Harden, K. Uridine nucleotide selectivity of three phospholipase C-activating P2 receptors: Identification of a UDP-selective, a UTP-selective, and an ATP- and UTP-specific receptor. Mol. Pharmacol. 1996, 50, 224–229. [Google Scholar]
- Sample Availability: Samples of the compounds 3a–g and 10a–g are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
