Identification and Characterization of Two New Degradation Products of Saikosaponin A under Acid Hydrolytic Conditions
Abstract
:1. Introduction
2. Results and Discussion
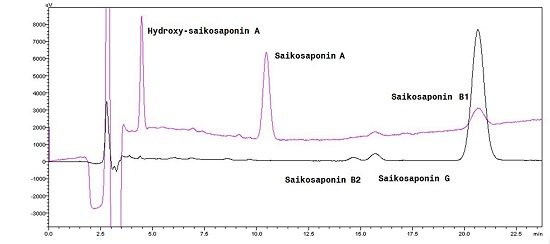
2.1. Identification of New Acid Degradation Products
2.2. Characterization of the Two New Degradation Compounds
2.2.1. Degradation Compound of Peak 1
2.2.2. Degradation Compound of Peak 3
2.3. Structural Transformation Mechanism of Hydroxy-Saikosaponin A
2.4. Structural Transformation Mechanism of SS-B2
2.5. Structural Transformation Path of SS-A under Acid Conditions
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Preparation of Standards and Sample Solutions
3.2.1. Standard Solutions
3.2.2. Sample Solutions
3.3. HPLC Analysis
3.4. Isolation by Semi-Preparative HPLC
3.5. NMR Spectra
4. Conclusions
Supplementary Materials
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Huang, H.Q.; Zhang, X.; Lin, M.; Shen, Y.H.; Yan, S.K.; Zhang, W.D. Characterization and identification of saikosaponins in crude extracts from three Bupleurum species using LC-ESI-MS. J. Sep. Sci. 2008, 31, 3190–3201. [Google Scholar] [CrossRef] [PubMed]
- Pan, S.L.; Kang, Y. Botany: Macroscopical Characters of the Medicinal Species of Bupleurum and Their Distribution in China. In Bupleurum Species–Scientific Evaluation and Clinical Application; Pan, S.L., Ed.; Taylor and Francis Group: Boca Raton, FL, USA, 2006; pp. 23–25. [Google Scholar]
- Tang, Y.H.; Zhang, Y.Y.; Zhu, H.Y.; Huang, C.G. A high-performance liquid chromatographic method for saikosaponin a quantification in rat plasma. Biomed. Chromatogr. 2007, 21, 458–462. [Google Scholar] [CrossRef] [PubMed]
- Yoon, K.D.; Kim, J. Application of centrifugal partition chromatography coupled with evaporative light scattering detection for the isolation of saikosaponins-A and -C from Bupleurum falcatum roots. J. Sep. Sci. 2009, 32, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Ye, M.; Bi, Y.F.; Ding, L.; Zhu, W.W.; Gao, W. Saikosaponin A functions as anti-epileptic effect in pentylenetetrazol induced rats through inhibiting mTOR signaling pathway. Biomed. Pharmacother. 2016, 81, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.H.; Xie, W.; Bao, Y.; Li, H.M.; Hu, S.J.; Xing, J.L. Saikosaponin A mediates the anticonvulsant properties in the HNC models of AE and SE by inhibiting NMDA receptor current and persistent sodium current. PLoS ONE 2012, 11. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Bao, Y. Dynamic observation of the effect of saikosaponin A on mice with experimental seizure. J. Guangdong Pharm. Univ. 2006, 22, 65–67. [Google Scholar]
- Lu, C.N.; Yuan, Z.G.; Zhang, X.L.; Yan, R.; Zhao, Y.Q.; Liao, M.; Chen, J.X. Saikosaponin a and its epimer saikosaponin d exhibit anti-inflammatory activity by suppressing activation of NF-kB signaling pathway. Int. Immunopharmacol. 2012, 14, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Hu, X.; Cao, Y.; Zhang, Z.; Zhang, N. Saikosaponin A inhibits lipopolysaccharide-oxidative stress and inflammation in Human umbilical vein endothelial cells via preventing TLR4 translocation into lipid rafts. Free Radic. Biol. Med. 2015, 89, 777–785. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.O.; Park, J.Y.; Jeon, S.Y.; Yang, C.H.; Kim, M.R. Saikosaponin A, an active compound of Radix Bupleuri, attenuates inflammation in hypertrophied 3T3-L1 adipocytes via ERK/NF-kB signaling pathways. Int. J. Mol. Med. 2015, 35, 1126–1132. [Google Scholar] [PubMed]
- Zhu, J.; Luo, C.; Wang, P.; He, Q.; Zhou, J.; Peng, H. Saikosaponin A mediates the inflammatory response by inhibiting the MAPK and NF-kB pathways in LPS-stimulated RAW 264.7 cells. Exp. Ther. Med. 2013, 5, 1345–1350. [Google Scholar] [PubMed]
- Ravi, T.K.; Gandhimathi, M.; Suganthi, A.; Sarovar, S. Forced-degradation study of valdecoxib as bulk drug and in tablet formulation by HPTLC. J. Sep. Sci. 2006, 11, 1647–1652. [Google Scholar] [CrossRef]
- Shabir, G. Validation of high-performance liquid chromatography methods for pharmaceutical analysis: Understanding the differences and similarities between validation requirements of the US Food and Drug Administration, the US Pharmacopeia and the International Conference on Harmonization. J. Chromatogr. A 2003, 987, 57–66. [Google Scholar] [PubMed]
- Li, X.Q.; Gao, Q.T.; Chen, X.H.; Bi, K.S. High Performance Liquid Chromatographic Assay of Saikosaponins from Radix Bupleuri in China. Biol. Pharm. Bull. 2005, 28, 1736–1742. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, K.; Amagaya, S.; Ogihara, Y. Structural transformation of saikosaponins by gastric juice and intestinal flora. J. Pharmacobio-Dyn. 1985, 8, 718–725. [Google Scholar] [CrossRef]
- Li, J.; Jiang, H.; Zhang, Y.P.; Wang, X.S.; Li, X.M. Determination of Saikosaponin B1 and B2 in Chai-Huang Tablet by HPLC. Chin. Pharmacol. J. 2011, 46, 61–63. [Google Scholar]
- Ohtake, N.; Nakai, Y.; Yamamoto, M.; Sakakibara, I. Separation and isolation methods for analysis of the active principles of Shosaikoto (SST) oriental medicine. J. Chromatogr. B 2004, 812, 135–148. [Google Scholar] [CrossRef]
- Sumino, M.; Saito, Y.; Ikegami, F.; Hirasaki, Y.; Namiki, T. Extraction efficiency of shosaikoto (xiaochaihu tang) and investigation of the major constituents in the residual crude drugs. Evid. Based Complement. Altern. Med. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Junwal, M.; Modhe, G.; Tiwari, H.; Kurmi, M.; Parashar, N. Forced degradation studies to assess the stability of drugs and products. Trends Anal. Chem. 2013, 49, 71–88. [Google Scholar] [CrossRef]
- Zhao, L.C.; Li, W.; He, Y.F.; Li, X.H.; Wang, Z.; Liu, W.C.; Zheng, Y.N.; Liang, J. Fast and Sensitive LC-DAD-ESI/MS Method for Analysis of Saikosaponins c, a, and d from the Roots of Bupleurum falcatum (Sandaochaihu). Molecules 2011, 16, 1533–1543. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.S.; Hu, P.Y.; Li, D.X.; He, M.Z.; Rao, X.; Luo, X.J.; Wang, Y.S.; Wang, Y.R. Formulations, Hemolytic and Pharmacokinetic Studies on Saikosaponin A and Saikosaponin D Compound Liposomes. Molecules 2015, 20, 5889–5907. [Google Scholar] [CrossRef] [PubMed]
- Ishii, H.; Nakamura, M.; Seo, S.; Tori, K.; Tozyo, T.; Yoshimura, Y. Isolation, characterization, and nuclear magnetic resonance spectra of new saponins from the roots of Bupleurum falcatum L. Chem. Pharm. Bull. 1980, 28, 2367–2383. [Google Scholar] [CrossRef]
- Yu, J.Q.; Deng, A.J.; Wu, L.Q.; Zhang, Z.H.; Liu, Y.; Wang, W.J.; Qin, H.L. Osteoclast-inhibiting saikosaponin derivatives from Bupleurum chinense. Fitoterapia 2013, 85, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Jiang, H.; Zhang, Y.P.; Zhang, Q.; Xu, Y. Structure of secondary saikosaponins in saiko decoction. Chin. J. Chin. Mater. Med. 2012, 37, 3078–3082. [Google Scholar]
- Fang, W.; Yang, Y.J.; Guo, B.L.; Cen, S. Triterpenoid saponins from Bupleurum marginatum var. stenophyllum. Chin. J. Chin. Mater. Med. 2016, 41, 1251–1256. [Google Scholar]
- Liang, H.; Zhao, Y.Y.; Bai, Y.J.; Zhang, R.Y.; Tu, G.Z. A new saikosaponin from Bupleurum chinense DC. Acta Pharmacol. Sin. 1998, 33, 282–285. [Google Scholar]
- Sample Availability: Not available.






| No. | Peak Label | Retention Time (min) | Peak Information |
|---|---|---|---|
| 1 | Peak-1 | 4.50 | Unknown impurity |
| 2 | Peak-2 | 11.50 | Saikosaponin A |
| 3 | Peak-3 | 14.75 | Unknown impurity |
| 4 | Peak-4 | 16.00 | Saikosaponin G |
| 5 | Peak-5 | 21.50 | Saikosaponin B1 |
| No. | Peak1 | SS-B3 | Peak3 | SS-B1 | No. | Peak1 | SS-B3 | Peak3 | SS-B1 |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 41.5 | 40.1 | 38.4 | 38.4 | 23 | 64.3 | 64.2 | 64.7 | 64.0 |
| 2 | 26.8 | 26.5 | 26.1 | 26.1 | 24 | 13.7 | 13.6 | 13.1 | 13.1 |
| 3 | 81.8 | 81.7 | 81.6 | 81.5 | 25 | 18.3 | 18.2 | 18.4 | 18.9 |
| 4 | 43.9 | 43.9 | 43.8 | 43.7 | 26 | 18.3 | 18.5 | 17.4 | 17.0 |
| 5 | 47.8 | 47.6 | 47.3 | 47.3 | 27 | 26.8 | 26.5 | 21.8 | 22.0 |
| 6 | 17.8 | 17.9 | 18.9 | 18.2 | 28 | 68.7 | 68.5 | 64.7 | 64.0 |
| 7 | 33.3 | 33.3 | 32.5 | 32.6 | 29 | 33.5 | 33.3 | 25.1 | 24.9 |
| 8 | 41.0 | 41.0 | 41.2 | 40.5 | 30 | 24.0 | 24.0 | 32.3 | 32.4 |
| 9 | 55.7 | 54.0 | 54.0 | 54.5 | CH3O | 52.1 | |||
| 10 | 36.9 | 36.8 | 36.7 | 36.5 | 1′ | 106.1 | 106.0 | 106.0 | 106.0 |
| 11 | 66.8 | 75.9 | 126.3 | 127.0 | 2′ | 71.6 | 71.8 | 71.8 | 71.7 |
| 12 | 128.1 | 122.5 | 126.3 | 125.7 | 3′ | 85.2 | 85.3 | 85.5 | 85.4 |
| 13 | 145.2 | 148.3 | 136.2 | 136.4 | 4′ | 71.9 | 71.9 | 71.9 | 71.7 |
| 14 | 43.9 | 43.9 | 42.0 | 44.2 | 5′ | 71.1 | 71.0 | 71.1 | 71.1 |
| 15 | 38.2 | 37.0 | 32.6 | 35.0 | 6′ | 17.4 | 17.2 | 17.2 | 17.2 |
| 16 | 66.4 | 66.2 | 67.9 | 76.5 | 1′′ | 106.8 | 106.7 | 106.7 | 106.7 |
| 17 | 43.9 | 44.0 | 45.4 | 44.4 | 2′′ | 75.9 | 75.9 | 75.9 | 75.8 |
| 18 | 43.9 | 44.0 | 133.1 | 133.3 | 3′′ | 78.9 | 78.8 | 78.7 | 78.8 |
| 19 | 46.5 | 47.0 | 39.1 | 38.4 | 4′′ | 72.2 | 72.2 | 72.2 | 72.2 |
| 20 | 31.0 | 31.1 | 32.7 | 32.6 | 5′′ | 78.5 | 78.5 | 78.5 | 78.8 |
| 21 | 34.2 | 34.2 | 35.6 | 35.1 | 6′′ | 62.8 | 62.7 | 62.9 | 62.7 |
| 22 | 26.0 | 26.2 | 24.6 | 30.0 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, J.; Xu, Q.; Jiang, H. Identification and Characterization of Two New Degradation Products of Saikosaponin A under Acid Hydrolytic Conditions. Molecules 2016, 21, 1232. https://doi.org/10.3390/molecules21091232
Li J, Xu Q, Jiang H. Identification and Characterization of Two New Degradation Products of Saikosaponin A under Acid Hydrolytic Conditions. Molecules. 2016; 21(9):1232. https://doi.org/10.3390/molecules21091232
Chicago/Turabian StyleLi, Jun, Qiang Xu, and Hua Jiang. 2016. "Identification and Characterization of Two New Degradation Products of Saikosaponin A under Acid Hydrolytic Conditions" Molecules 21, no. 9: 1232. https://doi.org/10.3390/molecules21091232