Separation of Alkyne Enantiomers by Chiral Column HPLC Analysis of Their Cobalt-Complexes
Abstract
:1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. General Methods
3.2. Preparation of Alkynes and Compound Characterization
3.3. General Precedure for Preparation of Co2(CO)6–Alkyne Complexes and Compound Characterization
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Trost, B.M.; Li, C.-J. Modern Alkyne Chemistry: Catalytic and Atom-Economic Transformations, 1st ed.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2015. [Google Scholar]
- Trost, B.M.; Masters, J.T. Transition metal-catalyzed couplings of alkynes to 1,3-enynes: Modern methods and synthetic applications. Chem. Soc. Rev. 2016, 45, 2212–2238. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Shi, L.L. Exploring the complexity-generating features of the Pauson–Khand reaction from a synthetic perspective. Eur. J. Org. Chem. 2016, 14, 2356–2368. [Google Scholar]
- Kacprzak, K.; Skiera, I.; Piasecka, M.; Paryzek, Z. Alkaloids and isoprenoids modification by copper(I)-catalyzed Huisgen 1,3-dipolar cycloaddition (click chemistry): Toward new functions and molecular architectures. Chem. Rev. 2016, 116, 2767–2770. [Google Scholar] [CrossRef] [PubMed]
- Nagle, D.G.; Geralds, R.-S.; Yoo, H.-D.; Gerwick, W.H.; Kim, T.-S.; Nambu, M.; White, J.D. Absolute configuration of curacin A, a novel antimitotic agent from the tropical marine cyanobacterium Lyngbya majuscula. Tetrahedron Lett. 1995, 36, 1189–1192. [Google Scholar] [CrossRef]
- White, J.D.; Kim, T.-S.; Nambu, M. Synthesis of curacin A: A powerful antimitotic from the cyanobacterium Lyngbya majuscule. J. Am. Chem. Soc. 1995, 117, 5612–5613. [Google Scholar] [CrossRef]
- White, J.D.; Kim, T.-S.; Nambu, M. Absolute configuration and total synthesis of (+)-curacin A, an antiproliferative agent from the cyanobacterium Lyngbya majuscula. J. Am. Chem. Soc. 1997, 119, 103–111. [Google Scholar] [CrossRef]
- Codesido, E.M.; Cid, M.M.; Castedo, L.; Mouriño, A.; Granja, J.R. Synthesis of vitamin D analogues with a 2-hydroxy-3-deoxy ring A. Tetrahedron Lett. 2000, 41, 5861–5864. [Google Scholar] [CrossRef]
- García-Fandiño, R.; Aldegunde, M.J.; Codesido, E.M.; Castedo, L.; Granja, J.R. RCM for the construction of novel steroid-like polycyclic systems. 1. Studies on the Synthesis of a PreD3-D3 transition state analogue. J. Org. Chem. 2005, 70, 8281–8290. [Google Scholar] [CrossRef] [PubMed]
- Nicolaou, K.C.; Leung, G.Y.C.; Dethe, D.H.; Guduru, R.; Sun, Y.-P.; Lim, C.S.; Chen, D.Y.-K. Chemical synthesis and biological evaluation of palmerolide A analogues. J. Am. Chem. Soc. 2008, 130, 10019–10023. [Google Scholar] [CrossRef] [PubMed]
- Ngai, M.H.; Yang, P.-Y.; Liu, K.; Shen, Y.; Wenk, M.R.; Yao, S.Q.; Lear, M.J. Click-based synthesis and proteomic profiling of lipstatin analogues. Chem. Commun. 2010, 46, 8335–8337. [Google Scholar] [CrossRef] [PubMed]
- Shibata, H.; Tsuchikawa, H.; Hayashi, T.; Matsumori, N.; Murata, M.; Usui, T. Modification of bafilomycin structure to efficiently synthesize solid-state NMR probes that selectively bind to vacuolar-type ATPase. Chem. Asian J. 2015, 10, 915–924. [Google Scholar] [CrossRef] [PubMed]
- Iseki, K.; Mizuno, S.; Kuroki, Y.; Kobayashi, Y. A chiral formamide: Design and application to catalytic asymmetric synthesis. Tetrahedron Lett. 1998, 39, 2767–2770. [Google Scholar] [CrossRef]
- Seco, J.M.; Quinoa, E.; Riguera, R. The assignment of absolute configuration by NMR. Chem. Rev. 2004, 104, 17–118. [Google Scholar] [CrossRef]
- Miyamoto, H.; Hirano, T.; Okawa, Y.; Nakazaki, A.; Kobayashi, S. Stereoselective synthesis of spirocyclic oxindoles based on a one-pot Ullmann coupling/Claisen rearrangement and its application to the synthesis of a hexahydropyrrolo[2,3-b]indole alkaloid. Tetrahedron 2013, 69, 9481–9493. [Google Scholar] [CrossRef]
- Wang, P.; Lee, H.K. Recent applications of high-performance liquid chromatography to the analysis of metal complexes. J. Chromatogr. A 1997, 789, 437–451. [Google Scholar] [CrossRef]
- Nicholas, K.M.; Pettit, R. An alkyne protecting group. Tetrahedron Lett. 1971, 12, 9481–9493. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 2a–k are available from the authors.


| Alkyne (1) | Co-Complex (2) | HPLC Chart | HPLC Conditions (CHIRALPAK–IB Column 350 nm, 25 °C) |
|---|---|---|---|
 |  |  | 2-PrOH/n-hexane = 0.4:99.6 1 mL·min−1 |
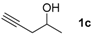 |  |  | 2-PrOH/n-hexane = 0.4:99.6 1 mL·min−1 |
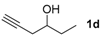 |  |  | 2-PrOH/n-hexane = 0.4:99.6 1 mL·min−1 |
 | 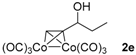 |  | 2-PrOH/n-hexane = 0.4:99.6 1 mL·min−1 |
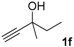 |  | 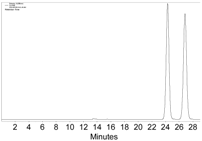 | 2-PrOH/n-hexane = 0.4:99.6 1 mL·min−1 a |
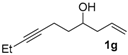 | 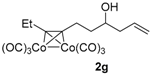 |  | 2-PrOH/n-hexane = 0.5:99.5 1 mL·min−1 |
 |  |  | 2-PrOH/n-hexane = 0.3:99.7 1 mL·min−1 |
 |  |  | EtOAc/n-hexane = 0.05:99.95 0.3 mL·min−1 b |
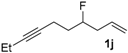 |  | 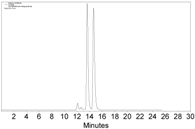 | CH2Cl2/n-hexane = 0.05:99.95 0.4 mL·min−1 c |
 |  |  | 2-PrOH/n-hexane = 0.1:99.9 1 mL·min−1 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Q.; Wang, J.; Li, J.; Wang, X.; Lu, S.; Li, X.; Gong, Y.; Xu, S. Separation of Alkyne Enantiomers by Chiral Column HPLC Analysis of Their Cobalt-Complexes. Molecules 2017, 22, 466. https://doi.org/10.3390/molecules22030466
Liu Q, Wang J, Li J, Wang X, Lu S, Li X, Gong Y, Xu S. Separation of Alkyne Enantiomers by Chiral Column HPLC Analysis of Their Cobalt-Complexes. Molecules. 2017; 22(3):466. https://doi.org/10.3390/molecules22030466
Chicago/Turabian StyleLiu, Qiaoyun, Jing Wang, Junfei Li, Xiaolei Wang, Shichao Lu, Xuan Li, Yaling Gong, and Shu Xu. 2017. "Separation of Alkyne Enantiomers by Chiral Column HPLC Analysis of Their Cobalt-Complexes" Molecules 22, no. 3: 466. https://doi.org/10.3390/molecules22030466
APA StyleLiu, Q., Wang, J., Li, J., Wang, X., Lu, S., Li, X., Gong, Y., & Xu, S. (2017). Separation of Alkyne Enantiomers by Chiral Column HPLC Analysis of Their Cobalt-Complexes. Molecules, 22(3), 466. https://doi.org/10.3390/molecules22030466






