Mini Review of Phytochemicals and Plant Taxa with Activity as Microbial Biofilm and Quorum Sensing Inhibitors
Abstract
:1. Introduction
2. Model Organism Bioassays
3. Phytochemicals as QS and Biofilm Inhibitors
3.1. Phenolics
3.1.1. Phenylpropenoids
3.1.2. Benzoic Acid Derivatives
| Active Constituents (Source Plants) | Biological Activities | Ref. |
|---|---|---|
| Phenylpropenoids | ||
eugenol (various species)  |
| [23] [23] [24] [24] [25] |
| cinnamaldehyde (Cinnamomum sp.—Lauraceae)  |
| [24] [26] [24] [27] [28] |
| Benzoic Acid Derivatives | ||
| vanillin (Vanilla planifolia Jacks. ex Andrews—Orchidaceae)  |
| [29] [30] [31] |
| gallic acid (various species)  |
| [31] [33] [34] [35] |
| ellagic acid (various species)  |
| [17] [17] [32] [36] [37] |
| Tannins | ||
| 1,2,3,4,6-penta-O-galloyl-ß-d-glucopyranose (various species)  |
| [38] |
| punicalagin (Punica granatum L. (Lythraceae) and Combretaceae species)  |
| [39] |
| hamamelitannin (Hamamelis virginiana L.—Hamamelidaceae)  |
| [40] |
| tannic acid (various species)  |
| [17] [17] [32] [32] [41] [42] [42] |
| Stilbenes | ||
| resveratrol (various species)  |
| [33] [42] [43] |
| pterostilbene (Vitis sp.—Vitaceae and Ericaceae species)  |
| [44] |
| Flavonoids | ||
| quercetin (various species)  |
| [45] [46] [46] |
| (−)-catechin (Camellia sinensis (L.) Kuntze—Theaceae and others)  |
| [47] |
| (−)-epicatechin (Camellia sinensis (L.) Kuntze—Theaceae and others)  |
| [31] [31] [35] [35] [47] |
| (–)-gallocatechin (Camellia sinensis (L.) Kuntze—Theaceae)  |
| [34] |
| (–)-epigallocatechin (Camellia sinensis (L.) Kuntze—Theaceae and others)  |
| [34] |
| (–)-catechin gallate (Camellia sinensis (L.) Kuntze—Theaceae) 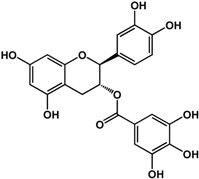 |
| [34] |
| (–)-epicatechin gallate (Camellia sinensis (L.) Kuntze—Theaceae and others)  |
| [34] |
| (–)-gallocatechin gallate (Camellia sinensis (L.) Kuntze—Theaceae)  |
| [34] |
| (−)-epigallocatechin gallate (Camellia sinensis (L.) Kuntze—Theaceae)  |
| [17] [17] [34] |
| Diarylheptanoids | ||
| curcumin (Curcuma longa L.—Zingiberaceae)  |
| [27] [48] [48] [49] [50] [51] [52] [53] [53] [53] [54] |
3.1.3. Tannins
3.1.4. Stilbenes and Flavonoids
3.1.5. Diarylheptanoids
3.2. Terpenoids
| Active Constituents (Source Plants) | Biological Activities | Ref. |
|---|---|---|
| Monoterpenes | ||
| thymol (Thymus vulgaris L.—Lamiaceae)  |
| [24] [24] [56] [57] |
| carvacrol (Thymus vulgaris L. and other Lamiaceae species)  |
| [24] [24] [56] |
| Sesquiterpenes | ||
| salvipisone (Salvia sclarea L.—Lamiaceae)  |
| [58,59] |
| acanthospermolide (Acanthospermum hispidum DC.—Asteraceae)  |
| [60] |
| Triterpenoids | ||
| isolimonic acid (Citrus × aurantium L.—Rutaceae)  |
| [61] |
| ichangin (Citrus × aurantium L.—Rutaceae)  |
| [61] |
| betulinic acid (various species)  |
| [42] |
| ursolic acid (various species)  |
| [21] [21] [62] |
| gymnemic acids (Gymnema sylvestre (Retz.) R.Br. ex Sm.—Apocynaceae and Asclepiadaceae species)  |
| [63] |
3.3. Sulfur-Containing Phytochemicals
| Active Constituents (Source Plants) | Biological Activities | Ref. |
|---|---|---|
| Sulfur-Containing Compounds | ||
allicin (Allium sativum L.—Amaryllidaceae)  |
| [32] [64] [65] |
ajoene (Allium sativum L.—Amaryllidaceae) 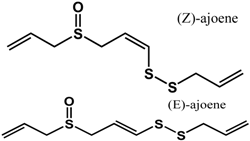 |
| [66] [66] |
sulforaphane (Brassicaceae species)  |
| [67] |
| Sulfur-Containing Compounds | ||
allyl isothiocyanate (Brassicaceae species) 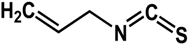 |
| [35] [68] |
| Coumarins | ||
aesculetin (various species)  |
| [18] [36] [69] |
umbelliferone (Apiaceae species)  |
| [69] [69] [70] |
| Quninones | ||
chrysophanol (various species)  |
| [71] |
emodin (various species)  |
| [71] |
shikonin (Boraginaceae species)  |
| [71] |
purpurin (Rubia tinctorum L.—Rubiaceae)  |
| [72] |
| Alkaloids | ||
berberine (Berberidaceae species and others)  |
| [25] [73] |
chelerythrine (Papaveraceae species)  |
| [74] |
sanguinarine (Papaveraceae species)  |
| [74] |
| Alkaloids | ||
reserpine (Rauwolfia sp.—Apocynaceae)  |
| [25] |
3.4. Coumarins
3.5. Quinones
3.6. Alkaloids
4. Taxa and Habitats
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AHL | N-acyl-homoserine lactone |
| AIPs | autoinducing peptides for signaling |
| EPS | extrapolymeric substance |
| GFP | green fluorescent protein |
| HPLC-DAD | High performance liquid chromatography, diode array detection |
| QS | quorum sensing |
References
- Davies, D.G.; Parsek, M.R.; Pearson, J.P.; Iglewski, B.H.; Costerton, J.W.; Greenberg, E.P. The Involvement of Cell-to-Cell Signals in the Development of a Bacterial Biofilm. Science 1998, 280, 295–298. [Google Scholar] [CrossRef] [PubMed]
- Davey, M.E.; O’Toole, G.A. Microbial Biofilms: From Ecology to Molecular Genetics. Microbiol. Mol. Biol. Rev. 2000, 64, 847–867. [Google Scholar] [CrossRef] [PubMed]
- Donlan, R.M.; Costerton, J.W. Biofilms: Survival Mechanisms of Clinically Relevant Microorganisms. Clin. Microbiol. Rev. 2002, 15, 167–193. [Google Scholar] [CrossRef] [PubMed]
- Hall-Stoodley, L.; Costerton, J.W.; Stoodley, P. Bacterial Biofilms: From the Natural Environment to Infectious Diseases. Nat. Rev. Microbiol. 2004, 2, 95–108. [Google Scholar] [CrossRef] [PubMed]
- Raffa, R.B.; Iannuzzo, J.R.; Levine, D.R.; Saeid, K.K.; Schwartz, R.C.; Sucic, N.T.; Terleckyj, O.D.; Young, J.M. Bacterial Communication (“Quorum Sensing”) Via Ligands and Receptors: A Novel Pharmacologic Target for the Design of Antibiotic Drugs. J. Pharmacol. Exp. Ther. 2005, 312, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Alem, M.A.; Oteef, M.D.; Flowers, T.H.; Douglas, L.J. Production of Tyrosol by Candida albicans Biofilms and its Role in Quorum Sensing and Biofilm Development. Eukaryot. Cell 2006, 5, 1770–1779. [Google Scholar] [CrossRef] [PubMed]
- Sudoh, M.; Nagahashi, S.; Doi, M.; Ohta, A.; Takagi, M.; Arisawa, M. Cloning of the Chitin Synthase 3 Gene from Candida albicans and its Expression during Yeast-Hyphal Transition. Mol. Gen. Genet. 1993, 241, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Cramton, S.E.; Gerke, C.; Schnell, N.F.; Nichols, W.W.; Götz, F. The Intercellular Adhesion (Ica) Locus is Present in Staphylococcus aureus and is Required for Biofilm Formation. Infect. Immun. 1999, 67, 5427–5433. [Google Scholar] [PubMed]
- Singh, P.K.; Schaefer, A.L.; Parsek, M.R.; Moninger, T.O.; Welsh, M.J.; Greenberg, E.P. Quorum-Sensing Signals Indicate that Cystic Fibrosis Lungs are Infected with Bacterial Biofilms. Nature 2000, 407, 762–764. [Google Scholar] [CrossRef] [PubMed]
- Huber, B.; Riedel, K.; Hentzer, M.; Heydorn, A.; Gotschlich, A.; Givskov, M.; Molin, S.; Eberl, L. The Cep Quorum-Sensing System of Burkholderia cepacia H111 Controls Biofilm Formation and Swarming Motility. Microbiology 2001, 147, 2517–2528. [Google Scholar] [CrossRef] [PubMed]
- Borucki, M.K.; Peppin, J.D.; White, D.; Loge, F.; Call, D.R. Variation in Biofilm Formation among Strains of Listeria monocytogenes. Appl. Environ. Microbiol. 2003, 69, 7336–7342. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.K.; Chandra, J. Candida Biofilm Resistance. Drug Resist. Updat. 2004, 7, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Rabin, N.; Zheng, Y.; Opoku-Temeng, C.; Du, Y.; Bonsu, E.; Sintim, H.O. Agents that inhibits bacterial biofilm formation. Future Med. Chem. 2015, 7, 647–671. [Google Scholar] [CrossRef] [PubMed]
- Nealson, K.H.; Platt, T.; Hastings, J.W. Cellular Control of the Synthesis and Activity of the Bacterial Luminescent System. J. Bacteriol. 1970, 104, 313–322. [Google Scholar] [PubMed]
- Meighen, E.A. Molecular Biology of Bacterial Bioluminescence. Microbiol. Rev. 1991, 55, 123–142. [Google Scholar] [PubMed]
- Adonizio, A.L.; Downum, K.; Bennett, B.C.; Mathee, K. Anti-Quorum Sensing Activity of Medicinal Plants in Southern Florida. J. Ethnopharmacol. 2006, 105, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Huber, B.; Eberl, L.; Feucht, W.; Polster, J. Influence of Polyphenols on Bacterial Biofilm Formation and Quorum-Sensing. Z. Naturforsch. C 2003, 58, 879–884. [Google Scholar] [CrossRef] [PubMed]
- Brackman, G.; Hillaert, U.; van Calenbergh, S.; Nelis, H.J.; Coenye, T. Use of Quorum Sensing Inhibitors to Interfere with Biofilm Formation and Development in Burkholderia Multivorans and Burkholderia Cenocepacia. Res. Microbiol. 2009, 160, 144–151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Girennavar, B.; Cepeda, M.L.; Soni, K.A.; Vikram, A.; Jesudhasan, P.; Jayaprakasha, G.K.; Pillai, S.D.; Patil, B.S. Grapefruit Juice and its Furocoumarins Inhibits Autoinducer Signaling and Biofilm Formation in Bacteria. Int. J. Food Microbiol. 2008, 125, 204–208. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhang, B.; Li, P. Purification and Identification of Quorum Sensing Signal Peptide AIP of Lactobacillus Pentosus. J. Chin. Inst. Food Sci. Technol. 2015, 15, 201–206. [Google Scholar]
- Ren, D.; Zuo, R.; Barrios, A.F.G.; Bedzyk, L.A.; Eldridge, G.R.; Pasmore, M.E.; Wood, T.K. Differential Gene Expression for Investigation of Escherichia Coli Biofilm Inhibition by Plant Extract Ursolic Acid. Appl. Environ. Microbiol. 2005, 71, 4022–4034. [Google Scholar] [CrossRef] [PubMed]
- Dewick, P.M. Medicinal Natural Products: A Biosynthetic Approach, 2nd ed.; John Wiley & Sons, Ltd.: West Sussex, UK, 2002. [Google Scholar]
- Zhou, L.; Zheng, H.; Tang, Y.; Yu, W.; Gong, Q. Eugenol Inhibits Quorum Sensing at Sub-Inhibitory Concentrations. Biotechnol. Lett. 2013, 35, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, A.; Upadhyaya, I.; Kollanoor-Johny, A.; Venkitanarayanan, K. Antibiofilm Effect of Plant Derived Antimicrobials on Listeria monocytogenes. Food Microbiol. 2013, 36, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Magesh, H.; Kumar, A.; Alam, A.; Priyam; Sekar, U.; Sumantran, V.N.; Vaidyanathan, R. Identification of Natural Compounds which Inhibit Biofilm Formation in Clinical Isolates of Klebsiella Pneumoniae. Indian J. Exp. Biol. 2013, 51, 764–772. [Google Scholar] [PubMed]
- Brackman, G.; Defoirdt, T.; Miyamoto, C.; Bossier, P.; van Calenbergh, S.; Nelis, H.; Coenye, T. Cinnamaldehyde and Cinnamaldehyde Derivatives Reduce Virulence in Vibrio spp. by Decreasing the DNA-Binding Activity of the Quorum Sensing Response Regulator LuxR. BMC Microbiol. 2008, 8. [Google Scholar] [CrossRef] [PubMed]
- Sharma, G.; Raturi, K.; Dang, S.; Gupta, S.; Gabrani, R. Combinatorial Antimicrobial Effect of Curcumin with Selected Phytochemicals on Staphylococcus epidermidis. J. Asian Nat. Prod. Res. 2014, 16, 535–541. [Google Scholar] [CrossRef] [PubMed]
- Amalaradjou, M.A.R.; Venkitanarayanan, K. Effect of trans-cinnamaldehyde on Inhibition and Inactivation of Cronobacter sakazakii Biofilm on Abiotic Surfaces. J. Food Prot. 2011, 74, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Ponnusamy, K.; Paul, D.; Kweon, J.H. Inhibition of Quorum Sensing Mechanism and Aeromonas hydrophila Biofilm Formation by Vanillin. Environ. Eng. Sci. 2009, 26, 1359–1363. [Google Scholar] [CrossRef]
- Kappachery, S.; Paul, D.; Yoon, J.; Kweon, J.H. Vanillin, a Potential Agent to Prevent Biofouling of Reverse Osmosis Membrane. Biofouling 2010, 26, 667–672. [Google Scholar] [CrossRef] [PubMed]
- Plyuta, V.; Zaitseva, J.; Lobakova, E.; Zagoskina, N.; Kuznetsov, A.; Khmel, I. Effect of Plant Phenolic Compounds on Biofilm Formation by Pseudomonas aeruginosa. APMIS 2013, 121, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Ta, C.A.; Freundorfer, M.; Mah, T.F.; Otarola-Rojas, M.; Garcia, M.; Sanchez-Vindas, P.; Poveda, L.; Maschek, J.A.; Baker, B.J.; Adonizio, A.L.; et al. Inhibition of Bacterial Quorum Sensing and Biofilm Formation by Extracts of Neotropical Rainforest Plants. Planta Med. 2014, 80, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Morán, A.; Gutiérrez, S.; Martínez-Blanco, H.; Ferrero, M.; Monteagudo-Mera, A.; Rodríguez-Aparicio, L. Non-Toxic Plant Metabolites Regulate Staphylococcus Viability and Biofilm Formation: A Natural Therapeutic Strategy Useful in the Treatment and Prevention of Skin Infections. Biofouling 2014, 30, 1175–1182. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, T.; Nakahara, A.; Minnatul, K.M.; Noiri, Y.; Ebisu, S.; Kato, A.; Azakami, H. The Inhibitory Effects of Catechins on Biofilm Formation by the Periodontopathogenic Bacterium, Eikenella Corrodens. Biosci. Biotechnol. Biochem. 2010, 74, 2445–2450. [Google Scholar] [CrossRef] [PubMed]
- Borges, A.; Serra, S.; Cristina Abreu, A.; Saavedra, M.J.; Salgado, A.; Simões, M. Evaluation of the Effects of Selected Phytochemicals on Quorum Sensing Inhibition and in Vitro Cytotoxicity. Biofouling 2014, 30, 183–195. [Google Scholar] [CrossRef] [PubMed]
- Dürig, A.; Kouskoumvekaki, I.; Vejborg, R.M.; Klemm, P. Chemoinformatics-Assisted Development of New Anti-Biofilm Compounds. Appl. Microbiol. Biotechnol. 2010, 87, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Bakkiyaraj, D.; Nandhini, J.R.; Malathy, B.; Pandian, S.K. The Anti-Biofilm Potential of Pomegranate (Punica granatum L.) Extract Against Human Bacterial and Fungal Pathogens. Biofouling 2013, 29, 929–937. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.H.; Chang, F.R.; Hua, M.Y.; Wu, Y.C.; Liu, S.T. Inhibitory Effects of 1,2,3,4,6-Penta-O-Galloyl-Beta-d-Glucopyranose on Biofilm Formation by Staphylococcus aureus. Antimicrob. Agents Chemother. 2011, 55, 1021–1027. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Yan, C.; Xu, Y.; Feng, Y.; Wu, Q.; Lv, X.; Yang, B.; Wang, X.; Xia, X. Punicalagin Inhibits Salmonella Virulence Factors and Has Anti-Quorum-Sensing Potential. Appl. Environ. Microbiol. 2014, 80, 6204–6211. [Google Scholar] [CrossRef] [PubMed]
- Kiran, M.D.; Adikesavan, N.V.; Cirioni, O.; Giacometti, A.; Silvestri, C.; Scalise, G.; Ghiselli, R.; Saba, V.; Orlando, F.; Shoham, M.; et al. Discovery of a Quorum-Sensing Inhibitor of Drug-Resistant Staphylococcal Infections by Structure-Based Virtual Screening. Mol. Pharmacol. 2008, 73, 1578–1586. [Google Scholar] [CrossRef] [PubMed]
- Payne, D.E.; Martin, N.R.; Parzych, K.R.; Rickard, A.H.; Underwood, A.; Boles, B.R. Tannic Acid Inhibits Staphylococcus aureus Surface Colonization in an IsaA-Dependent Manner. Infect. Immun. 2013, 81, 496–504. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.S.; Lee, J.; Ryu, S.Y.; Joo, S.W.; Cho, M.H.; Lee, J. Inhibition of Pseudomonas aeruginosa and Escherichia coli O157: H7 Biofilm Formation by Plant Metabolite ε-Viniferin. J. Agric. Food Chem. 2013, 61, 7120–7126. [Google Scholar] [CrossRef] [PubMed]
- Coenye, T.; Brackman, G.; Rigole, P.; de Witte, E.; Honraet, K.; Rossel, B.; Nelis, H.J. Eradication of Propionibacterium Acnes Biofilms by Plant Extracts and Putative Identification of Icariin, Resveratrol and Salidroside as Active Compounds. Phytomedicine 2012, 19, 409–412. [Google Scholar] [CrossRef] [PubMed]
- Li, D.D.; Zhao, L.X.; Mylonakis, E.; Hu, G.H.; Zou, Y.; Huang, T.K.; Yan, L.; Wang, Y.; Jiang, Y.Y. In Vitro and in Vivo Activities of Pterostilbene against Candida albicans Biofilms. Antimicrob. Agents Chemother. 2014, 58, 2344–2355. [Google Scholar] [CrossRef] [PubMed]
- Vikram, A.; Jayaprakasha, G.; Jesudhasan, P.; Pillai, S.; Patil, B. Suppression of Bacterial cell-cell Signalling, Biofilm Formation and Type III Secretion System by Citrus Flavonoids. J. Appl. Microbiol. 2010, 109, 515–527. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Park, J.; Cho, H.S.; Joo, S.W.; Cho, M.H.; Lee, J. Anti-Biofilm Activities of Quercetin and Tannic Acid against Staphylococcus aureus. Biofouling 2013, 29, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Vandeputte, O.M.; Kiendrebeogo, M.; Rajaonson, S.; Diallo, B.; Mol, A.; El Jaziri, M.; Baucher, M. Identification of Catechin as One of the Flavonoids from Combretum Albiflorum Bark Extract that Reduces the Production of Quorum-Sensing-Controlled Virulence Factors in Pseudomonas aeruginosa PAO1. Appl. Environ. Microbiol. 2010, 76, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Rudrappa, T.; Bais, H.P. Curcumin, a Known Phenolic from Curcuma Longa, Attenuates the Virulence of Pseudomonas aeruginosa PAO1 in Whole Plant and Animal Pathogenicity Models. J. Agric. Food Chem. 2008, 56, 1955–1962. [Google Scholar] [CrossRef] [PubMed]
- Pattiyathanee, P.; Vilaichone, R.; Chaichanawongsaroj, N. Effect of Curcumin on Helicobacter Pylori Biofilm Formation. Afr. J. Biotechnol. 2009, 8, 5106–5115. [Google Scholar]
- Hu, P.; Huang, P.; Chen, M.W. Curcumin reduces Streptococcus mutans biofilm formation by inhibiting sortase A activity. Arch. Oral Biol. 2013, 58, 1343–1348. [Google Scholar] [CrossRef] [PubMed]
- Neelakantan, P.; Subbarao, C.; Sharma, S.; Subbarao, C.V.; Garcia-Godoy, F.; Gutmann, J.L. Effectiveness of Curcumin against Enterococcus Faecalis Biofilm. Acta Odontol. Scand. 2013, 71, 1453–1457. [Google Scholar] [CrossRef] [PubMed]
- Packiavathy, I.A.S.V.; Sasikumar, P.; Pandian, S.K.; Ravi, A.V. Prevention of Quorum-Sensing-Mediated Biofilm Development and Virulence Factors Production in Vibrio spp. by Curcumin. Appl. Microbiol. Biotechnol. 2013, 97, 10177–10187. [Google Scholar] [CrossRef] [PubMed]
- Packiavathy, I.A.S.V.; Priya, S.; Pandian, S.K.; Ravi, A.V. Inhibition of Biofilm Development of Uropathogens by curcumin—An Anti-Quorum Sensing Agent from Curcuma Longa. Food Chem. 2014, 148, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Shahzad, M.; Sherry, L.; Rajendran, R.; Edwards, C.; Combet, E.; Ramage, G. Inhibitory effect of plant (poly)phenolics on growth and biofilm formation by Candida albicans. Proc. Nutr. Soc. 2014, 73, E28. [Google Scholar] [CrossRef]
- Karaman, M.; Firinci, F.; Ayyildiz, Z.A.; Bahar, İ.H. Pseudomonas aeruginosa Suşlarında Imipenem, Tobramisin ve Curcuminin Biyofilm Oluşumu Üzerine Etkisi. Mikrobiyol. Bul. 2013, 47, 192–194. [Google Scholar] [CrossRef] [PubMed]
- Soumya, E.A.; Saad, I.K.; Hassan, L.; Ghizlane, Z.; Hind, M.; Adnane, R. Carvacrol and Thymol Components Inhibiting Pseudomonas aeruginosa Adherence and Biofilm Formation. Afr. J. Microbiol. Res. 2011, 5, 3229–3232. [Google Scholar]
- Qiu, J.; Wang, D.; Xiang, H.; Feng, H.; Jiang, Y.; Xia, L.; Dong, J.; Lu, J.; Yu, L.; Deng, X. Subinhibitory Concentrations of Thymol Reduce Enterotoxins A and B and α-Hemolysin Production in Staphylococcus aureus Isolates. PLoS ONE 2010, 5, e9736. [Google Scholar] [CrossRef] [PubMed]
- Kuźma, L.; Rózalski, M.; Walencka, E.; Rózalska, B.; Wysokińska, H. Antimicrobial Activity of Diterpenoids from Hairy Roots of Salvia Sclarea L.: Salvipisone as a Potential Anti-Biofilm Agent Active Against Antibiotic Resistant Staphylococci. Phytomedicine 2007, 14, 31–35. [Google Scholar] [CrossRef] [PubMed]
- Walencka, E.; Rozalska, S.; Wysokinska, H.; Rozalski, M.; Kuzma, L.; Rozalska, B. Salvipisone and Aethiopinone from Salvia Sclarea Hairy Roots Modulate Staphylococcal Antibiotic Resistance and Express Anti-Biofilm Activity. Planta Med. 2007, 73, 545–551. [Google Scholar] [CrossRef] [PubMed]
- Cartagena, E.; Colom, O.A.; Neske, A.; Valdez, J.C.; Bardón, A. Effects of Plant Lactones on the Production of Biofilm of Pseudomonas aeruginosa. Chem. Pharm. Bull. 2007, 55, 22–25. [Google Scholar] [CrossRef] [PubMed]
- Vikram, A.; Jesudhasan, P.R.; Jayaprakasha, G.K.; Pillai, S.D.; Patil, B.S. Citrus Limonoids Interfere with Vibrio harveyi Cell-Cell Signalling and Biofilm Formation by Modulating the Response Regulator LuxO. Microbiology 2011, 157, 99–110. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Garo, E.; Goering, M.G.; Pasmore, M.; Yoo, H.; Esser, T.; Sestrich, J.; Cremin, P.A.; Hough, G.W.; Perrone, P. Bacterial Biofilm Inhibitors from Diospyros d Endo. J. Nat. Prod. 2006, 69, 118–120. [Google Scholar] [CrossRef] [PubMed]
- Vediyappan, G.; Dumontet, V.; Pelissier, F.; D’Enfert, C. Gymnemic Acids Inhibit Hyphal Growth and Virulence in Candida albicans. PLoS ONE 2013, 8, e74189. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Wang, J.; Yu, J.; Li, Y.; Liu, G. Effects of Allicin on the Formation of Pseudomonas aeruginosa Biofilm and the Production of Quorum-Sensing Controlled Virulence Factors. Pol. J. Microbiol. 2013, 62, 243–251. [Google Scholar]
- Pérez-Giraldo, C.; Cruz-Villalón, G.; Sánchez-Silos, R.; Martínez-Rubio, R.; Blanco, M.; Gómez-García, A. In Vitro Activity of Allicin against Staphylococcus epidermidis and Influence of Subinhibitory Concentrations on Biofilm Formation. J. Appl. Microbiol. 2003, 95, 709–711. [Google Scholar] [CrossRef] [PubMed]
- Jakobsen, T.H.; van Gennip, M.; Phipps, R.K.; Shanmugham, M.S.; Christensen, L.D.; Alhede, M.; Skindersoe, M.E.; Rasmussen, T.B.; Friedrich, K.; Uthe, F.; et al. Ajoene, a Sulfur-Rich Molecule from Garlic, Inhibits Genes Controlled by Quorum Sensing. Antimicrob. Agents Chemother. 2012, 56, 2314–2325. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ganin, H.; Rayo, J.; Amara, N.; Levy, N.; Krief, P.; Meijler, M.M. Sulforaphane and Erucin, Natural Isothiocyanates from Broccoli, Inhibit Bacterial Quorum Sensing. Med. Chem. Commun. 2012, 4, 175–179. [Google Scholar] [CrossRef]
- Borges, A.; Simões, L.; Serra, C.; Saavedra, M.; Simões, M. Activity of Allylisothiocyanate and 2-Phenylethylisothiocyanate on Motility and Biofilm Prevention of Pathogenic Bacteria. In Worldwide Research Efforts in the Fighting against Microbial Pathogens: From Basic Research to Technological Developments; Méndez-Vilas, E., Ed.; Brown Walker Press: Boca Raton, FL, USA, 2013; pp. 8–12. [Google Scholar]
- Lee, J.; Kim, Y.; Cho, H.S.; Ryu, S.Y.; Cho, M.H.; Lee, J. Coumarins Reduce Biofilm Formation and the Virulence of Escherichia coli O157: H7. Phytomedicine 2014, 21, 1037–1042. [Google Scholar] [CrossRef] [PubMed]
- Monte, J.; Abreu, A.C.; Borges, A.; Simões, L.C.; Simões, M. Antimicrobial Activity of Selected Phytochemicals against Escherichia coli and Staphylococcus aureus and Their Biofilms. Pathogens 2014, 3, 473–498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ding, X.; Yin, B.; Qian, L.; Zeng, Z.; Yang, Z.; Li, H.; Lu, Y.; Zhou, S. Screening for Novel Quorum-Sensing Inhibitors to Interfere with the Formation of Pseudomonas aeruginosa Biofilm. J. Med. Microbiol. 2011, 60, 1827–1834. [Google Scholar] [CrossRef] [PubMed]
- Tsang, P.W.; Bandara, H.; Fong, W. Purpurin Suppresses Candida albicans Biofilm Formation and Hyphal Development. PLoS ONE 2012, 7, e50866. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, X.; Yao, X.; Zhu, Z.; Tang, T.; Dai, K.; Sadovskaya, I.; Flahaut, S.; Jabbouri, S. Effect of Berberine on Staphylococcus epidermidis Biofilm Formation. Int. J. Antimicrob. Agents 2009, 34, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Artini, M.; Papa, R.; Barbato, G.; Scoarughi, G.; Cellini, A.; Morazzoni, P.; Bombardelli, E.; Selan, L. Bacterial Biofilm Formation Inhibitory Activity Revealed for Plant Derived Natural Compounds. Bioorg. Med. Chem. 2012, 20, 920–926. [Google Scholar] [CrossRef] [PubMed]
- The Plant List. Available online: http://www.theplantlist.org/ (accessed on 25 November 2015).
- Ta, C.A.K. Bacterial Biofilm Inhibition and Antifungal Activity of Neotropical Plants. Ph.D. Thesis, University of Ottawa, Ottawa, ON, Canada, 2015. [Google Scholar]
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ta, C.A.K.; Arnason, J.T. Mini Review of Phytochemicals and Plant Taxa with Activity as Microbial Biofilm and Quorum Sensing Inhibitors. Molecules 2016, 21, 29. https://doi.org/10.3390/molecules21010029
Ta CAK, Arnason JT. Mini Review of Phytochemicals and Plant Taxa with Activity as Microbial Biofilm and Quorum Sensing Inhibitors. Molecules. 2016; 21(1):29. https://doi.org/10.3390/molecules21010029
Chicago/Turabian StyleTa, Chieu Anh Kim, and John Thor Arnason. 2016. "Mini Review of Phytochemicals and Plant Taxa with Activity as Microbial Biofilm and Quorum Sensing Inhibitors" Molecules 21, no. 1: 29. https://doi.org/10.3390/molecules21010029





