Abstract
Cancer is the second leading cause of death in the United States, and those who survive cancer may experience lasting difficulties, including treatment side effects, as well as physical, cognitive, and psychosocial struggles. Naturally-occurring agents from dietary fruits and vegetables have received considerable attention for the prevention and treatment of cancers. These natural agents are safe and cost efficient in contrast to expensive chemotherapeutic agents, which may induce significant side effects. The pomegranate (Punica granatum L.) fruit has been used for the prevention and treatment of a multitude of diseases and ailments for centuries in ancient cultures. Pomegranate exhibits strong antioxidant activity and is a rich source of anthocyanins, ellagitannins, and hydrolysable tannins. Studies have shown that the pomegranate fruit as well as its juice, extract, and oil exert anti-inflammatory, anti-proliferative, and anti-tumorigenic properties by modulating multiple signaling pathways, which suggest its use as a promising chemopreventive/chemotherapeutic agent. This review summarizes preclinical and clinical studies highlighting the role of pomegranate in prevention and treatment of skin, breast, prostate, lung, and colon cancers.
1. Introduction
Cancer is a disease of unrestricted cell proliferation. Normally considered a disease of genetic origin, research over the last several decades has established beyond doubt that various epigenetic/environmental factors play an important role in the development and/or progression of cancer [1,2]. With no single defined cause and a number of risk factors, including smoking, alcohol consumption, poor diet, and obesity, etc., cancer is widely accepted as a lifestyle disease [3,4]. Almost 1,685,210 new cancer cases will be detected in 2016 in the United States alone, and nearly 595,690 people will die of cancer [5]. Despite the considerable advancement in treatment options, the incidence and mortality from cancer continues to increase [3,5]. It is estimated that there will be virtually 20 million cancer patients by the year 2025 [5]. Therefore, attention is being focused on prevention as an ultimate strategy for the management of cancer [6,7]. It is currently estimated that two-thirds of cancer-related deaths may well be prevented through lifestyle variation, mostly through dietary means [7,8].
Almost 2500 years ago, Hippocrates recognized the importance of food for overall health. Medicinal systems across different cultures have professed and promoted the use of edible substances, especially those derived from plants, to prevent/treat diseases over last several centuries, hence, creating awareness of the potential of natural agents also known as phytochemicals as cancer chemopreventive/chemotherapeutic agents. As of today almost 47% of the available anticancer drugs in the market are derivatives of natural products or natural product mimics [9]. There is a great deal of scientific evidence to show that daily consumption of a diet rich in fruits and vegetables reduces the risk of cancer [10,11,12] and, in recent years, considerable interest has been focused on plant foods containing polyphenolic compounds [13,14]. Reports suggest that, except for 5%–10% of all cancer cases, the remaining 90%–95% are caused by environment and lifestyle [15]. So far, nearly 25,000 different phytochemicals have been identified in fruits and vegetables that have tremendous anticancer properties [15]. These phytochemicals are nontoxic and generally target multiple signaling pathways [6,14].
Pomegranate (Punica granatum L.) is obtained from a deciduous tree belonging to the family Lythraceae. There are reports that indicate that it first originated in modern day from Iran and has been cultivated through the Mediterranean region and Northern India since ancient times [16]. Today, it is cultivated in North Africa and tropical Africa, North and South America, and even in Europe for its fruit crop and also as decorative trees and shrubs. Pomegranate fruit is a rounded berry with a thick reddish skin covering approximately 200–1400 white to deep red or purple seeds. Pomegranate seeds are edible and hold strong antioxidant and anti-inflammatory properties due to their high content of hydrolysable tannins and anthocyanins [17]. As compared to the antioxidant activity of vitamin E, β-carotene, and ascorbic acid, the pomegranate antioxidants appear unique due to combinations of a wide array of polyphenols, having a broader range of action against several types of free radicals [18]. As compared to the recognized antioxidants in red wine and green tea, anthocyanins from pomegranate fruit possess significantly higher antioxidant activity [19].
Pomegranate has been used in various medicinal systems of medicine for the treatment and therapy of a multitude of diseases and ailments. In the ancient Indian medicinal system, i.e., in Ayurvedic medicine, the pomegranate was considered to be a whole pharmacy unto itself. It was recommended to be used as an antiparasitic agent and to treat diarrhea and ulcers [20,21]. The Unani system of medicine, which is another traditional system of medicine, recognizes the importance of pomegranate in the treatment of diabetes [22]. The medicinal properties of pomegranate have sparked significant interest in today’s scientific community as evidenced by the scientific research relating to health benefits of pomegranate that have been published in last few decades [14,23]. Remarkably, it is not just the pomegranate fruit itself, but other parts of the plant as well, including the bark, leaves, and roots of the pomegranate tree, that are rich in molecular constituents with therapeutic properties [21,24].
Studies have shown that pomegranate and its constituents can efficiently affect multiple signaling pathways involved in inflammation, cellular transformation, hyperproliferation, angiogenesis, initiation of tumorigenesis, and eventually suppressing the final steps of tumorigenesis and metastasis [14,23]. The pomegranate constituents are shown to modulate transcription factors, pro-apoptotic proteins, anti-apoptotic proteins, cell cycle regulator molecules, protein kinases, cell adhesion molecules, pro-inflammatory mediators, and growth factors in various cancers (Table 1). In this review article, we first discussed some of the polyphenolic constituents and mineral ions present in pomegranate, and we then discussed studies on chemopreventive/chemotherapeutic properties of pomegranate against different types of cancer, such as skin, breast, prostate, colon, and lung cancers in cell culture systems, animal models and humans.

Table 1.
Molecular targets of pomegranate in cancers.
2. Pomegranate Chemical Constituents
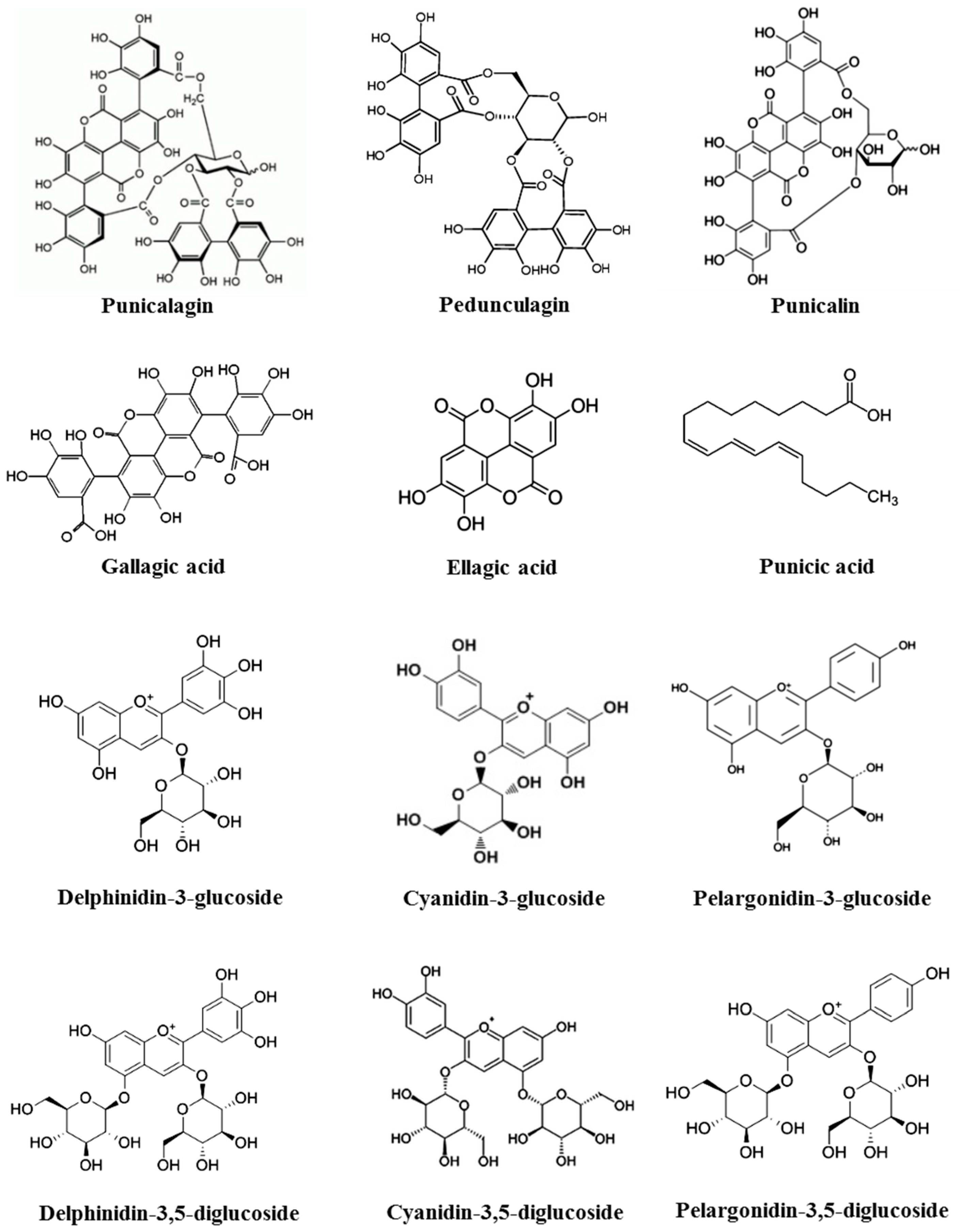
The pomegranate fruit consists of white to deep purple seeds embedded in a white spongy astringent membrane surrounded by a thick reddish skin, or pericarp. Pericarp constitutes almost 50% of the fruit weight and is a rich source of bioactive constituents, such as phenolics, flavonoids, ellagitannins, and proanthocyanidin compounds. It also contains various minerals, mainly potassium (K), nitrogen (N), calcium (Ca), phosphorus (P), magnesium (Mg), and sodium (Na), as well as complex polysaccharides. The remaining 50% of the fruit consists of seeds (constituting 10% of the fruit weight) and arils (constituting 40% of the fruit weight) [17]. Pomegranate seeds hold strong antioxidant and anti-inflammatory properties due to the high content of hydrolysable tannins (punicalagin, pedunculagin, punicalin, gallagic acid, ellagic acid, and esters of glucose) and anthocyanins (delphinidin-3-glucoside, cyanidin-3-glucoside, delphinidin-3,5-diglucoside, cyanidin-3,5-diglucoside, pelargonidin-3,5-diglucoside, and pelargonidin-3-glucoside) (Figure 1) [34,62]. Various organic acids, such as ascorbic acid, citric acid, and malic acid, etc., are also reported to be present in the seed coat [17], while the arils contain water (85%), sugars (10%), mainly fructose and glucose, and pectin (1.5%). Arils are a rich source of bioactive compounds such as phenolics and flavonoids, principally anthocyanins. Pomegranate seed oil consists of mainly conjugated linolenic acid. Interestingly, punicic acid, a conjugated isomer of linolenic acid found uniquely in pomegranate oil, constitutes 70%–76% of the seed oil [63]. Sterols, steroids, and cerebroside, a key component of mammalian myelin sheaths, constitute the minor share of the seed oil [64].
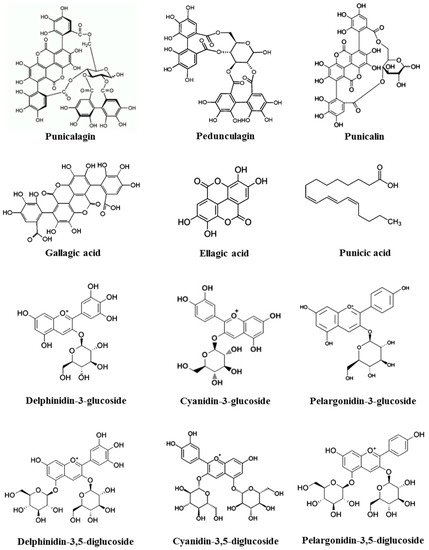
Figure 1.
Chemical structures of major constituents present in pomegranate.
Pomegranate leaves contain some unique tannins in addition to containing glycosides of apigenin, which is flavone with progestinic and anxiolytic characters [65,66]. Leaves also represent a rich source of elements such as N, K, Ca, and Fe, with levels of elements varying with the season and the stage and maturity of the plant [24]. For example, K content is reported to be high in young leaves, while levels of Ca and Fe are considered to be highest in old leaves. Medium-age plants have high N content. The content of N, however, is reduced at the time of flowering and setting of the fruit. N content is further reported to decline with the fruit maturity [67,68].
3. Pomegranate and Skin Cancer
Skin cancer is the most common form of cancer in fair skinned individuals. It is estimated that, in the United States alone, nearly 83,510 new skin cancer cases will be diagnosed in the year 2016 that will result in nearly 13,650 deaths [5]. Considering this grim statistics, it is essential to develop novel and effective skin cancer chemopreventive/chemotherapeutic strategies. Sun exposure is the major known environmental factor influencing the development of skin cancer of all types. Ultraviolet B (UVB) radiation coming from the sun represents a major risk factor for the development of skin cancer [69,70]. At the molecular level, exposure of skin to UVB radiation leads to activation of multiple signaling pathways in the skin. These pathways control DNA damage repair, oxidative balance, inflammation, immune responsiveness, and cell survival or cell death [69,70]. Pomegranate fruit extract (PFE), pomegranate juice (PJ), and pomegranate seed oil (PSO) have been tested in cell culture, reconstituted human skin models, and animal models of skin cancer and exhibit immense potential for preventing UVB-induced skin cancer.
PFE was shown to inhibit UVB-induced phosphorylation of the mitogen-activated protein kinases (MAPK) in normal human epidermal keratinocytes (NHEK) [25]. Pretreatment of NHEK with PFE resulted in a dose- and time-dependent inhibition of UVB-induced phosphorylation of ERKl/2, JNK1/2, and p38 proteins. PFE was also found to inhibit UVB-mediated activation of the nuclear factor kappa B (NFκB) pathway, an effect that was accompanied with reduced phosphorylation of IκBα, increased stabilization of IκBα protein, and reduced activation of IKKα protein [25]. The photo-protective effects of PFE extend against the harmful effects of UVA radiation as well, shown in a study wherein PFE was evaluated for its effects against UVA-mediated activation of signal transducer and activator of transcription 3 (STAT3), AKT, and ERK1/2 in NHEK [29]. While UVA was shown to result in increased phosphorylation of STAT3, AKT, mTOR, and ERK1/2 in NHEK, pretreatment with PFE resulted in inhibition of these events in a dose-dependent manner. Interestingly, PFE treatment to NHEK resulted in a significant inhibition of UVA-induced expression in Ki-67 and PCNA, and it also led to an enhanced expression of pro-apoptotic Bax and Bad with downregulation of antiapoptotic Bcl-xL protein [29]. Similarly, polyphenol-enriched pomegranate extract (POMx) was evaluated for its effect on UVB-mediated oxidative stress and markers of photoaging in immortalized human HaCaT keratinocytes, and it was found that pretreatment protects the cells from UVB-induced oxidative stress and markers for photoaging [26]. Treatment of HaCaT cells with POMx prior to UVB irradiation resulted in inhibition of UVB-mediated decrease in glutathione content as well as UVB-induced lipid peroxidation [26]. POMx treatment was also found to protect HaCaT cells against UVB-induced photoaging as evidenced by reduction in expression of UVB-induced upregulation of matrix metalloproteinases (MMPs) (such as MMP-1, -2, -7, and -9) and phosphorylation of MAPK [26]. Another recent study carried out on HaCaT cells evaluated photoprotective effects of a nanoemulsion of PSO against UVB radiation and found that PSO protected cells against UVB-induced DNA damage in a dose-dependent manner [30]. Similarly, PFE was shown to protect human fibroblast cells from the UVA- and UVB-induced damage by reducing activation of NFκB, downregulating active caspase 3, and increasing cells in G0/G1 phase [31]. Pomegranate-derived products, such as PJ, POMx, and PSO, were tested for their UVB protective effects in reconstituted human skin [32]. Pretreatment of EpiDerm with pomegranate-derived products inhibited UVB-induced DNA damage as well as activation of MMPs in the EpiDerm, thus indicating the usefulness of pomegranate-derived products against UVB-induced damage to human skin [32].
The chemopreventive properties of PFE were further evaluated in mice exposed to UVB radiation. Afaq et al. [33] evaluated the effects of PFE administration via drinking water against the early biomarkers of UVB-induced skin cancer in SKH-1 hairless mice that were exposed to a single dose of UVB (180 mJ/cm2) irradiation. It was observed that PFE treatment augmented UVB-mediated increase in the protein expression of p21 and p53, but also resulted in inhibition of NFκB signaling as evidenced by reduced nuclear translocation of NFκB, reduced activation of IKKα, as well as decreased phosphorylation and degradation of IκBα. Photochemopreventive effects of PFE administered via drinking water were further evaluated in mice exposed to multiple UVB irradiations [27]. Oral administration of PFE inhibited UVB-induced epidermal hyperplasia, leukocyte infiltration, and protein oxidation. Oral administration of PFE also attenuated UVB-induced activation of key inflammatory and cell proliferative pathways such as NFκB and MAPK. Reduction in UVB-induced protein expression of COX-2, iNOS, PCNA, cyclin D1, and MMPs in mouse skin further supported anti-inflammatory and anti-proliferative effects of PFE [27]. More importantly, oral administration of PFE in drinking water reduced UVB-induced skin tumor incidence and tumor multiplicity in SKH-1 hairless mice [28]. PFE treatment resulted in inhibition of UVB-induced phosphorylation of STAT3, and NFκB/p65 with a concomitant decrease in the protein expressions of iNOS, and COX-2 in uninvolved skin from tumor-bearing mice and skin tumors compared to non-PFE-treated animals. These data suggest that PFE protects against UVB-induced skin tumorigenesis, at least in part, by modulating transcription factors STAT3 and NFκB.
PFE’s capability to inhibit skin cancer was also determined in 7,12-dimethylbenz(a)anthracene (DMBA) initiated and 12-O-tetradecanoylphorbol-3-acetate (TPA) promoted chemical carcinogenesis model. Topical application of PFE to mouse skin resulted in delayed onset of skin tumor formation, as well as a significant reduction in tumor incidence and tumor burden in mice [34]. PFE was found to inhibit TPA-induced skin edema, thus highlighting PFE’s anti-inflammatory effects. It was further observed that topical application of PFE inhibited TPA-induced activation of NFκB and IKKα, phosphorylation and degradation of IκBα, as well as phosphorylation of ERK1/2, p38 and JNK1/2. Hora et al. [35] also demonstrated the anti-skin tumorigenic effect of PSO by using chemical carcinogenesis protocol (DMBA initiated and TPA promoted) in CD-1 mice. A significant reduction in tumor incidence and tumor multiplicity was observed in PSO-treated mice compared to the untreated mice. To further improve the anticancer effects of PFE, George et al. [71] carried out a combinatorial phytochemical treatment approach and administered PFE and diallyl sulfide (DAS), alone and in combination in chemical carcinogenesis model. It was observed that PFE and DAS exerted inhibition of tumor development synergistically. While PFE and DAS reduced tumor incidence by ~55% and ~45%, respectively, even more potent reduction (~84%) of tumor incidence was observed in mice that received both PFE and DAS. These data suggest that PFE and PSO exhibit chemopreventive effects against skin tumorigenesis.
4. Pomegranate and Breast Cancer
Breast cancer is the second leading cause of cancer-related deaths in women. In 2016, an estimated 246,660 new cases of invasive breast cancer are expected to be diagnosed in women in the Unites States, along with 61,000 new cases of non-invasive (in situ) breast cancer [5]. Old age, family history of breast cancer, early age at menarche, late age of menopause, long-term use of estrogen-replacement therapy, and later age at birth of first-born child are some of the common established risk factors for the development of breast cancer. Steroid hormones, particularly estrogens, are believed to play a central role in development of breast cancer [72]. In recent years, the link between dietary factors and breast cancer risk has been a significant area of research. Studies have shown the beneficial effects of pomegranates in breast cancer [73]. Kim et al. [74] reported that polyphenols from fermented PJ, pericarp, and PSO inhibited aromatase, which converts androgen to estrogen and plays an important role in breast carcinogenesis. It was shown that polyphenols derived from fermented PJ, pericarp, and PSO were also able to inhibit 17-β-hydroxysteroid dehydrogenase, an estrogen biosynthetic enzyme. Consistent with their anti-estrogenic effects, polyphenols from fermented PJ and pericarp exerted a cell growth inhibitory effect against both MCF-7 and MB-MDA-231 breast cancer cell lines. Polyphenols from fermented PJ also inhibited DMBA-induced cancerous lesion formation in a murine mammary gland organ culture [74]. Another study revealed the potential of pomegranate ellagitannins-derived compounds exhibiting anti-proliferative and anti-aromatase activities in breast cancer cells [36]. The pomegranate ellagitannin-derived compounds including ellagic acid, gallagic acid, and urolithins A and B (acetylated, methylated, and sulfated analogues) were investigated for their anti-aromatase activity by using placental microsome aromatase assay and a live cell based assay. It was observed that urolithin A, methylated urolithin A, urolithin B, methylated urolithin B, acetylated urolithin B, urolithin B sulfate, and gallagic acid significantly inhibited aromatase activity in the placental microsomes. When these active compounds were further compared in an aromatase over-expressing cell line (MCF-7aro), urolithin B was found to be the most potent aromatase inhibitor. It significantly inhibited aromatase activity at 2.35 μM (p ≤ 0.05) and 4.7 μM (p ≤ 0.01). Gallagic acid was observed to exhibit anti-aromatase activity as well; it inhibited aromatase activity significantly at a dose of 4.7 μM (p ≤ 0.01). Urolithins were further tested for their effects against testosterone-induced cell proliferation, and it was found that urolithin B inhibits testosterone-induced cell proliferation. Urolithin B was followed by gallagic acid for the anti-proliferative effects [36]. These data suggest that intake of pomegranate may be a beneficial strategy for breast cancer chemoprevention.
The methanolic extract of pomegranate pericarp (PME) was shown to possess a selective estrogen receptor modulator (SERM) property in human breast cancer cell lines and in vivo models of estrogen deprivation [37]. SERMs are ligands for the estrogen receptor (ER) and may exert an agonist or an antagonist function depending on the type of tissue. SERMs are frequently used for the therapy of estrogen-dependent breast cancers. PME treatment led to significant dose-dependent inhibition of cell growth in MCF-7 cell line that are ER+, while there was no effect on the proliferation of ER− MDA MB-231 cells. PME also inhibited 17β-estradiol-induced proliferation in MCF-7 cells. In addition, PME was found to downregulate the expression of estrogen responsive genes such as ERα, pS2, and PR in the MCF-7 cells. Finally, the lack of esterogenicity of PME was confirmed in ovariectomized (OVX) mice, wherein uterine wet weights and epithelial heights were assessed as markers of esterogenicity. It was observed that while 17β-estradiol increased absolute and normalized uterine wet weight in OVX animals by approximately two times, there was no significant difference in weight of uterus between the groups that received PME and the vehicle-treated OVX control group, indicating the lack of estrogenecity of PME on uterine endometrium. Similarly, from the uterine histology it was clear that while 17β-estradiol induced proliferation of uterine epithelium, there was no luminal epithelial proliferation in PME treated OVX mice [37]. Further, Rocha et al. [38] tested PJ and its components for their effects on a number of precarious processes involved in breast cancer metastasis. They used two breast cancer cell lines, MDA-MB-231 cells (ER−) and MCF-7 (ER+), and the non-neoplastic cell line MCF10A, and showed that PJ, or a combination of its components, luteolin plus ellagic acid plus punicic acid, increased cancer cell adhesion, decreased cancer cell migration, and reduced growth of the breast cancer cells, without affecting the normal cells. PJ and the three components also prevented the production of pro-inflammatory cytokines/chemokines in the cancer cells. Interestingly, the study also revealed that PJ and its components promoted expression of genes involved in increased adhesion, inhibited cell migration stimulating genes, and prevented chemotaxis of the cancer cells to stromal cell-derived factor 1α.
Several studies investigating the chemopreventative potential of pomegranate against breast cancer have highlighted the importance of pro-apoptotic and antioxidant properties manifested in the PFE and its components [6,14,73]. Punicic acid, a polyunsaturated fatty acid found in PSO, was reported to significantly inhibit growth, as well as induce apoptosis of estrogen sensitive and insensitive breast cancer cell lines, namely MDA-MB-231 and MDA-ER-7 cells [39]. Methanolic extract of PFE was shown to reduce proliferation of MCF-7 breast cancer cells while increasing the number of apoptotic cells in a dose-dependent manner [40]. These effects of PFE were associated with an increased expression of pro-apoptotic gene Bax, and a reduced expression of anti-apoptotic gene Bcl2. Costantini et al. [41] identified punicic acid and its congeners as the most abundant compounds of the hydrophilic fraction (80% aqueous methanol extract) from PSO and evaluated their possible anti-inflammatory effects on breast cancer lines (MCF-7 and MDA-MB-231). The study indicated that the hydrophilic extract treatment resulted in a significant decrease in cell viability in both breast cancer cell lines with an increase in G0/G1 phase of the cell cycle compared to untreated cells and with no significant increase in apoptosis in these two breast cancer cell lines. This study also indicated that with increasing amounts of the hydrophilic extracts of PSO, there was a decrease in the levels of VEGF and pro-inflammatory cytokines (IL-2, IL-6, IL-12, IL-17, CXCL10, MIP-1α, MIP-1β, MCP-1, and TNF-α).
More recently, a study examining the anti-breast cancer properties of PFE has focused on gene expression changes that occur at the whole genome level in the MCF-7 cells [42]. It was observed that the reduced proliferation of MCF-7 cells by PFE treatment led to differential expression of 903 genes, of which 505 genes were upregulated, while 398 genes were downregulated. A majority of the genes that were upregulated were involved in regulation of apoptosis, while the genes that were downregulated included genes involved in mitosis, chromosomal organization, RNA processing, DNA damage response, and DNA repair. Genes such as MRE11, RAD50, NBS1, RAD51, BRCA1, BRCA2, BRCC3, and MSH6 that are involved in DNA damage response and repair were found to be downregulated [42]. Another cDNA microarray based study for understanding the molecular mechanisms underlying the ellagic acid-induced growth inhibition on MCF-7 cells proposes that ellagic acid inhibits the growth of breast cancer cells by cell cycle arrest and inhibition of proliferation [43]. It was observed that changes in genes that belong to TGF-β/Smads signaling pathway as a molecular mechanism of ellagic acid regulated cell cycle arrest in MCF-7 cells. TGF-β is known to be a strong tumor suppressor that promotes cell growth inhibition, apoptosis, and differentiation [75,76].
Studies evaluating the chemopreventive effect of orally administered pomegranate emulsion (PE) against breast cancer were performed in DMBA-induced mammary tumorigenesis in female Sprague-Dawley rats [77]. Rats that were administered PE exhibited reduction in both tumor incidence and cumulative tumor burden compared to control rats. PE-treated tumors exhibited almost normal ductal and alveolar structure with uniform epithelial cells without any sign of hyperplasia when compared with tumors from control rats that showed extensive epithelial proliferation histologically. PE exerted its chemopreventive effect against DMBA-initiated mammary tumors by reducing cell proliferation and inducing apoptosis [77]. Mechanistic information underlying the chemopreventive effects of PE was further evaluated in another study from the same group showing that PE-treated tumors showed reduced expression of ER-α and ER-β, as well as reduced expression, cytoplasmic accumulation, and nuclear translocation of β-catenin [44]. These data suggest that PE-induced disruption of ER and Wnt/β-catenin signaling pathways is the molecular basis of its chemopreventive effect against DMBA-inflicted rat mammary tumorigenesis.
5. Pomegranate and Prostate Cancer
Prostate cancer (PCa) is the second major cause of cancer related deaths in men in the United States. The latest count for new PCa diagnoses is estimated to be 180,890 with 26,120 estimated death cases in United States [5]. Pomegranate has been shown to exhibit beneficial effects against PCa in cell culture and animal studies. Lansky et al. [78] reported that ellagic acid, caffeic acid, luteolin, and punicic acid that are found in substantial amounts in the peels, PJ, and PSO of the pomegranate fruit reduced the invasive potential of PC-3 cells. A supradditive inhibition in PC-3 cell invasion was observed when caffeic acid, luteolin, and punicic acid were equally combined at the same gross dose when compared to individual agents. Albrecht et al. [79] examined the effects of pomegranate-derived fractions, namely pomegranate pericarp polyphenols, fermented PJ polyphenols, and cold-pressed PSO on PCa growth, apoptosis, invasion, and tumor growth. Treatment of human PCa cells with PSO, fermented PJ polyphenols, and pomegranate pericarp polyphenols reduced cell proliferation, increased cells in G2/M phase, and induced apoptosis. Pomegranate-derived fractions treatment reduced PC-3 invasion and also inhibited tumor growth in athymic mice implanted with PC-3 cells. Malik et al. [45] reported that the modulation of cdk is the key mechanism involved in the pro-apoptotic and anti-proliferative effects of PFE. Treatment of highly aggressive PC-3 cells with PFE resulted in a cell growth inhibition and induction of apoptosis. The study documented that PFE essentially downregulated cyclins D1, D2, E, cdk2, cdk4, and cdk6 and upregulated p21 and p27. PFE-induced apoptosis in PC-3 cells was accompanied with an increase in cleaved PARP, a decrease in Bcl-2, and a concomitant increase in Bax. Additionally, oral administration of PFE in drinking water to athymic nude mice implanted with CWR22Rν1 cells resulted in a significant inhibition in tumor growth that was associated with a reduction in the secretion of prostate-specific antigen (PSA) in the serum [45]. Treatment of LNCaP cells with ellagic acid, a component of PJ, induced apoptosis by increasing the Bax/Bcl-2 ratio and cleavage of caspase 3. Ellagic acid treatment increased the expression of p21 and p27, whereas expression of cyclin D1 and cdk1 was decreased [80]. These data indicate that ellagic acid is a potential chemotherapeutic agent against PCa.
Studies have demonstrated that PFE exhibits beneficial effects by reducing proliferation and inducing apoptosis in PCa cells by targeting multiple signaling pathways. Treatment of human metastatic castration-resistant PCa cells with POMx induced cell death by reducing the expression of survivin and inhibiting STAT3. In this study, POMx treatment also enhanced the efficacy of docetaxel in reducing C4-2 tumor growth in athymic nude mice [46]. Oral administration of PJ in drinking water to transgenic rats for adenocarcinoma of the prostate resulted in a decrease in the incidence of adenocarcinoma in the lateral prostate as compared to the control group. Ellagic acid also reduced the progression of prostatic lesions or adenocarcinoma in lateral prostate. Both PJ and ellagic acid suppressed prostate carcinogenesis by activation of caspase 3-mediated apoptosis. Insulin-like growth factor-1 (IGF-1) is upregulated in several cancers including PCa, and exhibits mitogenic and anti-apoptotic effects. IGF binding protein (IGFBP)-3 is the most abundant of the IGFBPs and it binds to IGF-1 and regulates the availability and ligand function of IGF-1 to IGF-1 receptor [81]. Administration of PFE in drinking water to TRAMP mice inhibited prostate carcinogenesis by downregulating IGF-1/Akt/mTOR pathways [49]. Treatment of LAPC4 PCa cells with POMx resulted in the inhibition of cell proliferation and induction of apoptosis. Co-treatment of LAPC4 cells with POMx and IGFBP-3 resulted in the additive inhibition of cell growth and synergistic activation of apoptosis. In addition, co-treatment with IGF-1 and POMx blocked apoptosis in 22Rν1 cells induced by POMx. However, the effects of IGF-1 in inhibiting POMx-induced apoptosis was abolished in IGF-1 receptor null MEF cells, indicating the significance of the IGF1 receptor in antagonizing the effects of POMx [50]. PFE treatment of androgen independent DU145 cells with constitutive activation of NFκB resulted in inhibition of cell proliferation and induction of apoptosis by blockade of NFκB. In addition, PFE treatment inhibited the growth of androgen-sensitive and androgen-independent PCa that lack basal NFκB activity. These data suggest that PFE inhibits the growth of PCa cells in NFκB-dependent and -independent manner. Dietary supplementation of PFE to castrated immunodeficient mice delayed the emergence of LAPC4 androgen-independent xenografts by inhibition of NFκB activity [47]. A proteomics study evaluating the effects of PJ on DU145 cells demonstrated that PJ potentially limits PCa by modulating the expression of genes associated with apoptosis, the NFκB signaling pathway, invasion/metastasis, angiogenesis, and cytoskeleton [48].
Androgens and their receptors are crucial factors contributing to PCa development, growth, and progression [82]. Treatment of androgen-dependent (LNCaP) and androgen-independent (LNCaP-AR and DU145) human PCa cell lines with PFE and PJ displayed decreased expression of genes involved in androgen biosynthesis, such as 3β-hydroxysteroid dehydrogenase type 2, aldo-keto reductase family 1 member C3, steroid 5α reductase type 1, and AR [51]. These findings suggest that the polyphenols present in pomegranate may be useful in androgen-independent PCa and in subsets of PCa where there is up-regulation of AR. The cytochrome P450 (CYP) proteins are responsible for bioactivation of xenobiotics and endobiotics. The CYP1 isoforms activate a number of polycyclic aromatic hydrocarbons to exert their detrimental effects. Studies have shown that CYP1B1 plays an important role in the initiation and promotion of cancer and, therefore, represents an attractive target for cancer chemoprevention. It was observed that systemically available metabolites of PJ could effectively inhibit enzyme activity/expression of CYP1B1 [52]. Previous studies have shown that polymorphisms in CYP1B1 and PSA genes increased the risk of PCa [83]. Therefore, these studies suggest that consumption of PJ may reduce the incidence of PCa. A significant amount of chemopreventive studies explicitly suggest that the potential protective effect of PJ against PCa is largely attributed to ellagitanins, representing the most abundant polyphenols present in PJ. In this context, the main metabolite to concentrate in the human prostate gland upon consumption of PJ was urolithin A glucuronide, (3,8-dihydroxy-6H-dibenzo[b,d]pyran-6-one glucuronide), together with traces of urolithin B glucuronide, (3-hydroxy-6H-dibenzo[b,d]pyran-6-one glucuronide) and dimethyl ellagic acid [84]. These data indicate urolithin glucuronides and dimethyl ellagic acid may be the bioactive metabolites accounting for the chemopreventive effects of PJ against PCa.
Pantuck et al. [85] performed the first clinical trial of PJ in PCa patients following surgery and radiation. The study reported that oral consumption of PJ had no adverse effects and significantly increased PSA doubling times (PSADT) in men with PCa. A randomized, multi-center, double-blind phase II study was performed to determine the biological activity of two doses of POMx in PCa patients by monitoring PSADT following initial therapy for localized PCa. Treatment of PCa patients with POMx increased the PSADT by almost six months in both the treatment arms [86]. To determine the effects of PFE treatment on PSADT in PCa patients with rising PSA after primary therapy, a randomized, double-blind, placebo-controlled study was performed. It was observed that PFE did not significantly prolong the PSADT in patients with rising PSA after primary therapy compared to the placebo-treated group. In addition, this study indicated that patients with MnSOD AA genotype receiving PFE may be more sensitive in prolonging PSADT [87]. A phase II, randomized double-blind trial of men with PCa undergoing radical prostatectomy showed that there was no significant reduction in the level of 8-hydroxy-2′-deoxyguanosine in POMx treated group compared to the placebo-treated group. In addition, there were no differences in expression of pS6, NFκB, or Ki67 within PCa tissues between arms [88]. Stenner-Liewen et al. [89] evaluated the therapeutic impact of PJ as an adjunct intervention in a cohort of more advanced or metastatic PCa, of which 68% had castration-resistant PCa. The patients continued their baseline treatment, such as androgen deprivation therapy. The study concluded that consumption of PJ did not result in a significant decline in PSA levels compared to placebo.
6. Pomegranate and Lung Cancer
Lung cancer is the leading cause of cancer-related mortality worldwide. According to statistics, an estimated 224,390 new cases and 158,080 deaths are expected to be caused by lung and bronchus tumors in both sexes in the United States in 2016 [5]. Current research has documented the potential of PFE in inhibiting the growth of lung cancer cells in culture. PFE treatment resulted in a substantial decrease in the viability of human lung carcinoma A549 cells but had minimal effects on normal human bronchial epithelial cells. PFE treated A549 cells displayed a dose-dependent arrest of cells in the G0/G1 phase of the cell cycle, which was linked to induction of WAF1/p21 and KIP1/p27 and a decrease in the expression of cyclins and cdks. Furthermore, PFE treatment inhibited several signaling pathways, including MAPK, PI3K/AKT, and NFκB [53]. Using punicalagin isolated from the pomegranate husk, Aqil et al. [90] showed that punicalagin possesses strong antioxidant activity by decreasing the accumulation of oxidative DNA products and displays strong anti-proliferative activity against lung cancer cells. Punicalagin and ellagic acid, the major constituents of the pomegranate peel, were shown to possess strong anti-proliferative activities. Both A549 and H1299 lung cancer cell lines displayed comparable levels of sensitivity to the tested compounds [54]. A recent study evaluated anti-proliferative properties of pomegranate peel against different cancer cells including lung cancer. This study indicated that the anti-proliferative properties of pomegranate are not solely confined to the edible part of the pomegranate fruit [91]. Another study showed that pomegranate leaf extract (PLE) reduced cell proliferation of non-small cell lung carcinoma cell lines (A549, H1299) and mouse Lewis lung carcinoma cell line LL/2. PLE treatment reduced H1299 cell migration and invasion, indicating usefulness of the PLE in reducing metastasis [92].
The chemopreventive efficacy of PFE was evaluated using benzo(a)pyrene [B(a)P] and N-nitroso-tris-chloroethylurea (NTCU) induced lung tumor models of A/J mice. It was found that compared to the control mice that were exposed to B(a)P and NTCU, mice that received PFE in drinking water had statistically significant lower lung tumor multiplicities. PFE-treated mice showed decreased activation of NFκB, MAPK, and PI3K pathways leading to reduced cell proliferation and angiogenesis in lungs of B(a)P- and NTCU-treated mice [55]. Another study revealed that oral consumption of PFE in drinking water reduced tumor growth in athymic nude mice implanted with A549 cells [53]. Punicalagin and ellagic acid were shown to possess strong anti-mutagenic and anti-proliferative activities in B(a)P-induced lung cancer model [54]. The results from these studies suggest the usefulness of PFE as a chemopreventive/chemotherapeutic agent against human lung cancer. Pomegranate peel aqueous extract was evaluated for the antioxidant and anti-inflammatory properties and it was found that it inhibited neutrophil myeloperoxidase (MPO) activity. Although it showed no effect on superoxide generation, it attenuated lipopolysaccharide-induced lung inflammation in mice. Inhibition of MPO activity by pomegranate peel aqueous extract could be attributed to its anti-inflammatory action [56]. Similarly, Husari et al. [57] tested the antioxidant activity of PJ in response to hyperoxia and observed that rats exposed to hyperoxia displayed increased ROS production and increased levels of pro-inflammatory cytokines (IL-1β and IL-6) in the lungs. Administration of PJ in drinking water resulted in significant attenuation of these effects of hyperoxia, thus indicating that PJ possesses strong anti-inflammatory activities besides possessing strong antioxidant properties. Recently it has been shown that PFE possesses strong antioxidant activity in methotrexate-treated rats. Methotrexate-treated rats exhibited a significant increase in malondialdehyde levels, total oxidant status, and oxidative stress index in the serum and lung; however, pretreatment of rats with PFE reversed these effects [58].
7. Pomegranate and Colon Cancer
Colorectal cancer is the third most commonly diagnosed cancer and the third leading cause of cancer deaths in both men and women in the United States. According to the American Cancer Society’s most current estimates, approximately 134,490 new cases of colorectal cancer will be diagnosed in the United States in 2016 [5]. Increasing evidence supports that regular consumption of fruits, vegetables, and grains that are rich in polyphenols may reduce the risk of colon cancer [93,94,95]. Pomegranates have also been studied for their protective effects against colon cancer. Seeram et al. [96] studied the effect of PJ and its purified polyphenols on human colon cancer cell lines (HT-29, HCT116, SW480, SW620), and found that PJ displayed the highest anti-proliferative and pro-apoptotic effects compared to its purified polyphenols. Thus, this study suggests that separation of individual polyphenols from PJ may decrease the overall anti-proliferative activity, owing to the requirement of multiple compounds for chemical synergy and multifactorial effects compared to single purified agents. Treatment of HT-29 cancer cells with PJ inhibited TNFα-induced COX-2 protein expression and also suppressed NFκB DNA binding and AKT activity [59]. These studies indicate that PJ plays an important role in downregulating inflammatory signaling pathways in colon cancer cells. Larrosa et al. [97] studied the induction of apoptosis in Caco-2 colon cancer cells by punicalagin and ellagic acid from PJ. Their study revealed that treatment of Caco-2 cells with these agents resulted in the release of mitochondrial cytochrome c into the cytosol, activation of caspase-3 and -9, and down-regulation of anti-apoptotic Bcl-xL. Both punicalagin and ellagic acid treated Caco-2 cells resulted in decreased protein expression of cyclins, as well as arrest of cells in S phase of the cell-cycle. The authors suggest that the anti-carcinogenic effect of pomegranate ellagitannins could largely be due to their hydrolysis product ellagic acid, which induced apoptosis in colon cancer cells.
The consumption of PSO rich in conjugated linolenic acid in the diet was found to significantly inhibit the growth of azoxymethane-induced colonic adenocarcinomas in male F344 rats without causing any adverse effects [64]. Furthermore, the dietary intake of PSO deceased the multiplicity of colonic carcinomas in the colon of rats. Additionally, PJ was studied for the colon cancer chemopreventive effects in vivo on azoxymethane (AOM)-induced aberrant crypt foci in Fisher 344 male rats that were administered 20% PJ before and after treatment with azoxymethane [60]. The histopathology of the rat colon studied after the 17th week of treatment revealed a significant decrease in the number of large crypts in PJ-fed rats. The protective effect of the PJ was also evident from the PJ-fed rats’ increased food intake and weight gain. Further, the activity of hepatic glutathione S-transferase was significantly higher in rats fed with PJ, which probably supports the mechanism of pomegranate chemopreventive activities. These results suggest favorable effects of pomegranate against the development of colonic tumors in rats.
Earlier studies have shown that urolithins with potential cancer chemopreventive effects can reach high concentrations in the colon of animals following ellagitannins intake [98]. Recently, an interesting study based on the pilot trial with colorectal cancer (CRC) patients that needed surgical resection of colon tissues revealed significant levels of ellagic acid derivatives and urolithins found in human colon tissues from CRC patients after consumption of pomegranate extract (PE) [99]. The study was completed by twenty-six patients assigned in two PE groups: PEs with low (PE-1) and high (PE-2) punicalagin:ellagic acid ratios. Both PEs were well tolerated by the patients with no adverse effects, such as dyspepsia, allergic reactions, etc., reported during the study. Free ellagic acid, various ellagic acid conjugates, as well as gallic acid, and up to 12 different urolithins, were found in colon tissues, but no ellagitannins were found. The normal colon tissues had higher levels of individual and total metabolites than in malignant colon tissues. The main urolithins produced were urolithin A or isourolithin A (54% and 46% patients with urolithin A or isourolithin A phenotype). The study found higher urolithin formation after intake of the extract with higher free ellagic acid. Additional studies based on a randomized, double-blind, controlled trial with 35 CRC patients consuming 900 mg PE daily before surgery and control (no PE intake, n = 10) CRC patients, found an in vivo specific modulatory effect of the intake of PEs enriched in ellagic acid or ellagitannins (punicalagin) on specific miRs (miR-646, miR-1249, miR-135b-5p, miR-135b-3p,miR-92b-5p, miR-765, miR-496, miR-181c-3p, and miR-18a-3p) in human malignant and normal colon tissues from CRC patients [61]. The data revealed no direct relationship between the observed changes in expression levels of specific miRs and the presence of ellagic acid derivatives and urolithins in human colon tissues. Although a preliminary in silico analysis showed a potential involvement of the modulated miRs with cancer-related genes and pathways, the significance of these results in relation to cancer prevention is yet to be understood, warranting further research.
8. Conclusions
In the current setting, cancer prevention via dietary agents is quite a promising arena of oncology that has drawn a significant amount of attention from both scientists in basic and clinical sciences and the general masses due to dietary agents’ proven ability to prevent or suppress cancers, their low cost, and easy availability. However, current challenges relate to establishing the key component of these dietetic agents responsible for the anticancer effects and the mechanisms through which they suppress cancer. Accumulating research provides extensive evidence related to biological activities of pomegranate-derived products particularly with respect to their anticancer properties. Studies have suggested the whole pomegranate fruit, as well as its juice and oil, as promising chemopreventive/chemotherapeutic agents, as they exert anti-inflammatory, anti-proliferative, and anti-tumorigenic effects by modulating multiple signaling pathways. More in vitro and in vivo studies are needed to assess the combinatorial effect of pomegranate with other compounds to determine whether additive or synergistic, or even antagonistic, effects are observed. Considerable data demonstrates the in vitro and in vivo efficacy of pomegranate against cancer growth and promotion; however, well-designed human clinical trials are necessary to validate the usefulness of these natural agents either alone or in combination with current therapy for the prevention and treatment of skin, breast, prostate, lung, and colon cancers.
Acknowledgments
This work was supported by the funds from NCI/NIH (CA173043) and ACS-IRG (IRG-60-001-53).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Pogribny, I.P.; Rusyn, I. Environmental toxicants, epigenetics, and cancer. Adv. Exp. Med. Biol. 2013, 754, 215–232. [Google Scholar] [PubMed]
- Minamoto, T.; Mai, M.; Ronai, Z. Environmental factors as regulators and effectors of multistep carcinogenesis. Carcinogenesis 1999, 20, 519–527. [Google Scholar] [CrossRef] [PubMed]
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Afaq, F.; Mukhtar, H. Lifestyle as risk factor for cancer: Evidence from human studies. Cancer Lett. 2010, 293, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2016. CA Cancer J. Clin. 2016, 66, 7–30. [Google Scholar] [CrossRef] [PubMed]
- DiMarco-Crook, C.; Xiao, H. Diet-based strategies for cancer chemoprevention: The role of combination regimens using dietary bioactive components. Annu. Rev. Food Sci. Technol. 2015, 6, 505–526. [Google Scholar] [CrossRef] [PubMed]
- Key, T.J.; Schatzkin, A.; Willett, W.C.; Allen, N.E.; Spencer, E.A.; Travis, R.C. Diet, nutrition and the prevention of cancer. Public Health Nutr. 2004, 7, 187–200. [Google Scholar] [CrossRef] [PubMed]
- Barnard, R.J. Prevention of cancer through lifestyle changes. Evid. Based Complement. Altern. Med. 2004, 1, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the last 25 years. J. Nat. Prod. 2007, 70, 461–477. [Google Scholar] [CrossRef] [PubMed]
- Boeing, H.; Bechthold, A.; Bub, A.; Ellinger, S.; Haller, D.; Kroke, A.; Leschik-Bonnet, E.; Müller, M.J.; Oberritter, H.; Schulze, M.; et al. Critical review: Vegetables and fruit in the prevention of chronic diseases. Eur. J. Nutr. 2012, 51, 637–663. [Google Scholar] [CrossRef] [PubMed]
- Sauvaget, C.; Nagano, J.; Hayashi, M.; Spencer, E.; Shimizu, Y.; Allen, N. Vegetables and fruit intake and cancer mortality in the Hiroshima/Nagasaki Life Span Study. Br. J. Cancer 2003, 88, 689–694. [Google Scholar] [CrossRef] [PubMed]
- Gundgaard, J.; Nielsen, J.N.; Olsen, J.; Sørensen, J. Increased intake of fruit and vegetables: Estimation of impact in terms of life expectancy and healthcare costs. Public Health Nutr. 2003, 6, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.Y.; Cai, Y.Z.; Zhang, Y. Natural phenolic compounds from medicinal herbs and dietary plants: Potential use for cancer prevention. Nutr. Cancer 2010, 62, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Afaq, F.; Mukhtar, H. Cancer chemoprevention through dietary antioxidants: Progress and promise. Antioxid. Redox Signal. 2008, 10, 475–510. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.; Kunnumakkara, A.B.; Sundaram, C.; Harikumar, K.B.; Tharakan, S.T.; Lai, O.S.; Sung, B.; Aggarwal, B.B. Cancer is a preventable disease that requires major lifestyle changes. Pharm. Res. 2008, 25, 2097–2116. [Google Scholar] [CrossRef] [PubMed]
- Jurenka, J.S. Therapeutic applications of pomegranate (Punica granatum L.): A review. Altern. Med. Rev. 2008, 13, 128–144. [Google Scholar] [PubMed]
- Viuda-Martos, M.; Fernández-López, J.; Pérez-Álvarez, J.A. Pomegranate and its many functional components as related to human health: A review. Compr. Rev. Food Sci. Food Saf. 2010, 9, 635–654. [Google Scholar] [CrossRef]
- Aviram, M.; Kaplan, M.; Rosenblat, M.; Fuhrman, B. Dietary antioxidants and paraoxonases against LDL oxidation and atherosclerosis development. Handb. Exp. Pharmacol. 2005, 170, 263–300. [Google Scholar]
- Gil, M.I.; Tomas-Barberan, F.A.; Hess-Pierce, B.; Holcroft, D.M.; Kader, A.A. Antioxidant activity of pomegranate juice and its relationship with phenolic composition and processing. J. Agric. Food Chem. 2000, 48, 4581–4589. [Google Scholar] [CrossRef] [PubMed]
- Caceres, A.; Giron, L.M.; Alvarado, S.R.; Torres, M.F. Screening of antimicrobial activity of plants popularly used in Guatemala for the treatment of dermatomucosal diseases. J. Ethnopharmacol. 1987, 20, 223–237. [Google Scholar] [CrossRef]
- Naqvi, S.A.; Khan, M.S.; Vohora, S.B. Antibacterial, antifungal and anthelmintic studies on Ochrocarpus longifolius. Planta Med. 1976, 29, 98–100. [Google Scholar] [CrossRef] [PubMed]
- Saxena, A.; Vikram, N.K. Role of selected Indian plants in management of type 2 diabetes: A review. J. Altern. Complement. Med. 2004, 10, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Faria, A.; Calhau, C. The bioactivity of pomegranate: Impact on health and disease. Crit. Rev. Food Sci. Nutr. 2011, 51, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Lansky, E.P.; Newman, R.A. Punica granatum (pomegranate) and its potential for prevention and treatment of inflammation and cancer. J. Ethnopharmacol. 2007, 109, 177–206. [Google Scholar] [CrossRef] [PubMed]
- Afaq, F.; Malik, A.; Syed, D.; Maes, D.; Matsui, M.S.; Mukhtar, H. Pomegranate fruit extract modulates UV-B-mediated phosphorylation of mitogen-activated protein kinases and activation of nuclear factor kappa B in normal human epidermal keratinocytes. Photochem. Photobiol. 2005, 81, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Zaid, M.A.; Afaq, F.; Syed, D.N.; Dreher, M.; Mukhtar, H. Inhibition of UVB-mediated oxidative stress and markers of photoaging in immortalized HaCaT keratinocytes by pomegranate polyphenol extract POMx. Photochem. Photobiol. 2007, 83, 882–888. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Syed, D.N.; Pal, H.C.; Mukhtar, H.; Afaq, F. Pomegranate fruit extract inhibits UVB-induced inflammation and proliferation by modulating NF-κB and MAPK signaling pathways in mouse skin. Photochem. Photobiol. 2012, 88, 1126–1134. [Google Scholar] [CrossRef] [PubMed]
- Afaq, F.; Zaid, M.; Khan, N.; Syed, D.N.; Yun, J.-M.; Sarfaraz, S.; Suh, Y.; Mukhtar, H. Inhibitory effect of oral feeding of pomegranate fruit extract on UVB-induced skin carcinogenesis in SKH-1 hairless mice. Proc. Am. Assoc. Cancer Res. 2008, 49, 1246. [Google Scholar]
- Syed, D.N.; Malik, A.; Hadi, N.; Sarfaraz, S.; Afaq, F.; Mukhtar, H. Photochemopreventive effect of pomegranate fruit extract on UVA-mediated activation of cellular pathways in normal human epidermal keratinocytes. Photochem. Photobiol. 2006, 82, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Baccarin, T.; Mitjans, M.; Ramos, D.; Lemos-Senna, E.; Vinardell, M.P. Photoprotection by Punica granatum seed oil nanoemulsion entrapping polyphenol-rich ethyl acetate fraction against UVB-induced DNA damage in human keratinocyte (HaCaT) cell line. J. Photochem. Photobiol. B 2015, 153, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Pacheco-Palencia, L.A.; Noratto, G.; Hingorani, L.; Talcott, S.T.; Mertens-Talcott, S.U. Protective effects of standardized pomegranate (Punica granatum L.) polyphenolic extract in ultraviolet-irradiated human skin fibroblasts. J. Agric. Food Chem. 2008, 56, 8434–8441. [Google Scholar] [CrossRef] [PubMed]
- Afaq, F.; Zaid, M.A.; Khan, N.; Dreher, M.; Mukhtar, H. Protective effect of pomegranate-derived products on UVB-mediated damage in human reconstituted skin. Exp. Dermatol. 2009, 18, 553–561. [Google Scholar] [CrossRef] [PubMed]
- Afaq, F.; Khan, N.; Syed, D.N.; Mukhtar, H. Oral feeding of pomegranate fruit extract inhibits early biomarkers of UVB radiation-induced carcinogenesis in SKH-1 hairless mouse epidermis. Photochem. Photobiol. 2010, 86, 1318–1326. [Google Scholar] [CrossRef] [PubMed]
- Afaq, F.; Saleem, M.; Krueger, C.G.; Reed, J.D.; Mukhtar, H. Anthocyanin- and hydrolyzable tannin-rich pomegranate fruit extract modulates MAPK and NF-kappaB pathways and inhibits skin tumorigenesis in CD-1 mice. Int. J. Cancer 2005, 113, 423–433. [Google Scholar] [CrossRef] [PubMed]
- Hora, J.J.; Maydew, E.R.; Lansky, E.P.; Dwivedi, C. Chemopreventive effects of pomegranate seed oil on skin tumor development in CD1 mice. J. Med. Food 2003, 6, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Adams, L.S.; Zhang, Y.; Seeram, N.P.; Heber, D.; Chen, S. Pomegranate ellagitannin-derived compounds exhibit antiproliferative and antiaromatase activity in breast cancer cells in vitro. Cancer Prev. Res. 2010, 3, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Sreeja, S.; Santhosh Kumar, T.R.; Lakshmi, B.S. Pomegranate extract demonstrate a selective estrogen receptor modulator profile in human tumor cell lines and in vivo models of estrogen deprivation. J. Nutr. Biochem. 2012, 23, 725–732. [Google Scholar] [CrossRef] [PubMed]
- Ocha, A.; Wang, L.; Penichet, M.; Martins-Green, M. Pomegranate juice and specific components inhibit cell and molecular processes critical for metastasis of breast cancer. Breast Cancer Res. Treat. 2012, 136, 647–658. [Google Scholar]
- Grossmann, M.E.; Mizuno, N.K.; Schuster, T.; Cleary, M.P. Punicic acid is an omega-5 fatty acid capable of inhibiting breast cancer proliferation. Int. J. Oncol. 2010, 36, 421–426. [Google Scholar] [PubMed]
- Dikmen, M.; Ozturk, N.; Ozturk, Y. The antioxidant potency of Punica granatum L. Fruit peel reduces cell proliferation and induces apoptosis on breast cancer. J. Med. Food 2011, 14, 1638–1646. [Google Scholar] [CrossRef] [PubMed]
- Costantini, S.; Rusolo, F.; de Vito, V.; Moccia, S.; Picariello, G.; Capone, F.; Guerriero, E.; Castello, G.; Volpe, M.G. Potential anti-inflammatory effects of the hydrophilic fraction of pomegranate (Punica granatum L.) seed oil on breast cancer cell lines. Molecules 2014, 19, 8644–8660. [Google Scholar] [CrossRef] [PubMed]
- Shirode, A.B.; Kovvuru, P.; Chittur, S.V.; Henning, S.M.; Heber, D.; Reliene, R. Antiproliferative effects of pomegranate extraction MCF-7 breast cancer cells are associated with reduced DNA repair gene expression and induction of double strand breaks. Mol. Carcinog. 2014, 53, 458–470. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.S.; Bai, M.H.; Zhang, T.; Li, G.D.; Liu, M. Ellagic acid induces cell cycle arrest and apoptosis through TGF-beta/Smad3 signaling pathway in human breast cancer MCF-7 cells. Int. J. Oncol. 2015, 46, 1730–1738. [Google Scholar] [PubMed]
- Mandal, A.; Bishayee, A. Mechanism of Breast Cancer Preventive Action of Pomegranate: Disruption of Estrogen Receptor and Wnt/beta-Catenin Signaling Pathways. Molecules 2015, 20, 22315–22328. [Google Scholar] [CrossRef] [PubMed]
- Malik, A.; Afaq, F.; Sarfaraz, S.; Adhami, V.M.; Syed, D.N.; Mukhtar, H. Pomegranate fruit juice for chemoprevention and chemotherapy of prostate cancer. Proc. Natl. Acad. Sci. USA 2005, 102, 14813–14818. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, S.; Iqbal, S.; Chen, Z.; Wang, X.; Wang, Y.A.; Liu, D.; Bai, K.; Ritenour, C.; Kucuk, O.; et al. Pomegranate extract inhibits the bone metastatic growth of human prostate cancer cells and enhances the in vivo efficacy of docetaxel chemotherapy. Prostate 2013, 74, 497–508. [Google Scholar] [CrossRef] [PubMed]
- Rettig, M.B.; Heber, D.; An, J.; Seeram, N.P.; Rao, J.Y.; Liu, H.; Klatte, T.; Belldegrun, A.; Moro, A.; Henning, S.M.; et al. Pomegranate extract inhibits androgen-independent prostate cancer growth through a nuclear factor-kappaB-dependent mechanism. Mol. Cancer Ther. 2008, 7, 2662–2671. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.T.; Wu, Y.L.; Chien, L.H.; Chen, S.T.; Tzeng, Y.K.; Wu, T.F. Proteomic exploration of the impacts of pomegranate fruit juice on the global gene expression of prostate cancer cell. Proteomics 2012, 12, 3251–3262. [Google Scholar] [CrossRef] [PubMed]
- Adhami, V.M.; Siddiqui, I.A.; Syed, D.N.; Lall, R.K.; Mukhtar, H. Oral infusion of pomegranate fruit extract inhibits prostate carcinogenesis in the TRAMP model. Carcinogenesis 2012, 33, 644–651. [Google Scholar] [CrossRef] [PubMed]
- Koyama, S.; Cobb, L.J.; Mehta, H.H.; Seeram, N.P.; Heber, D.; Pantuck, A.J.; Cohen, P. Pomegranate extract induces apoptosis in human prostate cancer cells by modulation of the IGF-IGFBP axis. Growth Horm. IGF Res. 2010, 20, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Hong, M.Y.; Seeram, N.P.; Heber, D. Pomegranate polyphenols down-regulate expression of androgen-synthesizing genes in human prostate cancer cells overexpressing the androgen receptor. J. Nutr. Biochem. 2008, 19, 848–855. [Google Scholar] [CrossRef] [PubMed]
- Kasimsetty, S.G.; Bialonska, D.; Reddy, M.K.; Thornton, C.; Willett, K.L.; Ferreira, D. Effects of pomegranate chemical constituents/intestinal microbial metabolites on CYP1B1 in 22Rv1 prostate cancer cells. J. Agric. Food Chem. 2009, 57, 10636–10644. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Hadi, N.; Afaq, F.; Syed, D.N.; Kweon, M.H.; Mukhtar, H. Pomegranate fruit extract inhibits prosurvival pathways in human A549 lung carcinoma cells and tumor growth in athymic nude mice. Carcinogenesis 2007, 28, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Zahin, M.; Ahmad, I.; Gupta, R.C.; Aqil, F. Punicalagin and ellagic acid demonstrate antimutagenic activity and inhibition of benzo[a]pyrene induced DNA adducts. Biomed. Res. Int. 2014, 2014, 467465. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Afaq, F.; Kweon, M.H.; Kim, K.; Mukhtar, H. Oral consumption of pomegranate fruit extract inhibits growth and progression of primary lung tumors in mice. Cancer Res. 2007, 67, 3475–3482. [Google Scholar] [CrossRef] [PubMed]
- Bachoual, R.; Talmoudi, W.; Boussetta, T.; Braut, F.; El-Benna, J. An aqueous pomegranate peel extract inhibits neutrophil myeloperoxidase in vitro and attenuates lung inflammation in mice. Food Chem. Toxicol. 2011, 49, 1224–1228. [Google Scholar] [CrossRef] [PubMed]
- Husari, A.; Khayat, A.; Bitar, H.; Hashem, Y.; Rizkallah, A.; Zaatari, G.; El Sabban, M. Antioxidant activity of pomegranate juice reduces acute lung injury secondary to hyperoxia in an animal model. BMC Res. Notes 2014, 7, 664. [Google Scholar] [CrossRef] [PubMed]
- Selimoglu Sen, H.; Sen, V.; Bozkurt, M.; Turkcu, G.; Guzel, A.; Sezgi, C.; Abakay, O.; Kaplan, I. Carvacrol and pomegranate extract in treating methotrexate-induced lung oxidative injury in rats. Med. Sci. Monit. 2014, 20, 1983–1990. [Google Scholar] [PubMed]
- Adams, L.S.; Seeram, N.P.; Aggarwal, B.B.; Takada, Y.; Sand, D.; Heber, D. Pomegranate juice, total pomegranate ellagitannins, and punicalagin suppress inflammatory cell signaling in colon cancer cells. J. Agric. Food Chem. 2006, 54, 980–985. [Google Scholar] [CrossRef] [PubMed]
- Boateng, J.; Verghese, M.; Shackelford, L.; Walker, L.T.; Khatiwada, J.; Ogutu, S.; Williams, D.S.; Jones, J.; Guyton, M.; Asiamah, D.; et al. Selected fruits reduce azoxymethane (AOM)-induced aberrant crypt foci (ACF) in Fisher 344 male rats. Food Chem. Toxicol. 2007, 45, 725–732. [Google Scholar] [CrossRef] [PubMed]
- Nunez-Sanchez, M.A.; Davalos, A.; Gonzalez-Sarrias, A.; Casas-Agustench, P.; Visioli, F.; Monedero-Saiz, T.; Garcia-Talavera, N.V.; Gomez-Sanchez, M.B.; Sanchez-Alvarez, C.; Garcia-Albert, A.M.; et al. MicroRNAs expression in normal and malignant colon tissues as biomarkers of colorectal cancer and in response to pomegranate extracts consumption: Critical issues to discern between modulatory effects and potential artefacts. Mol. Nutr. Food Res. 2015, 59, 1973–1986. [Google Scholar] [CrossRef] [PubMed]
- Elfalleh, W.; Hannachi, H.; Tlili, N.; Yahia, Y.; Nasri, N.; Ferchichi, A. Total phenolic contents and antioxidant activities of pomegranate peel, seed, leaf and flower. J. Med. Plants Res. 2012, 6, 4724–4730. [Google Scholar] [CrossRef]
- Viladomiu, M.; Hontecillas, R.; Lu, P.; Bassaganya-Riera, J. Preventive and prophylactic mechanisms of action of pomegranate bioactive constituents. Evid. Based Complement. Altern. Med. 2013, 2013, 789764. [Google Scholar] [CrossRef] [PubMed]
- Kohno, H.; Suzuki, R.; Yasui, Y.; Hosokawa, M.; Miyashita, K.; Tanaka, T. Pomegranate seed oil rich in conjugated linolenic acid suppresses chemically induced colon carcinogenesis in rats. Cancer Sci. 2004, 95, 481–486. [Google Scholar] [CrossRef] [PubMed]
- Paladini, A.C.; Marder, M.; Viola, H.; Wolfman, C.; Wasowski, C.; Medina, J.H. Flavonoids and the central nervous system: From forgotten factors to potent anxiolytic compounds. J. Pharm. Pharmacol. 1999, 51, 519–526. [Google Scholar] [CrossRef] [PubMed]
- Zand, R.S.; Jenkins, D.J.; Diamandis, E.P. Steroid hormone activity of flavonoids and related compounds. Breast Cancer Res. Treat. 2000, 62, 35–49. [Google Scholar] [CrossRef] [PubMed]
- Munde, S.S.; Patil, V.K.; Chavan, S.D. Standardization of leaf sampling procedure in pomegranate (Punica granatum Linn.). J. Maharashtra Agric. Univ. 1980, 5, 90–91. [Google Scholar]
- Munde, S.S.; Patil, V.K.; Chavan, S.D. Chemical composition of pomegranate (Punica granatum L.) leaves sampled during different stages of crop. Food Farming Agric. 1981, 13, 177–181. [Google Scholar]
- Afaq, F. Natural agents: Cellular and molecular mechanisms of photoprotection. Arch. Biochem. Biophys. 2011, 508, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Afaq, F.; Katiyar, S.K. Polyphenols: Skin photoprotection and inhibition of photocarcinogenesis. Mini Rev. Med. Chem. 2011, 11, 1200–1215. [Google Scholar] [CrossRef] [PubMed]
- George, J.; Singh, M.; Srivastava, A.K.; Bhui, K.; Shukla, Y. Synergistic growth inhibition of mouse skin tumors by pomegranate fruit extract and diallyl sulfide: Evidence for inhibition of activated MAPKs/NF-kappaB and reduced cell proliferation. Food Chem. Toxicol. 2011, 49, 1511–1520. [Google Scholar] [CrossRef] [PubMed]
- Eliassen, A.H.; Hankinson, S.E. Endogenous hormone levels and risk of breast, endometrial and ovarian cancers: Prospective studies. Adv. Exp. Med. Biol. 2008, 630, 148–165. [Google Scholar] [PubMed]
- Sturgeon, S.R.; Ronnenberg, A.G. Pomegranate and breast cancer: Possible mechanisms of prevention. Nutr. Rev. 2010, 68, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.D.; Mehta, R.; Yu, W.; Neeman, I.; Livney, T.; Amichay, A.; Poirier, D.; Nicholls, P.; Kirby, A.; Jiang, W.; et al. Chemopreventive and adjuvant therapeutic potential of pomegranate (Punica granatum) for human breast cancer. Breast Cancer Res. Treat. 2002, 71, 203–217. [Google Scholar] [CrossRef] [PubMed]
- De Caestecker, M.P.; Piek, E.; Roberts, A.B. Role of transforming growth factor-beta signaling in cancer. J. Natl. Cancer Inst. 2000, 92, 1388–1402. [Google Scholar] [CrossRef] [PubMed]
- Moustakas, A.; Souchelnytskyi, S.; Heldin, C.H. Smad regulation in TGF-beta signal transduction. J. Cell Sci. 2001, 114, 4359–4369. [Google Scholar] [PubMed]
- Bishayee, A.; Mandal, A.; Bhattacharyya, P.; Bhatia, D. Pomegranate exerts chemoprevention of experimentally induced mammary tumorigenesis by suppression of cell proliferation and induction of apoptosis. Nutr. Cancer 2016, 68, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Lansky, E.P.; Harrison, G.; Froom, P.; Jiang, W.G. Pomegranate (Punica granatum) pure chemicals show possible synergistic inhibition of human PC-3 prostate cancer cell invasion across Matrigel. Investig. New Drugs 2005, 23, 121–122. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, M.; Jiang, W.; Kumi-Diaka, J.; Lansky, E.P.; Gommersall, L.M.; Patel, A.; Mansel, R.E.; Neeman, I.; Geldof, A.A.; Campbell, M.J. Pomegranate extracts potently suppress proliferation, xenograft growth, and invasion of human prostate cancer cells. J. Med. Food 2004, 7, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Naiki-Ito, A.; Chewonarin, T.; Tang, M.; Pitchakarn, P.; Kuno, T.; Ogawa, K.; Asamoto, M.; Shirai, T.; Takahashi, S. Ellagic acid, a component of pomegranate fruit juice, suppresses androgen-dependent prostate carcinogenesis via induction of apoptosis. Prostate 2015, 75, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Tao, Y.; Pinzi, V.; Bourhis, J.; Deutsch, E. Mechanisms of disease: Signaling of the insulin-like growth factor 1 receptor pathway--therapeutic perspectives in cancer. Nat. Clin. Pract. Oncol. 2007, 4, 591–602. [Google Scholar] [CrossRef] [PubMed]
- Klein, K.A.; Reiter, R.E.; Redula, J.; Moradi, H.; Zhu, X.L.; Brothman, A.R.; Lamb, D.J.; Marcelli, M.; Belldegrun, A.; Witte, O.N.; et al. Progression of metastatic human prostate cancer to androgen independence in immunodeficient SCID mice. Nat. Med. 1997, 3, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Cicek, M.S.; Liu, X.; Casey, G.; Witte, J.S. Role of androgen metabolism genes CYP1B1, PSA/KLK3, and CYP11alpha in prostate cancer risk and aggressiveness. Cancer Epidemiol. Biomark. Prev. 2005, 14, 2173–2177. [Google Scholar] [CrossRef] [PubMed]
- González-Sarrías, A.; Giménez-Bastida, J.A.; García-Conesa, M.T.; Gómez-Sánchez, M.B.; García-Talavera, N.V.; Gil-Izquierdo, A.; Sánchez-Alvarez, C.; Fontana-Compiano, L.O.; Morga-Egea, J.P.; Pastor-Quirante, F.A.; et al. Occurrence of urolithins, gut microbiota ellagic acid metabolites and proliferation markers expression response in the human prostate gland upon consumption of walnuts and pomegranate juice. Mol. Nutr. Food Res. 2010, 54, 311–322. [Google Scholar] [CrossRef] [PubMed]
- Pantuck, A.J.; Leppert, J.T.; Zomorodian, N.; Aronson, W.; Hong, J.; Barnard, R.J.; Seeram, N.; Liker, H.; Wang, H.; Elashoff, R.; et al. Phase II study of pomegranate juice for men with rising prostate-specific antigen following surgery or radiation for prostate cancer. Clin. Cancer Res. 2006, 12, 4018–4026. [Google Scholar] [CrossRef] [PubMed]
- Paller, C.J.; Ye, X.; Wozniak, P.J.; Gillespie, B.K.; Sieber, P.R.; Greengold, R.H.; Stockton, B.R.; Hertzman, B.L.; Efros, M.D.; Roper, R.P.; et al. A randomized phase II study of pomegranate extract for men with rising PSA following initial therapy for localized prostate cancer. Prostate Cancer Prostatic Dis. 2013, 16, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Pantuck, A.J.; Pettaway, C.A.; Dreicer, R.; Corman, J.; Katz, A.; Ho, A.; Aronson, W.; Clark, W.; Simmons, G.; Heber, D. A randomized, double-blind, placebo-controlled study of the effects of pomegranate extract on rising PSA levels in men following primary therapy for prostate cancer. Prostate Cancer Prostatic Dis. 2015, 18, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Freedland, S.J.; Carducci, M.; Kroeger, N.; Partin, A.; Rao, J.Y.; Jin, Y.; Kerkoutian, S.; Wu, H.; Li, Y.; Creel, P.; et al. A double-blind, randomized, neoadjuvant study of the tissue effects of POMx pills in men with prostate cancer before radical prostatectomy. Cancer Prev. Res. 2013, 6, 1120–1127. [Google Scholar] [CrossRef] [PubMed]
- Stenner-Liewen, F.; Liewen, H.; Cathomas, R.; Renner, C.; Petrausch, U.; Sulser, T.; Spanaus, K.; Seifert, H.H.; Strebel, R.T.; Knuth, A.; et al. Daily Pomegranate Intake Has No Impact on PSA Levels in Patients with Advanced Prostate Cancer—Results of a Phase IIb Randomized Controlled Trial. J. Cancer 2013, 4, 597–605. [Google Scholar] [CrossRef] [PubMed]
- Aqil, F.; Munagala, R.; Vadhanam, M.V.; Kausar, H.; Jeyabalan, J.; Schultz, D.J.; Gupta, R.C. Anti-proliferative activity and protection against oxidative DNA damage by punicalagin isolated from pomegranate husk. Food Res. Int. 2012, 49, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Modaeinama, S.; Abasi, M.; Abbasi, M.M.; Jahanban-Esfahlan, R. Anti Tumoral Properties of Punica Granatum (Pomegranate) Peel Extract on Different Human Cancer Cells. Asian Pac. J. Cancer Prev. 2015, 16, 5697–5701. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yang, F.; Zheng, W.; Hu, M.; Wang, J.; Ma, S.; Deng, Y.; Luo, Y.; Ye, T.; Yin, W. Punica granatum (pomegranate) leaves extract induces apoptosis through mitochondrial intrinsic pathway and inhibits migration and invasion in non-small cell lung cancer in vitro. Biomed. Pharmacother. 2016, 80, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Gosse, F.; Guyot, S.; Roussi, S.; Lobstein, A.; Fischer, B.; Seiler, N.; Raul, F. Chemopreventive properties of apple procyanidins on human colon cancer-derived metastatic SW620 cells and in a rat model of colon carcinogenesis. Carcinogenesis 2005, 26, 1291–1295. [Google Scholar] [CrossRef] [PubMed]
- Ramos, S. Effects of dietary flavonoids on apoptotic pathways related to cancer chemoprevention. J. Nutr. Biochem. 2007, 18, 427–442. [Google Scholar] [CrossRef] [PubMed]
- Bobe, G.; Albert, P.S.; Sansbury, L.B.; Lanza, E.; Schatzkin, A.; Colburn, N.H.; Cross, A.J. Interleukin-6 as a potential indicator for prevention of high-risk adenoma recurrence by dietary flavonols in the polyp prevention trial. Cancer Prev. Res. 2010, 3, 764–775. [Google Scholar] [CrossRef] [PubMed]
- Seeram, N.P.; Adams, L.S.; Henning, S.M.; Niu, Y.; Zhang, Y.; Nair, M.G.; Heber, D. In vitro antiproliferative, apoptotic and antioxidant activities of punicalagin, ellagic acid and a total pomegranate tannin extract are enhanced in combination with other polyphenols as found in pomegranate juice. J. Nutr. Biochem. 2005, 16, 360–367. [Google Scholar] [CrossRef] [PubMed]
- Larrosa, M.; Tomas-Barberan, F.A.; Espin, J.C. The dietary hydrolysable tannin punicalagin releases ellagic acid that induces apoptosis in human colon adenocarcinoma Caco-2 cells by using the mitochondrial pathway. J. Nutr. Biochem. 2006, 17, 611–625. [Google Scholar] [CrossRef] [PubMed]
- Espin, J.C.; Larrosa, M.; Garcia-Conesa, M.T.; Tomas-Barberan, F. Biological significance of urolithins, the gut microbial ellagic Acid-derived metabolites: The evidence so far. Evid. Based Complement. Altern. Med. 2013, 2013, 270418. [Google Scholar] [CrossRef] [PubMed]
- Nunez-Sanchez, M.A.; Garcia-Villalba, R.; Monedero-Saiz, T.; Garcia-Talavera, N.V.; Gomez-Sanchez, M.B.; Sanchez-Alvarez, C.; Garcia-Albert, A.M.; Rodriguez-Gil, F.J.; Ruiz-Marin, M.; Pastor-Quirante, F.A.; et al. Targeted metabolic profiling of pomegranate polyphenols and urolithins in plasma, urine and colon tissues from colorectal cancer patients. Mol. Nutr. Food Res. 2014, 58, 1199–1211. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).