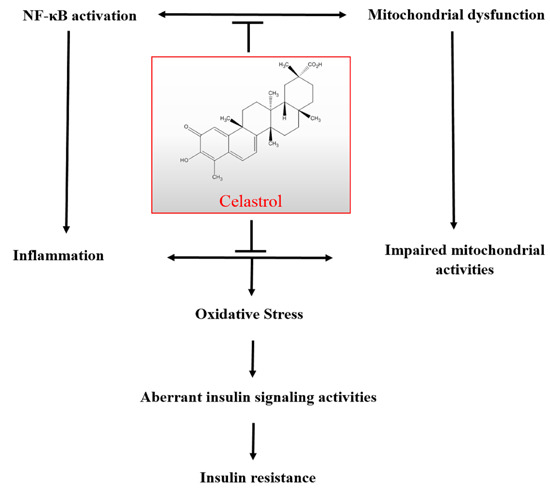
Celastrol Protects against Antimycin A-Induced Insulin Resistance in Human Skeletal Muscle Cells
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effects of AMA and Celastrol Treatment on Cell Viability (Dose and Time-Dependent)


2.2. Celastrol Increased Glucose Uptake in Insulin-Resistant Myotubes through the PI3K/Akt Pathway

2.3. Celastrol Improves Mitochondrial Activity and Energy Production in AMA-Induced Insulin-Resistant Human Skeletal Muscle Cells

2.4. Celastrol Attenuates Oxidative Stress in AMA-Treated Human Skeletal Muscle Cells

2.5. Celastrol Affects Mitochondrial Dynamics via Mitochondrial Fusion and Fission in AMA-Treated Skeletal Muscle Cells

2.6. Effect of Celastrol on the Production of Pro-Inflammatory Cytokines IL-6, TNF-α and IL-1β in AMA-Induced Mitochondrial Dysfunction Human Skeletal Muscle Cells

2.7. Effect of AMA and Celastrol on NF-κB and IκBα Expression Protein Activity


2.8. Effects of Celastrol on Protein Expression of Insulin Signaling Pathways and GLUT4 in AMA-Treated Human Skeletal Muscle Cells


2.9. Celastrol-Regulated pAMPK Expression and Inhibition of PKC θ in AMA-Treated Skeletal Muscle Cells

2.10. Discussion

3. Experimental Section
3.1. Cell Culture
3.2. AMA and Celastrol Treatment
3.3. Assessment of Cell Viability
3.4. Glucose Uptake Assay
3.5. Mitochondrial Superoxide Production
3.6. Intracellular ATP Concentration
3.7. Mitochondrial Membrane Potential (ΔΨm)
3.8. Citrate Synthase Activity
3.9. Measurement of DNA Oxidative Damage, Protein Carbonylation and Lipid Peroxidation
3.10. Cytokine ELISA for IL-6, TNF-α and IL-1β
3.11. Western Immunoblotting
3.12. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- IDF Diabetes Atlas, 6th ed.; International Diabetes Federation: Brussels, Belgium, 2013; p. 160.
- Abu Bakar, M.H.; Sarmidi, M.R.; Cheng, K.K.; Ali Khan, A.; Chua, L.S.; Zaman Huri, H.; Yaakob, H. Metabolomics-The Complementary Field in Systems Biology: A Review on Obesity and Type 2 Diabetes. Mol. Biosyst. 2015. [Google Scholar] [CrossRef]
- Hafizi Abu Bakar, M.; Kian Kai, C.; Wan Hassan, W.N.; Sarmidi, M.R.; Yaakob, H.; Zaman Huri, H. Mitochondrial dysfunction as a central event for mechanisms underlying insulin resistance: The roles of long chain fatty acids. Diabetes Metab. Res. Rev. 2014. [Google Scholar] [CrossRef]
- Bondia-Pons, I.; Ryan, L.; Martinez, J.A. Oxidative stress and inflammation interactions in human obesity. J. Physiol. Biochem. 2012, 68, 701–711. [Google Scholar] [CrossRef] [PubMed]
- Petersen, K.F.; Dufour, S.; Befroy, D.; Garcia, R.; Shulman, G.I. Impaired Mitochondrial Activity in the Insulin-Resistant Offspring of Patients with Type 2 Diabetes. N. Engl. J. Med. 2004, 350, 664–671. [Google Scholar] [CrossRef] [PubMed]
- Gaster, M.; Rustan, A.C.; Aas, V.; Beck-Nielsen, H. Reduced Lipid Oxidation in Skeletal Muscle From Type 2 Diabetic Subjects May Be of Genetic Origin: Evidence From Cultured Myotubes. Diabetes 2004, 53, 542–548. [Google Scholar] [CrossRef] [PubMed]
- Szendroedi, J.; Phielix, E.; Roden, M. The role of mitochondria in insulin resistance and type 2 diabetes mellitus. Nat. Rev. Endocrinol. 2012, 8, 92–103. [Google Scholar] [CrossRef]
- Wang, C.H.; Wang, C.C.; Wei, Y.H. Mitochondrial dysfunction in insulin insensitivity: Implication of mitochondrial role in type 2 diabetes. Ann. N. Y. Acad. Sci. 2010, 1201, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.R.; Nachbar, R.T.; Gorjao, R.; Vinolo, M.A.; Festuccia, W.T.; Lambertucci, R.H.; Cury-Boaventura, M.F.; Silveira, L.R.; Curi, R.; Hirabara, S.M. Mechanisms underlying skeletal muscle insulin resistance induced by fatty acids: Importance of the mitochondrial function. Lipids Health Dis. 2012, 11, 30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, H.; Zhang, H.M.; Wu, L.P.; Tan, D.X.; Kamat, A.; Li, Y.Q.; Katz, M.S.; Abboud, H.E.; Reiter, R.J.; Zhang, B.X. Impaired mitochondrial complex III and melatonin responsive reactive oxygen species generation in kidney mitochondria of db/db mice. J. Pineal Res. 2011, 51, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, M.; Guhathakurta, I.; Behera, P.; Ranjan, K.R.; Khanna, M.; Mukhopadhyay, S.; Chakrabarti, S. Mitochondrial bioenergetics is not impaired in nonobese subjects with type 2 diabetes mellitus. Metabolism 2011, 60, 1702–1710. [Google Scholar] [CrossRef] [PubMed]
- Lanza, I.R.; Sreekumaran Nair, K. Regulation of skeletal muscle mitochondrial function: Genes to proteins. Acta Physiol. 2010, 199, 529–547. [Google Scholar] [CrossRef]
- Roden, M. How Free Fatty Acids Inhibit Glucose Utilization in Human Skeletal Muscle. Physiology 2004, 19, 92–96. [Google Scholar] [CrossRef]
- Yuzefovych, L.V.; Schuler, A.M.; Chen, J.; Alvarez, D.F.; Eide, L.; LeDoux, S.P.; Wilson, G.L.; Rachek, L.I. Alteration of mitochondrial function and insulin sensitivity in primary mouse skeletal muscle cells isolated from transgenic and knockout mice: Role of OGG1. Endocrinology 2013. [Google Scholar] [CrossRef]
- Hernández-Aguilera, A.; Rull, A.; Rodríguez-Gallego, E.; Riera-Borrull, M.; Luciano-Mateo, F.; Camps, J.; Menéndez, J.A.; Joven, J. Mitochondrial Dysfunction: A Basic Mechanism in Inflammation-Related Non-Communicable Diseases and Therapeutic Opportunities. Mediat. Inflamm. 2013, 2013. Article ID 135698. [Google Scholar]
- Morita, T. Celastrol: A New Therapeutic Potential of Traditional Chinese Medicine. Am. J. Hypertens. 2010, 23, 821. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Koo, T.H.; Yoon, H.; Jung, H.S.; Jin, H.Z.; Lee, K.; Hong, Y.S.; Lee, J.J. Inhibition of NF-κB activation through targeting IκB kinase by celastrol, a quinone methide triterpenoid. Biochem. Pharmacol. 2006, 72, 1311–1321. [Google Scholar] [CrossRef] [PubMed]
- Sethi, G.; Ahn, K.S.; Pandey, M.K.; Aggarwal, B.B. Celastrol, a novel triterpene, potentiates TNF-induced apoptosis and suppresses invasion of tumor cells by inhibiting NF-κB–regulated gene products and TAK1-mediated NF-κB activation. Blood 2006, 109, 2727–2735. [Google Scholar]
- Shao, L.; Zhou, Z.; Cai, Y.; Castro, P.; Dakhov, O.; Shi, P.; Bai, Y.; Ji, H.; Shen, W.; Wang, J. Celastrol Suppresses Tumor Cell Growth through Targeting an AR-ERG-NF-κB Pathway in TMPRSS2/ERG Fusion Gene Expressing Prostate Cancer. PLoS ONE 2013, 8, e58391. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.E.; Lee, M.H.; Nam, D.H.; Song, H.K.; Kang, Y.S.; Lee, J.E.; Kim, H.W.; Cha, J.J.; Hyun, Y.Y.; Han, S.Y.; et al. Celastrol, an NF-κB Inhibitor, Improves Insulin Resistance and Attenuates Renal Injury in db/db Mice. PLoS ONE 2013, 8, e62068. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Tao, W.; Jiang, F.; Li, C.; Lin, J.; Liu, C. Celastrol Attenuates Hypertension-Induced Inflammation and Oxidative Stress in Vascular Smooth Muscle Cells via Induction of Heme Oxygenase-1. Am. J. Hypertens. 2010, 23, 895–903. [Google Scholar] [CrossRef] [PubMed]
- Der Sarkissian, S.; Cailhier, J.-F.; Borie, M.; Stevens, L.M.; Gaboury, L.; Mansour, S.; Hamet, P.; Noiseux, N. Celastrol protects ischaemic myocardium through a heat shock response with up-regulation of haeme oxygenase-1. Br. J. Pharmacol. 2014, 171, 5265–5279. [Google Scholar] [CrossRef] [PubMed]
- Abu Bakar, M.H.; Sarmidi, M.R.; Kian Kai, C.; Zaman Huri, H.; Yaakob, H. Amelioration of Mitochondrial Dysfunction-Induced Insulin Resistance in Differentiated 3T3-L1 Adipocytes via Inhibition of NF-κB Pathways. Int. J. Mol. Sci. 2014, 15, 22227–22257. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.W.; Kim, M.S.; Kim, H.S.; Kim, Y.; Shin, D.; Park, J.H. Y.; Kang, Y.H. Celastrol attenuates adipokine resistin-associated matrix interaction and migration of vascular smooth muscle cells. J. Cell. Biochem. 2013, 114, 398–408. [Google Scholar] [CrossRef] [PubMed]
- Ju, S.M.; Cho, Y.S.; Park, J.S. Celastrol ameliorates cytokine toxicity and pro-inflammatory immune responses by suppressing NF-kB activation in RINm5F beta cells. Biochem. Mol. Biol. Rep. 2015, 48, 172–177. [Google Scholar]
- Hansen, J.; Palmfeldt, J.; Vang, S.; Corydon, T.J.; Gregersen, N.; Bross, P. Quantitative Proteomics Reveals Cellular Targets of Celastrol. PLoS ONE 2011, 6, e26634. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.H.; Wang, C.C.; Huang, H.C.; Wei, Y.H. Mitochondrial dysfunction leads to impairment of insulin sensitivity and adiponectin secretion in adipocytes. FEBS J. 2013, 280, 1039–1050. [Google Scholar] [CrossRef] [PubMed]
- Parra, V.; Verdejo, H.E.; Iglewski, M.; del Campo, A.; Troncoso, R.; Jones, D.; Zhu, Y.; Kuzmicic, J.; Pennanen, C.; Lopez-Crisosto, C.; et al. Insulin Stimulates Mitochondrial Fusion and Function in Cardiomyocytes via the Akt-mTOR-NFκB-Opa-1 Signaling Pathway. Diabetes 2014, 63, 75–88. [Google Scholar] [CrossRef] [PubMed]
- Schultze, S.M.; Hemmings, B.A.; Niessen, M.; Tschopp, O. PI3K/AKT, MAPK and AMPK signalling: Protein kinases in glucose homeostasis. Expert Rev. Mol. Med. 2012, 14, e1. [Google Scholar] [CrossRef] [PubMed]
- Cogswell, P.C.; Kashatus, D.F.; Keifer, J.A.; Guttridge, D.C.; Reuther, J.Y.; Bristow, C.; Roy, S.; Nicholson, D.W.; Baldwin, A.S. NF-κB and IκBα Are Found in the Mitochondria: Evidence for Regulation of Mitochondrial Gene Expression by NF-κB. J. Biol. Chem. 2003, 278, 2963–2968. [Google Scholar] [CrossRef] [PubMed]
- Larsen, S.; Nielsen, J.; Hansen, C.N.; Nielsen, L.B.; Wibrand, F.; Stride, N.; Schroder, H.D.; Boushel, R.; Helge, J.W.; Dela, F.; et al. Biomarkers of mitochondrial content in skeletal muscle of healthy young human subjects. J. Physiol. 2012, 590, 3349–3360. [Google Scholar] [CrossRef] [PubMed]
- Pagel-Langenickel, I.; Bao, J.; Pang, L.; Sack, M.N. The role of mitochondria in the pathophysiology of skeletal muscle insulin resistance. Endocr. Rev. 2010, 31, 25–51. [Google Scholar] [CrossRef] [PubMed]
- Gruzman, A.; Babai, G.; Sasson, S. Adenosine Monophosphate-Activated Protein Kinase (AMPK) as a New Target for Antidiabetic Drugs: A Review on Metabolic, Pharmacological and Chemical Considerations. Rev. Diabet. Stud. 2009, 6, 13–36. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Soos, T.J.; Li, X.; Wu, J.; DeGennaro, M.; Sun, X.; Littman, D.R.; Birnbaum, M.J.; Polakiewicz, R.D. Protein Kinase C θ Inhibits Insulin Signaling by Phosphorylating IRS1 at Ser1101. J. Biol. Chem. 2004, 279, 45304–45307. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.; Kim, Y.B. Molecular mechanism of insulin resistance in obesity and type 2 diabetes. Korean J. Intern. Med. 2010, 25, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Højlund, K.; Mogensen, M.; Sahlin, K.; Beck-Nielsen, H. Mitochondrial Dysfunction in Type 2 Diabetes and Obesity. Endocrinol. Metab. Clin. North Am. 2008, 37, 713–731. [Google Scholar] [CrossRef] [PubMed]
- Shenouda, S.M.; Widlansky, M.E.; Chen, K.; Xu, G.; Holbrook, M.; Tabit, C.E.; Hamburg, N.M.; Frame, A.A.; Caiano, T.L.; Kluge, M.A.; et al. Altered mitochondrial dynamics contributes to endothelial dysfunction in diabetes mellitus. Circulation 2011, 124, 444–453. [Google Scholar] [CrossRef] [PubMed]
- Jheng, H.F.; Tsai, P.J.; Guo, S.M.; Kuo, L.H.; Chang, C.S.; Su, I.J.; Chang, C.R.; Tsai, Y.S. Mitochondrial fission contributes to mitochondrial dysfunction and insulin resistance in skeletal muscle. Mol. Cell. Biol. 2012, 32, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Mariappan, N.; Elks, C.M.; Sriramula, S.; Guggilam, A.; Liu, Z.; Borkhsenious, O.; Francis, J. NF-κB-induced oxidative stress contributes to mitochondrial and cardiac dysfunction in type II diabetes. Cardiovasc. Res. 2010, 85, 473–483. [Google Scholar] [CrossRef] [PubMed]
- Vaamonde-García, C.; Valcarcel-Ares, N.; Riveiro-Naveira, R.; Lema, B.; Blanco, F.J.; López-Armada, M.J. Inflammatory response is modulated by mitochondrial dysfunction in cultured normal human chondrocytes. Ann. Rheum. Dis. 2010, 69, A15–A16. [Google Scholar] [CrossRef]
- Ungvari, Z.; Orosz, Z.; Labinskyy, N.; Rivera, A.; Xiangmin, Z.; Smith, K.; Csiszar, A. Increased mitochondrial H2O2 production promotes endothelial NF-κB activation in aged rat arteries. Am. J. Physiol. Hear. Circ. Physiol. 2007, 293, H37–H47. [Google Scholar] [CrossRef]
- Vaamonde-García, C.; Riveiro-Naveira, R.R.; Valcárcel-Ares, M.N.; Hermida-Carballo, L.; Blanco, F.J.; López-Armada, M.J. Mitochondrial dysfunction increases inflammatory responsiveness to cytokines in normal human chondrocytes. Arthritis Rheum. 2012, 64, 2927–2936. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Jin, P.; LiqunYu; Wang, Y.; Han, L.; Shi, T.; Li, X. Impaired Mitochondrial Dynamics and Bioenergetics in Diabetic Skeletal Muscle. PLoS ONE 2014, 9, e92810. [Google Scholar] [CrossRef] [PubMed]
- Guest, C.B.; Park, M.J.; Johnson, D.R.; Freund, G.G. The implication of proinflammatory cytokines in type 2 diabetes. Front. Biosci. 2008, 13, 5187–5194. [Google Scholar] [CrossRef] [PubMed]
- Baker, R.G.; Hayden, M.S.; Ghosh, S. NF-κB, inflammation and metabolic disease. Cell Metab. 2011, 13, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Ni, H.; Zhao, W.; Kong, X.; Li, H.; Ouyang, J. NF-Kappa B Modulation Is Involved in Celastrol Induced Human Multiple Myeloma Cell Apoptosis. PLoS ONE 2014, 9, e95846. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Fu, Y.; Zhuang, L.; Gai, R.; Ma, J.; Lou, J.; Zhu, H.; He, Q.; Yang, B. Simultaneous NF-κB inhibition and E-cadherin upregulation mediate mutually synergistic anticancer activity of celastrol and SAHA in vitro and in vivo. Int. J. Cancer 2014, 135, 1721–1732. [Google Scholar] [CrossRef] [PubMed]
- Youn, G.S.; Kwon, D.J.; Ju, S.M.; Rhim, H.; Bae, Y.S.; Choi, S.Y.; Park, J. Celastrol ameliorates HIV-1 Tat-induced inflammatory responses via NF-kappaB and AP-1 inhibition and heme oxygenase-1 induction in astrocytes. Toxicol. Appl. Pharmacol. 2014, 280, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Chiang, K.C.; Tsui, K.H.; Chung, L.C.; Yeh, C.N.; Chen, W.T.; Chang, P.L.; Juang, H.H. Celastrol Blocks Interleukin-6 Gene Expression via Downregulation of NF-κB in Prostate Carcinoma Cells. PLoS ONE 2014, 9, e93151. [Google Scholar] [CrossRef] [PubMed]
- Yuzefovych, L.; Wilson, G.; Rachek, L. Different effects of oleate vs. palmitate on mitochondrial function, apoptosis, and insulin signaling in L6 skeletal muscle cells: Role of oxidative stress. Am. J. Physiol. Endocrinol. Metab. 2010, 299, E1096–E1105. [Google Scholar] [CrossRef] [PubMed]
- Houstis, N.; Rosen, E.D.; Lander, E.S. Reactive oxygen species have a causal role in multiple forms of insulin resistance. Nature 2006, 440, 944–948. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, A.; Levchenko, A.; Scott, M.L.; Baltimore, D. The IκB-NF-κB Signaling Module: Temporal Control and Selective Gene Activation. Science 2002, 298, 1241–1245. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, A.; Baltimore, D. Circuitry of nuclear factor κB signaling. Immunol. Rev. 2006, 210, 171–186. [Google Scholar] [CrossRef] [PubMed]
- Plomgaard, P.; Bouzakri, K.; Krogh-Madsen, R.; Mittendorfer, B.; Zierath, J.R.; Pedersen, B.K. Tumor Necrosis Factor-α Induces Skeletal Muscle Insulin Resistance in Healthy Human Subjects via Inhibition of Akt Substrate 160 Phosphorylation. Diabetes 2005, 54, 2939–2945. [Google Scholar] [CrossRef] [PubMed]
- Werner, E.D.; Lee, J.; Hansen, L.; Yuan, M.; Shoelson, S.E. Insulin Resistance Due to Phosphorylation of Insulin Receptor Substrate-1 at Serine 302. J. Biol. Chem. 2004, 279, 35298–35305. [Google Scholar] [CrossRef] [PubMed]
- Aguirre, V.; Werner, E.D.; Giraud, J.; Lee, Y.H.; Shoelson, S.E.; White, M.F. Phosphorylation of Ser307 in Insulin Receptor Substrate-1 Blocks Interactions with the Insulin Receptor and Inhibits Insulin Action. J. Biol. Chem. 2002, 277, 1531–1537. [Google Scholar] [CrossRef] [PubMed]
- Hirosumi, J.; Tuncman, G.; Chang, L.; Gorgun, C.Z.; Uysal, K.T.; Maeda, K.; Karin, M.; Hotamisligil, G.S. A central role for JNK in obesity and insulin resistance. Nature 2002, 420, 333–336. [Google Scholar] [CrossRef] [PubMed]
- Barja, G. Mitochondrial Oxygen Radical Generation and Leak: Sites of Production in States 4 and 3, Organ Specificity, and Relation to Aging and Longevity. J. Bioenerg. Biomembr. 1999, 31, 347–366. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, E.C.; Long, Y.C.; Martinsson, S.; Glund, S.; Garcia-Roves, P.; Svensson, L.T.; Andersson, L.; Zierath, J.R.; Mahlapuu, M. Opposite Transcriptional Regulation in Skeletal Muscle of AMP-activated Protein Kinase γ3 R225Q Transgenic Versus Knock-out Mice. J. Biol. Chem. 2006, 281, 7244–7252. [Google Scholar] [CrossRef] [PubMed]
- Kahn, N.Y.; Accili, C.R.; Domenico; Kitamura, A. Mouse Models of Insulin Resistance. Physiol. Rev. 2004, 84, 623–647. [Google Scholar] [CrossRef] [PubMed]
- Ciaraldi, T.P.; Mudaliar, S.; Barzin, A.; Macievic, J.A.; Edelman, S.V.; Park, K.S.; Henry, R.R. Skeletal Muscle GLUT1 Transporter Protein Expression and Basal Leg Glucose Uptake Are Reduced in Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2005, 90, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Gaster, M.; Staehr, P.; Beck-Nielsen, H.; Schrøder, H.D.; Handberg, A. GLUT4 Is Reduced in Slow Muscle Fibers of Type 2 Diabetic Patients: Is Insulin Resistance in Type 2 Diabetes a Slow, Type 1 Fiber Disease? Diabetes 2001, 50, 1324–1329. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.L.T.; Giannocco, G.; Furuya, D.T.; Lima, G.A.; Moraes, P.A.C.; Nachef, S.; Bordin, S.; Britto, L.R.G.; Nunes, M.T.; Machado, U.F. NF-κB, MEF2A, MEF2D and HIF1-a involvement on insulin- and contraction-induced regulation of GLUT4 gene expression in soleus muscle. Mol. Cell. Endocrinol. 2005, 240, 82–93. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, H.M. AMPK and Exercise: Glucose Uptake and Insulin Sensitivity. Diabetes Metab. J. 2013, 37, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Fujii, N.; Jessen, N.; Goodyear, L.J. AMP-activated protein kinase and the regulation of glucose transport. Am. J. Physiol.—Endocrinol. Metab. 2006, 291, E867–E877. [Google Scholar] [CrossRef] [PubMed]
- Lihn, A.S.; Pedersen, S.B.; Lund, S.; Richelsen, B. The anti-diabetic AMPK activator AICAR reduces IL-6 and IL-8 in human adipose tissue and skeletal muscle cells. Mol. Cell. Endocrinol. 2008, 292, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Green, C.J.; Pedersen, M.; Pedersen, B.K.; Scheele, C. Elevated NF-κB Activation Is Conserved in Human Myocytes Cultured From Obese Type 2 Diabetic Patients and Attenuated by AMP-Activated Protein Kinase. Diabetes 2011, 60, 2810–2819. [Google Scholar] [CrossRef] [PubMed]
- Barma, P.; Bhattacharya, S.; Bhattacharya, A.; Kundu, R.; Dasgupta, S.; Biswas, A.; Bhattacharya, S.; Roy, S.S.; Bhattacharya, S. Lipid induced overexpression of NF-κB in skeletal muscle cells is linked to insulin resistance. Biochim. Biophys. Acta—Mol. Basis Dis. 2009, 1792, 190–200. [Google Scholar] [CrossRef]
- Yang, H.; Chen, D.; Cui, Q.C.; Yuan, X.; Dou, Q.P. Celastrol, a Triterpene Extracted from the Chinese “Thunder of God Vine,” Is a Potent Proteasome Inhibitor and Suppresses Human Prostate Cancer Growth in Nude Mice. Cancer Res. 2006, 66, 4758–4765. [Google Scholar] [CrossRef] [PubMed]
- Im, A.R.; Kim, Y.H.; Uddin, M.R.; Chae, S.W.; Lee, H.W.; Jung, W.S.; Kim, Y.H.; Kang, B.J.; Kim, Y.S.; Lee, M.Y. Protection from antimycin A-induced mitochondrial dysfunction by Nelumbo nucifera seed extracts. Environ. Toxicol. Pharmacol. 2013, 36, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Tamrakar, A.K.; Jaiswal, N.; Yadav, P.P.; Maurya, R.; Srivastava, A.K. Pongamol from Pongamia pinnata stimulates glucose uptake by increasing surface GLUT4 level in skeletal muscle cells. Mol. Cell. Endocrinol. 2011, 339, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the celastrol is available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abu Bakar, M.H.; Cheng, K.-K.; Sarmidi, M.R.; Yaakob, H.; Huri, H.Z. Celastrol Protects against Antimycin A-Induced Insulin Resistance in Human Skeletal Muscle Cells. Molecules 2015, 20, 8242-8269. https://doi.org/10.3390/molecules20058242
Abu Bakar MH, Cheng K-K, Sarmidi MR, Yaakob H, Huri HZ. Celastrol Protects against Antimycin A-Induced Insulin Resistance in Human Skeletal Muscle Cells. Molecules. 2015; 20(5):8242-8269. https://doi.org/10.3390/molecules20058242
Chicago/Turabian StyleAbu Bakar, Mohamad Hafizi, Kian-Kai Cheng, Mohamad Roji Sarmidi, Harisun Yaakob, and Hasniza Zaman Huri. 2015. "Celastrol Protects against Antimycin A-Induced Insulin Resistance in Human Skeletal Muscle Cells" Molecules 20, no. 5: 8242-8269. https://doi.org/10.3390/molecules20058242