3.2. General Synthetic Procedure for 5a,b‒11a,b
A mixture of lawsone (2.87 mmol), NaI (3.16 mmol), triethylamine (3.16 mmol) and alkyl or terpenyl bromide (2.87 mmol) were stirred in DMF (5 mL) at 40 °C for three days. After the reaction was cooled to room temperature, 1 N HCl was added and the solution was poured into a separatory funnel with water, dichloromethane was added and the organic layer was separated from the aqueous layer. The organic layer was collected, dried over anhydrous magnesium sulfate (MgSO4) and concentrated under reduced pressure to remove the solvent, and then was subjected to column chromatography (ethyl acetate/n-hexane = 1/7) to produce yellow solid compounds, which were purified by recrystallization from n-hexane.
2-Hydroxy-3-methyl-1,
4-naphthoquinone (
5a). The reaction produced
5a in 45.3% as a yellow solid; mp 173.4–174.1 °C (lit. [
30] 173–174 °C).
1H-NMR (500 MHz, CDCl
3) δ
H 2.09 (s, 3H, CH
3), 7.29 (bs, 1H, 2-OH), 7.66 (dt,
J = 1.2, 7.5 Hz, 1H, H-7), 7.73 (dt,
J = 1.4, 7.6 Hz, 1H, H-6), 8.06 (dd,
J = 1.2, 7.7 Hz, 1H, H-8), 8.10 (dd,
J = 0.7, 7.7 Hz, 1H, H-5);
13C-NMR (125 MHz, CDCl
3) δ
C 8.91 (CH
3), 120.75 (C-3), 126.36 (C-8), 126.97 (C-5), 129.64 (C-9), 133.13 (C-6), 132.94 (C-10), 134.83 (C-7), 153.13 (C-2) 181.19 (C=O), 185.02 (C=O); LC-MS (ESI
−,
m/
z) calculated for C
11H
8O
3: 188.05, found for 186.98 [M − H]
−; HRMS-ESI:
m/
z calculated for C
11H
8O
3: 188.0473 [M]
+, found for 188.0471.
2-Methoxy-1,
4-naphthoquinone (
5b). The reaction produced
5b in 27.0% as a yellow solid; mp 187.0–187.8 °C (lit. [
31] 186–189 °C).
1H-NMR (500 MHz, CDCl
3) δ
H 3.89 (s, 3H, CH
3), 6.16 (s, 1H, H-3), 7.70 (dt,
J = 1.3, 7.5 Hz, 1H, H-7), 7.74 (dt,
J = 1.3, 7.5 Hz, 1H, H-6), 8.07 (dd,
J = 1.2, 7.5 Hz, 1H, H-8), 8.11 (dd,
J = 1.2, 7.5 Hz, 1H, H-5);
13C-NMR (125 MHz, CDCl
3) δ
C 56.65 (CH
3), 110.14 (C-3), 126.43 (C-8), 126.96 (C-5), 131.29 (C-9), 132.28 (C-10), 133.58 (C-6), 134.57 (C-7), 160.67 (C-2), 180.36 (C=O), 185.08 (C=O); LC-MS (ESI
+,
m/
z) calculated for C
11H
8O
3: 188.05, found for 189.09 [M + H]
+; HRMS-ESI:
m/
z calculated for C
11H
8O
3: 188.0473 [M]
+, found for 188.0476.
2-Hydroxy-3-butyl-1,
4-naphthoquinone (
6a). The reaction produced
6a in 36.6% as a yellow solid; mp 97.1–98.0 °C (lit. [
32] 93–94 °C).
1H-NMR (500 MHz, CDCl
3) δ
H 0.92 (t,
J = 7.5 Hz, 3H, CH
3), 1.39 (p,
J = 7.5 Hz 2H, H-3′), 1.50 (p,
J = 7.5 Hz, 2H, H-2′), 2.58 (t,
J = 7.5 Hz, 2H, H-1′), 7.30 (s, 1H, 2-OH), 7.65 (t,
J = 7.5 Hz, 1H, H-7), 7.73 (t,
J = 7.5 Hz, 1H, H-6), 8.05 (d,
J = 7.5 Hz, 1H, H-8), 8.10 (d,
J = 7.5 Hz, 1H, H-5);
13C-NMR (125 MHz, CDCl
3) δ
C 14.12 (CH
3), 23.09 (C-1′), 29.91 (C-3′), 30.64 (C-2′), 125.04 (C-3), 126.26 (C-8), 126.99 (C-5), 129.67 (C-9), 133.06 (C-6), 133.18 (C-10), 135.04 (C-7), 153.23 (C-2), 181.70 (C=O), 184.96 (C=O); LC-MS (ESI
−,
m/
z) calculated for C
14H
14O
3: 230.09, found for 229.05 [M − H]
−; HRMS-ESI:
m/
z calculated for C
14H
14O
3: 230.0943 [M]
+, found for 230.0948.
2-Butyloxy-1,
4-naphthoquinone (
6b). The reaction produced
6b in 5.5% as a yellow solid; mp 111.5–112.1 °C (lit. [
33] 97–98 °C).
1H-NMR (500 MHz, CDCl
3) δ
H 0.99 (t,
J = 7.4 Hz, 3H, CH
3), 1.53 (p,
J = 7.5 Hz 2H, H-3′), 1.88 (p,
J = 7.1 Hz, 2H, H-2′), 4.02 (t,
J = 6.6 Hz, 2H, H-1′), 6.16 (s, 1H, H-3), 7.70 (dt,
J = 1.0, 7.5 Hz, 1H, H-7), 7.74 (dt,
J = 1.0, 7.5 Hz, 1H, H-6), 8.07 (d,
J = 7.3 Hz, 1H, H-8), 8.11 (d,
J = 7.3 Hz, 1H, H-5);
13C-NMR (125 MHz, CDCl
3) δ
C 13.70 (CH
3), 19.13 (C-3′), 30.25 (C-2′), 69.39 (C-1′), 110.19 (C-3), 126.11 (C-8), 126.68 (C-5), 131.20 (C-9), 132.04 (C-10), 133.26 (C-6), 134.21 (C-7), 159.93 (C-2), 180.18 (C=O), 185.10 (C=O); LC-MS (ESI
−,
m/
z) calculated for C
14H
14O
3: 230.09, found for 229.52 [M − H]
−; HRMS-ESI:
m/
z calculated for C
14H
14O
3: 230.0943 [M]
+, found for 230.0941.
2-Hydroxy-3-octyl-1,
4-naphthoquinone (
7a). The reaction produced
7a in 36.2% as a yellow solid; mp 91.1–91.6 °C (lit. [
34] 88–89 °C).
1H-NMR (500 MHz, CDCl
3) δ
H 0.86 (t,
J = 7.5 Hz, 3H, CH
3), 1.23–1.36 (m, 8H, H-4′, H-5′, H-6′, H-7′), 1.39 (p,
J = 8.0 Hz, 2H, H-3′), 1.51 (p,
J = 7.6 Hz, 2H, H-2′), 2.58 (t,
J = 7.7 Hz, 2H, H-1′), 7.26 (s, 1H, 2-OH), 7.66 (dt,
J = 1.0, 7.6 Hz, 1H, H-7), 7.73 (dt,
J = 1.0, 7.6 Hz, 1H, H-6), 8.05 (d,
J = 7.5 Hz, 1H, H-8), 8.10 (d,
J = 7.6 Hz, 1H, H-5);
13C-NMR (125 MHz, CDCl
3) δ
C 14.32 (CH
3), 22.88 (C-1′), 28.53 (C-7′), 29.46 (C-2′), 29.64 (C-3′), 29.92 (C-4′), 30.41 (C-5′), 32.10 (C-6′), 125.09 (C-3), 126.27 (C-8), 127.01 (C-5), 129.69 (C-9), 133.06 (C-6), 133.21 (C-10), 135.04 (C-7), 153.20 (C-2), 181.72 (C=O), 184.94 (C=O); LC-MS (ESI
+,
m/
z) calculated for C
18H
22O
3: 286.16, found for 309.35 [M + Na]
+; HRMS-ESI:
m/
z calculated for C
18H
22O
3: 286.1569 [M]
+, found for 286.1568.
2-Octyloxy-1,4-naphthoquinone (7b). The reaction produced 7b in 3.8% as a yellow solid; mp 94.1–95.8 °C. 1H-NMR (500 MHz, CDCl3) δH 0.86 (t, J = 6.9, 3H, CH3), 1.23–1.35 (m, 8H, H-4′, H-5′, H-6′, H-7′), 1.44 (p, J = 7.6 Hz, 2H, H-3′), 1.87 (p, J = 7.2 Hz, 2H, H-2′), 3.98 (t, J = 6.7 Hz, 2H, H-1′), 6.12 (s, 1H, H-3), 7.68 (dt, J = 1.5, 7.5 Hz, 1H, H-7), 7.71 (dt, J = 1.0, 7.5 Hz, 1H, H-6), 8.05 (dd, J = 1.0, 7.0 Hz, 1H, H-8), 8.10 (dd, J = 1.5, 7.5 Hz, 1H, H-5); 13C-NMR (125 MHz, CDCl3) δC 14.08 (CH3), 22.63 (C-7′), 25.85 (C-2′), 28.26 (C-3′), 29.14 (C-4′), 29.20 (C-5′), 31.75 (C-6′), 69.69 (C-1′), 110.19 (C-3), 126.11 (C-8), 126.67 (C-5), 131.20 (C-9), 132.05 (C-10), 133.24 (C-6), 134.20 (C-7), 159.92 (C-2), 180.18 (C=O), 185.09 (C=O); LC-MS (ESI+, m/z) calculated for C18H22O3: 286.16, found for 309.25 [M + Na]+; HRMS-ESI: m/z calculated for C18H22O3: 286.1569 [M]+, found for 286.1564.
2-Hydroxy-3-dodectyl-1,
4-naphthoquinone (
8a). The reaction produced
8a in 56.3% as a yellow solid; mp 88.4–88.9 °C (lit. [
35] 85–87 °C).
1H-NMR (500 MHz, CDCl
3) δ
H 0.86 (t,
J = 7.0 Hz, 3H, CH
3), 1.23–1.35 (m, 18H, H-3′, H-4′, H-5′, H-6′, H-7′, H-8′, H-9′, H-10′, H-11′), 1.51 (p,
J = 7.5 Hz, 2H, H-2′), 2.58 (t,
J = 7.7 Hz, 2H, H-1′), 7.26 (s, 1H, 2-OH), 7.61 (dt,
J = 0.8, 7.5 Hz, 1H, H-7), 7.73 (dt,
J = 0.8, 7.5 Hz, 1H, H-6), 8.00 (d,
J = 7.5 Hz, 1H, H-8), 8.05 (d,
J = 7.5 Hz, 1H, H-5);
13C-NMR (125 MHz, CDCl
3) δ
C 14.33 (CH
3), 22.91 (C-1′), 23.62 (C-11′), 28.53 (C-2′), 29.57 (C-3′), 29.68 (C-4′), 29.80 (C-5′), 29.86 (C-6′), 29.88 (C-7′), 29.92 (C-8′), 30.02 (C-9′), 32.14 (C-10′), 125.09 (C-3), 126.26 (C-8), 127.00 (C-5), 129.69 (C-9), 133.05 (C-6), 133.21 (C-10), 135.04 (C-7), 153.20 (C-2), 181.71 (C=O), 184.93 (C=O); LC-MS (ESI
−,
m/
z) calculated for C
22H
30O
3: 342.22, found for 341.38 [M − H]
−; HRMS-ESI:
m/
z calculated for C
22H
30O
3: 342.2195 [M]
+, found for 342.2189.
2-Dodectyloxy-1,4-naphthoquinone (8b). The reaction produced 8b in 6.9% as a yellow solid; mp 96.1–96.7 °C. 1H-NMR (500 MHz, CDCl3) δH 0.86 (t, J = 7.0 Hz, 3H, CH3), 1.24–1.33 (m, 16H, H-4′, H-5′, H-6′, H-7′, H-8′, H-9′, H-10′, H-11′), 1.44 (p, J = 7.5 Hz, 2H, H-3′), 1.87 (p, J = 7.1 Hz, 2H, H-2′), 3.98 (t, J = 6.7 Hz, 2H, H-1′), 6.13 (s, 1H, H-3), 7.68 (t, J = 7.3 Hz, 1H, H-7), 7.72 (t, J = 7.3 Hz, 1H, H-6), 8.05 (d, J = 7.4 Hz, 1H, H-8), 8.10 (d, J = 7.5 Hz, 1H, H-5); 13C-NMR (125 MHz, CDCl3) δC 14.33 (CH3), 22.90 (C-7′), 26.06 (C-2′), 28.48 (C-3′), 29.46 (C-4′), 29.56 (C-5′), 29.69 (C-6′), 29.77 (C-7′), 29.83 (C-8′), 29.84 (C-9′), 32.13 (C-10′), 69.90 (C-1′), 110.40 (C-3), 126.32 (C-8), 126.89 (C-5), 131.42 (C-9), 132.26 (C-10), 133.46 (C-6), 134.42 (C-7), 160.13 (C-2), 180.40 (C=O), 185.31 (C=O); LC-MS (ESI−, m/z) calculated for C22H30O3: 342.22, found for 341.30 [M − H]−; HRMS-ESI: m/z calculated for C22H30O3: 342.2195 [M]+, found for 342.2197.
2-Hydroxy-3-isoprenyl-1,
4-naphthoquinone (
9a). The reaction produced
9a in 58.2% as a yellow solid; mp 142.5–144.0 °C (lit. [
36] 141–143 °C).
1H-NMR (500 MHz, CDCl
3) δ
H 1.66 (s, 3H, CH
3), 1.77 (s, 3H, CH
3), 3.29 (d,
J = 7.4 Hz, 2H, H-1′), 5.17–5.21 (m, 2H, H-2′), 7.27 (s, 1H, 2-OH), 7.66 (dt,
J = 1.1, 7.4 Hz, 1H, H-7), 7.73 (dt,
J = 1.4, 7.6 Hz, 1H, H-6), 8.05 (dd,
J = 1.0, 7.4 Hz, 1H, H-8), 8.10 (dd,
J = 1.0, 7.7 Hz, 1H, H-5);
13C-NMR (125 MHz, CDCl
3) δ
C 18.12 (CH
3), 22.84 (CH
3), 25.98 (C-1′), 119.85 (C-3), 123.68 (C-2′), 126.28 (C-8), 127.01 (C-5), 129.65 (C-9), 133.09 (C-6), 133.14 (C-10), 134.09 (C-3ʹ), 135.08 (C-7), 152.87 (C-2), 181.93 (C=O), 184.79 (C=O); LC-MS (ESI
−,
m/
z) calculated for C
15H
14O
3: 242.09, found for 241.16 [M − H]
–; HRMS-ESI:
m/
z calculated for C
15H
14O
3: 242.0943 [M]
+, found for 242.0948.
2-Isoprenyloxy-1,
4-naphthoquinone (
9b). The reaction produced
9b in 4.8% as a yellow solid; mp 158.0–159.6 °C (lit. [
37] 150–151 °C).
1H-NMR (500 MHz, CDCl
3) δ
H 1.74 (s, 3H, CH
3), 1.79 (s, 3H, CH
3), 4.57 (d,
J = 7.8 Hz, 2H, H-1′), 5.46–5.49 (m, 2H, H-2′), 6.14 (s, 1H, H-3), 7.68 (dt,
J = 1.5, 7.5 Hz, 1H, H-7), 7.72 (dt,
J = 1.5, 7.5 Hz, 1H, H-6), 8.06 (dd,
J = 1.5, 7.5 Hz, 1H, H-8), 8.10 (dd,
J = 1.5, 7.5 Hz, 1H, H-5);
13C-NMR (125 MHz, CDCl
3) δ
C 18.58 (CH
3), 26.05 (CH
3), 66.63 (C-1′), 110.73 (C-3), 117.41 (C-2′), 126.33 (C-8), 126.92 (C-5), 131.41 (C-9), 132.26 (C-10), 133.46 (C-6), 134.43 (C-7), 140.73 (C-3′), 159.80 (C-2), 180.51 (C=O), 185.28 (C=O); LC-MS (ESI
−,
m/
z) calculated for C
15H
14O
3: 242.09, found for 241.23 [M − H]
−; HRMS-ESI:
m/
z calculated for C
15H
14O
3: 242.0943 [M]
+, found for 242.0941.
2-Hydroxy-3-geranyl-1,
4-naphthoquinone (
10a). The reaction produced
10a in 50.8% as a yellow solid; mp 112.0–112.5 °C [
38].
1H-NMR (500 MHz, CDCl
3) δ
H 1.59 (s, 3H, CH
3), 1.65 (s, 3H, CH
3), 1.81 (s, 3H, CH
3), 1.98–2.01 (m, 2H, H-5′), 2.06–2.09 (m, 2H, H-6′), 3.34 (d,
J = 7.5 Hz, 2H, H-1′), 5.06–5.09 (m, 1H, H-7′), 5.23 (dt,
J = 1.0, 7.5 Hz, 1H, H-2′), 7.31 (s, 1H, 2-OH), 7.70 (dt,
J = 1.0, 7.5 Hz, 1H, H-7), 7.78 (dt,
J = 1.0, 7.5 Hz, 1H, H-6), 8.10 (dd,
J = 1.5, 7.5 Hz, 1H, H-8), 8.15 (dd,
J = 1.0, 7.5 Hz, 1H, H-5);
13C-NMR (125 MHz, CDCl
3) δ
C 16.47 (CH
3), 17.88 (CH
3), 22.77 (CH
3), 25.87 (C-1′), 26.82 (C-6′), 39.96 (C-5′), 119.67 (C-3), 123.76 (C-2′), 124.41 (C-7′), 126.28 (C-8), 127.00 (C-5), 129.65 (C-8′), 131.61 (C-9), 133.08 (C-6), 133.15 (C-10), 135.08 (C-7), 137.61 (C-3′), 152.88 (C-2), 181.94 (C=O), 184.74 (C=O); LC-MS (ESI
−,
m/
z) calculated for C
20H
22O
3: 310.16, found for 309.12 [M − H]
−; HRMS-ESI:
m/
z calculated for C
20H
22O
3: 310.1569 [M]
+, found for 310.1572.
2-Geranyloxy-1,4-naphthoquinone (10b). The reaction produced 10b in 5.2% as a pale yellow liquid. 1H-NMR (500 MHz, CDCl3) δH 1.59 (s, 3H, CH3), 1.65 (s, 3H, CH3), 1.81 (s, 3H, CH3), 1.99–2.02 (m, 2H, H-5′), 2.07–2.10 (m, 2H, H-6′), 4.58 (d, J = 6.6 Hz, 2H, H-1´), 5.06–5.10 (m, 1H, H-7´), 5.46 (dt, J = 0.8, 6.6 Hz, 1H, H-2′), 6.14 (s, 1H, H-3), 7.68 (dt, J = 1.0, 7.5 Hz, 1H, H-7), 7.74 (dt, J = 1.0, 7.5 Hz, 1H, H-6), 8.06 (dd, J = 1.5, 7.5 Hz, 1H, H-8), 8.10 (dt, J = 1.0, 7.5 Hz, 1H, H-5); 13C-NMR (125 MHz, CDCl3) δC 16.47 (CH3), 17.68 (CH3), 25.70 (CH3), 26.71 (C-6′), 39.65 (C-5′), 66.51 (C-1′), 110.56 (C-3), 117.22 (C-2′), 124.37 (C-7′), 126.23 (C-8), 126.88 (C-5), 131.18 (C-8′), 132.06 (C-9), 133.18 (C-6), 134.26 (C-10), 135.73 (C-7), 141.56 (C-3′), 159.56 (C-2), 180.29 (C=O), 185.06 (C=O); LC-MS (ESI−, m/z) calculated for C20H22O3: 310.16, found for 309.06 [M − H]−; HRMS-ESI: m/z calculated for C20H22O3: 310.1569 [M]+, found for 310.1574.
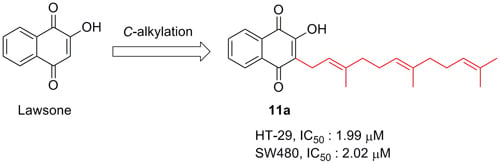
2-Hydroxy-3-farnesyl-1,
4-naphthoquinone (
11a). The reaction produced
11a in 35.2% as a yellow solid; mp 84.4–84.9 °C (lit. [
39] 68–69 °C).
1H-NMR (500 MHz, CDCl
3) δ
H 1.54 (s, 3H, CH
3), 1.54 (s, 6H, CH
3), 1.77 (s, 3H, CH
3), 1.92–1.95 (m, 2H, H-5′), 1.97–2.00 (m, 4H, H-9′, H-10′) 2.02–2.07 (m, 2H, H-6′), 3.30 (d,
J = 7.5 Hz, 2H, H-1′), 5.00–5.05 (m, 2H, H-7′, H-12′) 5.20 (t, 1H,
J = 7.3 Hz, 1H, H-2′), 7.28 (s, 1H, 2-OH), 7.65 (t,
J = 7.5 Hz, 1H, H-7), 7.73 (t,
J = 7.5 Hz, 1H, H-6), 8.05 (d,
J = 7.5 Hz, 1H, H-8), 8.11 (d,
J = 7.5 Hz, 1H, H-5);
13C-NMR (125 MHz, CDCl
3) δ
C 16.22 (CH
3), 16.48 (CH
3), 17.86 (CH
3), 22.77 (C-1′), 25.90 (CH
3), 26.69 (C-6′), 26.94 (C-11′), 39.89 (C-5′), 39.95 (C-9′), 119.69 (C-3), 123.75 (C-2′), 124.24 (C-7′), 124.57 (C-11′), 126.27 (C-8), 127.00 (C-5), 129.66 (C-9), 131.43 (C-10), 133.06 (C-6), 133.14 (C-12′), 135.06 (C-7), 135.24 (C-8′), 137.60 (C-3′), 152.89 (C-2), 181.92 (C=O), 184.74 (C=O); LC-MS (ESI
−,
m/
z) calculated for C
25H
30O
3: 378.22, found for 377.23 [M − H]
−; HRMS-ESI:
m/
z calculated for C
25H
30O
3: 378.2195 [M]
+, found for 378.2193.
2-Farnesyloxy-1,4-naphthoquinone (11b). The reaction produced 11b in 7.4% as a yellow liquid. 1H-NMR (500 MHz, CDCl3) δH 1.56 (s, 3H, CH3), 1.58 (s, 6H, CH3), 1.74 (s, 3H, CH3), 1.92–1.95 (m, 2H, H-5′), 2.00–2.03 (m, 2H, H-6′) 2.08–2.12 (m, 4H, H-9′, H-10′), 4.60 (d, J = 6.6 Hz, 2H, H-1′), 5.04–5.08 (m, 2H, H-7′, H-12′) 5.47 (dt, 1H, J = 0.8, 6.6 Hz, 1H, H-2′), 6.14 (s, 1H, H-3), 7.69 (dt, J = 1.5, 7.5 Hz, 1H, H-7), 7.72 (dt, J = 1.5, 7.5 Hz, 1H, H-6), 8.06 (dd, J = 1.5, 7.5 Hz, 1H, H-8), 8.10 (dd, J = 1.5, 7.5 Hz, 1H, H-5); 13C-NMR (125 MHz, CDCl3) δC 16.05 (CH3), 16.88 (CH3), 17.68 (CH3), 25.70 (CH3), 26.09 (C-6′), 26.71 (C-11′), 39.53 (C-5′), 39.67 (C-9′), 66.50 (C-1′), 110.55 (C-3), 116.91 (C-2′), 123.39 (C-7′), 124.28 (C-11′), 126.12 (C-8), 126.70 (C-5), 131.20 (C-9), 131.33 (C-10), 132.04 (C-12′), 133.24 (C-6), 134.21 (C-7), 135.72 (C-8′), 143.63 (C-3′), 159.58 (C-2), 180.31 (C=O), 185.04 (C=O); LC-MS (ESI−, m/z) calculated for C25H30O3: 378.22, found for 377.26 [M − H]−; HRMS-ESI: m/z calculated for C25H30O3: 378.2195 [M]+, found for 378.2190.
3.4. General Proceed Amination of Juglone with Alkyl Amine
A suspension of juglone (200 mg, 1.15 mmol) and alkyl amine (1.15 mmole) in 50 mL of ethanol was stirred for 1 h under reflux. The mixture was poured into water, 2 mL of 1 N hydrochloric acid was added, the precipitate was filtered off, washed with water, dried, and dissolved in chloroform, and the solution was applied to a column charged with silica gel. The column was eluted with ethyl acetate/n-hexane (1/5) to give the 2-amination and 3-amination products, respectively.
2-Butylamino-5-hydroxy-1,
4-naphthoquinone (
13a). The reaction produced
13a in 13.9% as an orange red solid; mp 154.6–155.4 °C (lit. [
41] 156–157 °C).
1H-NMR (500 MHz, CDCl
3) δ
H 0.96 (t,
J = 7.4 Hz, 3H, CH
3), 1.43 (p,
J = 7.5 Hz, 2H, H-2′), 1.67 (p,
J = 7.5 Hz, 2H, H-3′), 3.17 (q,
J = 6.9 Hz, 2H, H-1′), 5.61 (s, 1H, H-3), 6.04 (s, 1H, -NH), 7.22 (dd,
J = 0.8, 8.3 Hz, 1H, H-8), 7.44 (t,
J = 8.0 Hz, 1H, H-7), 7.56 (dd, 1H,
J = 0.8, 7.6 Hz, H-6), 13.08 (s, 1H, 5-OH);
13C-NMR (125 MHz, CDCl
3) δ
C 13.90 (CH
3), 20.40 (C-2′), 30.43 (C-3′), 42.63 (C-1′), 99.81 (C-3), 115.19 (C-9), 119.25 (C-6), 126.18 (C-8), 130.70 (C-10), 134.05 (C-7), 148.90 (C-2), 161.33 (C-5), 181.41 (C=O), 189.14 (C=O); LC-MS (ESI
+,
m/
z) calculated for C
14H
15NO
3: 245.11, found for 268.24 [M + Na]
+; HRMS-ESI:
m/
z calculated for C
14H
16O
3N: 246.1125 [M + H]
+, found for 246.1120.
3-Butylamino-5-hydroxy-1,
4-naphthoquinone (
13b). The reaction produced
13b in 6.9% as an orange red solid; mp 125.3–126.5 °C (lit. [
41] 111–112 °C).
1H-NMR (500 MHz, CDCl
3) δ
H 0.96 (t,
J = 7.5 Hz, 3H, CH
3), 1.42 (h,
J = 7.5 Hz, 2H, H-3′), 1.67 (p,
J = 7.5 Hz, 2H, H-2′), 3.17 (q,
J = 6.9 Hz, 2H, H-1′), 5.70 (s, 1H, H-3), 5.85 (bs, 1H, -NH), 7.12 (dd,
J = 2.3, 7.0 Hz, 1H, H-8), 7.57–7.62 (m, 2H, H-6, H-7), 11.52 (s, 1H, 5-OH);
13C-NMR (125 MHz, CDCl
3) δ
C 13.91 (CH
3), 20.42 (C-2′), 30.45 (C-3′), 42.56 (C-1′), 101.50 (C-3), 114.31 (C-9), 118.95 (C-6), 122.37 (C-8), 133.82 (C-10), 137.91 (C-7), 147.97 (C-2), 161.89 (C-5), 182.42 (C=O), 186.36 (C=O); LC-MS (ESI
+,
m/
z) calculated for C
14H
15NO
3: 245.11, found for 268.29 [M + Na]
+; HRMS-ESI:
m/
z calculated for C
14H
16O
3N: 246.1125 [M + H]
+, found for 246.1121.
2-Hexylamino-5-hydroxy-1,4-naphthoquinone (14a). The reaction produced 14a in 15.1% as an orange red solid; mp 126.0–127.5 °C. 1H-NMR (500 MHz, CDCl3) δH 0.88 (t, J = 7.5 Hz, 3H, CH3), 1.30–1.32 (m, 4H, H-4′, H-5′), 1.36–1.40 (m, 2H, H-3′), 1.67 (p, J = 7.3 Hz, 2H, H-2′), 3.16 (q, J = 6.8 Hz, 2H, H-1′), 5.60 (s, 1H, H-3), 6.05 (bs, 1H, -NH), 7.22 (d, J = 8.4 Hz, 1H, H-8), 7.44 (t, J = 7.9 Hz, 1H, H-7), 7.56 (d, J = 7.2 Hz, 1H, H-6), 13.08 (s, 1H, 5-OH); 13C-NMR (125 MHz, CDCl3) δC 14.19 (CH3), 22.72 (C-5′), 26.88 (C-2′), 28.38 (C-3′), 31.59 (C-4′), 42.93 (C-1′), 99.78 (C-3), 115.18 (C-9), 119.24 (C-6), 126.16 (C-8), 130.68 (C-10), 134.03 (C-7), 148.87 (C-2), 161.31 (C-5), 181.40 (C=O), 189.12 (C=O); LC-MS (ESI+, m/z) calculated for C16H19NO3: 273.14, found for 296.29 [M + Na]+; HRMS-ESI: m/z calculated for C16H20O3N: 274.1438 [M+H]+, found for 274.1435
3-Hexylaminp-5-hydroxy-1,4-naphthoquinone (14b). The reaction produced 14b in 24.1% as an orange red solid; mp 110.2–111.1 °C. 1H-NMR (500 MHz, CDCl3) δH 0.88 (t, J = 6.4 Hz, 3H, CH3), 1.31–1.32 (m, 4H, H-4′, H-5′), 1.39 (p, J = 6.9 Hz, 2H, H-3′), 1.69 (p, J = 7.3 Hz, 2H, H-2′), 3.16 (q, J = 6.6 Hz, 2H, H-1′), 5.69 (s, 1H, H-2), 5.87 (bs, 1H, -NH), 7.11 (dd, J = 2.0, 7.2 Hz, 1H, H-6), 7.57–7.61 (m, 2H, H-7, H-8), 11.52 (s, 1H, 5-OH); 13C-NMR (125 MHz, CDCl3) δC 13.97 (CH3), 22.50 (C-5′), 26.68 (C-2′), 28.17 (C-3′), 31.38 (C-4′), 42.63 (C-1′), 101.24 (C-2), 114.07 (C-9), 118.71 (C-6), 122.14 (C-8), 133.58 (C-10), 137.67 (C-7), 147.73 (C-3), 161.66 (C-5), 182.18 (C=O), 186.11 (C=O); LC-MS (ESI+, m/z) calculated for C16H19NO3: 273.14, found for 296.26 [M + Na]+; HRMS-ESI: m/z calculated for C16H20O3N: 274.1438 [M + H]+, found for 274.1433.
2-Octylamino-5-hydroxy-1,4-naphthoquinone (15a). The reaction produced 15a in 14.1% as an orange red solid; mp 128.2–129.5 °C. 1H-NMR (500 MHz, CDCl3) δH 0.87 (t, J = 6.6 Hz, 3H, CH3), 1.26–1.29 (m, 6H, H-5′, H-6′, H-7′), 1.36–1.39 (m, 4H, H-3′, H-4′), 1.67 (p, J = 7.3 Hz, 2H, H-2′), 3.16 (q, J = 6.6 Hz, 2H, H-1′), 5.60 (s, 1H, H-3), 6.04 (bs, 1H, -NH), 7.22 (d, J = 8.0 Hz, 1H, H-8), 7.44 (t, J = 7.9 Hz, 1H, H-7), 7.57 (d, J = 7.5 Hz, 1H, H-6), 13.08 (s, 1H, 5-OH); 13C-NMR (125 MHz, CDCl3) δC 14.29 (CH3), 22.83 (C-7′), 27.22 (C-2′), 28.43 (C-3′), 29.35 (C-4′), 29.92 (C-5′), 31.95 (C-6′), 42.94 (C-1′), 99.80 (C-3), 115.20 (C-9), 119.25 (C-6), 126.18 (C-8), 130.70 (C-10), 134.04 (C-7), 148.88 (C-2), 161.33 (C-5), 181.42 (C=O), 189.14 (C=O); LC-MS (ESI+, m/z) calculated for C18H23NO3: 301.17, found for 324.34 [M + Na]+; HRMS-ESI: m/z calculated for C18H24O3N: 302.1751 [M + H]+, found for 302.1746.
3-Octylamino-5-hydroxy-1,4-naphthoquinone (15b). The reaction produced 15b in 31.4% as an orange red solid; mp 119.5–120.3 °C. 1H-NMR (500 MHz, CDCl3) δH 0.87 (t, J = 6.3 Hz, 3H, CH3), 1.23–1.29 (m, 6H, H-5′, H-6′, H-7′), 1.37–1.39 (m, 2H, H-4′), 1.65–1.70 (m, 4H, H-2′, H-3′), 3.16 (q, J = 6.4 Hz, 2H, H-1′), 5.70 (s, 1H, H-2), 5.87 (bs, 1H, -NH), 7.11 (d, J = 7.3, 1H, H-8), 7.57–7.61 (m, 2H, H-6, H-7), 11.52 (s, 1H, 5-OH); 13C-NMR (125 MHz, CDCl3) δC 14.29 (CH3), 22.84 (C-7′), 27.24 (C-2′), 28.45 (C-3′), 29.36 (C-4′), 29.42 (C-5′), 31.96 (C-6′), 42.90 (C-1′), 101.46 (C-2), 114.31 (C-9), 118.98 (C-6), 122.40 (C-8), 133.81 (C-10), 137.93 (C-7), 148.02 (C-3), 161.93 (C-5), 182.40 (C=O), 186.33 (C=O); LC-MS (ESI+, m/z) calculated for C18H23NO3: 301.17, found for 324.36 [M + Na]+; HRMS-ESI: m/z calculated for C18H24O3N: 302.1751 [M + H]+, found for 302.1749.
2-Decylamino-5-hydroxy-1,4-naphthoquinone (16a). The reaction produced 16a in 19.2% as an orange red solid; mp 124.5–125.7 °C. 1H-NMR (500 MHz, CDCl3) δH 0.86 (t, J = 6.8 Hz, 3H, CH3), 1.23–1.30 (m, 8H, H-6′, H-7′, H-8′, H-9′), 1.35–1.40 (m, 6H, H-3′, H-4′, H-5′), 1.67 (p, J = 7.3 Hz, 2H, H-2′), 3.17 (q, J = 6.7 Hz, 2H, H-1′), 5.61 (s, 1H, H-3), 6.05 (bs, 1H, -NH), 7.22 (dd, J = 2.2, 7.3 Hz, 1H, H-8), 7.44 (dt, 1H, J = 1.0, 7.5 Hz, H-7), 7.56 (dd, 1H, J = 1.0, 7.5 Hz, H-6), 13.08 (s, 1H, 5-OH); 13C-NMR (125 MHz, CDCl3) δC 14.09 (CH3), 22.65 (C-9′), 26.98 (C-2′), 28.19 (C-3′), 29.20 (C-4′), 29.25 (C-5′), 29.46 (C-6′), 29.47 (C-7′), 31.85 (C-8′), 42.71 (C-1′), 99.57 (C-3), 114.97 (C-9), 119.02 (C-6), 125.95 (C-8), 130.47 (C-10), 133.82 (C-7), 148.66 (C-2), 161.10 (C-5), 181.20 (C=O), 188.91 (C=O); LC-MS (ESI+, m/z) calculated for C20H29NO3: 329.20, found for 352.47 [M + Na]+; HRMS-ESI: m/z calculated for C20H28O3N: 330.2064 [M + H]+, found for 330.2058.
3-Decylamino-5-hydroxy-1,4-naphthoquinone (16b). The reaction produced 16b in 39.5% as an orange red solid; mp 95.8–96.9 °C. 1H-NMR (500 MHz, CDCl3) δH 0.86 (t, J = 6.8 Hz, 3H, CH3), 1.23–1.31 (m, 10H, H-5′, H-6′, H-7′, H-8′, H-9′), 1.35–1.40 (m, 4H, H-3′, H-4′,), 1.67 (p, J = 7.3 Hz, 2H, H-2′′), 3.16 (q, J = 6.7 Hz, 2H, H-1′), 5.68 (s, 1H, H-2), 5.86 (bs, 1H, -NH), 7.11 (dd, J = 2.2, 7.3 Hz, 1H, H-8), 7.57–7.61 (m, 2H, H-6, H-7), 11.51 (s, 1H, 5-OH); 13C-NMR (125 MHz, CDCl3) δC 14.32 (CH3), 22.88 (C-9′), 27.24 (C-2′), 28.44 (C-3′), 29.45 (C-4′), 29.48 (C-5′), 29.70 (C-6′), 29.92 (C-7′), 32.08 (C-8′), 42.89 (C-1′), 101.45 (C-2), 114.30 (C-9), 118.97 (C-6), 122.39 (C-8), 133.80 (C-10), 137.91 (C-7), 148.02 (C-3), 161.92 (C-5), 182.38 (C=O), 186.31 (C=O); LC-MS (ESI+, m/z) calculated for C20H29NO3: 329.20, found for 330.26 [M + H]+; HRMS-ESI: m/z calculated for C20H28O3N: 330.2064 [M + H]+, found for 330.2059.
2-Dodecylamino-5-hydroxy-1,4-naphthoquinone (17a). The reaction produced 17a in 15.5% as an orange red solid; mp 121.8–122.2 °C. 1H-NMR (500 MHz, CDCl3) δH 0.86 (t, J = 6.8 Hz, 3H, CH3), 1.24–1.31 (m, 14H, H-5′, H-6′, H-7′, H-8′, H-9′, H-10′, H-11′), 1.34–1.40 (m, 4H, H-3′, H-4′), 1.67 (p, J = 7.3 Hz, 2H, H-2′), 3.16 (q, J = 6.6 Hz, 2H, H-1′), 5.60 (s, 1H, H-3), 6.05 (bs, 1H, -NH), 7.22 (d, J = 8.4 Hz, 1H, H-8), 7.44 (t, 1H, J = 7.9 Hz, H-7), 7.56 (d, 1H, J = 7.5 Hz, H-6), 13.08 (s, 1H, 5-OH); 13C-NMR (125 MHz, CDCl3) δC 14.34 (CH3), 22.90 (C-11′), 27.22 (C-2′), 28.42 (C-3′), 29.44 (C-4′), 29.55 (C-5′), 29.69 (C-6′), 29.75 (C-7′), 29.83 (C-8′), 29.92 (C-9′), 32.12 (C-10′), 42.94 (C-1′), 99.80 (C-3), 115.20 (C-9), 119.25 (C-6), 126.18 (C-8), 130.70 (C-10), 134.04 (C-7), 148.88 (C-2), 161.33 (C-5), 181.42 (C=O), 189.13 (C=O); LC-MS (ESI+, m/z) calculated for C22H31NO3: 357.23, found for 380.44 [M + Na]+; HRMS-ESI: m/z calculated for C22H32O3N: 358.2377 [M + H]+, found for 358.2371.
3-Dodecylamino-5-hydroxy-1,4-naphthoquinone (17b). The reaction produced 17b in 24.6% as an orange red solid; mp 98.0–98.5 °C. 1H-NMR (500 MHz, CDCl3) δH 0.86 (t, J = 6.7 Hz, 3H, CH3), 1.24–1.31 (m, 12H, H-6′, H-7′, H-8′, H-9′, H-10′, H-11′), 1.35–1.39 (m, 4H, H-4′, H-5′), 1.65–1.70 (m, 4H, H-2′, H-3′), 3.15 (t, J = 6.1 Hz, 2H, H-1′), 5.70 (s, 1H, H-2), 5.87 (bs, 1H, -NH), 7.11 (d, J = 7.4 Hz, 1H, H-8), 7.57–7.61 (m, 2H, H-6, H-7), 11.52 (s, 1H, 5-OH); 13C-NMR (125 MHz, CDCl3) δC 14.34 (CH3), 22.90 (C-11′), 27.24 (C-2′), 28.44 (C-3′), 29.45 (C-4′), 29.55 (C-5), 29.70 (C-6′), 29.76 (C-7′), 29.83 (C-8′), 29.92 (C-9′), 32.13 (C-10′), 42.87 (C-1′), 101.49 (C-2), 114.32 (C-9), 118.95 (C-6), 122.37 (C-8), 133.83 (C-10), 137.91 (C-7), 147.97 (C-3), 161.90 (C-5), 182.42 (C=O), 186.34 (C=O); LC-MS (ESI+, m/z) calculated for C22H31NO3: 357.23, found for 380.46 [M + Na]+; HRMS-ESI: m/z calculated for C22H32O3N: 358.2377 [M + H]+, found for 358.2374.
2-[(2-Aminoethyl)-morpholine]-5-hydroxy-1,4-naphthoquinone (18a). The reaction produced 18a in 38.9% as a red solid; mp 122.0–122.5 °C. 1H-NMR (500 MHz, CDCl3) δH 2.48 (t, J = 4.2 Hz, 4H, H-2′′, H-6′′), 2.67 (t, J = 6.0 Hz, 2H, H-2′), 3.22 (q, J = 5.7 Hz, 2H, H-1′), 3.72 (t, J = 4.6 Hz, 4H, H-3′′, H-5′′), 5.59 (s, 1H, H-3), 6.66 (bs, 1H, -NH), 7.22 (dd, J = 0.9, 8.4 Hz, 1H, H-8), 7.45 (t, J = 7.9 Hz, 1H, H-7), 7.57 (dd, J = 0.9, 7.2 Hz, 2H, H-6), 13.05 (s, 1H, 5-OH); 13C-NMR (125 MHz, CDCl3) δC 38.68 (C-1′), 53.39 (C-2′′, C-6′′), 55.68 (C-2′), 67.10 (C-3′′, C-5′′), 100.12 (C-2), 115.18 (C-9), 119.22 (C-6), 126.13 (C-8), 130.76 (C-10), 134.13 (C-7), 148.89 (C-3), 161.32 (C-5), 181.28 (C=O), 189.17 (C=O); LC-MS (ESI−, m/z) calculated for C16H18N2O4: 302.13, found for 301.08 [M − H]−; HRMS-ESI: m/z calculated for C16H18N2O4: 302.1267 [M]+, found for 302.1264.
3-[(2-Aminoethyl)-morpholine]-5-hydroxy-1,4-naphthoquinone (18b). The reaction produced 18b in 16.8% as a red solid; mp 150.2–151.4 °C. 1H-NMR (500 MHz, CDCl3) δH 2.49 (t, J = 8.5 Hz, 4H, H-2′′, H-6′′), 2.69 (t, J = 6.0 Hz, 2H, H-2′), 3.20 (q, J = 5.5 Hz, 2H, H-1′), 3.74 (t, J = 5.0 Hz, 4H, H-3′′, H-5′′), 5.66 (s, 1H, H-3), 6.49 (bs, 1H, -NH), 7.12 (dd, J = 2.7, 6.8 Hz, 1H, H-8), 7.57–7.61 (m, 2H, H-6, H-7), 11.56 (s, 1H, 5-OH); 13C-NMR (125 MHz, CDCl3) δC 38.36 (C-1′), 53.16 (C-2′′, C-6′′), 55.49 (C-2′), 66.65 (C-3′′, C-5′′), 99.97 (C-2), 114.94 (C-9), 119.05 (C-6), 125.92 (C-8), 130.55 (C-10), 133.96 (C-7), 148.65 (C-2), 161.11 (C-5), 181.01 (C=O), 188.97 (C=O); LC-MS (ESI−, m/z) calculated for C16H18N2O4: 302.13, found for 301.16 [M − H]−; HRMS-ESI: m/z calculated for C16H18N2O4: 302.1267 [M]+, found for 302.1261.
2-[(3-Aminopropyl)-morpholine]-5-hydroxy-1,4-naphthoquinone (19a). The reaction produced 19a in 29.2% as a red solid; mp 84.5–85.6 °C. 1H-NMR (500 MHz, CDCl3) δH 1.84 (p, J = 5.9 Hz, 2H, H-2′), 2.49 (bs, 4H, H-2′′, H-6′′), 2.54 (t, J = 5.8 Hz, 2H, H-3′), 3.27 (q, J = 5.7 Hz, 2H, H-1′), 3.84 (t, J = 4.6 Hz, 4H, H-3′′, H-5′′), 5.55 (s, 1H, H-3), 7.20 (dd, J = 0.8, 8.0 Hz, 1H, H-8), 7.43 (t, J = 7.9 Hz, 1H, H-7), 7.56 (dd, J = 0.9, 7.6 Hz, 1H, H-6), 8.08 (bs, 1H, -NH), 13.17 (s, 1H, 5-OH); 13C-NMR (125 MHz, CDCl3) δC 23.21 (C-2′), 43.15 (C-1′), 53.83 (C-2′′, C-6′′), 57.91 (C-3′), 66.76 (C-3′′, C-5′′), 99.04 (C-2), 115.12 (C-9), 118.89 (C-6), 125.70 (C-8), 130.69 (C-10), 133.70 (C-7), 149.46 (C-3), 161.06 (C-5), 181.21 (C=O), 188.84 (C=O); LC-MS (ESI−, m/z) calculated for C17H20N2O4: 316.14, found for 315.10 [M − H]−; HRMS-ESI: m/z calculated for C17H20O4N2: 316.1423 [M]+, found for 316.1428.
2-[(3-Aminopropyl)-morpholine]-5-hydroxy-1,4-naphthoquinone (19b). The reaction produced 19b in 30.2% as a red solid; mp 101.6–102.7 °C. 1H-NMR (500 MHz, CDCl3) δH 1.84 (p, J = 6.0 Hz, 2H, H-2′), 2.50 (bs, 4H, H-2′′, H-6′′), 2.55 (t, J = 5.8 Hz, 2H, H-3′), 3.25 (q, J = 5.5 Hz, 2H, H-1′), 3.84 (t, J = 4.6 Hz, 4H, H-3′′, H-5′′), 5.63 (s, 1H, H-3), 7.10 (dd, J = 2.2, 7.3 Hz, 1H, H-8), 7.56–7.60 (m, 2H, H-6, H-7), 7.99 (bs, 1H, -NH), 11.63 (s, 1H, 5-OH); 13C-NMR (125 MHz, CDCl3) δC 23.15 (C-2′), 43.24 (C-1′), 53.83 (C-2′′, C-6′′), 58.05 (C-3′), 66.78 (C-3′′, C-5′′), 100.77 (C-3), 114.22 (C-9), 118.60 (C-6), 122.05 (C-8), 133.76 (C-10), 137.55 (C-7), 148.55 (C-2), 161.69 (C-5), 182.17 (C=O), 186.51 (C=O); LC-MS (ESI−, m/z) calculated for C17H20N2O4: 316.14, found for 315.11 [M − H]−; HRMS-ESI: m/z calculated for C17H20O4N2: 316.1423 [M]+, found for 316.1428.