K2S2O8-Promoted Aryl Thioamides Synthesis from Aryl Aldehydes Using Thiourea as the Sulfur Source
Abstract
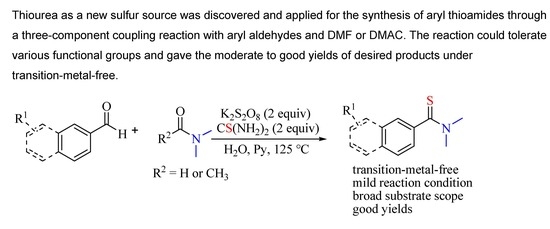
:1. Introduction
2. Results and Discussion
3. Materials and Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Petrov, K.A.; Andreev, L.N. The Chemical Properties of Thioamides. Russ. Chem. Rev. 1971, 40, 505–524. [Google Scholar] [CrossRef]
- Cremlyn, R.J. An Introduction to Organo-Sulfur Chemistry; Wiley & Sons: New York, NY, USA, 1996. [Google Scholar]
- Jiang, W.; Li, Y.; Wang, Z. Heteroarenes as high performance organic semiconductors. Chem. Soc. Rev. 2013, 42, 6113–6127. [Google Scholar] [CrossRef] [PubMed]
- Jagodziński, T.S. Thioamides as Useful Synthons in the Synthesis of Heterocycles. Chem. Rev. 2003, 103, 197–228. [Google Scholar] [CrossRef] [PubMed]
- Lincke, T.; Behnken, S.; Ishida, K.; Roth, M.; Hertweck, C. Closthioamide: An Unprecedented Polythioamide Antibiotic from the Strictly Anaerobic Bacterium Clostridium cellulolyticum. Angew. Chem. Int. Ed. 2010, 49, 2011–2013. [Google Scholar] [CrossRef] [PubMed]
- Shen, C.; Zhang, P.F.; Sun, Q.; Bai, S.Q.; Andy Hor, T.S.; Liu, X.G. Recent advances in C–S bond formation via C–H bond functionalization and decarboxylation. Chem. Soc. Rev. 2015, 44, 291–314. [Google Scholar] [CrossRef] [PubMed]
- Anthony, J.E. Functionalized Acenes and Heteroacenes for Organic Electronics. Chem. Rev. 2006, 106, 5028–5048. [Google Scholar] [CrossRef] [PubMed]
- Ashfaq, M.; Shah, S.S.A.; Najam, T.; Ahmad, M.M.; Tabassum, R.; Rivera, G. Synthetic Thioamide, Benzimidazole, Quinolone and Derivatives with Carboxylic Acid and Ester Moieties: A Strategy in the Design of Antituberculosis Agents. Curr. Med. Chem. 2014, 21, 911–931. [Google Scholar] [CrossRef] [PubMed]
- Zoumpoulakis, P.; Camoutsis, C.; Pairas, G.; Sokovic, M.; Glamoclija, J.; Potamitis, C.; Pitsas, A. Synthesis of novel sulfonamide-1,2,4-triazoles, 1,3,4-thiadiazoles and 1,3,4-oxadiazoles, as potential antibacterial and antifungal agents. Biological evaluation and conformational analysis studies. Bioorg. Med. Chem. 2012, 20, 1569–1583. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Fu, Y.Z. A Perspective on Energy Densities of Rechargeable Li-S Batteries and Alternative Sulfur-Based Cathode Materials. Energy Environ. Mater. 2018, 1, 20–27. [Google Scholar] [CrossRef]
- Polshettiwar, V. Phosphorus Pentasulfide (P4S10). Synlett 2004, 12, 2245–2246. [Google Scholar] [CrossRef]
- Ozturk, T.; Ertas, E.; Mert, O. Use of Lawesson’s Reagent in Organic Syntheses. Chem. Rev. 2007, 107, 5210–5278. [Google Scholar] [CrossRef] [PubMed]
- Vásquez-Céspedes, S.; Ferry, A.; Candish, L.; Glorius, F. Heterogeneously Catalyzed Direct C–H Thiolation of Heteroarenes. Angew. Chem., Int. Ed. 2015, 54, 5772–5776. [Google Scholar] [CrossRef] [PubMed]
- Jiao, J.; Wei, L.; Ji, X.M.; Hu, M.L.; Tang, R.Y. Direct Introduction of Dithiocarbamates onto Imidazoheterocycles under Mild Conditions. Adv. Synth. Catal. 2016, 358, 268–275. [Google Scholar] [CrossRef]
- Rafique, J.; Saba, S.; Rosário, A.R.; Braga, A.L. Regioselective, Solvent- and Metal-Free Chalcogenation of Imidazo[1,2-a] pyridines by Employing I2/DMSO as the Catalytic Oxidation System. Chem. Eur. J. 2016, 22, 11854–11862. [Google Scholar] [CrossRef] [PubMed]
- Ding, Q.P.; Cao, B.P.; Yuan, J.J.; Liu, X.J.; Peng, Y.Y. Synthesis of thioethers via metal-free reductive coupling of tosylhydrazones with thiols. Org. Biomol. Chem. 2011, 9, 748–751. [Google Scholar] [CrossRef] [PubMed]
- Ravi, C.; Mohan, D.C.; Adimurthy, S. N-Chlorosuccinimide-Promoted Regioselective Sulfenylation of Imidazoheterocycles at Room Temperature. Org. Lett. 2014, 16, 2978–2981. [Google Scholar] [CrossRef] [PubMed]
- Hiebel, M.A.; Berteina-Raboin, S. Iodine-catalyzed regioselective sulfenylation of imidazoheterocycles in PEG400. Green Chem. 2015, 17, 937–944. [Google Scholar] [CrossRef]
- Siddaraju, Y.; Prabhu, K.R. Iodine-Catalyzed Cross Dehydrogenative Coupling Reaction: A Regioselective Sulfenylation of Imidazoheterocycles Using Dimethyl Sulfoxide as an Oxidant. J. Org. Chem. 2016, 81, 7838–7846. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.L.; Tian, S.K. Iodine-Catalyzed Regioselective Sulfenylation of Indoles with Sulfonyl Hydrazides. Angew. Chem. Int. Ed. 2013, 52, 4929–4932. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhang, S.; Tang, L.; Hu, Y.B.; Zha, Z.G.; Wang, Z.Y. Catalyst-free thiolation of indoles with sulfonyl hydrazides for the synthesis of 3-sulfenylindoles in water. Green Chem. 2016, 18, 2609–2613. [Google Scholar] [CrossRef]
- Singh, R.; Allam, K.B.; Singh, N.; Kumari, K.; Singh, S.K. A Direct Metal-Free Decarboxylative Sulfono Functionalization (DSF) of Cinnamic Acids to α, β-Unsaturated Phenyl Sulfones. Org. Lett. 2015, 17, 2656–2659. [Google Scholar] [CrossRef] [PubMed]
- Senadi, G.C.; Guo, B.C.; Hu, W.P.; Wang, J.J. Iodine-promoted cyclization of N-propynyl amides and N-allyl amides via sulfonylation and sulfenylation. Chem. Commun. 2016, 52, 11410–11413. [Google Scholar] [CrossRef] [PubMed]
- Handa, S.; Fennewald, J.C.; Lipshutz, B.H. Aerobic Oxidation in Nanomicelles of Aryl Alkynes, in Water at Room Temperature. Angew. Chem. Int. Ed. 2014, 53, 3432–3435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rao, W.H.; Shi, B.F. Copper(II)-Catalyzed Direct Sulfonylation of C(sp2)–H Bonds with Sodium Sulfinates. Org. Lett. 2015, 17, 2784–2787. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Wu, W.; Zhao, W.; Li, Y.; Xie, P.; Huang, Y.; Liu, Y.; Zhou, A. Generation of thioethers via direct C–H functionalization with sodium benzenesulfinate as a sulfur source. Org. Biomol. Chem. 2016, 14, 1428–1431. [Google Scholar] [CrossRef] [PubMed]
- Xiao, F.; Chen, S.; Tian, J.; Huang, H.; Liu, Y.; Deng, G.J. Chemoselective cross-coupling reaction of sodium sulfinates with phenols under aqueous conditions. Green Chem. 2016, 18, 1538–1546. [Google Scholar] [CrossRef]
- Guo, Y.J.; Lu, S.; Tian, L.L.; Huang, E.L.; Hao, X.Q.; Zhu, X.J.; Shao, T.; Song, M.P. Iodine-Mediated Difunctionalization of Imidazopyridines with Sodium Sulfinates: Synthesis of Sulfones and Sulfides. J. Org. Chem. 2018, 83, 338–349. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Liu, Y.; Chen, J.F.; Yao, E.; Cheng, J. Multicomponent Coupling Reactions of Two N-Tosyl Hydrazones and Elemental Sulfur: Selective Denitrogenation Pathway toward Unsymmetric 2,5-Disubstituted 1,3,4-Thiadiazoles. Org. Lett. 2016, 18, 5268–5271. [Google Scholar] [CrossRef] [PubMed]
- Ravi, C.; Reddy, N.N.K.; Pappula, V.; Samanta, S.; Adimurthy, S. Copper-Catalyzed Three-Component System for Arylsulfenylation of Imidazopyridines with Elemental Sulfur. J. Org. Chem. 2016, 81, 9964–9972. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.R.; Liao, Y.Y.; Deng, J.C.; Feng, K.Y.; Zhang, M.; Ning, Y.Y.; Lin, Z.W.; Tang, R.Y. Oxidative dual C–H thiolation of imidazopyridines with ethers or alkanes using elemental sulphur. Chem. Commun. 2017, 53, 7784–7787. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.M.; Yang, Y.Z.; Xiao, G.H.; Song, J.X.; Liang, Y.; Deng, G.B. Double C–S bond formation via C–H bond functionalization: Synthesis of benzothiazoles and naphtho[2,1-d]thiazoles from N-substituted arylamines and elemental sulfur. Chem. Commun. 2017, 53, 11917–11920. [Google Scholar] [CrossRef] [PubMed]
- Bergman, J. Comparison of Two Reagents for Thionations. Synthesis 2018, 50, 2323–2328. [Google Scholar] [CrossRef]
- Hurd, R.N.; Delamater, G. The Preparation and Chemical Properties of Thionamides. Chem. Rev. 1961, 61, 45–86. [Google Scholar] [CrossRef]
- Wei, J.P.; Li, Y.M.; Jiang, X.F. Aqueous Compatible Protocol to Both Alkyl and Aryl Thioamide Synthesis. Org. Lett. 2016, 18, 340–343. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Yu, J.T.; Zhou, Y.N.; Jiang, Y.; Cheng, J. Aqueous MCRs of quaternary ammoniums, N-substituted formamides and sodium disulfide towards aryl thioamides. Org. Chem. Front. 2017, 4, 413–416. [Google Scholar] [CrossRef]
- Xu, K.; Li, Z.Y.; Cheng, F.Y.; Zuo, Z.Z.; Wang, T.; Wang, M.C.; Liu, L.T. Transition-Metal-Free Cleavage of C–C Triple Bonds in Aromatic Alkynes with S8 and Amides Leading to Aryl Thioamides. Org. Lett. 2018, 20, 2228–2231. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Vanjari, R.; Guntreddi, T.; Singh, K.N. Sulfur promoted decarboxylative thioamidation of carboxylic acids using formamides as amine proxy. Tetrahedron 2016, 72, 2012–2017. [Google Scholar] [CrossRef]
- Nguyen, T.B.; Tran, M.Q.; Ermolenko, L.; Al-Mourabit, A. Three-Component Reaction between Alkynes, Elemental Sulfur, and Aliphatic Amines: A General, Straightforward, and Atom Economical Approach to Thioamides. Org. Lett. 2014, 16, 310–313. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.M.; Jayalakshmi, M.; Reddy, R.S. Time-selective Hydrothermal Synthesis of SnS Nanorods and Nanoparticles by Thiourea Hydrolysis. Chem. Lett. 2004, 33, 1044–1045. [Google Scholar] [CrossRef]
- Jayalakshmi, M.; Rao, M.M. Synthesis of zinc sulphide nanoparticles by thiourea hydrolysis and their characterization for electrochemical capacitor applications. J. Power Sources 2006, 157, 624–629. [Google Scholar] [CrossRef]
- Zhang, K.; Han, Q.; Wang, X.; Zhu, J. One-Step Synthesis of Bi2S3/BiOX and Bi2S3/(BiO)2CO3 Heterojunction Photocatalysts by Using Aqueous Thiourea Solution as Both Solvent and Sulfur Source. ChemistrySelect 2016, 1, 6136–6145. [Google Scholar] [CrossRef]
- Manivel, P.; Prabakaran, K.; Krishnakumar, V.; Khan, F.N.; Maiyalagan, T. Thiourea-Mediated Regioselective Synthesis of Symmetrical and Unsymmetrical Diversified Thioethers. Ind. Eng. Chem. Res. 2014, 53, 7866–7870. [Google Scholar] [CrossRef]
- Niu, H.; Xia, C.; Qu, G.; Wu, S.; Jiang, Y.; Jin, X.; Guo, H. Microwave-Promoted “One-Pot” Synthesis of 4-Nitrobenzylthioinosine Analogues Using Thiourea as a Sulfur Precursor. Chem. Asian J. 2012, 7, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Firouzabadi, H.; Iranpoor, N.; Gholinejad, M. One-Pot Thioetherification of Aryl Halides Using Thiourea and Alkyl Bromides Catalyzed by Copper(I) Iodide Free from Foul-Smelling Thiols in Wet Polyethylene Glycol (PEG 200). Adv. Synth. Catal. 2010, 352, 119–124. [Google Scholar] [CrossRef]
- Mondal, J.; Modak, A.; Dutta, A.; Basu, S.; Jha, S.N.; Bhattacharyya, D.; Bhaumik, A. One-pot thioetherification of aryl halides with thiourea and benzylbromide in water catalyzed by Cu-grafted furfural imine-functionalized mesoporous SBA-15. Chem. Commun. 2012, 48, 8000–8002. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Yu, L.; Su, C.; Yang, Y.; Li, H.; Xu, Q. Efficient Generation of C-S Bonds via a By-Product-Promoted Selective Coupling of Alcohols, Organic Halides, and Thiourea. Adv. Synth. Catal. 2017, 359, 1649–1655. [Google Scholar] [CrossRef]
- Abbasi, M.; Khalifeh, R. One-pot odourless synthesis of thioesters via in situ generation of thiobenzoic acids using benzoic anhydrides and thiourea. Beilstein J. Org. Chem. 2015, 11, 1265–1273. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Swain, S.P.; Chou, Y.; Hou, D. Thioesterifications Free of Activating Agent and Thiol: A Three-Component Reaction of Carboxylic Acids, Thioureas, and Michael Acceptors. Adv. Synth. Catal. 2015, 357, 2644–2650. [Google Scholar] [CrossRef]
- Bezgubenko, L.V.; Pipko, S.E.; Sinitsa, A.D. Dichlorothiophosphoric acid and dichlorothiophosphate anion as thionating agents in the synthesis of thioamides. Russ. J. Gen. Chem. 2008, 78, 1341–1344. [Google Scholar] [CrossRef]
- Meltzer, R.I.; Lewis, A.D.; King, J.A. Antitubercular Substances. IV. Thioamides. J. Am. Chem. Soc. 1955, 77, 4062–4066. [Google Scholar] [CrossRef]
- Perregaard, J.; Lawesson, S.O. Studies on Organophosphoous Compounds. XI.* Oxidation of Aromatic Compounds with Sulfur in Hexamethylphosphoric Triamide (HMPA). A New Method for Preparation of N,N-Dimethylthiocarboxamides. Acta Chem. Scand. B 1975, 29, 604–608. [Google Scholar]
Sample Availability: Not available. |





| Entry | Oxidant (Equiv) | Concentration of H2O (M) | Yield (%) b |
|---|---|---|---|
| 1 | BPO (2) | 14 | 58 |
| 2 | BQ (2) | 14 | 0 |
| 3 | DTBP (2) | 14 | <5 |
| 4 | TBHP (2) | 14 | 20 |
| 5 | K2S2O8 (2) | 14 | 69 |
| 6 | (NH4)2S2O8 (2) | 14 | 55 |
| 7 | K2S2O8 (2) | 42 | 0 c |
| 8 | K2S2O8 (2) | 8 | 65 d |
| 9 | K2S2O8 (2) | 0 | 0 e |
| 10 | K2S2O8 (3) | 14 | 68 |
| 11 | K2S2O8 (1.8) | 14 | 61 |
| 12 | K2S2O8 (2) | 14 | 54 f |
| 13 | K2S2O8 (2) | 14 | 80 g |

| Entry | R | Yield (%) of 3 b |
|---|---|---|
| 1 | 4-Cl | 63 (3a) |
| 2 | H | 52 (3b) |
| 3 | 4-CH3 | 55 (3c) |
| 4 | 3-Br | 48 (3h) |
| 5 | 3-CH3 | 51 (3i) |
| 6 | 4-Ph | 60 (3n) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bian, Y.; Qu, X.; Chen, Y.; Li, J.; Liu, L. K2S2O8-Promoted Aryl Thioamides Synthesis from Aryl Aldehydes Using Thiourea as the Sulfur Source. Molecules 2018, 23, 2225. https://doi.org/10.3390/molecules23092225
Bian Y, Qu X, Chen Y, Li J, Liu L. K2S2O8-Promoted Aryl Thioamides Synthesis from Aryl Aldehydes Using Thiourea as the Sulfur Source. Molecules. 2018; 23(9):2225. https://doi.org/10.3390/molecules23092225
Chicago/Turabian StyleBian, Yongjun, Xingyu Qu, Yongqiang Chen, Jun Li, and Leng Liu. 2018. "K2S2O8-Promoted Aryl Thioamides Synthesis from Aryl Aldehydes Using Thiourea as the Sulfur Source" Molecules 23, no. 9: 2225. https://doi.org/10.3390/molecules23092225