Abstract
Total synthesis of naturally occurring spirobisnaphthalene palmarumycin CP17 and its methoxy analogues was first achieved through Friedel-Crafts acylation, Wolff-Kishner reduction, intramolecular cyclization, ketalization, benzylic oxidation, and demethylation using the inexpensive and readily available methoxybenzene, 1,2-dimethoxybenzene and 1,4-dimethoxybenzene and 1,8-dihydroxynaphthalene as raw materials. Demethylation with (CH3)3SiI at ambient temperature resulted in ring A aromatization and acetal cleavage to give rise to binaphthyl ethers. The antifungal activities of these spirobisnaphthalene derivatives were evaluated, and the results revealed that 5 and 9b exhibit EC50 values of 9.34 µg/mL and 12.35 µg/mL, respectively, against P. piricola.
1. Introduction
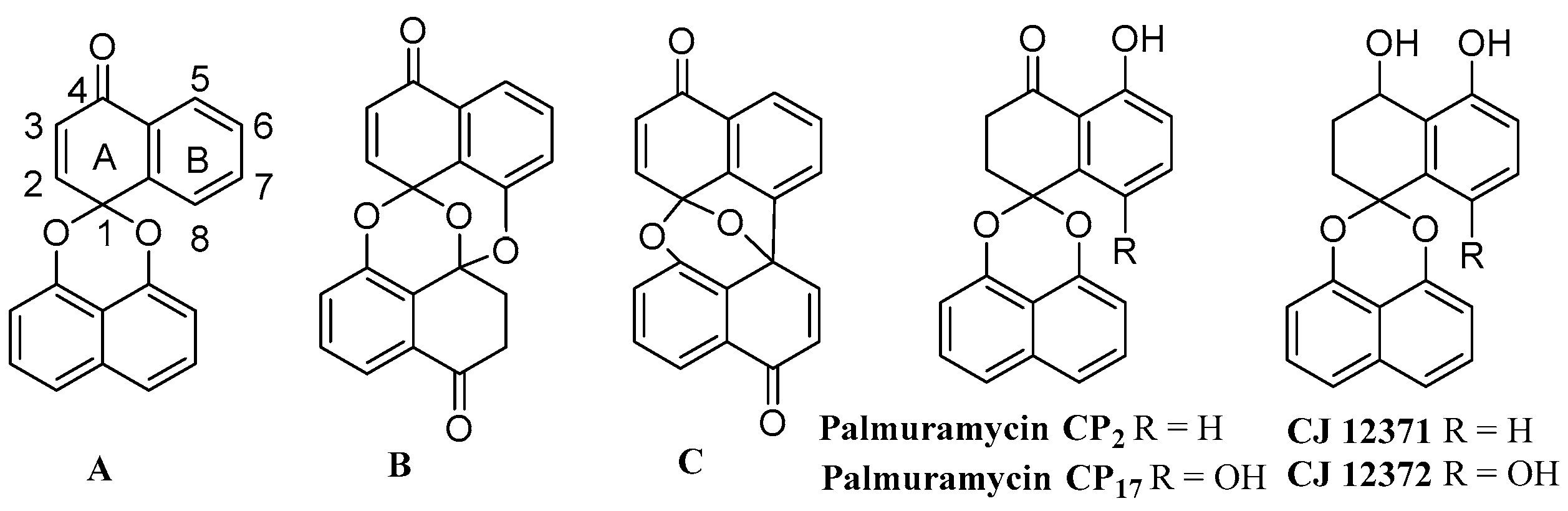
The spirobisnaphthalenes are a group of natural products, possessing a bisnaphthoquinone spiroketal moiety as a common structural feature, which display a variety of biological activities such as antibacterial, antifungal, antileishmanial, enzyme-inhibitory and antitumor activities [1]. The spirobisnaphthalenes have been classified into three subtypes based on their number of spiroketal units (Scheme 1). The most typical subtype, Type A, has a spiroketal structure composed of a 1,8-dihydroxy naphthalene and a naphthoquinone. In Type B, two naphthyl structural units are connected with an additional ether linkage to the core bisnaphthoquinone spiroketal structure. Finally, Type C has a binaphthyl moiety in place of the Type B′s binaphthyl ether structure [1]. Recently, a new spirobisnaphthalene subclass was isolated from the saprobic fungus Urnula craterium [2]. These are structurally distinct in that they contain a 1,8-dihydroxynaphthalene–derived spiroether unit bridged through a spiroether linkage. The spirobisnaphthalenes have attracted significant attention not only because of their unique structures, but also because they display a range of biological activities. In 1997, Wipf and Jung reported the first synthetic study toward diepoxin σ; their strategy has become a typical approach to synthesize this class of natural products [3]. A comprehensive review outlining synthetic approaches to these metabolites was published in 2005 [1]. Since then, several new spirobisnaphthalene natural products have been isolated and the total syntheses of some of these have been accomplished by different research groups. In addition, many non-natural derivatives have been prepared by synthesis and evaluated for their biological activities [4,5,6,7,8,9,10,11,12,13,14,15,16,17,18]. The spirobisnaphthalenes isolated from fungi along with their biological activities were further reviewed in 2010 [19,20]. We have explored the identification of spirobisnaphthalenes from the endophytic fungus Berkleasmium sp. Dzf12, which was an isolate from the medicinal plant Dioscorea zingiberensis. It followed that a variety of bioactive spirobisnaphthalenes and the preparative separation of spirobisnaphthalenes could be separated on a preparative basis by high-speed counter-current chromatography from extracts of Berkleasmium sp. Dzf12 [21,22,23]. It emerged also that the production of some of these metabolites could be enhanced in liquid culture by the addition of oligosaccharides, polysaccharides, or yeast extract, and that they could be most efficiently isolated by resin adsorption.
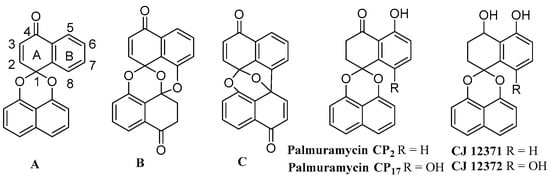
Scheme 1.
Three types of basic spirobisnaphthalene structures (A–C), palmarumycin CPs and CJs.
A racemic synthesis of the spiroxin framework using a desymmetrization approach has been reported [24]. This was followed by a reported catalytic asymmetric approach to construct the tertiary naphthoquinol stereogenic center present in the spiroxin framework [25]. Since the first synthesis of racemic spiroxin C and several preussomerin analogues, there have been no new syntheses of Type B and C spirobisnaphthalenes since 2004, due to their synthetic complexity [19,20,24], so attention remained focused on elaborations of the type A class and their bioactivity evaluation [16,17,18].
Palmarumycin CP17 is a new type A spirobisnaphthalene isolated from the Panamanian endophytic fungus Edenia sp and the endophytic fungus Dzf12 of Dioscorea zingiberensis. It has strong antileishmanial and antimicrobial activities [26,27,28]. Total syntheses of type A spirobisnaphthalenes have been achieved via four different approaches based on the different construction protocols of the core spiroketal structure. These include a biomimetic synthesis by oxidative cyclization to the binaphthyl ether [29,30], direct acetalization [31,32], a silver-mediated cationic cyclization following Suzuki-Miyamura cross-coupling [33], and a Diels-Alder approach in combination with the biomimetic oxidative cyclization of naphthyl phenyl ether [16]. Although the total syntheses of a range of similar palmarumycins, including CP1, CP2 and CJ-12371, was accomplished by direct acetalization as the key step [31,32,33,34,35], the existence of the sensitive 8-hydroxyl or 8-chlorine substituents found in type A spirobisnaphthalenes such as CJ 12372, ascochytain, palmarumycin B6, CP17, and CP18 offer a new challenge. In order to gain insights into the structure-activity relationships of both natural and non-natural spirobisnaphthalenes, we have used the direct acetalization approach, following the synthetic protocol of preussomerin G and I [31,32,36], to complete the total synthesis of palmarumycin CP17 (6a), its 8-methoxy analogue (6b), and 5,8-dimethoxy CJ 12372 (8) as well as the other 6-methoxy,7-methoxy and 6,7-dimethoxy spirobisnaphthalene derivatives (18–20) (Scheme 2, Scheme 3 and Scheme 4). These compounds were then evaluated for their antifungal activities. Here we present our results.
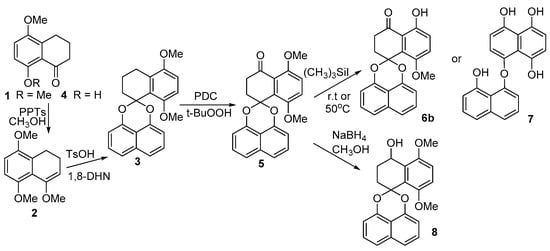
Scheme 2.
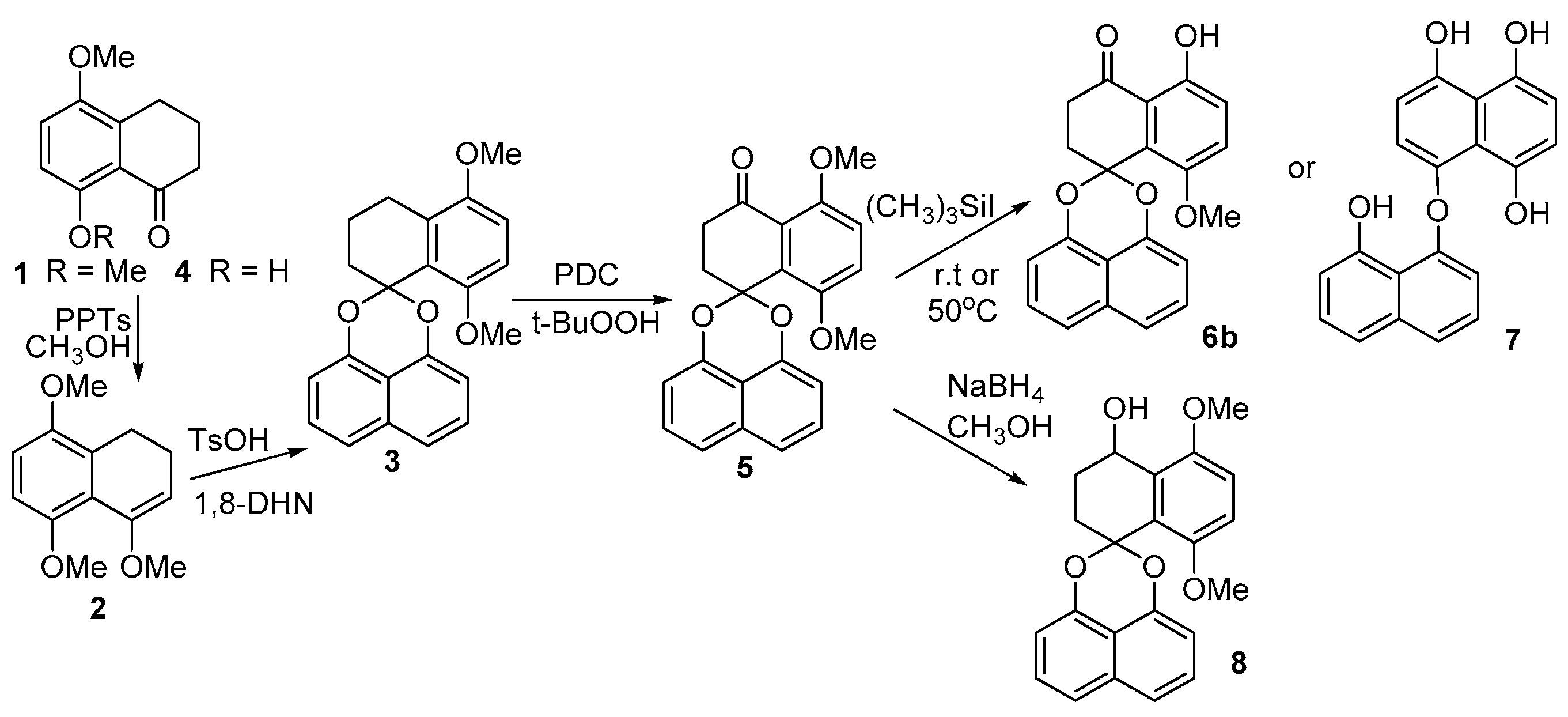
The synthesis of intermediates 3, 5, 8-methoxy analogue 6b, binaphthyl ether 7 and 5, 8-dimethoxy CJ 12372 8.
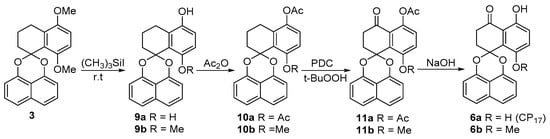
Scheme 3.
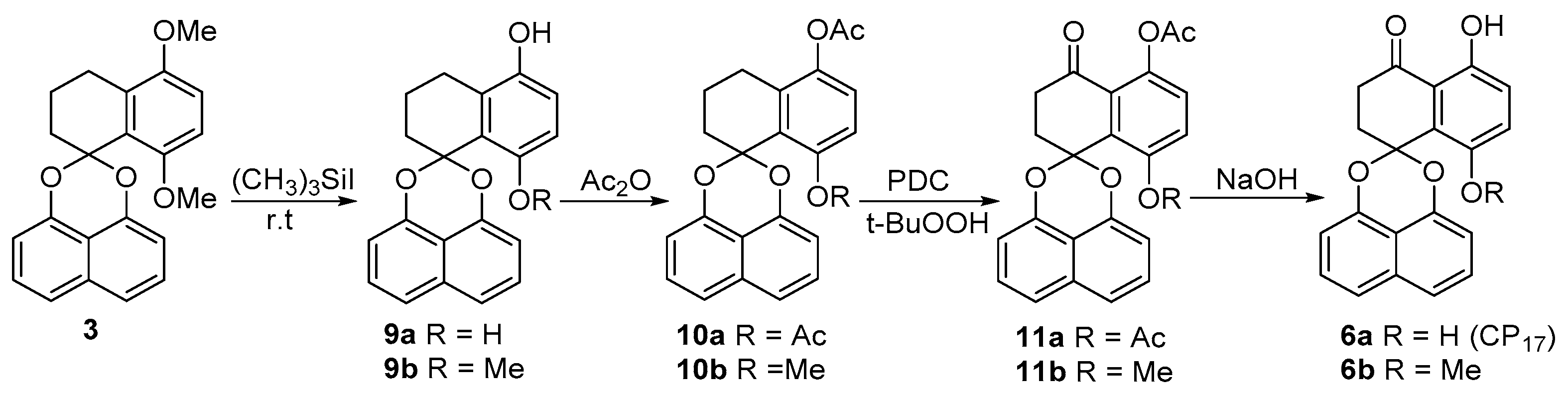
The synthesis of palmarumycin CP17 6a and 8-methoxy analogue 6b.
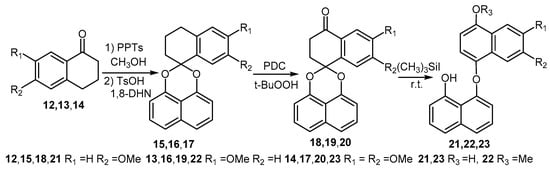
Scheme 4.
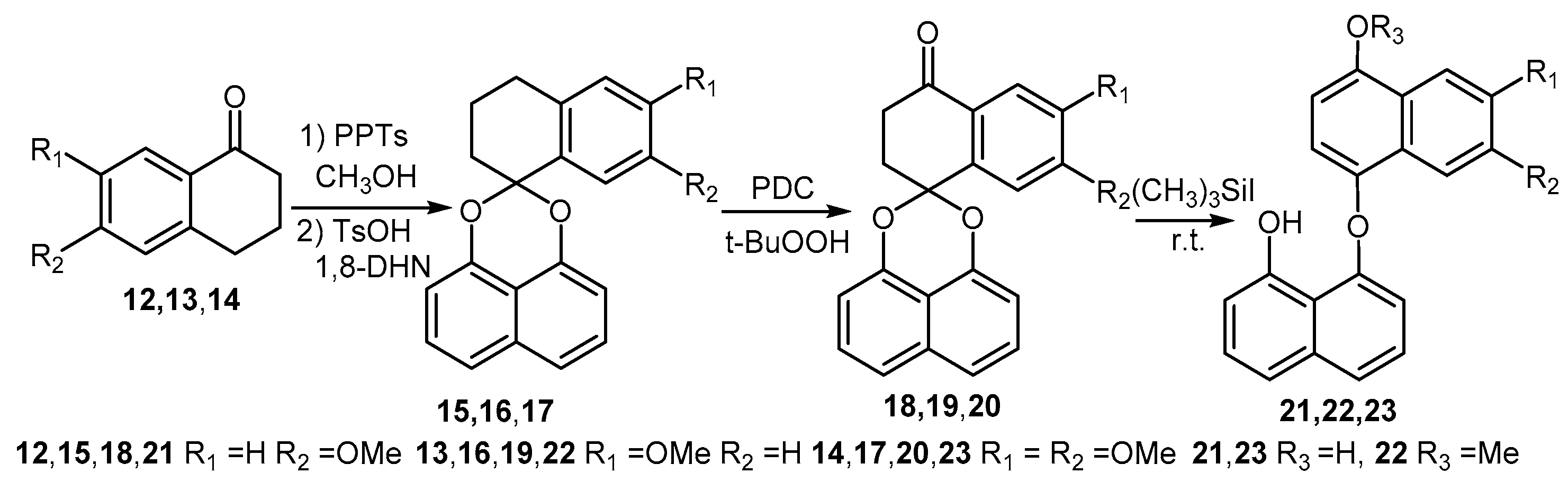
Synthesis of palmarumycin CP17 analogues 18–20 and demethylation products of 5, 18 and 19.
2. Results and Discussion
2.1. Synthesis of Palmarumycin CP17 and Its Methoxy Analogues
Of the four main approaches to the construction of the core spiroketal structure, direct acetalization offers the most economical approach to palmarumycin CP17 due to the available starting materials. The presence of the C8 hydroxyl group renders the direct acetalization between tetralone 1 and 1,8-dihydroxynaphthalene (1,8-DHN) problematic and the spiroketal core 3 of palmarumycin CP17 was not accessible in this way. Similarly, the reaction between 5-methoxy-8-hydroxytetralone 4 and 1,8-DHN was unsuccessful. Tetralone 4 was obtained by the demethylation of 5,8-dimethoxy tetralone with (CH3)3SiI; however, the intramolecular hydrogen bonding is probably responsible for this failure. Nonetheless, we found that 1 could be easily transferred into enol ether 2 in high yield with pyridine p-toluenesulfonate (PPTs) as a catalyst and under an atmosphere of N2. Product 2 required isolation over neutral Al2O3 chromatography. The reaction of ketoenol ether 2 with 1,8-DHN in the presence of TsOH under a strict oxygen and moisture-free atmosphere readily afforded the key spirobisnaphthalene 3 (Scheme 2) [37]. The benzylic oxidation of 3 using pyridine dichromate (PDC) and t-BuOOH gave spirobisnaphthalene 5 successfully. Selective mono-demethylation of 5 with (CH3)3SiI under ambient condition generated only the 8-methoxy analogue (6b) of palmarumycin CP17 (73% yield), but not the desired palmarumycin CP17 (6a) (Scheme 2). However, demethylation of 5 with (CH3)3SiI at 50 °C unexpectedly resulted in ring A aromatization and acetal cleavage to afford binaphthyl ether 7, which was the same as the case reported in the reference (Scheme 2) [31,32]. The NaBH4 reduction of 5 produced 5,8-dimethoxy CJ 12372 (8) in 97% yield (Scheme 2); however, demethylation of 8 with (CH3)3SiI and BBr3, in an attempt to prepare CJ 12372, proved unsuccessful.
Based on the above observations, an alternative reaction sequence was explored (Scheme 3). In the new sequence, compounds 9a and 9b could be readily obtained in high yields without ring A aromatization and acetal cleavage when demethylation of 3 was carried out with a different quantity of (CH3)3SiI (Scheme 3) [29,30]. After the acetylation to generate 10a and 10b, benzylic oxidation with pyridine dichromate (PDC) and t-BuOOH afforded ketones 11a (69%) and 11b (55%), respectively (Scheme 3). Finally, spirobisnaphthalenes 11a and 11b were deacetylated to give palmarumycin CP17 6a (91%) and 8-methoxy analogue 6b (93%), respectively. After the accomplishment of 6a and 6b, the spirobisnaphthalenes 15–17 were prepared by 1,8-DHN coupling with tetralones 12–14, following the procedure carried out for 3, and the benzylic oxidation as before gave spirobisnaphthalenes 18–20 (Scheme 4). However, demethylation of spirobisnaphthalenes 18 and 19 with (CH3)3SiI, even at room temperature, resulted in ring A aromatization and acetal cleavage to give rise to binaphthyl ethers 21–23 (Scheme 4), while demethylation of 20 with (CH3)3SiI at room temperature gave a complex product mixture, which could not be satisfactorily purified. Based on these results, we deduce that the hydroxyl and carbonyl groups at C4, the locations and numbers of the methoxyl groups on ring B and the reaction temperature are three factors which influence ring A aromatization and acetal cleavage to generate binaphthyl ethers. Nonetheless, with 5, 10a and 10b, and 11a and 11b in hand, we have demonstrated a convenient method which should be amenable to the preparation of the other more complex natural products with a similar structure skeleton. Such targets are in progress in our laboratory.
2.2. Antifungal Activity of Spirobisnaphthalene Palmarumycin CP17 and Its Methoxy Analogues
The antifungal activity of the prepared compounds was evaluated using the mycelial growth rate test and the results are listed in Table 1 and Table 2. The data shows that these spirobisnaphthalenes exhibit higher inhibition against P. piricola, R. solani, B. cinerea and C. arachidicolahori in comparison to F. oxysporum, F. graminearum and P. asaragisace. The data in Table 1 illustrated that the hydroxyl and carbonyl groups at C4 play a crucial role for antifungal activity, while the demethylated or deactylated products exhibit much weaker activity than those with methoxy or acetyl groups. Palmarumycin CP17 6a and 8-methoxyl analogue 6b had very similar inhibition potency against all seven phytopathogens, which indicates the hydroxyl/methoxyl groups at C8 have no significant impact on the antifungal activities. The inhibitory rates for 5 and 9b against P. piricola were 73.7% and 75.0%, respectively, and the inhibition rate of 11b against R. solani was 71.3%. In addition, the EC50 values of 5, 9b and 11b were further determined to be 9.34, 12.35 and 11.18 µg/mL, respectively, and illustrated further the significant antifungal activities of these phytopathogens.

Table 1.
The antifungal activities of compounds against phytopathogens (inhibitory rate, %) at 50 µg/mL a.

Table 2.
The EC50 values for compounds 5, 9b and 11b against different phytopathogens.
3. Materials and Methods
3.1. General Information
All reactions were performed under an N2 atmosphere. Unless otherwise stated, all reagents were purchased from commercial suppliers and used without further purification. Organic solvents were concentrated under reduced pressure using a rotary evaporator or oil pump. Flash column chromatography was performed using Qingdao Haiyang flash silica gel (200–300 mesh). Meting points were measured on a Yanagimoto apparatus and uncorrected. Infrared spectra were recorded using a Shimadzu IR-435 instrument with KBr plates. 1H- and 13C-NMR spectra were obtained on Bruker DPX 300 spectrometer (Bruker Biospin Co., Stuttgart, Germany) with CDCl3 as the solvent and TMS as the internal standard. HR-MS were obtained on a Brüker Apex II mass spectrometer using nitrobenzoyl alcohol and sodium chloride as matrix (ThermoFisher scientific Inc., Waltham, MA, USA).
3.2. Synthesis of Spirobisnaphthalene Palmarumycin CP17 and Their Methoxy Analogues
5,8-Dimethoxytetralone (1)
It was synthesized in 43% overall yield using p-dimethoxybenzene as starting material according to the procedures in reference [27].
5,8-Dimethoxytetralone methyl enol ether (2)
It was obtained in 93% yield by the reaction of 5,8-dimethoxytetralone and CH(OCH3)3 in the presence of PPTs according to the protocol in reference [28], which was directly utilized to the next reaction without further purification.
5,8-Dimethoxy-1,2,3,4-tetrahydrospiro[naphtha-ene-1,2′-naphtho-[1,8-de][1,3]dioxine (3)
First 7.0 g (32 mmol) of 5,8-dimethoxytetralone methyl enol ether (2), 5.6 g (35 mmol) 1,8-dihydroxynaphthalene, 700 mg (4 mmol) p-TsOH and 350 mL toluene were added into a three-necked flask, stirred and heated to reflux under N2 atmosphere and kept for 42 h. After the reaction finished, the solution was diluted with 150 mL diethyl ether, washed with NaHCO3 solution, dried over anhydrous Na2SO4, filtered the solid. The organic phase was concentrated under the reduced pressure, the residue was put into a neutral Al2O3 chromatograph column and eluted with petroleum–ethyl acetate (8:1) to afford a white solid 7.0 g, 64% yield, m.p. 163–164 °C. IR υ (cm−1): 3054, 3004, 2946, 2827, 1601, 1580, 1484, 1466, 1411, 1378, 1305. 1H-NMR (300 MHz, CDCl3) δ: 1.79–1.87 (m, 2H), 2.13–2.17 (m, 2H), 2.77 (t, J = 6.5 Hz, 2H), 3.71 (s, 3H), 3.82 (s, 3H), 6.87–6.91 (m, 4H), 7.38–7.47 (m, 4H); 13C-NMR (75 MHz, CDCl3) δ: 18.93, 24.12, 32.86, 55.87, 57.28, 101.03, 108.77, 111.43, 111.99, 113.02, 119.62, 124.75, 127.28, 129.78, 134.17, 148.04, 150.87, 153.68; ESI-MS(+) m/z: 349 [M + H]+.
5,8-Dimethoxy-2,3-dihydrospiro[naphthalene-1,2′-naphtho[1,8-de][1,3]dioxine-4-one (5)
First 1.50 g (4.3 mmol) 3, 4.5 g PDC, 8.6 g celite and 40 mL benzene were added into a 200 mL flask in an ice-water bath, stirred and injected 6 mL 5–6 M t-BuOOH with syringe in 15 m. Then moved the ice-water bath, stirred at room temperature 24 h, and again put 2 mL 5–6 M t-BuOOH with syrup, continued to react 24 h in stirring condition. The solution was diluted with ethyl acetate and filtered to remove the solid, the organic phase was washed two times with 25 mL 1 M HCl solution and water, dried over anhydrous Na2SO4. The organic phase was concentrated under the reduced pressure, the residue was put into a neutral Al2O3 chromatograph column and eluted with petroleum–ethyl acetate (10:1) to afford a white solid 0.59 g, 65% yield, m.p. 174–176 °C. IR υ (cm−1): 3060, 2964, 2936, 2837, 1695, 1606, 1578, 1479, 1410, 1378, 1279, 1154 , 1037; 1H-NMR (300 MHz, CDCl3) δ: 2.48–2.53 (m, 2H), 2.72–2.77 (m, 2H), 3.75 (s, 3H), 3.91 (s, 3H), 6.94 (d, J = 7.2 Hz, 2H), 7.12 (d, J = 9.2 Hz, 1H), 7.28 (d, J = 9.2 Hz, 1H), 7.40–7.50 (m, 4H); 13C-NMR (75 MHz, CDCl3) δ: 31.37, 36.90, 56.91, 57.88, 99.98, 109.02, 112.74, 115.57, 120.26, 121.59, 122.76, 127.41, 129.50, 134.25, 147.38, 152.72, 153.43, 196.07; ESI-MS (+) m/z: 385 [M + Na]+.
5-Hydroxy-8-methoxy-2,3-dihydrospiro[naphthalene-1,2′-naphtho-[1,8-de][1,3]dioxine-4-one (6b)
First 60 mg (0.16 mmol) 5, 15 mL CHCl3, and 0.3 mL (CH3)3SiI were added into a 25 mL flask, stirred at room temperature 12 h. Then removed the solvent under the reduced pressure, the residue was put into a silica gel chromatograph column and eluted with petroleum–ethyl acetate (4:1) to afford a little yellow solid 42 mg, 73% yield, m.p. 206–207 °C. IR υ (cm−1): 3427, 3062, 2959, 2920, 2851, 1693, 1611, 1588, 1474, 1409, 1378, 1295, 1266, 1057; 1H-NMR (300 MHz, CDCl3) δ: 2.49–2.53 (m, 2H), 2.67–2.71 (m, 2H), 3.92 (s, 3H), 7.06 (dd, J = 0.7, 7.5 Hz, 2H), 7.13 (d, J = 9.2 Hz, 1H), 7.24 (d, J = 9.2 Hz, 1H), 7.47 (t, J = 8.4 Hz, 2H), 7.59 (dd, J = 0.7, 8.4 Hz, 2H), 7.64 (s, 1H); 13C-NMR (75 MHz, CDCl3) δ: 29.46, 35.60, 56.98, 102.45, 110.50, 113.59, 116.93, 121.50, 121.77, 123.67, 124.81, 127.57, 134.18, 146.34, 148.92, 153.71, 194.94. HR-ESI-MS: calcd for [M + H]+ C21H17O5: 349.1070, found: 349.1071.
8-Hydroxynaphthalen-1-yl-4,5,8-trihydroxynaphthalene ether (7)
First 0.72 g (2 mmol) 5, First 60 mL CHCl3, and 3.0 mL (CH3)3SiI were added into a 150 mL flask, stirred at 50 °C in an oil bath for 24 h. Then added 50 mL CHCl3, and washed with 5% Na2S2O3 solution and water, removed the solvent under the reduced pressure, the residue was put into a silica gel chromatograph column and eluted with petroleum–ethyl acetate (10:1) to afford a little yellow solid 0.31 g, 44% yield, m.p. 206–207 °C. 1H-NMR (300 MHz, CDCl3) δ: 6.98 (dd, J = 1.1, 7.6 Hz, 1H), 7.25–7.44 (m, 6H), 7.7.51–7.58 (m, 2H), 7.77 (d, J = 8.8 Hz, 1H), 8.88 (s, 1H), 12.42 (s, 1H). ESI-MS (+) m/z: 335 [M + H]+.
8-((8-Hydroxynaphthalen-1-yl)oxy)naphthalene-1,4,5-triol (8)
In a 25 mL flask, 250 mg 5 (0.69 mmol) was dissolved in 10 mL methanol, 27 mg NaBH4 (0.7 mmol) was added into the flask at ambient temperature and stirred for 30 min. The solvent was removed in vacuo, 20 mL water was added, and extracted with ethyl acetate (3 × 20 mL). The organic phase was combined and dried over anhydrous Na2SO4, then the solvent was removed under reduced pressure to produce a white solid 242 mg, yield 97%, m.p. 189–190 °C. IR υ (cm−1): 3053, 3004, 2934, 2827, 1603, 1582, 1486, 1411, 1380, 1307, 1273, 1261, 1061, 906; 1H-NMR (300 MHz, CDCl3) δ: 2.02 (d, J = 15.3 Hz, 1H), 2.15–2.11 (m, 2H), 2.34 (dd, J = 3.0, 12.0 Hz, 1H), 3.06 (br, 1H), 3.71 (s, 3H), 3.89 (s, 3H), 5.11 (t, J = 4.5 Hz, 1H), 6.87–6.93 (m, 2H), 6.98 (d, J = 1.2 Hz, 2H), 7.37–7.48 (m, 4H); 13C-NMR (75 MHz, CDCl3): δ: 26.10, 27.40, 55.00, 57.50, 63.10, 100.61, 108.80, 112.64, 114.59, 119.78, 124.70, 127.29, 147.98, 151.40, 153.70. HRMS: calcd for C22H21O5, 365.1384, found: [M + H]+, 365.1382.
5,8-Dihydroxy-1,2,3,4-tetrahydrospiro[naphthalene-1,2′-naphtho-[1,8-de][1,3]dioxine (9a) and 5-Hydroxy-8-methoxy-1,2,3,4-tetrahydrospiro[naphthalene-1,2′-naphtho-[1,8-de][1,3]dioxine (9b)
First 1.0 g (2.9 mmol) 3, 125 mL CHCl3, and 4.3 mL (CH3)3SiI (30 mmol) were added into a 250 mL flask, stirred at 50 °C in an oil bath for 72 h. Then added 10 mL CH3OH and stirred 0.5 h at room temperature, diluted with 50 mL CHCl3, washed with 5% Na2S2O3 solution and water, dried over anhydrous Na2SO4. The solvent was removed under the reduced pressure, the residue was put into a silica gel chromatograph column and eluted with petroleum–ethyl acetate (5:1) to afford a white solid 9a 0.79 g, 86% yield, m.p. 170–171 °C. IR υ (cm−1): 3057, 2928, 1610, 1579, 1466, 1409, 1388, 1346. 1H-NMR (300 MHz, CDCl3) δ: 1.87–1.94 (m, 2H), 2.15–2.19 (m, 2H), 2.77 (t, J = 6.4 Hz, 2H), 4.44 (s, 1H), 6.80 (dd, J = 8.7, 11.0 Hz, 2H), 6.99 (d, J = 0.9 Hz, 2H), 7.02 (s, 1H), 7.45 (dd, J = 7.5, 8.4 Hz, 2H), 7.55 (dd, J = 0.9, 8.4 Hz, 2H); 13C-NMR (75 MHz, CDCl3) δ:18.72, 23.55, 30.79, 103.29, 110.39, 113.91, 115.70, 117.56, 120.33, 121.22, 125.78, 127.47, 134.14, 146.02, 147.05, 150.38. When the quantity of (CH3)3SiI was 5 eq. molar of 3 without changing the other condition, a little yellow solid 9b was obtained in 58% yield, 164–166 °C. IR υ (cm−1): 3440, 3060, 2998, 2983, 2944, 2927, 2864, 2831, 1606, 1582, 1477, 1466, 1410, 1378, 1263; 1H-NMR (300 MHz, CDCl3) δ: 1.83–1.89 (m, 2H), 2.13–2.17 (m, 2H), 2.78 (t, J = 6.1 Hz, 2H), 3.82 (s, 3H), 6.88 (dd, J = 8.9, 10.8 Hz, 2H), 6.99–7.02 (m, 3H), 7.45 (dd, J = 7.6, 8.4 Hz, 2H), 7.55 (dd, J = 0.8, 8.4 Hz, 2H); HR-ESI-MS: calcd for [M + H]+ C21H19O4: 335.1278, found: 335.1278.
5,8-Diacetoxy-1,2,3,4-tetrahydrospiro[naphthalene-1,2’-naphtho-[1,8-de][1,3]dioxine (10a)
First 1.95 g (6.1 mmol) 9a, 1.8 mL (12.9 mmol) Et3N and 50 mL CH2Cl2 were added into a 150 mL flask and sealed, injected 1.8 mL Ac2O with syringe in 1 h, and stirred another 1 h at room temperature. Then removed the solvent, the residue was put into a silica gel chromatograph column and eluted with petroleum–ethyl acetate (6:1) to afford a white solid 10a 1.65 g, 68% yield, m.p. 151–153 °C. IR υ (cm−1): 2943, 2878, 1763, 1693, 1640, 1469, 1375, 1195; 1H-NMR (300 MHz, CDCl3) δ: 1.75 (s, 3H), 1.84–1.88 (m, 2H), 2.14–2.18 (m, 2H), 2.34 (s, 3H), 2.69 (t, J = 12.2 Hz, 2H), 6.92 (dd, J = 1.0 Hz, 7.2 Hz, 2H), 7.04 (d, J = 8.7 Hz, 1H), 7.17 (d, J = 8.7 Hz, 1H), 7.40–7.51 (m, 4H); 13C-NMR (75MHz, CDCl3) δ: 18.62, 20.59, 20.74, 24.15, 31.92, 100.10, 109.14, 112.68, 120.28, 123.25, 123.56, 127.44, 128.17, 133.09, 134.17, 145.98, 147.28, 147.70, 168.74, 169.61; HR-ESI-MS: calcd for [M + H]+ C24H21O6: 405.1333, found: 405.1332.
5-Acetoxy-8-methoxy-1,2,3,4-tetrahydrospiro[naphthalene-1,2′-naphtho-[1,8-de][1,3]dioxine (10b)
When 140 mg 9b was used as raw material to react in the similar way as 9a without changing the other condition, a white solid 10b 150 mg was obtained in 95% yield, m.p. 148–150 °C. IR υ (cm−1): 3055, 2952, 2936, 2837, 1760, 1603, 1583, 1477, 1438, 1413, 1380, 1276, 1209, 1065, 1021; 1H-NMR (300 MHz, CDCl3) δ: 1.56 (s, 3H), 1.81–1.89 (m, 2H), 2.12–2.16 (m, 2H), 2.77 (t, J = 6.3 Hz, 2H), 3.86 (s, 3H), 6.90–6.93 (m, 3H), 6.99 (d, J = 5.8 Hz, 1H), 7.39–7.49 (m, 4H); 13C-NMR (75 MHz, CDCl3) δ: 18.77, 20.62, 23.75, 31.97, 55.78, 100.49, 109.08, 110.83, 112.77, 120.14, 122.58, 127.41, 129.67, 134.18, 142.89, 147.48, 154.53, 170.23. HR-ESI-MS: calcd for [M + H]+ C23H21O5: 377.1383, found: 377.1384.
5,8-Diacetoxy-2,3-dihydrospiro[naphthalene-1,2′-naphtho[1,8-de][1,3]dioxine-4-one (11a) and 5-Acetoxy-8-methoxy-2,3-dihydrospiro[naphthalene-1,2′-naphtho[1,8-de][1,3]dioxine-4-one (11b)
First 210 mg (0.56 mmol) 10b, 0.4 g PDC, 1.2 g celite and 6 mL benzene were added into a 50 mL flask in a ice-water bath, stirred and injected 1 mL 5–6 M t-BuOOH with syringe in 15 min. Then moved the ice-water bath, stirred at room temperature 24 h, and again injected 0.5 mL 5–6 M t-BuOOH with syringe, continued to react in stirring condition 24 h. The solution was diluted with ethyl acetate and filtered to remove the solids, the organic phase was washed two times with 10 mL 1 M HCl solution and water, dried over anhydrous Na2SO4. The organic phase was concentrated under the reduced pressure, the residue was put into a neutral Al2O3 chromatograph column and eluted with petroleum–ethyl acetate (5:1) to afford a white solid 11b 152 mg, 69% yield, m.p. 148–150 °C. IR υ (cm−1): 2925, 2853, 1748, 1583, 1478, 1412, 1379, 1310, 1276, 1113; 1H-NMR (300 MHz, CDCl3) δ: 1.83 (s, 3H), 2.48–2.52 (m, 2H), 2.72–2.77 (m, 2H), 3.97 (s, 3H), 6.96 (dd, J = 1.0, 7.4 Hz, 2H), 7.18 (d, J = 9.3 Hz, 1H), 7.35 (d, J = 9.1 Hz, 1H), 7.45 (dd, J = 7.4, 8.3 Hz, 2H), 7.53 (dd, J = 1.0, 8.5 Hz, 2H); 13C-NMR (75 MHz, CDCl3) δ: 20.51, 30.50, 36.40, 56.71, 99.41, 109.33, 112.49, 114.70, 120.78, 122.24, 127.54, 131.30, 132.59, 134.19, 141.78, 146.76, 157.28, 169.92,194.86. HR-ESI-MS: calcd for [M + H]+ C23H19O6: 391.1176, found: 391.1177. In the similar way, 11a was obtained in 55% yield, m.p. 136–137 °C. IR υ (KBr): 2945, 2859, 1750, 1585, 1482, 1408, 1375, 1304, 1278, 1115 cm−1; 1H-NMR ((300 MHz, CDCl3) δ: 1.81 (s, 3H), 2.28 (s, 3H), 2.49–2.53 (m, 2H), 2.71–2.77 (m, 2H), 6.95 (dd, J = 1.0 Hz, 7.4 Hz, 2H), 7.18 (d, J = 9.3 Hz, 1H), 7.36 (d, J = 9.1 Hz, 1H), 7.47 (dd, J = 7.4 Hz, 8.3 Hz, 2H), 7.55 (dd, J = 1.0 Hz, 8.5 Hz, 2H); 13C-NMR (75 MHz, CDCl3) δ: 20.50, 21.81, 30.61, 36.52, 99.52, 109.43, 112.51, 114.68, 120.76, 122.25, 127.54, 131.35, 132.64, 134.22, 141.83, 146.79, 157.26, 169.95, 194.88. HR-MS (ESI): calcd for C24H19O7 [M + H]+: 419.1131, found: 419.1132.
5,8-Dihydroxy-2,3-dihydrospiro[naphthalene-1,2′-naphtho[1,8-de][1,3]dioxine-4-one (Palmarumycin CP17, 6a) and 5-Hydroxy-8-methoxy-2,3-di-hydrospiro[naphthalene-1,2′-naphtho[1,8-de][1,3]-dioxine-4-one (6b)
The 5 mL methanol solution of 168 mg (0.40 mmol) 11a was added a 25 mL flask, stirred and added 9 mL 1% NaOH solution at room temperature to react 1 h. Then adjusted pH value to 5–6 with 1 M HCl, removed the methanol under the reduced pressure. Extracted with ethyl acetate (3 × 10 mL) and dried over anhydrous Na2SO4. The solvent was removed from the organic phase to afford a white solid 6a 122 mg, 91% yield, m.p. 173–174 °C. IR υ (cm−1): 3435, 3064, 2969, 2924, 2856, 1695, 1610, 1585, 1473, 1407, 1376, 1290, 1264, 1058; 1H-NMR (300 MHz, CDCl3) δ: 2.49–2.53 (m, 2H), 2.74–2.78 (m, 2H), 7.04–7.07 (m, 3H), 7.24–7.27 (m, 1H), 7.48 (t, J = 7.4 Hz, 2H), 7.59 (d, J = 7.5 Hz, 2H), 7.65 (s, 1H), 12.36 (s, 1H); 13C-NMR (75 MHz, CDCl3) δ: 28.98, 33.85, 102.06, 110.46, 113.53, 114.49, 119.49, 121.85, 127.61, 128.87, 134.22, 146.29, 147.68, 157.20, 202.41. HR-MS (ESI): calcd for [M − H]− C21H17O5: 333.0768, found: 333.0769. These data were consistent with that of naturally occurring compounds [19]. In the similar way, 145 mg (0.37 mmol) 11b was transferred into 121 mg of 6b in 93% yield, the m.p., 1H-NMR, 13C-NMR and MS data were identical with that of the demethylation product of compound 5.
7-Methoxytetralone (13) and 6,7-Dimethoxytetralone (14)
They were prepared by the similar methods [27] as 1 using methoxybenzene and 1,2-dimethoxybenzene as the starting materials. Compound 13, a white solid, 85% yield, m.p. 59–60 °C. 1H NMR (300 MHz, CDCl3) δ: 2.07–2.16 (m, 2H), 2.64 (t, J = 6.4 Hz, 2H), 2.90 (t, J = 6.4 Hz, 2H), 3.83 (s, 3H), 7.05 (dd, J = 2.8, 8.7 Hz, 1H), 7.17 (d, J = 8.7 Hz, 1H), 7.52 (d, J = 2.8 Hz, 1H); ESI-MS m/z: 177 [M + H]+, 199 [M + Na]+. Compound 14, a white solid, 75% yield, m.p. 94–95 °C. 1H-NMR (300 MHz, CDCl3) δ: 2.08–2.17 (m, 2H), 2.60 (t, J = 6.5 Hz, 2H), 2.90 (t, J = 6.3 Hz, 2H), 3.92 (s, 3H), 3.94 (s, 3H), 6.68 (s, 1H), 7.52 (s, 1H); ESI-MS m/z: 207 [M + H]+, 229 [M + Na]+. 6-Methoxytetralone (12) was purchased from Acros Organic Co.
6-Methoxy-3,4-dihydro-2H-spironaphthalene-1,2′-naphtho[1,8-de][1,3]dioxine (15), 7-Methoxy-3,4-dihydro-2H-spironaphthalene-1,2′-naphtho[1,8-de][1,3]dioxine (16) and 6,7-Dimethoxy-3,4-dihydro-2H-spironaphthaene-1,2′-naphtho[1,8-de][1,3]dioxine (17)
These enol methyl ether intermediates were prepared in the similar methods as 2 using 12, 13 and 14 as raw materials, respectively. Then compounds 15, 16 and 17 were obtained by reaction of these enol methyl ethers with 1,8-dihydroxynaphthalene in the catalysis of p-TsOH in toluene as compound 3. Compound 15, a white solid, 61% yield, m.p. 146–147 °C. 1H-NMR (300 MHz, CDCl3) δ: 1.88–1.96 (m, 2H), 2.12–2.16 (m, 2H), 2.88 (t, J = 6.2 Hz, 2H), 3.83 (s, 3H), 6.72 (d, J = 2.6 Hz, 1H), 6.88–6.92 (m, 3H), 7.39–7.50 (m, 4H), 7.77 (d, J = 8.7 Hz, 1H); 13C-NMR (75 MHz, CDCl3) δ: 19.81, 29.70, 31.11, 55.31, 100.69, 109.25, 112.86, 113.15, 113.69, 120.16, 127.35, 127.87, 129.05, 134.18, 139.77, 148.44, 160.22; ESI-MS: m/z 319 [M + H]+. Compound 16, a white solid, 65% yield, m.p. 105–107 °C. 1H-NMR (300 MHz, CDCl3) δ: 1.89–1.95 (m, 2H), 2.13–2.17 (m, 2H), 2.85 (t, J = 6.3 Hz, 2H), 3.81 (s, 3H), 6.92–6.97 (m, 3H), 7.14 (d, J = 8.5 Hz, 1H), 7.36 (d, J = 2.7 Hz, 1H), 7.40–7.51 (m, 4H); 13C-NMR (75 MHz, CDCl3) δ: 28.56, 29.69, 31.98, 55.42, 100.68, 109.35, 110.84, 113.70, 117.26, 120.27, 127.38, 129.81, 130.20, 134.20, 135.92, 148.23, 158.37; ESI-MS: m/z 319 [M + H]+. Compound 17, a white solid, 67% yield, m.p. 143–145 °C. 1H-NMR (CDCl3) δ: 1.90–1.96 (m , 2H), 2.12–2.16 (m, 2H), 2.84 (t, J = 6.0 Hz, 2H), 3.89 (s, 3H), 3.91 (s, 3H), 6.67 (s, 1H), 6.92–6.95 (m, 1H), 7.29 (d, J = 6.0 Hz, 1H), 7.41–7.51 (m, 4H); 13C-NMR (75MHz, CDCl3) δ: 19.96, 29.06, 31.01, 55.94, 55.95, 100.82, 109.34, 109.52, 110.71, 113.90, 120.23, 127.13, 127.28, 130.99, 134.20, 146.02, 148.38, 150.08; ESI-MS: m/z 349 [M + H]+.
6-Methoxy-2,3-dihydrospiro[naphthalene-1,2′-naphtho[1,8-de][1,3]dioxine-4-one (18), 7-Methoxy-2,3-dihydrospiro[naphthalene-1,2′-naphtho-[1,8-de][1,3]dioxine-4-one (19) and 6,7-Dimethoxy-2,3-di-hydrospiro[naphthalene-1,2′-naphtho-[1,8-de][1,3]dioxine-4-one (20)
The benzylic oxidation of three intermediates 15–17 by PDC and t-BuOOH as 5 afforded 18, 19 and 20. Compound 18, a white solid, 68% yield, m.p. 145–146 °C. 1H-NMR (300 MHz, CDCl3) δ: 2.51 (t, J = 6.3 Hz, 2H), 2.80 (t, J = 6.3 Hz, 2H), 3.91 (s, 3H), 6.96 (dd, J = 7.4, 1.0 Hz, 2H), 7.24–7.28 (m, 1H), 7.42–7.59 (m, 5H), 7.89 (d, J = 8.7 Hz, 1H); 13C-NMR (75 MHz, CDCl3) δ: 29.85, 34.42, 55.66, 98.80, 109.31, 109.35, 113.42, 120.72, 121.93, 127.48, 127.93, 132.95, 134.20, 147.73, 160.87, 196.19; ESI-MS m/z: 333 [M + H]+. Compound 19, a white solid, 63% yield, m.p. 147–148 °C. 1H-NMR (300 MHz, CDCl3) δ: 2.50 (t, J = 6.3 Hz, 2H), 2.75 (t, J = 6.3 Hz, 2H), 3.90 (s, 3H), 6.98 (d, J = 7.4 Hz, 2H), 7.08 (dd, J = 2.6, 8.7 Hz, 1H), 7.43–7.55 (m, 5H), 8.10 (d, J = 8.7 Hz, 1H); 13C-NMR (75 MHz, CDCl3) δ: 29.82, 34.95, 55.69, 98.70, 109.45, 109.91, 113.42, 116.77, 120.83, 125.13, 129.57, 134.20, 142.69, 147.52, 164.43, 194.99; ESI-MS m/z: 333 [M + H]+. Compound 20, a white solid, 49% yield, m.p. 148–149 °C. 1H-NMR (300 MHz, CDCl3) δ: 2.51 (t, J = 6.3 Hz, 2H), 2.77 (t, J = 6.3 Hz, 2H), 3.99 (s, 3H), 4.01 (s, 3H), 6.99 (dd, J = 7.4, 1.0 Hz, 2H), 7.41–7.59 (m, 6H); 13C-NMR (75 MHz, CDCl3) δ: 30.03, 34.12, 56.23, 56.32, 98.88, 107.84, 108.29, 109.49, 113.46, 120.83, 125.48, 127.52, 134.25, 135.06, 147.70, 150.43, 154.21, 195.15; ESI-MS m/z: 363 [M + H]+.
8-Hydroxynaphthalen-1-yl-4-hydroxy-6-methoxynaphthalene ether (21), 8-Hydroxynaphthalen-1-yl-4-hydroxy-7-methoxynaphthalene ether (22) and 8-Hydroxy-naphthalen-1-yl-4,7-dimethoxynaphthalene ether (23)
The (CH3)3SiI demethylation of compound 18 was carried out as 5 at room temperature to afford compound 21 as a little yellow oily liquid, yield 95%. 1H-NMR (300 MHz, CDCl3) δ: 3.97 (s, 3H), 5.50 (brs, 1H), 6.43 (dd, J = 7.8, 1.0 Hz, 1H), 6.81 (d, J = 8.1Hz, 1H), 7.00–7.15 (m, 4H), 7.36–7.53 (m, 4H), 7.85 (d, J = 8.7 Hz, 1H), 9.32 (brs, 1H); 13C-NMR (75 MHz, CDCl3) δ: 55.44, 100.76, 108.54, 108.69, 110.74, 115.11, 115.44, 119.22, 120.20, 122.79, 123.42, 123.50, 125.62, 126.82, 127.79, 136.98, 143.46, 148.53, 154.12, 156.52, 158.12. ESI-MS m/z: 355 [M + Na]+. Similarly, compounds 22 and 23 were obtained using 19 as starting material in same process. Compound 22, a little yellow solid, yield 48%, m.p. 128–130 °C. 1H-NMR (300 MHz, CDCl3) δ: 3.73 (s, 3H), 5.76 (brs, 1H), 6.49 (dd, J = 7.8, 1.0 Hz, 1H), 6.66 (d, J = 8.1 Hz, 1H), 7.02 (dd, J = 7.4, 1.8 Hz, 1H), 7.10–7.25 (m, 4H), 7.37–7.48 (m, 3H), 8.16 (d, J = 8.7 Hz, 1H), 9.39 (brs, 1H); 13C-NMR (75 MHz, CDCl3) δ: 55.35, 99.81, 105.96, 108.83, 110.70, 115.24, 118.77, 119.27, 120.71, 122.85, 124.28, 125.72, 127.76, 129.61, 136.96, 142.59, 149.83, 154.06, 156.28, 158.98. ESI-MS m/z: 355 [M + Na]+. Compound 23, a little yellow solid, m.p. 138–139 °C, yield 35%. 1H-NMR (300 MHz, CDCl3) δ: 3.73 (s, 3H), 4.01 (s, 3H), 6.49 (dd, J = 7.8, 1.0 Hz, 1H), 6.67 (d, J = 8.1 Hz, 1H), 7.02 (dd, J = 7.4, 1.8 Hz, 1H), 7.10–7.25 (m, 4H), 7.37–7.48 (m, 3H), 8.23 (d, J = 8.7 Hz, 1H), 9.37 (brs, 1H). ESI-MS m/z: 369 [M + Na]+.
1H-, 13C-NMR and HR-MS spectra of compounds 1–23 can be found in the Supplementary Materials.
3.3. Bioassay of Spirobisnaphthalene Palmarumycin CP17 and Its Methoxy Analogues
Antifungal activities of the compounds against P. piricola, B. cinerea, A.solani, C. arachidicolahori, F. oxysporurm, F. graminearum and P. asaragisace were evaluated using the mycelial growth rate test [38]. The culture media with known concentration of the test compounds were obtained by mixing the solution in acetone with potato dextrose agar (PDA), on which fungus cakes were placed. The blank test was made using acetone and carbendazim was used as the positive control. The culture was incubated at 25 ± 0.5 °C. Three replicates were performed. After the mycelia in the blank grew completely, the diameter of the mycelia was measured, and the inhibition rate was calculated according to the formula in reference [38].
4. Conclusions
The total syntheses of palmarumycin CP17 6a and its 8-methoxy analogue 6b were carried out in nine steps, using p-dimethoxybenzene as the starting material, with 4.9% and 5.8% overall yields, respectively. The direct acetalization of enol ether and 1,8-dihydroxy naphthalene, and benzylic oxidation were used as the key steps in constructing the core spiroketal and the carbonyl group at C4. The spirobisnaphthalenes 18–20 were also prepared with a similar protocol, but their demethylation with (CH3)3SiI at ambient temperature resulted in ring A aromatization and acetal cleavage to afford binaphthyl ethers. The EC50 values of 5 and 9b against P. piricola and 11b against R. solani were 9.34, 12.35 and 11.18 µg/mL, which showed their strong antifungal activities on these phytopathogens.
Supplementary Materials
Supplementary materials can be accessed at: http://www.mdpi.com/1420-3049/21/5/600/s1.
Acknowledgments
This project was co-founded by National Hi-Tech R & D Program of China (No. 2011AA10A202), and Natural Science Foundation of China (No. 21172254 and 31071710).
Author Contributions
R. Wang, G. Liu and M. Yang synthesized all of the compounds; R. Wang performed the bioassay of compounds and statistics analysis; M. Wang and L. Zhou started the project, designed the molecules, wrote and revised the paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Miyashita, K.; Imanishi, T. Syntheses of natural products having an epoxyquinone structure. Chem. Rev. 2005, 105, 4515–4536. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Schwan, W.R.; Volk, T.J.; Rott, M.; Liu, M.; Huang, P.; Liu, Z.; Wang, Y.; Zitomer, N.C.; Sleger, C.; et al. Antibacterial spirobisnaphthalenes from the North American cup fungus Urnula craterium. J. Nat. Prod. 2012, 75, 1534–1538. [Google Scholar] [CrossRef] [PubMed]
- Wipf, P.; Jung, J.K. Long-range electrostatic effects in synthesis: Dipole-controlled nucleophilic addition to a naphthoquinone acetal in model studies toward diepoxin sigma. Angew. Chem. Int. Ed. 1997, 36, 764–767. [Google Scholar] [CrossRef]
- Jiao, P.; Swenson, D.; Gloer, J.; Campbell, J.; Shearer, C. Decaspirones A–E, new bioactive spirodioxynaphthalenes from the freshwater aquatic fungus decaisnella thyridioides. J. Nat. Prod. 2006, 69, 1667–1671. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Guo, H.; Li, E.; Liu, X.; Zhou, Y.; Che, Y. Decaspirones F–I, bioactive secondary metabolites from the saprophytic fungus helicoma viridis. J. Nat. Prod. 2006, 69, 1672–1675. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Krohn, K.; Elsasser, B.; Florke, U.; Draeger, S.; Schulz, B. Metabolic products of the endophytic fungus Microsphaeropsis sp. from Larix decidua. Eur. J. Org. Chem. 2007, 29, 4845–4854. [Google Scholar] [CrossRef]
- Kanoh, K.; Okada, A.; Adachi, K.; Imagawa, H.; Nishizawa, M.; Matsuda, S.; Shizuri, Y.; Utsumi, R. Ascochytatin, a novel bioactive spirodioxynaphthalene metabolite producted by the marine-derived fungus Ascochyta sp. NGB4. J. Antibiot. 2008, 61, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Shi, Q.; Lin, G.; Guo, S.; Yang, J. Spirobisnaphthalene analogues from the endophytic fungus Preussia sp. J. Nat. Prod. 2009, 72, 1712–1715. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Kurtan, T.; Miao, Z.; Mandi, A.; Istvan, K.; Liu, H.; Ding, J.; Guo, Y. Palmarumycins BG1–BG7 and Preussomerin BG1: Establishment of their absolute configurations using theoretical calculations of electronic circular dichroism spectra. J. Org. Chem. 2011, 76, 1821–1831. [Google Scholar] [CrossRef] [PubMed]
- Macías-Rubalcava, M.; Sobrino, M.; Hernández-Bautista, C.; Hernández-Ortega, S. Naphthoquinone spiroketals and organic extracts from the endophytic fungus Edenia gomezpompae as potential herbicides. J. Agric. Food Chem. 2014, 62, 3553–3562. [Google Scholar] [CrossRef] [PubMed]
- Macías-Rubalcava, M.; Hernández-Bautista, B.; Jiménez-Estrada, M.; González, M.; Glenn, A.; Hanlin, R. Naphthoquinone spiroketal with allelochemical activity from the newly discovered endophytic fungus Edenia gomezpompae. Phytochemistry 2008, 69, 1185–1196. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Song, H.; Li, J.; Tang, Y.; Sun, R.; Wang, L. Ymf 1029A–E, preussomerin analogues from the fresh-water-derived fungus YMF 1.01029. J. Nat. Prod. 2008, 71, 952–956. [Google Scholar] [CrossRef] [PubMed]
- Dersar, S.A.; Blunt, J.W.; Munro, M.H.G. Spiro-mamakone A: A unique relative of the spirobisnaphthalene class of compounds. Org. Lett. 2006, 8, 2059–2061. [Google Scholar]
- Pudhom, K.; Teerawatananond, T.; Chookpaiboon, S. Spirobisnaphthalenes from the mangrove-derived fungus Rhytidhysteron sp. AS21B. Mar. Drugs 2014, 12, 1271–1280. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.-M.; Mafezoli, J.; Oliveira, M.C.F.; U’Ren, J.M.; Arnold, A.E.; Gunatilaka, A.A.L. Anteaglonialides A−F and Palmarumycins CE1−CE3 from Anteaglonium sp. FL0768, a fungal endophyte of the spikemoss Selaginella arenicola. J. Nat. Prod. 2015, 78, 2738–2747. [Google Scholar] [CrossRef] [PubMed]
- Krohn, K.; Wang, S.; Ahmed, I.; Altun, S.; Aslan, A.; Florke, U.; Kock, I.; Schlummer, S. Flexible route to Palmarumycin CP1 and CP2 and CJ-12.371 methyl ether. Eur. J. Org. Chem. 2010, 4476–4481. [Google Scholar] [CrossRef]
- Wipf, P.; Lynch, S.; Powis, G.; Birmingham, A.; Englund, E. Synthesis and biological activity of prodrug inhibitors of the thioredoxin–thioredoxin reductase system. Org. Biomol. Chem. 2005, 3, 3880–3882. [Google Scholar] [CrossRef] [PubMed]
- Aslan, A.; Altun, S.; Ahmed, I.; Florke, U.; Schulz, B.; Krohn, K. Synthesis of biologically active nonnatural palmarumycin derivatives. Eur. J. Org. Chem. 2011, 1176–1188. [Google Scholar] [CrossRef]
- Zhou, L.; Zhao, J.; Shan, X.; Cai, X.; Peng, Y. Spirobisnaphthalenes from fungi and their biological activities. Mini-Rev. Med. Chem. 2010, 10, 977–989. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.S.; Guo, Y.W.; Krohn, K. Structure, bioactivities, biosynthetic relationships and chemical synthesis of the spirodioxynaphthalenes. Nat. Prod. Rep. 2010, 27, 1840–1870. [Google Scholar] [CrossRef] [PubMed]
- Shan, T.; Tian, J.; Wang, X.; Mou, Y.; Mao, Z.; Lai, D.; Dai, J.; Peng, Y.; Zhou, L.; Wang, M. Bioactive spirobisnaphthalenes from the endophytic fungus Berkleasmium sp. J. Nat. Prod. 2014, 77, 2151–2160. [Google Scholar] [CrossRef] [PubMed]
- Mou, Y.; Luo, H.; Mao, Z.; Shan, T.; Sun, W.; Zhou, K.; Zhou, L. Enhancement of palmarumycins C12 and C13 production in liquid culture of endophytic fungus Berkleasmium sp. Dzf12 after treatments with metal ions. Int. J. Mol. Sci. 2013, 14, 979–998. [Google Scholar] [CrossRef] [PubMed]
- Shan, T.; Lu, S.; Luo, C.; Luo, R.; Mou, Y.; Wang, M.; Peng, Y.; Zhou, L. Preparative separation of spirobisnaphthalenes from endophytic fungus Berkleasmium sp. Dzf12 by high-speed counter-current chromatography. Molecules 2013, 18, 12896–12908. [Google Scholar] [CrossRef] [PubMed]
- Nabatame, K.; Hirama, M.; Inoue, M. A simple desymmetrization approach to the spiroxin framework. Heterocycles 2008, 76, 1011–1016. [Google Scholar]
- Kwan, A.; Stein, J.; Carrico-Moniz, D. A catalytic asymmetric entry to enantioenriched tertiary naphthoquinols via a facile tandem oxidation/ring-opening sequence. Tetrahedron Lett. 2011, 52, 3426–3428. [Google Scholar] [CrossRef]
- Martinez-Luis, S.; Della-Togna, G.; Coley, P.; Kursar, T.; Gerwick, W.; Cubilla-Rios, L. Antileishmanial constituents of the Panamanian endophytic fungus Edenia sp. J. Nat. Prod. 2008, 71, 2011–2014. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Luis, S.; Cherigo, L.; Spadafora, C.; Gerwick, W.H.; Cubilla-Rios, L. Additional antileishmanial constituents of the Panamanian endophytic fungus Edenia sp. Rev. Latinoamer. Quim. 2009, 37, 104–114. [Google Scholar]
- Cai, X.; Shan, T.; Li, P.; Huang, Y.; Xu, L.; Zhou, L.; Wang, M.; Jiang, W. Spirobisnaphthalenes from the endophytic fungus Dzf12 of Dioscorea zingiberensis and their antimicrobial activities. Nat. Prod. Commun. 2009, 4, 1469–1472. [Google Scholar] [PubMed]
- Wipf, P.; Jung, J.K. Total synthesis of palmarumycin CP1 and (±)-deoxypreussomerin A. J. Org. Chem. 1998, 63, 3530–3531. [Google Scholar] [CrossRef]
- Karsten, K.; Karsten, B.; Hans-Jiirgen, A.; Siegfried, D.; Barbara, S.; Stefan, B.; Gerhard, B. Generation of the palmarumycin spiroacetal framework by oxidative cyclization of an open chain metabolite from coniothyrium palmarum. Liebigs Ann. Recueil 1997, 2531–2534. [Google Scholar]
- Ragot, J.; Alcaraz, M.; Taylor, R. Syntheses of palmarumycin CP1 and CP2, CJ-12,371 and novel analogues. Tetrahedron Lett. 1998, 39, 4921–4924. [Google Scholar] [CrossRef]
- Ragot, J.; Steeneck, C.; Alcaraz, M.; Taylor, R. The synthesis of 1,8-dihydroxynaphthalene-derived natural products: Palmarumycin CP1. palmarumycin CP2, palmarumycin C11, CJ-12,371, deoxypreussomerin A and novel analogues. J. Chem. Soc. Perkin Trans. 1 1999, 1073–1082. [Google Scholar] [CrossRef]
- Inoue, M.; Nabatame, K.; Hirama, M. Communication-a novel route to 1,8-dihydroxynaphthalene- derived natural products. Synthesis of (±)-CJ-12,372. Heterocycles 2003, 59, 87–92. [Google Scholar] [CrossRef]
- Barrett, A.G.M.; Hamprecht, D.; Meyer, T. Total syntheses of palmarumycins CP1 and CP2 and CJ-12,371: novel spiro-ketal fungal metabolites. Chem. Commun. 1998, 809–810. [Google Scholar] [CrossRef]
- Barrett, A.G.M.; Blaney, F.; Campbell, A.D.; Hamprecht, D.; Meyer, T.; White, A.J.P.; Witty, D.; Williams, D.J. Unified route to the palmarumycin and preussomerin natural products. Enantioselective Synthesis of (−)-Preussomerin G. J. Org. Chem. 2002, 67, 2735–2746. [Google Scholar] [CrossRef] [PubMed]
- Chi, S.; Heathcock, C.H. Total syntheses of (±)-preussomerins G and I. Org. Lett. 1999, 1, 3–5. [Google Scholar] [CrossRef] [PubMed]
- Wipf, P.; Jung, J. Formal total synthesis of (+)-diepoxin σ. J. Org. Chem. 2000, 65, 6319–6337. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.C. The Bioassay Technologies for Pesticides; Beijing Agricultural University Press: Beijing, China, 1991; pp. 161–162. [Google Scholar]
- Sample Availability: Samples of the compounds 1–23 are available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).