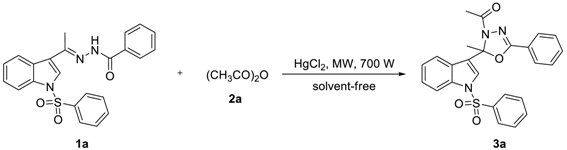
Abstract
A series of 2-alkyl-2-(N-arylsulfonylindol-3-yl)-3-N-acyl-5-aryl-1,3,4-oxadiazolines were expeditious prepared under microwave-assisted, catalyzed by HgCl2 and solvent-free conditions. This method has the advantage of low catalyst loading and recovering catalyst, ease reaction and repaid reaction times, easy separation products and excellent yields, and more conducive to the large-scale synthesis products. Furthermore, compounds 3s, 3y, 3a′, 3b′, 3f′, 3i′, 3q′, and 3r′ exhibited more potent anti-HIV-1 activity with EC50 values of 3.35, 6.12, 3.63, 9.54, 1.79, 0.51, 3.00, and 4.01 μg/mL, and TI values of 32.66, >32.68, 31.22, 13.94, 24.27, 39.59, 26.01, and 24.51, respectively. Especially compound 3i′ displayed the highest anti-HIV-1 activity with TI values of 39.59.
1. Introduction
Acquired immunodeficiency syndrome (AIDS) is mainly caused by human immunodeficiency virus type 1 (HIV-1) infection and has remained one of the most difficult medical barriers for human health since it was first reported in 1981 [1]. The reverse transcriptase (RT) of the HIV-1 plays a significant role in the viral replication process, which makes it a pivotal target for anti-HIV-1 inhibitor discovery [2,3]. Although numerous RT inhibitors, including primarily the nucleoside/nucleotide RT inhibitors (NRTIs) and non-nucleoside RT inhibitors (NNRTIs), have been developed, like other anti-HIV inhibitors, effectiveness of now approved NRTIs and NNRTIs have been hampered because of the fast development of resistance [4,5,6,7]. It is estimated that 36.9 million people (including 2.6 million children) were living with HIV infection in the year 2014 according to UNAIDS-2015 report, and 1.2 million people died due to HIV as well as related diseases [8]. To circumvent this challenge, it is urgent to discover and develop safe, green, efficient, selective, and novel anti-HIV inhibitors having significant potency against drug-resistant RT viral strains as well as less toxicity [8,9,10,11].
To the best of our knowledge, 1,3,4-oxadiazolines are excellent candidates for the research of anti-HIV-1 agents, antibacterial agents, antitumor agents, and chitin biosynthesis inhibitors [12,13,14,15]. As our ongoing work on research of anti-HIV-1 inhibitors, 2-alkyl-2-(N-arylsulfonylindol-3-yl)-3-N-acyl-5-aryl-1,3,4-oxadiazoline derivatives were further prepared through a modified route.
Generally, there are three ways to synthesize 1,3,4-oxadiazolines. The first kind is the traditional procedure, which is usually require excess of anhydride, and long reaction time [16,17,18]. The second is the ultrasonic irradiation assisted synthesis, it is with relatively mild reaction conditions and higher yield [19]. The third is the microwave irradiation assisted synthesis, which are under solvent-free conditions with short reaction time [15]. Although many advantages, there need to be further improved on the methods of the ultrasonic irradiation and microwave irradiation assisted synthesis of 1,3,4-oxadiazolines. On the other hand, since the first report of microwave irradiation assisted synthesis in 1986 [20,21], the technique has been accepted as a method for reducing reaction times and increasing yields of product compared to conventional procedure [22,23,24,25,26]. Especially, the use of microwave ovens as tools for synthetic chemistry is a fast growth area [27,28,29,30,31,32], so here we aimed to use this method to synthetize 2-alkyl-2-(N-arylsulfonylindol-3-yl)-3-N-acyl-5-aryl-1,3,4-oxadiazolines catalyzed by HgCl2 under solvent-free conditions.
2. Results and Discussion
2.1. Chemistry
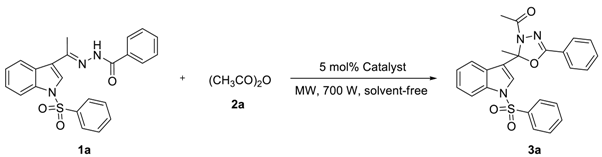
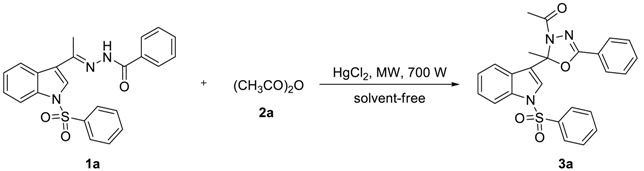
To find out the most compatible reaction conditions for synthesizing 2-alkyl-2-(N-arylsulfonylindol-3-yl)-3-N-acyl-5-aryl-1,3,4-oxadiazolines, the reaction of N-benzenesulfonyl-3-acetylindole benzoyl hydrazone (1a) with acetic anhydride (2a) under microwave irradiation and solvent-free conditions was investigated, and a wide range of reaction catalysts were also tested. As can be clearly seen in Table 1, the cyclization reaction catalyzed by MgCl2.6H2O as a Lewis acid catalyst was found to be sluggish at best condition, providing 3a in 38% yield and recycling 1a in 37% yield after 10 × 3 min following purification by preparative thin-layer chromatography (entry 1). The reaction was accelerated dramatically by the presence of ZnCl2 providing the product 3a in 48% yield and recycling the raw material 1a in 23% yield after 10 × 3 min (entry 2). Although these experiments were not so efficient, the use of solvent-free reaction conditions does have some intrinsic ecological and chemical value. Inspired by these results, Lewis acid catalyst (entry 3) was found to be greatly enhancing the reaction in AlCl3 catalyzed, which providing 3a in 53% yield after 10 × 3 min following purification on silica. Similarly, reactions were improved by the presence of SnCl2.2H2O (entry 4) and FeCl3 (entry 5) providing the product 3a in 63% and 68% yield, respectively. However, the optimum conditions for this transformation employed HgCl2 as a Lewis acid catalyst, providing 3a in 91% yield after 10 × 3 min (entry 6).

Table 1.
Screening of the catalyst for the cyclization reaction between 1a and 2a.
In order to further optimize the reaction conditions, a wide range of reaction parameters were tested by altering the amount of 2a and catalyst as well as the reaction time in a test reaction of 1a and 2a (Table 2).

Table 2.
Optimization of the reaction conditions.
As can be clearly seen in Table 2, when 0.5 mmol of 1a and 2.5 mmol of 2a reacted by HgCl2 catalyst at the amount of 5 mol% under microwave irradiation, 3a was obtained in a 97% yield after 10 × 2 min (entry 1). When the amount of 2a reduced to 2.0 mmol, 3a was obtained in a 90% yield for 10 × 2 min (entry 2). Moreover, when the amount of 2a decreased to 1.5 mmol, 3a was obtained in a 91% yield if the reaction time was prolonged to 10 × 3 min (entry 3). Nevertheless, if we keep reducing the amount of 2a to 1.0 mmol, only providing 3a in 41% yield and recycling 1a in 55% yield after 10 × 4 min following purification by preparative thin-layer chromatography (entry 4). However, the cyclization reaction was terminated by the absence of HgCl2 even if the reaction time was prolonged to 10 × 12 min and the amount of 2a was increased to 2.5 mmol (entry 5). Inspired by these phenomena, we realized that the amount of HgCl2 as a Lewis acid catalyst was significant for enhancing the cyclization reaction. For example, when 0.5 mmol of 1a and 2.5 mmol of 2a reacted by HgCl2 catalyst at the amount of 2.5 mol% under microwave irradiation, 3a was synthesized in a 69% yield after 10 × 5 min (entry 6). When the amount of HgCl2 increased to 3.0 mol%, 3a was prepared in a 78% yield for 10 × 3 min (entry 7). To our delight, when the amount of HgCl2 increased to 4.0 mol%, the cyclization reaction was accelerated dramatically providing the product 3a in 91% yield after 10 × 3 min (entry 8). We concluded that when 0.5 mmol of 1a and 1.5 mmol of 2a reacted by HgCl2 catalyst at the amount of 4 mol% under microwave irradiation, 3a was smoothly synthesized in a 90% yield after 10 × 3 min (entry 9).
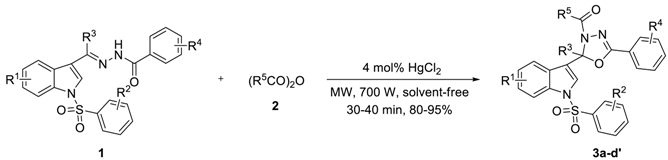
Based upon the above results, a wide range of N-arylsulfonyl-3-acylindole arylcarbonyl hydrazones (1, R1 = H, 6-Me, 5-CN or 5-NO2; R2 = H, p-Me, p-Cl, m-NO2 or p-OMe; R3 = Me, Et or n-Pr; R4 = H or m-Me) with anhydrides (2, R5 = Me or Et) was investigated to explore the scope of the reaction. As outlined in Table 3, 2-alkyl-2-(N-arylsulfonylindol-3-yl)-3-N-acyl-5-aryl-1,3,4-oxadiazolines (3a–d′) were prepared in 80–95% yields for 10 × 3–10 × 4 min (Of course, the reaction can also be carried out directly in one go for 30 or 40 min, with little impact on yield). The steric and electronic effects of substituents of 1 to the reaction were not very obvious.

Table 3.
Synthesis of 2-alkyl-2-(N-arylsulfonylindol-3-yl)-3-N-acyl-5-aryl-1,3,4-oxadiazolines (3a–d′) a.
We have previously developed an efficient method for the synthesis of 2-monosubstituted 3-N-acyl-5-phenyl-1,3,4-oxadiazolines 3e′–r′ under ultrasonic irradiation [19], however, under the above reaction conditions, 2,2-disubstituted 3-N-acyl-5-phenyl-1,3,4-oxadiazolines were not obtained at all even if the reaction time was prolonged. So we have previously described another a convenient, rapid, and high-yielding reaction for the synthesis of 2,2-disubstituted 3-N-acyl-5-phenyl-1,3,4-oxadiazolines 3a–w under microwave irradiation and solvent-free conditions [15]. On the other hand, compared with the previous procedure, the present methodology has the advantages of low catalyst loading and recovering catalyst, short reaction and repaid reaction times, easy separation products, and increased reaction yields (except 3i, 3j, 3o, 3r, 3t, 3u, and 3w), and more conducive to the large-scale synthesis products.
In order to further study the cyclization reaction, we have done the following study. As illustrated in Table 4, when the cyclization reaction of 1a (3 mmol) with 2a (9 mmol) was complete, the reaction mixture was cooled to room temperature. Then the mixture was filtered and the filter residue was washed with dichloromethane (3 × 10 mL). The HgCl2 catalyst was reused directly in the next reaction. For examples, when 3 mmol of 1a and 9 mmol of 2a reacted by HgCl2 catalyst at the amount of 4 mol% under microwave irradiation, 3a was smoothly synthesized in 98% yield after 5 min for the first time (entry 1). When the reaction time prolonged to 6 min, 3a was obtained in 98% yield for recovering HgCl2 catalyst was reused directly in the reaction (entry 2). Entries 3 and 4, 3a was obtained in 97% and 98% yield, respectively.

Table 4.
Typical procedure for recovering HgCl2 catalyst.
To our delight, in the case of reaction amplification, it was found that HgCl2 had better catalytic effect on the reaction. It may be that HgCl2 induces the cyclization reaction in which the turnover frequency is accelerated, and the yield of compound 3a increased significantly.
2.2. Biological Activities
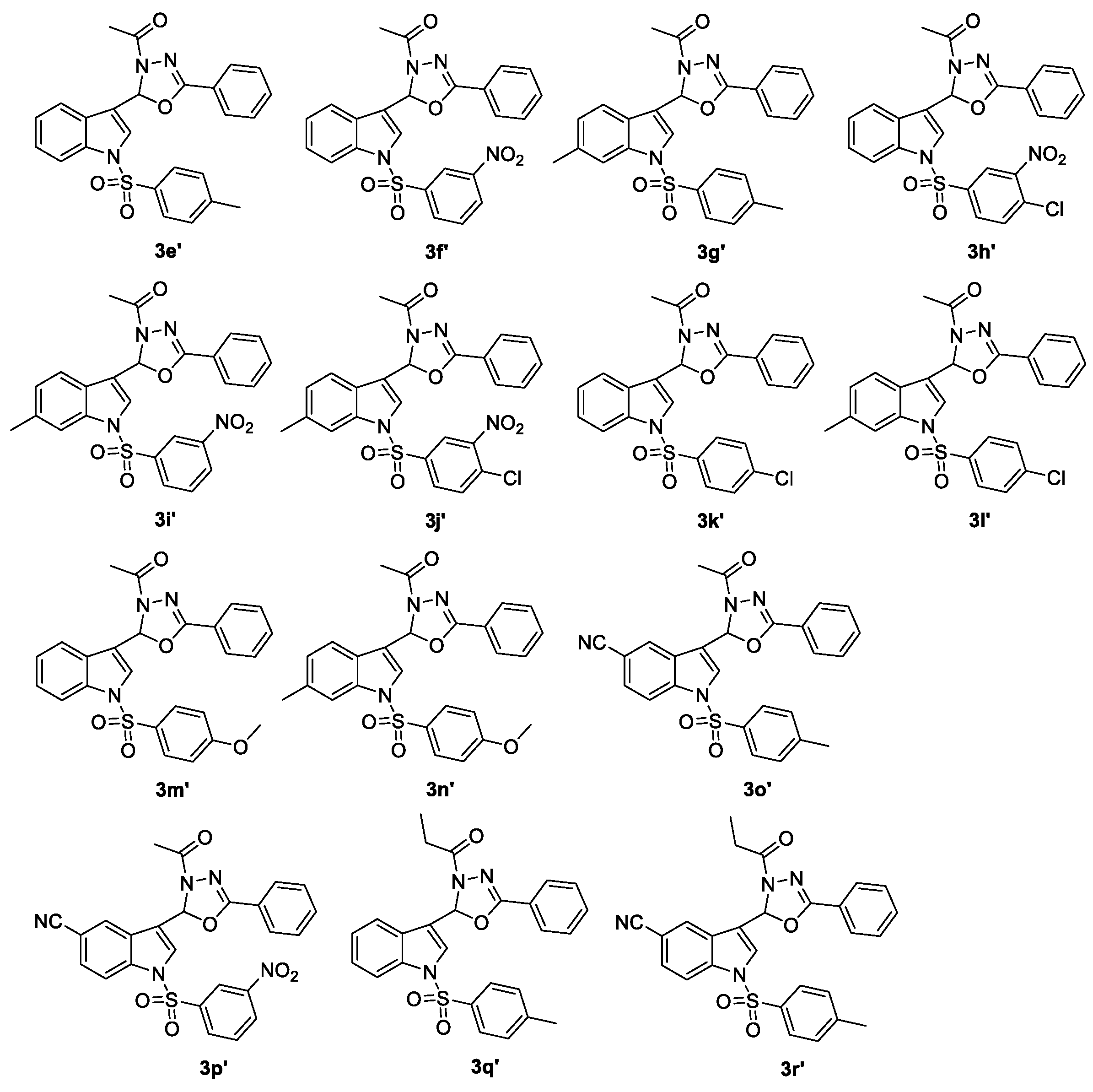
Fourteen compounds of 2-(N-arylsulfonylindol-3-yl)-3-N-acyl-5-aryl-1,3,4-oxadiazolines (3e′–r′, Figure 1) were prepared using our previously published method, and characterized by 1H-NMR, EI-MS or ESI-MS and HRMS [19]. Subsequently, 21 2-alkyl-2-(N-arylsulfonylindol-3-yl)-3-N-acyl-5-aryl-1,3,4-oxadiazolines (3s–r′) were tested in vitro for their anti-HIV-1 activity, and 3′-azido-3′-deoxythymidine (AZT) was used as a positive control illustrated in Table 5.
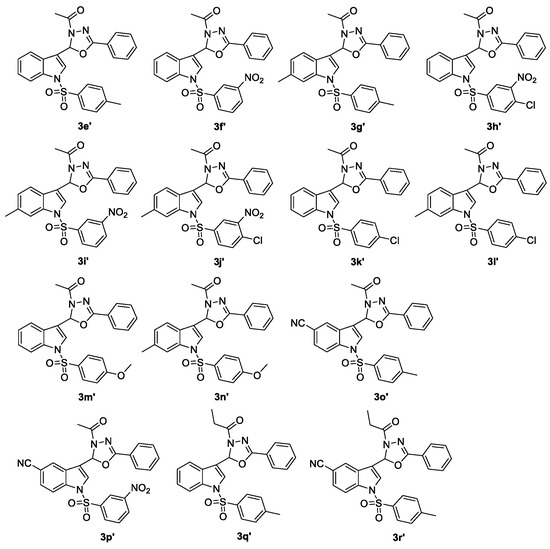
Figure 1.
Chemical structures of 2-(N-arylsulfonylindol-3-yl)-3-N-acyl-5-aryl-1,3,4-oxadiazolines 3e′–r′.

Table 5.
Anti-HIV-1 activity of 2-alkyl-2-(N-arylsulfonylindol-3-yl)-3-N-acyl-5-aryl-1,3,4-oxadiazolines 3s–r′ in vitro a.
Among these tested 2-alkyl-2-(N-arylsulfonylindol-3-yl)-3-N-acyl-5-aryl-1,3,4-oxadiazolines, compounds 3s, 3y, 3a′, 3b′, 3f′, 3i′, 3q′, and 3r′ exhibited more potent anti-HIV-1 activity with EC50 values of 3.35, 6.12, 3.63, 9.54, 1.79, 0.51, 3.00, and 4.01 μg/mL, and TI values of 32.66, >32.68, 31.22, 13.94, 24.27, 39.59, 26.01, and 24.51, respectively. Especially compound 3i′ displayed the highest anti-HIV-1 activity with TI values of 39.59. Meanwhile, a brief structure activity relationship (SAR) was determined. (1) When R1 = H or 6-Me, compound with R2 = m-NO2 was more vital for the anti-HIV-1 activities than R2 = m-NO2, p-Cl or p-Cl (3f′ and 3i′ vs. 3h′ and 3j′, EC50 values of 1.79, 0.51, 3.53, and 12.42 μg/mL, TI values of 24.27, 39.59, 1.36, and 0.18, respectively; that is, the TI value of 3f′ was close to 18 times of that of 3h′ and the TI value of 3i′ was close to 220 times of that of 3j′. 3f′ and 3i′ vs. 3k′ and 3l′; EC50 values of 1.79, 0.51, 19.37, and 57.49 μg/mL; TI values of 24.27, 39.59, 0.86, and 3.16, respectively; that is, the TI value of 3f′ was more than 28 times of that of 3k′ and the TI value of 3i′ was more than 12 times of that of 3l′). (2) The length of the chain of substituent R5 was significant for the anti-HIV-1 activity (3q′ vs. 3e′, EC50 values of 3.00 and 15.52 μg/mL, TI values of 26.01 and 0.97, respectively, especially the TI value of 3q′ was close to 27 times of that of 3e′; 3r′ vs. 3o′, EC50 values of 4.01 and 18.14 μg/mL, and TI values of 24.51 and 8.36, respectively). (3) When R1 = 5-NO2, compound with R2 = H was more important for the anti-HIV-1 activities than R2 = p-Me or p-Cl (3s vs. 3b′ and 3c′, EC50 values of 3.35, 9.54 and 6.01 μg/mL, TI values of 32.66, 13.94 and 8.89, respectively; that is, the TI value of 3s was more than two times of that of 3b′ and was more than three times of that of 3c′). When R1 = 5-CN, introduction of R2 as the p-Cl group could lead to the pronounced compound (3a′ vs. 3z, EC50 values of 3.63 and 17.57 μg/mL, TI values of 31.22 and 3.01, respectively, especially the TI value of 3a′ was more than 10 times of that of 3z).
3. Experimental Section
3.1. General Information
All solvents and reagents were used as obtained from commercial sources without further purification. Analytical thin-layer chromatography (TLC) and preparative thin-layer chromatography (PTLC) were performed with silica gel plates using silica gel 60 GF254 (Qingdao Haiyang Chemical Co., Ltd., Shandong, China). Melting points are uncorrected. Nuclear magnetic resonance spectra (NMR) were recorded on a Bruker Avance DMX 400 MHz instrument (Bruker Daltonik, Bremen, Germany) in CDCl3 (1H at 400 MHz and 13C at 100 MHz) using TMS (tetramethylsilane) as the internal standard. Electrospray ion trap mass spectrometry (ESI-TRAP-MS) was carried out with a Bruker ESI-TRAP Esquire 6000 plus mass spectrometry instrument (Bruker, Germany) Microwave irradiation was performed in a Galanz microwave oven, WG700CTL20II-K6 (Galanz, Guangdong, China).
3.2. Preparation of 2-Alkyl-2-(N-arylsulfonylindol-3-yl)-3-N-acyl-5-aryl-1,3,4-oxadiazolines (3a–d′)
A mixture of 1 (0.5 mmol), 2 (1.5 mmol), and HgCl2 (0.02 mmol) was reacted under microwave heating at 700 W for 30–40 min. After allowing the mixture to cool to room temperature, the mixture was diluted with dichloromethane (30 mL) and filtered. The filtrate was washed with saturation sodium bicarbonate (2 × 20 mL), and brine (1 × 10 mL). Then the organic phase was dried over anhydrous Na2SO4, concentrated in vacuo and purified by preparative thin-layer chromatography (PTLC) to give the desired products 3a–d′ in 80–95% yields. Compounds 3a–w were known compounds and characterized by comparison of the data as described in our previous paper [15]. The NMRs, MS (see Supplementary Materials) and typical spectral data of compounds 3s–d′ were as follows.
2-Methyl-2-(N-benzenesulfonyl-5-nitroindol-3-yl)-3-N-acetyl-5-phenyl-1,3,4-oxadiazolines (3s): Yellow solid, m.p. 186–188 °C. 1H-NMR (400 MHz, CDCl3) δ: 8.42 (d, J = 2.0 Hz, 1H), 8.17–8.19 (m, 1H), 8.00–8.04 (m, 2H), 7.93–7.95 (m, 2H), 7.87–7.89 (m, 2H), 7.60–7.64 (m, 1H), 7.49–7.55 (m, 3H), 7.46 (t, J = 7.2 Hz, 2H), 2.36 (s, 3H), 2.33 (s, 3H). 13C-NMR (100 MHz, CDCl3) δ: 167.1, 154.0, 144.4, 137.9, 137.2, 134.7, 131.8, 129.7, 128.7, 128.4, 127.3, 127.0, 126.8, 124.0, 121.7, 120.1, 116.6, 113.9, 96.7, 23.6, 22.3. MS (ESI-TRAP), m/z (%): 527 ([M + Na]+, 100).
2-Methyl-2-(N-benzenesulfonyl-6-methylindol-3-yl)-3-N-acetyl-5-phenyl-1,3,4-oxadiazolines (3y): White solid, m.p. 142–144 °C. 1H-NMR (400 MHz, CDCl3) δ: 7.90 (d, J = 7.2 Hz, 2H), 7.86 (d, J = 7.2 Hz, 2H), 7.78 (s, 1H), 7.71 (s, 1H), 7.40–7.57 (m, 6H), 7.25–7.28 (m, 1H), 6.99 (d, J = 7.6 Hz, 1H), 2.40 (s, 3H), 2.31 (s, 3H), 2.27 (s, 3H). 13C-NMR (100 MHz, CDCl3) δ: 166.6, 153.9, 138.1, 135.6, 135.1, 133.9, 131.4, 129.3, 128.6, 126.8, 125.3, 124.6, 120.8, 119.4, 113.7, 97.2, 23.3, 22.2, 21.7. MS (ESI-TRAP), m/z (%): 496 ([M + Na]+, 100).
2-Methyl-2-(N-benzenesulfonyl-5-cyanoindol-3-yl)-3-N-acetyl-5-phenyl-1,3,4-oxadiazolines (3z): White solid, m.p. 208–210 °C. 1H-NMR (400 MHz, CDCl3) δ: 8.03 (d, J = 8.8 Hz, 1H), 7.97 (s, 1H), 7.94 (d, J = 7.6 Hz, 2H), 7.87 (d, J = 8.4 Hz, 2H), 7.75 (s, 1H), 7.60–7.64 (m, 1H), 7.51–7.56 (m, 4H), 7.43–7.47 (m, 2H), 2.31 (s, 6H). 13C-NMR (100 MHz, CDCl3) δ: 167.0, 153.9, 137.4, 136.8, 134.6, 131.8, 129.7, 128.7, 127.9, 127.8, 127.5, 127.0, 126.8, 125.0, 124.0, 120.8, 119.1, 114.5, 107.5, 96.7, 23.3, 22.2. MS (ESI-TRAP), m/z (%): 507 ([M + Na]+, 100).
2-Methyl-2-(N-p-chlorobenzenesulfonyl-5-cyanoindol-3-yl)-3-N-acetyl-5-phenyl-1,3,4-oxadiazolines (3a′): White solid, m.p. 128–130 °C. 1H-NMR (400 MHz, CDCl3) δ: 8.01 (d, J = 8.8 Hz, 1H), 7.93 (s, 1H), 7.84–7.87 (m, 4H), 7.75 (s, 1H), 7.44–7.57 (m, 6H), 2.31 (s, 6H). 13C-NMR (100 MHz, CDCl3) δ: 167.0, 153.9, 141.5, 136.7, 135.7, 131.9, 130.0, 128.7, 128.3, 128.1, 127.7, 127.6, 126.8, 125.1, 123.9, 121.2, 118.9, 114.5, 107.7, 96.5, 23.3, 22.2. MS (ESI-TRAP), m/z (%): 541 ([M + Na]+, 100).
2-Methyl-2-(N-p-methylbenzenesulfonyl-5-nitroindol-3-yl)-3-N-acetyl-5-phenyl-1,3,4-oxadiazolines (3b′): Yellow solid, m.p. 110–112 °C. 1H-NMR (400 MHz, CDCl3) δ: 8.41 (d, J = 2.0 Hz, 1H), 8.16–8.19 (m, 1H), 7.99–8.02 (m, 2H), 7.89 (d, J = 7.2 Hz, 2H), 7.83 (d, J = 8.4 Hz, 2H), 7.42–7.52 (m, 3H), 7.32 (d, J = 8.0 Hz, 2H), 2.37 (s, 3H), 2.35 (s, 3H), 2.33 (s, 3H). 13C-NMR (100 MHz, CDCl3) δ: 167.1, 154.0, 146.1, 144.3, 137.8, 134.2, 131.8, 130.3, 128.7, 128.4, 127.2, 127.1, 126.8, 124.0, 121.5, 120.0, 116.6, 113.8, 96.8, 23.6, 22.3, 21.6. MS (ESI-TRAP), m/z (%): 541 ([M + Na]+, 100).
2-Methyl-2-(N-p-chlorobenzenesulfonyl-5-nitroindol-3-yl)-3-N-acetyl-5-phenyl-1,3,4-oxadiazolines (3c′): Yellow solid, m.p. 108–110 °C. 1H-NMR (400 MHz, CDCl3) δ: 8.42 (d, J = 1.6 Hz, 1H), 8.18-8.21 (m, 1H), 7.97–8.02 (m, 2H), 7.85–7.89 (m, 4H), 7.43–7.52 (m, 5H), 2.35 (s, 3H), 2.33 (s, 3H). 13C-NMR (100 MHz, CDCl3) δ: 167.2, 154.0, 144.5, 141.6, 137.8, 135.6, 131.8, 130.1, 128.7, 128.4, 127.4, 126.9, 124.0, 122.1, 120.3, 116.8, 113.8, 96.6, 23.6, 22.3. MS (ESI-TRAP), m/z (%): 561 ([M + Na]+, 78).
2-Ethyl-2-(N-benzenesulfonylindol-3-yl)-3-N-acetyl-5-phenyl-1,3,4-oxadiazolines (3d′): White solid, m.p. 224–226 °C. 1H-NMR (400 MHz, CDCl3) δ: 7.85–7.93 (m, 6H), 7.41–7.56 (m, 7H), 7.25–7.29 (m, 1H), 7.13–7.17 (m, 1H), 3.05–3.11 (m, 1H), 2.54–2.59 (m, 1H), 2.31 (s, 3H), 1.03–1.07 (m, 3H). 13C-NMR (100 MHz, CDCl3) δ: 166.8, 154.5, 137.9, 135.1, 133.9, 131.5, 129.3, 128.6, 127.6, 126.9, 126.8, 125.8, 124.8, 124.3, 123.7, 120.9, 120.1, 113.7, 100.0, 28.4, 22.2, 6.5. MS (ESI-TRAP), m/z (%): 496 ([M + Na]+, 100).
3.3. Anti-HIV-1 Activity Assay
3.3.1. Virus and Cells
Cell line (C8166) and the laboratory-derived virus (HIV-1IIIB) were obtained from MRC, AIDS Reagent Project, London, UK. C8166 was maintainedin RPMI-1640 supplemented with 10% heat-inactivated newborn calf serum (Gibco, Grand Island, NY, USA). The cells used in all experiments were in log-phase growth. The 50% HIV-1IIIB tissue culture infectious dose (TCID50) in C8166 cells was determined and calculated by the Reed and Muench method. Virus stocks were stored in small aliquots at −70 °C.
3.3.2. MTT-Based Cytotoxicity Assay
Cellular toxicity of 2-alkyl-2-(N-arylsulfonylindol-3-yl)-3-N-acyl-5-aryl-1,3,4-oxadiazolines 3s–r′ on C8166 cells was assessed by MTT method as described previously. Briefly, cells were seeded on 96-well microtiter plate in the absence or presence of various concentrations of 2-alkyl-2-(N-arylsulfonylindol-3-yl)-3-N-acyl-5-aryl-1,3,4-oxadiazolines in triplicate and incubated at 37 °C in a humid atmosphere of 5% CO2 for 3 day. The supernatants were discarded and MTT reagent (5 mg/mL in PBS) was added to each wells, then incubated for 4 h, 100 μL of 50% N,N-dimethylformamide (DMF)-20% SDS was added. After the formazan was dissolved completely, the plates were read on a Bio-TekElx800 ELISA reader (BioTek, Winooski, VT, USA) at 595/630 nm. The cytotoxic concentration that caused the reduction of viable C8166 cells by 50% (CC50) was determined from dose–response curve.
3.3.3. Syncytia Assay
In the presence of 100 μL various concentrations of 2-alkyl-2-(N-arylsulfonylindol-3-yl)-3-N-acyl-5-aryl-1,3,4-oxadiazolines, C8166 cells (4 × 105/mL) were infected with virus HIV-1IIIB at a multiplicity of infection (M.O.I) of 0.06. The final volume per well was 200 μL. Control assays were performed without the testing compounds in HIV-1IIIB infected and uninfected cultures. After 3 days of culture, the cytopathic effect (CPE) was measured by counting the number of syncytia. Percentage inhibition of syncytia formation was calculated and 50% effective concentration (EC50) was calculated. AZT (Sigma-Aldrich, St. Louis, MO, USA) was used as a positive control. Therapeutic index (TI) = CC50/EC50.
4. Conclusions
Here we report a very superior method of the microwave-assisted expeditious synthesis of 2-alkyl-2-(N-arylsulfonylindol-3-yl)-3-N-acyl-5-aryl-1,3,4-oxadiazolines catalyzed by HgCl2 under solvent-free conditions. This method has the advantages of low catalyst loading and recovering catalyst, short reaction and repaid reaction times, easy separation products, excellent yields, and being more conducive to the large-scale synthesis products. Compound 3i′ especially displayed the highest anti-HIV-1 activity with TI values of 39.59. It implied that 3i′ might be regarded as the lead compound for further preparation of anti-HIV-1 agents.
Supplementary Materials
The following are available online. The NMRs and MS of the new compounds (3s–d′).
Author Contributions
Designed the experiments, synthesized the compounds, and analyzed the data: Z.C. Wrote the paper: Z.C., Y.T., S.L., J.J., M.H., and G.C. All authors approved the final manuscript.
Funding
The present research was supported by the National Natural Science Foundation of China (grant no. U1604105).
Acknowledgments
We would like to acknowledge the MRC AIDS Research Project and the NIH AIDS Research and Reference Reagent Program for providing cell lines and viruses.
Conflicts of interest
The authors have reported no conflict of interest.
References
- Gottlieb, M.S.; Schroff, R.; Schanker, H.M.; Weisman, J.D.; Fan, P.T.; Wolf, R.A.; Saxon, A. Pneumocystis carinii pneumonia and mucosal candidiasis in previously healthy homosexual men: Evidence of a new acquired cellular immunodeficiency. N. Engl. J. Med. 1981, 305, 1425–1431. [Google Scholar] [CrossRef] [PubMed]
- Jonckheere, H.; Anné, J.; De Clercq, E. The HIV-1 reverse transcription (RT) process as target for RT inhibitors. Med. Res. Rev. 2000, 20, 129–154. [Google Scholar] [CrossRef]
- Yisma, E.; Dessalegn, B.; Astatkie, A.; Fesseha, N.; Benagiano, G.; Bastianelli, C.; Brusamento, S.; Elmoniry, H.; van Velthoven, M.; Pape, U. Global report: UNAIDS report on the global AIDS epidemic 2013. Reprod. Health 2014, 10, 23. [Google Scholar] [CrossRef] [PubMed]
- Boone, L.R. Next-generation HIV-1 non-nucleoside reverse transcriptase inhibitors. Curr. Opin. Investig. Drugs 2006, 7, 128–135. [Google Scholar] [PubMed]
- De Clercq, E. New developments in anti-HIV chemotherapy. Biochim. Biophy. Acta 2002, 1587, 258–275. [Google Scholar] [CrossRef]
- Sluis-Cremer, N.; Wainberg, M.A.; Schinazi, R.F. Resistance to reverse transcriptase inhibitors used in the treatment and prevention of HIV-1 infection. Future Microbiol. 2015, 10, 1773–1782. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.Y.; Fan, E.K.; Wu, J.D.; Liu, X.Y. Recent advances in the DABOs family as potent HIV-1 non-nucleoside reverse transcriptase inhibitors. Curr. Med. Chem. 2011, 18, 2376–2385. [Google Scholar] [CrossRef] [PubMed]
- Chander, S.; Wang, P.; Ashok, P.; Yang, L.M.; Zheng, Y.T.; Murugesan, S. Rational design, synthesis, anti-HIV-1 RT and antimicrobial activity of novel 3-(6-methoxy-3,4-dihydroquinolin-1(2H)-yl)-1-(piperazin-1-yl) propan-1-one derivatives. Bioorg. Chem. 2016, 67, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Yu, D.L.; Ho, P.; Lee, K.H.; Chen, C.H. Synthesis and anti-HIV activity of bi-functional triterpene derivatives. Lett. Drug Des. Discov. 2007, 4, 471–478. [Google Scholar] [CrossRef]
- Polanski, J.; Niedbala, H.; Musiol, R.; Podeszwa, B.; Tabak, D.; Palka, A.; Mencel, A.; Finster, J.; Mouscadet, J.F.; Le Bret, M. 5-Hydroxy-6-quinaldic acid as a novel molecular scaffold for HIV-1 integrase inhibitors. Lett. Drug Des. Discov. 2006, 3, 175–178. [Google Scholar] [CrossRef]
- Safakish, M.; Hajimahdi, Z.; Zabihollahi, R.; Aghasadeghi, M.R.; Vahabpour, R.; Zarghi, A. Design, synthesis, and docking studies of new 2-benzoxazolinone derivatives as anti-HIV-1 agents. Med. Chem. Res. 2017, 26, 2718–2726. [Google Scholar] [CrossRef]
- Ke, S.Y.; Liu, F.Y.; Wang, N.; Yang, Q.; Qian, X.H. 1,3,4-Oxadiazoline derivatives as novel potential inhibitors targeting chitin biosynthesis: Design, synthesis and biological evaluation. Bioorg. Med. Chem. Lett. 2009, 19, 332–335. [Google Scholar] [CrossRef] [PubMed]
- Coley, H.M.; Sarju, J.; Wagner, G. Synthesis and characterization of platinum(II) oxadiazoline complexes and their in vitro antitumor activity in platinum-sensitive and -resistant cancer cell lines. J. Med. Chem. 2008, 51, 135–141. [Google Scholar] [CrossRef] [PubMed]
- El-Emam, A.A.; Al-Deeb, O.A.; Al-Omar, M.; Lehmann, J. Synthesis, antimicrobial, and anti-HIV-1 activity of certain 5-(1-adamantyl)-2-substituted thio-1,3,4-oxadiazoles and 5-(1-adamantyl)-3-substituted aminomethyl-1,3,4-oxadiazoline-2-thiones. Bioorg. Med. Chem. 2004, 12, 5107–5113. [Google Scholar] [CrossRef] [PubMed]
- Che, Z.P.; Huang, N.; Yu, X.; Yang, L.M.; Ran, J.Q.; Zhi, X.Y.; Xu, H.; Zheng, Y.T. Microwave-assisted combinatorial synthesis of 2-alkyl-2-(N-arylsulfonylindol-3-yl)-3-N-acyl-5-aryl-1,3,4-oxadiazolines as anti-HIV-1 agents. Comb. Chem. High Throughput Scr. 2013, 16, 400–407. [Google Scholar] [CrossRef]
- Joshi, S.D.; Vagdevi, H.M.; Vaidya, V.P.; Gadaginamath, G.S. Synthesis of new 4-pyrrol-1-yl benzoic acid hydrazide analogs and some derived oxadiazole, triazole and pyrrole ring systems: A novel class of potential antibacterial and antitubercular agents. Eur. J. Med. Chem. 2008, 43, 1989–1996. [Google Scholar] [CrossRef] [PubMed]
- Cottineau, B.; Renaux, S.; Chenault, J.; Guillaumet, G. New synthesis of pyrazolyl-1,3,4-oxadiazole and 1,3,4-oxadiazoline derivatives. Lett. Org. Chem. 2005, 2, 599–601. [Google Scholar] [CrossRef]
- Li, C.K.; Ma, Y.J.; Cao, L.H. Synthesis of novel 3-acetyl-2-aryl-5-(3-aryl-1-phenyl-pyrazol-4-yl)-2,3- dihydro-1,3,4-oxadiazoles. J. Chin. Chem. Soc. 2009, 56, 182–185. [Google Scholar] [CrossRef]
- Xu, H.; Che, Z.P.; Wang, Q. An efficiently sonochemical synthesis of 2-(N-aryl-sulfonylindol-3-yl)-3-N-acyl-5-phenyl-1,3,4-oxadiazolines. Heterocycles 2011, 42, 825–832. [Google Scholar] [CrossRef]
- Gedye, R.; Smith, F.; Westaway, K.; Humera, A.; Baldisera, L.; Laberge, L.; Rousell, L. The use of microwave ovens for rapid organic synthesis. Tetrahedron Lett. 1986, 27, 279–282. [Google Scholar] [CrossRef]
- Giguere, R.J.; Bray, T.L.; Duncan, S.M.; Majetich, G. Application of commercial microwave ovens to organic synthesis. Tetrahedron Lett. 1986, 27, 4945–4948. [Google Scholar] [CrossRef]
- Koziej, D.; Floryan, C.; Sperling, R.A.; Ehrlicher, A.J.; Issadore, D.; Westervelt, R.; Weitz, D.A. Microwave dielectric heating of non-aqueous droplets in a microfluidic device for nanoparticle synthesis. Nanoscale 2013, 5, 5468–5475. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, S.L.; Tofteng, A.P.; Malik, L.; Jensen, J. Microwave heating in solid-phase synthesis. Chem. Soc. Rev. 2012, 41, 1826–1844. [Google Scholar] [CrossRef] [PubMed]
- Westman, J. An efficient combination of microwave dielectric heating and the use of solid-supported triphenylphosphine for Wittig reactions. Org. Lett. 2001, 3, 3745–3747. [Google Scholar] [CrossRef] [PubMed]
- Mariappan, A.; Rajaguru, K.; Muthusubramanian, S.; Bhuvanesh, N. Microwave-assisted catalyst-free synthesis of tetrasubstituted pyrroles using dialkyl acetylenedicarboxylates and monophenacylanilines. Syn. Commun. 2016, 47, 805–812. [Google Scholar] [CrossRef]
- Wang, S.L.; Cheng, C.; Wu, F.Y.; Jiang, B.; Shi, F.; Tu, S.J.; Rajale, T.; Li, G. Microwave-assisted multi-component reaction in water leading to highly regioselective formation of benzo[f]azulen-1-ones. Tetrahedron 2011, 67, 4485–4493. [Google Scholar] [CrossRef] [PubMed]
- Vaddula, B.R.; Varma, R.S.; Leazer, J. Mixing with microwaves: Solvent-free and catalyst-free synthesis of pyrazoles and diazepines. Tetrahedron Lett. 2013, 54, 1538–1541. [Google Scholar] [CrossRef]
- De-la-Torre, P.; Osorio, E.; Alzate-Morales, J.H.; Caballero, J.; Trilleras, J.; Astudillo-Saavedra, L.; Brito, L.; Cárdenas, A.; Quiroga, J.; Gutiérrez, M. Ultrasound-assisted phase-transfer catalysis method in an aqueous medium to promote the Knoevenagel reaction: Advantages over the conventional and microwave-assisted solvent-free/catalyst-free method. Ultrason. Sonochem. 2014, 21, 1666–1674. [Google Scholar] [CrossRef] [PubMed]
- Driowya, M.; Saber, A.; Marzag, H.; Demange, L.; Bougrin, K.; Benhida, R. Microwave-assisted syntheses of bioactive seven-membered, macro-sized heterocycles and their fused derivatives. Molecules 2016, 21, 1032. [Google Scholar] [CrossRef] [PubMed]
- Vera, G.; Diethelm, B.; Terraza, C.A.; Recabarren-Gajardo, G. Suzuki-type cross-coupling reaction of unprotected 3-iodoindazoles with pinacol vinyl boronate: An expeditive C-3 vinylation of indazoles under microwave irradiation. Molecules 2018, 23, 2051. [Google Scholar] [CrossRef] [PubMed]
- Kantar, G.K.; Baltas, N.; Mentese, E.; Sasmaz, S. Microwave-assisted synthesis and investigation of xanthine oxidase inhibition of new phthalonitrile and phthalocyanines containing morpholino substituted 1,2,4-triazole-3-one. J. Organomet. Chem. 2015, 787, 8–13. [Google Scholar] [CrossRef]
- Saber, A.; Marzag, H.; Benhida, R.; Bougrin, K. Microwave-assisted cycloaddition reactions in carbo- and heterocyclic chemistry. Curr. Org. Chem. 2014, 18, 2139–2180. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 3a–r′ are available from the authors. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).