The Ubiquitin-Conjugating Enzyme Gene Family in Longan (Dimocarpus longan Lour.): Genome-Wide Identification and Gene Expression during Flower Induction and Abiotic Stress Responses
Abstract
:1. Introduction
2. Results
2.1. Identification and Characterization of UBC Family Genes in Longan
2.2. The Gene Structure and Motif Composition of the Longan UBC Gene Family
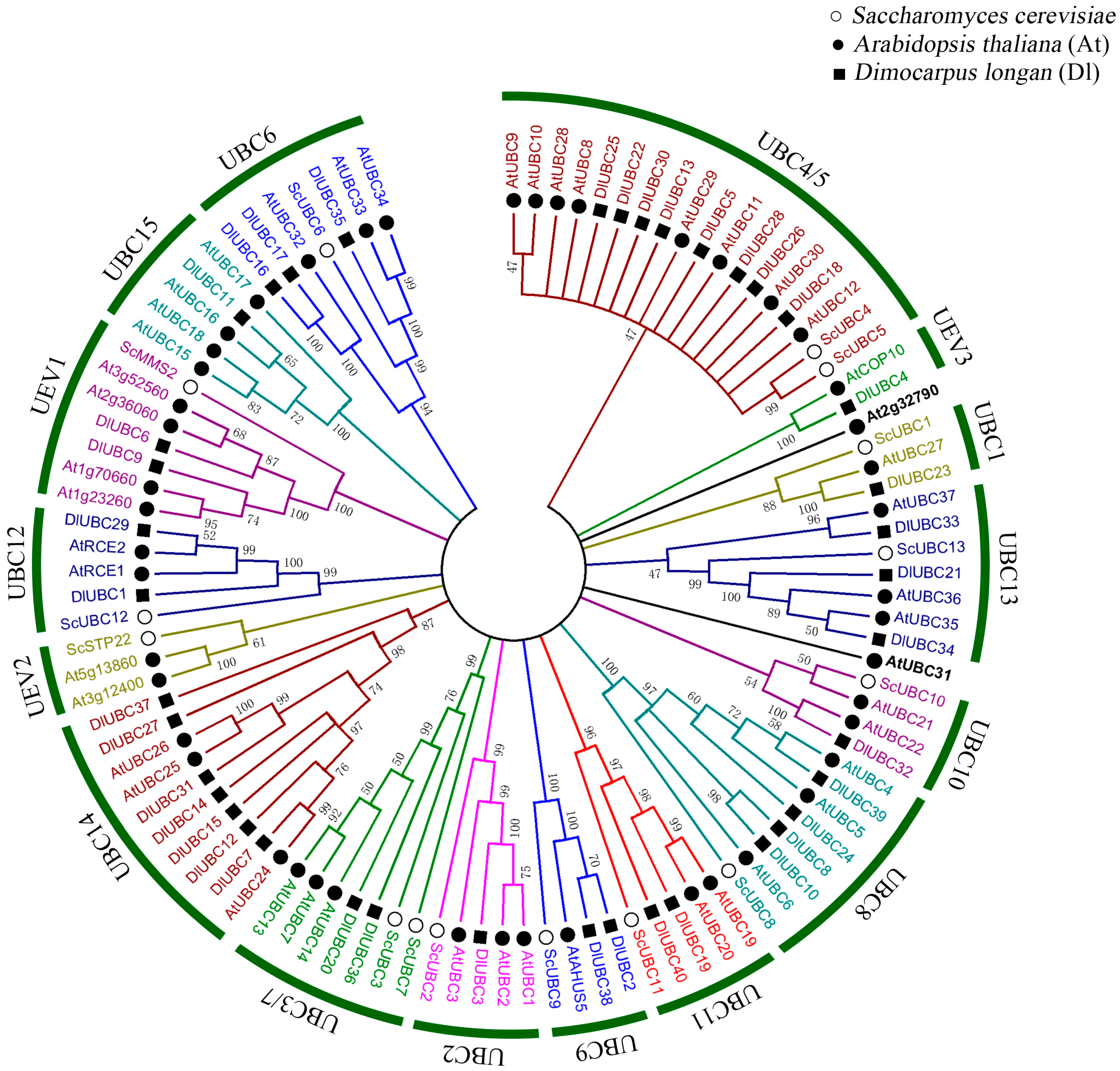
2.3. Phylogenetic Analysis of the DlUBC Genes
2.4. Tissue-Specific Expression Patterns of DlUBC in Longan
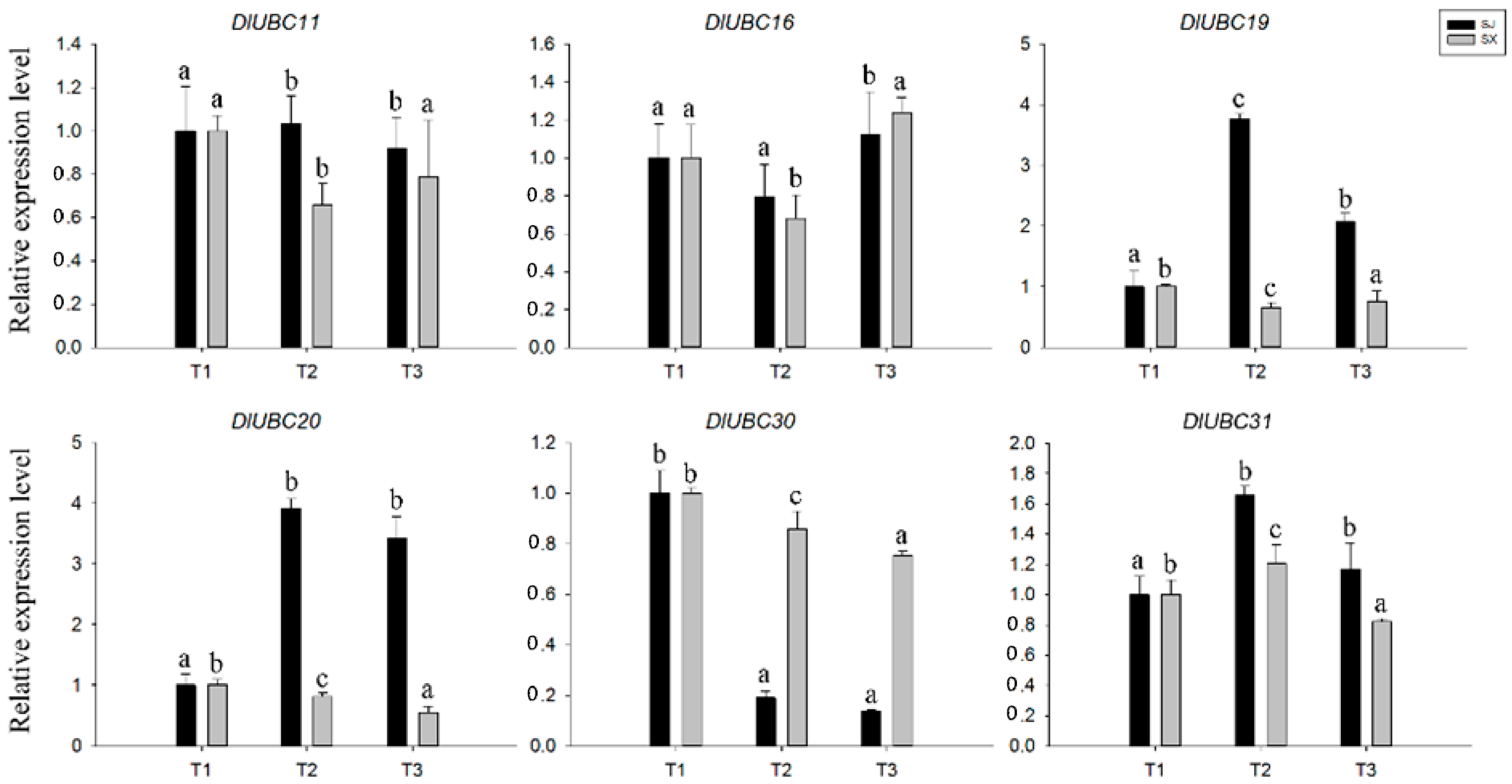
2.5. Comparative Expression Profiles of the Two Longan Species during Flowering
2.6. Differential Regulation of DlUBCs in Response to Stress and Hormonal Treatments
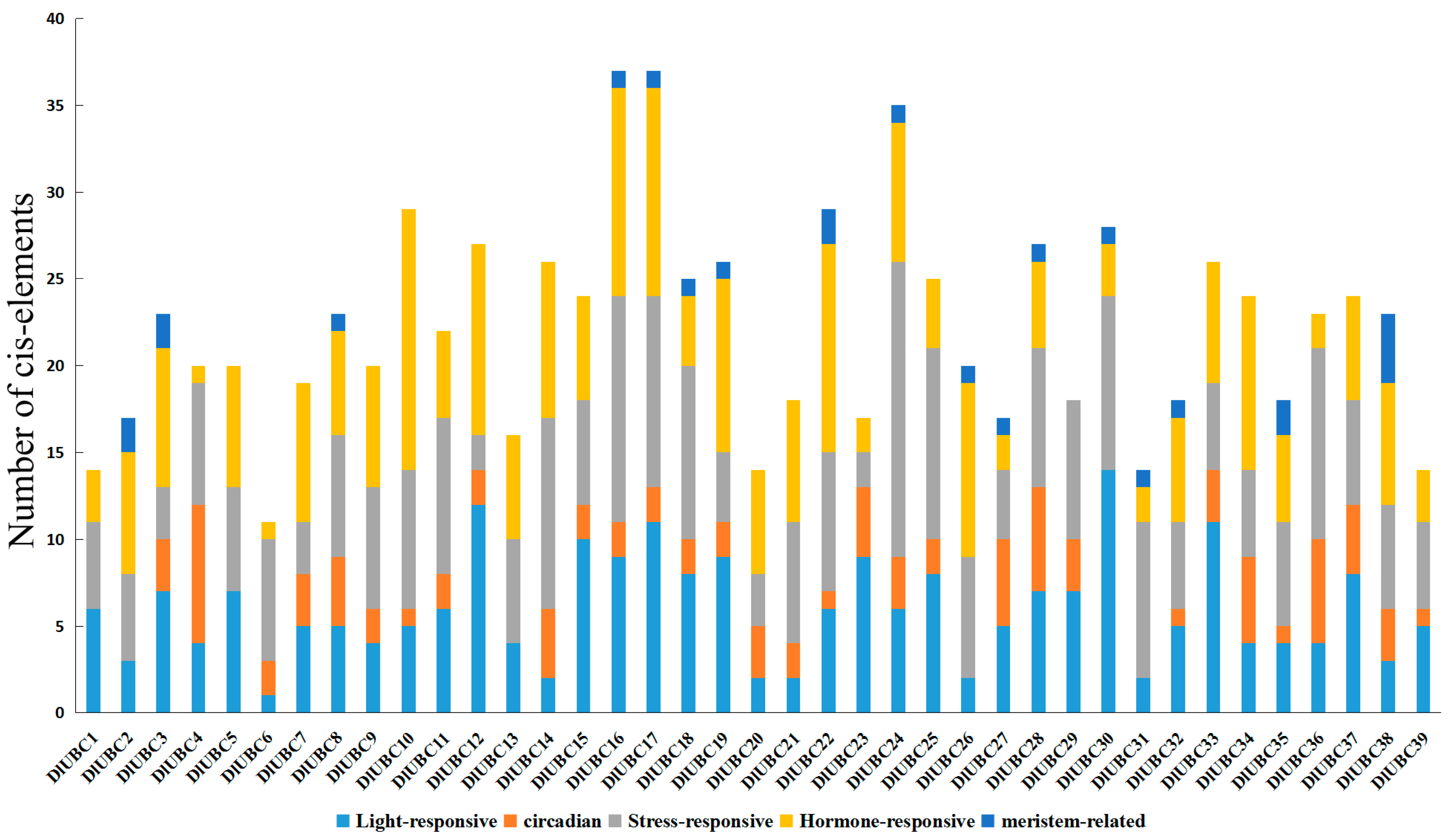
2.7. Analysis Related Cis-Elements in the Candidate DlUBC Genes
3. Discussion
4. Materials and Methods
4.1. Identification of the Longan Conjugating Enzyme Family Gene
4.2. Sequence Analysis
4.3. Sequence Alignment, Cis-Elements in the Promoters and Phylogenetic Analysis
4.4. Expression Analysis of Longan UBCs in Various Tissues and at Different Flowering Stages
4.5. Hormonal and Stress Treatments and Expression Profiling Using qRT-PCR
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Callis, J.; Carpenter, T.; Sun, C.-W.; Vierstra, R.D. Structure and evolution of genes encoding polyubiquitin and ubiquitin-like proteins in Arabidopsis thaliana ecotype Columbia. Genetics 1995, 139, 921–939. [Google Scholar] [PubMed]
- Hershko, A.; Ciechanover, A. The ubiquitin system. Ann. Rev. Biochem. 1998, 67, 425–479. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, D.; Riezman, H. Proteasome-independent functions of ubiquitin in endocytosis and signaling. Science 2007, 315, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, F.-R.; Wilson, G.; Day, C.L. The N-terminal extension of UBE2E ubiquitin-conjugating enzymes limits chain assembly. J. Mol. Biol. 2013, 425, 4099–4111. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, K.S.; Ellison, M.J.; Barber, K.R.; Williams, R.S.; Huzil, J.T.; McKenna, S.; Ptak, C.; Glover, M.; Shaw, G.S. Structure of a conjugating enzyme-ubiquitin thiolester intermediate reveals a novel role for the ubiquitin tail. Structure 2001, 9, 897–904. [Google Scholar] [CrossRef]
- Papaleo, E.; Casiraghi, N.; Arrigoni, A.; Vanoni, M.; Coccetti, P.; De Gioia, L. Loop 7 of E2 enzymes: An ancestral conserved functional motif involved in the E2-mediated steps of the ubiquitination cascade. PLoS ONE 2012, 7, e40786. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.; Hu, H.; Jue, D.; Zhao, Q.; Chen, H.; Xie, J.; Jia, L. The banana E2 gene family: Genomic identification, characterization, expression profiling analysis. Plant Sci. 2016, 245, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Jue, D.; Sang, X.; Lu, S.; Dong, C.; Zhao, Q.; Chen, H.; Jia, L. Genome-wide identification, phylogenetic and expression analyses of the ubiquitin-conjugating enzyme gene family in maize. PLoS ONE 2015, 10, e0143488. [Google Scholar] [CrossRef] [PubMed]
- Michelle, C.; Vourc’h, P.; Mignon, L.; Andres, C.R. What was the set of ubiquitin and ubiquitin-like conjugating enzymes in the eukaryote common ancestor? J. Mol. Evol. 2009, 68, 616–628. [Google Scholar] [CrossRef] [PubMed]
- Callis, J. The ubiquitination machinery of the ubiquitin system. Arabidopsis Book 2014, e0174. [Google Scholar] [CrossRef] [PubMed]
- Sadanandom, A.; Bailey, M.; Ewan, R.; Lee, J.; Nelis, S. The ubiquitin–proteasome system: Central modifier of plant signalling. New Phytol. 2012, 196, 13–28. [Google Scholar] [CrossRef] [PubMed]
- Welchman, R.L.; Gordon, C.; Mayer, R.J. Ubiquitin and ubiquitin-like proteins as multifunctional signals. Nat. Rev. Mol. Cell Biol. 2005, 6, 599. [Google Scholar] [CrossRef] [PubMed]
- Cui, F.; Liu, L.; Zhao, Q.; Zhang, Z.; Li, Q.; Lin, B.; Wu, Y.; Tang, S.; Xie, Q. Arabidopsis ubiquitin conjugase UBC32 is an ERAD component that functions in brassinosteroid-mediated salt stress tolerance. Plant Cell 2012, 24, 233–244. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Cao, L.; Wang, H. Arabidopsis ubiquitin-conjugating enzyme UBC22 is required for female gametophyte development and likely involved in Lys11-linked ubiquitination. J. Exp. Bot. 2016, 67, 3277–3288. [Google Scholar] [CrossRef] [PubMed]
- Mural, R.V.; Liu, Y.; Rosebrock, T.R.; Brady, J.J.; Hamera, S.; Connor, R.A.; Martin, G.B.; Zeng, L. The Tomato Fni3 Lysine-63–Specific Ubiquitin-Conjugating Enzyme and Suv Ubiquitin E2 Variant Positively Regulate Plant Immunity. Plant Cell 2013, 25, 3615–3631. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, W.; Cai, J.; Zhang, Y.; Qin, G.; Tian, S. Tomato nuclear proteome reveals the involvement of specific E2 ubiquitin-conjugating enzymes in fruit ripening. Genome Biol. 2014, 15, 548. [Google Scholar] [CrossRef] [PubMed]
- Millyard, L.; Lee, J.; Zhang, C.; Yates, G.; Sadanandom, A. The ubiquitin conjugating enzyme, TaU4 regulates wheat defence against the phytopathogen Zymoseptoria tritici. Sci. Rep. 2016, 6, 35683. [Google Scholar] [CrossRef] [PubMed]
- Turnbull, C. Long-distance regulation of flowering time. J. Exp. Bot. 2011, 62, 4399–4413. [Google Scholar] [CrossRef] [PubMed]
- Andrés, F.; Coupland, G. The genetic basis of flowering responses to seasonal cues. Nat. Rev. Genet. 2012, 13, 627. [Google Scholar] [CrossRef] [PubMed]
- Bluemel, M.; Dally, N.; Jung, C. Flowering time regulation in crops—What did we learn from Arabidopsis? Curr. Opin. Biotechnol. 2015, 32, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Dally, N.; Xiao, K.; Holtgräwe, D.; Jung, C. The B2 flowering time locus of beet encodes a zinc finger transcription factor. Proc. Natl. Acad. Sci. USA 2014, 111, 10365–10370. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.R.; Ai, X.Y.; Zhang, J.Z. Genetic regulation of flowering time in annual and perennial plants. Wiley Interdiscip. Rev. RNA 2014, 5, 347–359. [Google Scholar] [CrossRef] [PubMed]
- Srikanth, A.; Schmid, M. Regulation of flowering time: All roads lead to Rome. Cell. Mol. Life Sci. 2011, 68, 2013–2037. [Google Scholar] [CrossRef] [PubMed]
- Smaczniak, C.; Immink, R.G.; Muiño, J.M.; Blanvillain, R.; Busscher, M.; Busscher-Lange, J.; Dinh, Q.P.; Liu, S.; Westphal, A.H.; Boeren, S. Characterization of MADS-domain transcription factor complexes in Arabidopsis flower development. Proc. Natl. Acad. Sci. USA 2012, 109, 1560–1565. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.Y.; Kim, Y.; Kim, S.Y.; Lee, J.S.; Ahn, J.H. Control of flowering time and cold response by a NAC-domain protein in Arabidopsis. PLoS ONE 2007, 2, e642. [Google Scholar] [CrossRef] [PubMed]
- Shin, B.; Choi, G.; Yi, H.; Yang, S.; Cho, I.; Kim, J.; Lee, S.; Paek, N.C.; Kim, J.H.; Song, P.S. AtMYB21, a gene encoding a flower-specific transcription factor, is regulated by COP1. Plant J. 2002, 30, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Tong, Z.; Hong, B.; Yang, Y.; Li, Q.; Ma, N.; Ma, C.; Gao, J. Overexpression of two chrysanthemum DgDREB1 group genes causing delayed flowering or dwarfism in Arabidopsis. Plant Mol. Biol. 2009, 71, 115–129. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Liu, Z.; Wang, L.; Kim, S.G.; Seo, P.J.; Qiao, M.; Wang, N.; Li, S.; Cao, X.; Park, C.M. WRKY71 accelerates flowering via the direct activation of FLOWERING LOCUS T and LEAFY in Arabidopsis thaliana. Plant J. 2016, 85, 96–106. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zhao, D.; Zheng, C.; Chen, C.; Peng, X.; Cheng, Y.; Wan, H. Genomic Analysis of the ASMT Gene Family in Solanum lycopersicum. Molecules 2017, 22, 1984. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.-J.; Zhang, Y.-C.; Li, Q.-H.; Sang, Y.; Mao, J.; Lian, H.-L.; Wang, L.; Yang, H.-Q. COP1-mediated ubiquitination of CONSTANS is implicated in cryptochrome regulation of flowering in Arabidopsis. Plant Cell 2008, 20, 292–306. [Google Scholar] [CrossRef] [PubMed]
- McNellis, T.W.; Von Arnim, A.G.; Araki, T.; Komeda, Y.; Miséra, S.; Deng, X.-W. Genetic and molecular analysis of an allelic series of cop1 mutants suggests functional roles for the multiple protein domains. Plant Cell 1994, 6, 487–500. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.-W.; Rubio, V.; Lee, N.-Y.; Bai, S.; Lee, S.-Y.; Kim, S.-S.; Liu, L.; Zhang, Y.; Irigoyen, M.L.; Sullivan, J.A. COP1 and ELF3 control circadian function and photoperiodic flowering by regulating GI stability. Mol. Cell 2008, 32, 617–630. [Google Scholar] [CrossRef] [PubMed]
- Smalle, J.; Vierstra, R.D. The ubiquitin 26S proteasome proteolytic pathway. Annu. Rev. Plant Biol. 2004, 55, 555–590. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Ménard, R.; Berr, A.; Fuchs, J.; Cognat, V.; Meyer, D.; Shen, W.H. The E2 ubiquitin-conjugating enzymes, AtUBC1 and AtUBC2, play redundant roles and are involved in activation of FLC expression and repression of flowering in Arabidopsis thaliana. Plant J. 2009, 57, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.K. Genes uniquely expressed in vegetative and potassium chlorate induced floral buds of Dimocarpus longan. Plant Sci. 2006, 170, 500–510. [Google Scholar] [CrossRef]
- Jia, T.; Wei, D.; Meng, S.; Allan, A.C.; Zeng, L. Identification of regulatory genes implicated in continuous flowering of longan (Dimocarpus longan L.). PLoS ONE 2014, 9, e114568. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Shi, S.; Li, W.; Shu, B.; Liu, L.; Xie, J.; Wei, Y. Transcriptome analysis of ‘Sijihua’longan (Dimocarpus longan L.) based on next-generation sequencing technology. J. Hortic. Sci. Biotechnol. 2016, 91, 180–188. [Google Scholar] [CrossRef]
- Lin, Y.; Min, J.; Lai, R.; Wu, Z.; Chen, Y.; Yu, L.; Cheng, C.; Jin, Y.; Tian, Q.; Liu, Q. Genome-wide sequencing of longan (Dimocarpus longan Lour.) provides insights into molecular basis of its polyphenol-rich characteristics. GigaScience 2017, 6, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Kraft, E.; Stone, S.L.; Ma, L.; Su, N.; Gao, Y.; Lau, O.-S.; Deng, X.-W.; Callis, J. Genome analysis and functional characterization of the E2 and RING-type E3 ligase ubiquitination enzymes of Arabidopsis. Plant Physiol. 2005, 139, 1597–1611. [Google Scholar] [CrossRef] [PubMed]
- Bae, H.; Kim, W.T. The N-terminal tetra-peptide (IPDE) short extension of the U-box motif in rice SPL11 E3 is essential for the interaction with E2 and ubiquitin-ligase activity. Biochem. Biophys. Res. Commun. 2013, 433, 266–271. [Google Scholar] [CrossRef] [PubMed]
- Jue, D.; Sang, X.; Shu, B.; Liu, L.; Wang, Y.; Jia, Z.; Zou, Y.; Shi, S. Characterization and expression analysis of genes encoding ubiquitin conjugating domain-containing enzymes in Carica papaya. PLoS ONE 2017, 12, e0171357. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Xie, T.; Chen, C.; Luan, A.; Long, J.; Li, C.; Ding, Y.; He, Y. Genome-wide organization and expression profiling of the R2R3-MYB transcription factor family in pineapple (Ananas comosus). BMC Genom. 2017, 18, 503. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Jiang, W.; Liu, J.; Li, Y.; Gai, J.; Li, Y. Genome-wide characterization of the aldehyde dehydrogenase gene superfamily in soybean and its potential role in drought stress response. BMC Genom. 2017, 18, 518. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wen, P.; Lau, O.S.; Deng, X.W. Characterization of the Ubiquitin E2 enzyme variant gene family in Arabidopsis. J. Int. Plant Biol. 2007, 49, 120–126. [Google Scholar] [CrossRef]
- Jeon, E.H.; Pak, J.H.; Kim, M.J.; Kim, H.J.; Shin, S.H.; Lee, J.H.; Kim, D.H.; Oh, J.S.; Oh, B.-J.; Jung, H.W. Ectopic expression of ubiquitin-conjugating enzyme gene from wild rice, OgUBC1, confers resistance against UV-B radiation and Botrytis infection in Arabidopsis thaliana. Biochem. Biophys. Res. Commun. 2012, 427, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Schmidt, W. A lysine-63-linked ubiquitin chain-forming conjugase, UBC13, promotes the developmental responses to iron deficiency in Arabidopsis roots. Plant J. 2010, 62, 330–343. [Google Scholar] [CrossRef] [PubMed]
- Wen, R.; Wang, S.; Xiang, D.; Venglat, P.; Shi, X.; Zang, Y.; Datla, R.; Xiao, W.; Wang, H. UBC13, an E2 enzyme for Lys63-linked ubiquitination, functions in root development by affecting auxin signaling and Aux/IAA protein stability. Plant J. 2014, 80, 424–436. [Google Scholar] [CrossRef] [PubMed]
- Razafimandimbison, S.G.; Ekman, S.; McDowell, T.D.; Bremer, B. Evolution of growth habit, inflorescence architecture, flower size, and fruit type in rubiaceae: Its ecological and evolutionary implications. PLoS ONE 2012, 7, e40851. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wang, H.; Yu, D. The Arabidopsis WRKY transcription factors WRKY12 and WRKY13 oppositely regulate flowering under short-day conditions. Mol. Plant 2016, 9, 1492–1503. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Dai, Y.; Cui, S.; Ma, L. Histone H2B monoubiquitination in the chromatin of FLOWERING LOCUS C regulates flowering time in Arabidopsis. Plant Cell 2008, 20, 2586–2602. [Google Scholar] [CrossRef] [PubMed]
- Zhiguo, E.; Zhang, Y.; Li, T.; Wang, L.; Zhao, H. Characterization of the ubiquitin-conjugating enzyme gene family in rice and evaluation of expression profiles under abiotic stresses and hormone treatments. PLoS ONE 2015, 10, e0122621. [Google Scholar]
- Cho, S.K.; Ryu, M.Y.; Seo, D.H.; Kang, B.G.; Kim, W.T. The Arabidopsis RING E3 ubiquitin ligase AtAIRP2 plays combinatory roles with AtAIRP1 in abscisic acid-mediated drought stress responses. Plant Physiol. 2011, 157, 2240–2257. [Google Scholar] [CrossRef] [PubMed]
- Baranwal, V.K.; Negi, N.; Khurana, P. Genome-wide identification and structural, functional and evolutionary analysis of WRKY components of mulberry. Sci. Rep. 2016, 6, 30794. [Google Scholar] [CrossRef] [PubMed]
- Lippok, B.; Birkenbihl, R.P.; Rivory, G.; Brümmer, J.; Schmelzer, E.; Logemann, E.; Somssich, I.E. Expression of AtWRKY33 encoding a pathogen- or PAMP-responsive WRKY transcription factor is regulated by a composite DNA motif containing W box elements. Mol. Plant-Microbe Interact. 2007, 20, 420–429. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Patra, B.; Li, R.; Pattanaik, S.; Yuan, L. Promoter analysis reveals cis-regulatory motifs associated with the expression of the WRKY transcription factor CrWRKY1 in Catharanthus roseus. Planta 2013, 238, 1039–1049. [Google Scholar] [CrossRef] [PubMed]
- Finn, R.D.; Coggill, P.; Eberhardt, R.Y.; Eddy, S.R.; Mistry, J.; Mitchell, A.L.; Potter, S.C.; Punta, M.; Qureshi, M.; Sangrador-Vegas, A. The Pfam protein families database: Towards a more sustainable future. Nucleic Acids Res. 2016, 44, D279–D285. [Google Scholar] [CrossRef] [PubMed]
- Letunic, I.; Copley, R.R.; Schmidt, S.; Ciccarelli, F.D.; Doerks, T.; Schultz, J.; Ponting, C.P.; Bork, P. SMART 4.0: Towards genomic data integration. Nucleic Acids Res. 2004, 32 (Suppl. 1), D142–D144. [Google Scholar] [CrossRef] [PubMed]
- Gasteiger, E.; Gattiker, A.; Hoogland, C.; Ivanyi, I.; Appel, R.D.; Bairoch, A. ExPASy: The proteomics server for in-depth protein knowledge and analysis. Nucleic Acids Res. 2003, 31, 3784–3788. [Google Scholar] [CrossRef] [PubMed]
- Guo, A.-Y.; Zhu, Q.; Chen, X.; Luo, J. GSDS: A gene structure display server. Yi Chuan Hered. 2007, 29, 1023–1026. [Google Scholar] [CrossRef]
- Bailey, T.L.; Boden, M.; Buske, F.A.; Frith, M.; Grant, C.E.; Clementi, L.; Ren, J.; Li, W.W.; Noble, W.S. MEME SUITE: Tools for motif discovery and searching. Nucleic Acids Res. 2009, 37 (Suppl. 2), W202–W208. [Google Scholar] [CrossRef]
- Zhou, Q.; Guo, J.J.; He, C.T.; Shen, C.; Huang, Y.Y.; Chen, J.X.; Guo, J.H.; Yuan, J.G.; Yang, Z.Y. Comparative Transcriptome Analysis between Low- and High-Cadmium-Accumulating Genotypes of Pakchoi (Brassica chinensis L.) in Response to Cadmium Stress. Environ. Sci. Technol. 2016, 50, 6485–6494. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. MEGA6: Molecular evolutionary genetics analysis version 6.0. Mol. Biol. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef] [PubMed]
- Saeed, A.; Sharov, V.; White, J.; Li, J.; Liang, W.; Bhagabati, N.; Braisted, J.; Klapa, M.; Currier, T.; Thiagarajan, M. TM4: A free, open-source system for microarray data management and analysis. Biotechniques 2003, 34, 374. [Google Scholar] [PubMed]
- Qiu, J.; Wu, D. A study on flower differentiation of ‘Shixia’ longan (Dimocarpus longana lour.cv. Shixia). J. South China Agric. Univ. 2001, 1, 6. [Google Scholar]
Sample Availability: Samples of the compounds are available from the authors. |




| Gene Name | Gene Locus ID | Location | ORF (bp) | Size (aa) | UBCc Domain | PI | MW (KDa) | Intron | Full Length |
|---|---|---|---|---|---|---|---|---|---|
| DlUBC1 | Dlo_000106.1 | scaffold1:985697:987091 | 567 | 188 | 32–174 | 7.08 | 21.65 | 4 | 1395 |
| DlUBC2 | Dlo_000292.1 | scaffold1:3030633:3033521 | 483 | 160 | 8–158 | 8.42 | 18 | 4 | 2888 |
| DlUBC3 | Dlo_026265.1 | scaffold6:2172282:2176388 | 459 | 152 | 7–150 | 5.37 | 17.35 | 4 | 4106 |
| DlUBC4 | Dlo_032644.1 | scaffold9:121161:124354 | 519 | 172 | 29–172 | 6.82 | 18.74 | 3 | 3193 |
| DlUBC5 | Dlo_002066.1 | scaffold11:28768:34251 | 447 | 148 | 4–147 | 7.72 | 16.49 | 3 | 5483 |
| DlUBC6 | Dlo_009607.1 | scaffold19:759717:760407 | 330 | 109 | 1–107 | 6.50 | 12.75 | 2 | 691 |
| DlUBC7 | Dlo_011099.1 | scaffold21:38074:42650 | 2769 | 922 | 676–833 | 4.77 | 102.86 | 6 | 4576 |
| DlUBC8 | Dlo_012344.1 | scaffold23:232655:235481 | 528 | 175 | 1–139 | 4.18 | 19.99 | 5 | 2826 |
| DlUBC9 | Dlo_014547.1 | scaffold27:1628072:1630343 | 462 | 153 | 8–152 | 4.83 | 17.48 | 3 | 2272 |
| DlUBC10 | Dlo_016672.1 | scaffold32:473094:475341 | 537 | 178 | 1–139 | 4.16 | 20.08 | 5 | 2247 |
| DlUBC11 | Dlo_021485.1 | scaffold45:1169893:1172427 | 486 | 161 | 18–161 | 7.00 | 18.43 | 5 | 2534 |
| DlUBC12 | Dlo_024241.1 | scaffold54:747570:752709 | 3438 | 1145 | 894–1054 | 4.57 | 126.46 | 6 | 5139 |
| DlUBC13 | dlo_034965.1 | scaffold54:903777:904902 | 447 | 148 | 4–147 | 7.68 | 16.51 | 3 | 1125 |
| DlUBC14 | Dlo_024956.1 | scaffold56:49487:50629 | 1143 | 380 | 79–236 | 9.82 | 42.60 | 0 | 1142 |
| DlUBC15 | Dlo_024957.1 | scaffold56:57967:58685 | 582 | 193 | 1–136 | 9.08 | 21.98 | 1 | 719 |
| DlUBC16 | Dlo_024978.1 | scaffold56:225690:227300 | 915 | 304 | 14–174 | 5.89 | 34.00 | 4 | 1610 |
| DlUBC17 | Dlo_024979.1 | scaffold56:228547:229700 | 540 | 179 | 14–155 | 6.59 | 20.69 | 3 | 1153 |
| DlUBC18 | Dlo_026358.1 | scaffold60:843621:845935 | 447 | 148 | 4–147 | 7.69 | 16.39 | 3 | 2314 |
| DlUBC19 | Dlo_032160.2 | scaffold87:1001138:1002555 | 612 | 203 | 61–203 | 5.50 | 22.39 | 6 | 1417 |
| DlUBC20 | Dlo_033394.1 | scaffold94:238050:239736 | 330 | 109 | 1–107 | 5.50 | 12.44 | 3 | 1686 |
| DlUBC21 | Dlo_034066.1 | scaffold99:423532:424482 | 267 | 88 | 1–86 | 8.82 | 9.90 | 4 | 950 |
| DlUBC22 | Dlo_001351.1 | scaffold105:47204:51064 | 447 | 148 | 4–147 | 7.72 | 16.58 | 3 | 4250 |
| DlUBC23 | Dlo_005400.1 | scaffold139:303181:306533 | 585 | 194 | 5–150 | 4.72 | 21.26 | 4 | 3352 |
| DlUBC24 | Dlo_009107.1 | scaffold182:25126:28034 | 630 | 209 | 74–173 | 5.30 | 24.28 | 4 | 2908 |
| DlUBC25 | Dlo_009840.1 | scaffold192:53161:56243 | 447 | 148 | 4–147 | 7.72 | 16.55 | 3 | 3082 |
| DlUBC26 | Dlo_009841.1 | scaffold192:56889:58714 | 474 | 157 | 2–131 | 8.44 | 17.85 | 3 | 1825 |
| DlUBC27 | Dlo_010855.1 | scaffold204:275311:285221 | 2088 | 695 | 332–489 | 4.79 | 77.60 | 7 | 9890 |
| DlUBC28 | Dlo_013399.1 | scaffold247:138383:139816 | 447 | 148 | 4–147 | 7.72 | 16.46 | 3 | 1433 |
| DlUBC29 | Dlo_013592.1 | scaffold250:137505:139550 | 552 | 183 | 32–174 | 8.61 | 21.01 | 4 | 2045 |
| DlUBC30 | Dlo_015182.2 | scaffold286:460274:460720 | 447 | 148 | 29–142 | 8.55 | 17.22 | 0 | 446 |
| DlUBC31 | Dlo_016190.1 | scaffold303:303486:307073 | 1617 | 538 | 258–415 | 5.99 | 59.98 | 6 | 3587 |
| DlUBC32 | Dlo_017847.1 | scaffold347:238580:242477 | 735 | 244 | 1–132 | 9.60 | 26.47 | 4 | 3897 |
| DlUBC33 | Dlo_019295.1 | scaffold388:24595:27323 | 1731 | 576 | 9–161 | 8.39 | 63.39 | 5 | 2728 |
| DlUBC34 | Dlo_021725.2 | scaffold459:127808:133234 | 462 | 153 | 8–151 | 6.74 | 17.21 | 7 | 5426 |
| DlUBC35 | Dlo_031542.1 | scaffold832:7085:11030 | 717 | 238 | 8–162 | 8.78 | 26.98 | 8 | 3945 |
| DlUBC36 | Dlo_032039.1 | scaffold860:46363:48204 | 315 | 104 | 1–89 | 4.63 | 11.71 | 2 | 1841 |
| DlUBC37 | Dlo_001660.1 | scaffold1077:94570:95523 | 954 | 317 | 68–228 | 6.08 | 37.35 | 0 | 953 |
| DlUBC38 | Dlo_006580.1 | scaffold1489:71154:72782 | 483 | 160 | 8–158 | 8.39 | 18.06 | 4 | 1628 |
| DlUBC39 | Dlo_008135.1 | scaffold1681:29423:31993 | 510 | 169 | 4–134 | 4.33 | 19.25 | 4 | 2570 |
| DlUBC40 | Dlo_008607.1 | scaffold17588:7:484 | 243 | 80 | 2–68 | 4.12 | 9.04 | 2 | 478 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jue, D.; Sang, X.; Liu, L.; Shu, B.; Wang, Y.; Xie, J.; Liu, C.; Shi, S. The Ubiquitin-Conjugating Enzyme Gene Family in Longan (Dimocarpus longan Lour.): Genome-Wide Identification and Gene Expression during Flower Induction and Abiotic Stress Responses. Molecules 2018, 23, 662. https://doi.org/10.3390/molecules23030662
Jue D, Sang X, Liu L, Shu B, Wang Y, Xie J, Liu C, Shi S. The Ubiquitin-Conjugating Enzyme Gene Family in Longan (Dimocarpus longan Lour.): Genome-Wide Identification and Gene Expression during Flower Induction and Abiotic Stress Responses. Molecules. 2018; 23(3):662. https://doi.org/10.3390/molecules23030662
Chicago/Turabian StyleJue, Dengwei, Xuelian Sang, Liqin Liu, Bo Shu, Yicheng Wang, Jianghui Xie, Chengming Liu, and Shengyou Shi. 2018. "The Ubiquitin-Conjugating Enzyme Gene Family in Longan (Dimocarpus longan Lour.): Genome-Wide Identification and Gene Expression during Flower Induction and Abiotic Stress Responses" Molecules 23, no. 3: 662. https://doi.org/10.3390/molecules23030662





